Antibody data
- Antibody Data
- Antigen structure
- References [90]
- Comments [0]
- Validations
- Immunocytochemistry [11]
- Immunoprecipitation [1]
- Immunohistochemistry [3]
- Other assay [71]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 71-2700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Catenin Polyclonal Antibody (CAT-15)
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody can be used to specifically immunoprecipitate the ~ 92 kDa beta-catenin protein from native cell lysates. It is suitable for use in immunoprecipitation (IP) and IP/western applications. Note that when the antibody is used for straight western blotting, cross-reactivity with a ~100 kDa protein of unknown identity is sometimes observed. Suggested positive control lysates include HeLa and A431.
- Reactivity
- Human, Mouse, Rat, Chicken/Avian, Xenopus
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- CAT-15
- Vial size
- 100 μg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Shielding of actin by the endoplasmic reticulum impacts nuclear positioning.
Multi-Targeting Approach in Glioblastoma Using Computer-Assisted Drug Discovery Tools to Overcome the Blood-Brain Barrier and Target EGFR/PI3Kp110β Signaling.
A Drug Screening Reveals Minocycline Hydrochloride as a Therapeutic Option to Prevent Breast Cancer Cells Extravasation across the Blood-Brain Barrier.
PA1 participates in the maintenance of blood-testis barrier integrity via cooperation with JUN in the Sertoli cells of mice.
Food-Derived High Arginine Peptides Promote Spermatogenesis Recovery in Busulfan Treated Mice.
Somatic mutations of GNA11 and GNAQ in CTNNB1-mutant aldosterone-producing adenomas presenting in puberty, pregnancy or menopause.
Behind Brain Metastases Formation: Cellular and Molecular Alterations and Blood-Brain Barrier Disruption.
Pinhead antagonizes Admp to promote notochord formation.
Picturing Breast Cancer Brain Metastasis Development to Unravel Molecular Players and Cellular Crosstalk.
A β-catenin-driven switch in TCF/LEF transcription factor binding to DNA target sites promotes commitment of mammalian nephron progenitor cells.
Myocyte Enhancer Factor 2C as a New Player in Human Breast Cancer Brain Metastases.
IL-20 Cytokines Are Involved in Epithelial Lesions Associated with Virus-Induced COPD Exacerbation in Mice.
Ctdnep1 and Eps8L2 regulate dorsal actin cables for nuclear positioning during cell migration.
3,4,5-Tri-O-Caffeoylquinic Acid Promoted Hair Pigmentation Through β-Catenin and Its Target Genes.
Evaluation of morphokinetic characteristics of zona pellucida free mouse pre-implantation embryos using time-lapse monitoring system.
Sox17 and β-catenin co-occupy Wnt-responsive enhancers to govern the endoderm gene regulatory network.
Endogenously produced LG3/4/5-peptide protects testes against toxicant-induced injury.
Sevoflurane inhibits the proliferation and invasion of hepatocellular carcinoma cells through regulating the PTEN/Akt/GSK‑3β/β‑catenin signaling pathway by downregulating miR‑25‑3p.
Differential expression of cell-cell junction proteins in the testis, epididymis, and ductus deferens of domestic turkeys (Meleagris gallopavo) with white and yellow semen.
Astrocytes deliver CK1 to neurons via extracellular vesicles in response to inflammation promoting the translation and amyloidogenic processing of APP.
miR-187-3p increases gemcitabine sensitivity in breast cancer cells by targeting FGF9 expression.
Gfi1b regulates the level of Wnt/β-catenin signaling in hematopoietic stem cells and megakaryocytes.
β-catenin-mediated hair growth induction effect of 3,4,5-tri-O-caffeoylquinic acid.
Desmoplakin is required for epidermal integrity and morphogenesis in the Xenopus laevis embryo.
Planar cell polarity protein Dishevelled 3 (Dvl3) regulates ectoplasmic specialization (ES) dynamics in the testis through changes in cytoskeletal organization.
ERK1/2 Phosphorylation of FHOD Connects Signaling and Nuclear Positioning Alternations in Cardiac Laminopathy.
Fusobacterium nucleatum promotes colorectal cancer by inducing Wnt/β-catenin modulator Annexin A1.
[Di (2-ethylhexyl) phthalate-induced hypospadias in SD rats is related with Mafb expression: a transcriptome profiling-based study].
Dynein 1 supports spermatid transport and spermiation during spermatogenesis in the rat testis.
α-Catenin-dependent cytoskeletal tension controls Yap activity in the heart.
Downregulation of ROS1 enhances the therapeutic efficacy of arsenic trioxide in acute myeloid leukemia cell lines.
Development of a polarized pancreatic ductular cell epithelium for physiological studies.
Actin nucleator Spire 1 is a regulator of ectoplasmic specialization in the testis.
Secreted Giardia intestinalis cysteine proteases disrupt intestinal epithelial cell junctional complexes and degrade chemokines.
Loss of β-catenin in resident cardiac fibroblasts attenuates fibrosis induced by pressure overload in mice.
Intra-axonal Synthesis of SNAP25 Is Required for the Formation of Presynaptic Terminals.
Antagonistic Activities of Sox2 and Brachyury Control the Fate Choice of Neuro-Mesodermal Progenitors.
Bioengineered Submucosal Organoids for In Vitro Modeling of Colorectal Cancer.
Aroclor1254 disrupts the blood-testis barrier by promoting endocytosis and degradation of junction proteins via p38 MAPK pathway.
A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion.
Regulation of the blood-testis barrier by a local axis in the testis: role of laminin α2 in the basement membrane.
DTX3L is upregulated in glioma and is associated with glioma progression.
Perfluorooctanesulfonate (PFOS)-induced Sertoli cell injury through a disruption of F-actin and microtubule organization is mediated by Akt1/2.
Basement Membrane Laminin α2 Regulation of BTB Dynamics via Its Effects on F-Actin and Microtubule Cytoskeletons Is Mediated Through mTORC1 Signaling.
Genomic integration of Wnt/β-catenin and BMP/Smad1 signaling coordinates foregut and hindgut transcriptional programs.
A reductionist metastasis-on-a-chip platform for in vitro tumor progression modeling and drug screening.
Interleukin 1alpha-induced disruption of the Sertoli cell cytoskeleton affects gap junctional communication.
Polarity protein Crumbs homolog-3 (CRB3) regulates ectoplasmic specialization dynamics through its action on F-actin organization in Sertoli cells.
N-Cadherin Regulates Cell Migration Through a Rab5-Dependent Temporal Control of Macropinocytosis.
Hemodynamics associated with atrial fibrillation directly alters thrombotic potential of endothelial cells.
Glucose-Induced Oxidative Stress Reduces Proliferation in Embryonic Stem Cells via FOXO3A/β-Catenin-Dependent Transcription of p21(cip1).
Cucurbitacin B inhibits the stemness and metastatic abilities of NSCLC via downregulation of canonical Wnt/β-catenin signaling axis.
Overexpression of plastin 3 in Sertoli cells disrupts actin microfilament bundle homeostasis and perturbs the tight junction barrier.
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
Liver-Tumor Hybrid Organoids for Modeling Tumor Growth and Drug Response In Vitro.
Flutamide alters β-catenin expression and distribution, and its interactions with E-cadherin in the porcine corpus luteum of mid- and late pregnancy.
Flutamide alters the distribution of c-Src and affects the N-cadherin-β-catenin complex in the seminiferous epithelium of adult rat.
Musashi-1 maintains blood-testis barrier structure during spermatogenesis and regulates stress granule formation upon heat stress.
rpS6 regulates blood-testis barrier dynamics through Akt-mediated effects on MMP-9.
Environmental toxicants perturb human Sertoli cell adhesive function via changes in F-actin organization mediated by actin regulatory proteins.
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Intercellular adhesion molecule-2 is involved in apical ectoplasmic specialization dynamics during spermatogenesis in the rat.
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
Analysis of a Jup hypomorphic allele reveals a critical threshold for postnatal viability.
Filamin A is a regulator of blood-testis barrier assembly during postnatal development in the rat testis.
Regulation of the blood-testis barrier by coxsackievirus and adenovirus receptor.
Tumor necrosis factor α-mediated restructuring of the Sertoli cell barrier in vitro involves matrix metalloprotease 9 (MMP9), membrane-bound intercellular adhesion molecule-1 (ICAM-1) and the actin cytoskeleton.
Loss of cadherin-binding proteins β-catenin and plakoglobin in the heart leads to gap junction remodeling and arrhythmogenesis.
Focal adhesion kinase-Tyr407 and -Tyr397 exhibit antagonistic effects on blood-testis barrier dynamics in the rat.
Microtubule affinity-regulating kinase 4 (MARK4) is a component of the ectoplasmic specialization in the rat testis.
Drug transporters and blood--testis barrier function.
Opposing effects of Tcf3 and Tcf1 control Wnt stimulation of embryonic stem cell self-renewal.
A study to assess the assembly of a functional blood-testis barrier in developing rat testes.
c-Yes regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization in the seminiferous epithelium of rat testes.
Different responses in transformation of MDCK cells in 2D and 3D culture by v-Src as revealed by microarray techniques, RT-PCR and functional assays.
Immunofluorescent analysis of connexin-43 using monoclonal antibodies to its extracellular domain.
Immunofluorescent analysis of connexin-43 using monoclonal antibodies to its extracellular domain.
Drug transporter, P-glycoprotein (MDR1), is an integrated component of the mammalian blood-testis barrier.
The Wnt antagonists Frzb-1 and Crescent locally regulate basement membrane dissolution in the developing primary mouth.
N-cadherin haploinsufficiency affects cardiac gap junctions and arrhythmic susceptibility.
Rab4A GTPase catenin interactions are involved in cell junction dynamics in the testis.
C-terminal truncation of connexin43 changes number, size, and localization of cardiac gap junction plaques.
Disruption of Sertoli-germ cell adhesion function in the seminiferous epithelium of the rat testis can be limited to adherens junctions without affecting the blood-testis barrier integrity: an in vivo study using an androgen suppression model.
Blood-testis barrier dynamics are regulated by {alpha}2-macroglobulin via the c-Jun N-terminal protein kinase pathway.
TGF-beta3 regulates anchoring junction dynamics in the seminiferous epithelium of the rat testis via the Ras/ERK signaling pathway: An in vivo study.
Differential expression of E-cadherin, N-cadherin and beta-catenin in proximal and distal segments of the rat nephron.
Cadmium alters the localization of N-cadherin, E-cadherin, and beta-catenin in the proximal tubule epithelium.
Is the cadherin/catenin complex a functional unit of cell-cell actin-based adherens junctions in the rat testis?
Is the cadherin/catenin complex a functional unit of cell-cell actin-based adherens junctions in the rat testis?
Janota CS, Pinto A, Pezzarossa A, Machado P, Costa J, Campinho P, Franco CA, Gomes ER
Nature communications 2022 May 19;13(1):2763
Nature communications 2022 May 19;13(1):2763
Multi-Targeting Approach in Glioblastoma Using Computer-Assisted Drug Discovery Tools to Overcome the Blood-Brain Barrier and Target EGFR/PI3Kp110β Signaling.
Franco C, Kausar S, Silva MFB, Guedes RC, Falcao AO, Brito MA
Cancers 2022 Jul 19;14(14)
Cancers 2022 Jul 19;14(14)
A Drug Screening Reveals Minocycline Hydrochloride as a Therapeutic Option to Prevent Breast Cancer Cells Extravasation across the Blood-Brain Barrier.
Godinho-Pereira J, Lopes MD, Garcia AR, Botelho HM, Malhó R, Figueira I, Brito MA
Biomedicines 2022 Aug 16;10(8)
Biomedicines 2022 Aug 16;10(8)
PA1 participates in the maintenance of blood-testis barrier integrity via cooperation with JUN in the Sertoli cells of mice.
Liu B, Liu C, Ma B, Zhang R, Zhao Z, Xiao S, Cao W, Ma Y, Zhu G, Li W, Li Z
Cell & bioscience 2022 Apr 4;12(1):41
Cell & bioscience 2022 Apr 4;12(1):41
Food-Derived High Arginine Peptides Promote Spermatogenesis Recovery in Busulfan Treated Mice.
Liu W, Zhang L, Gao A, Khawar MB, Gao F, Li W
Frontiers in cell and developmental biology 2021;9:791471
Frontiers in cell and developmental biology 2021;9:791471
Somatic mutations of GNA11 and GNAQ in CTNNB1-mutant aldosterone-producing adenomas presenting in puberty, pregnancy or menopause.
Zhou J, Azizan EAB, Cabrera CP, Fernandes-Rosa FL, Boulkroun S, Argentesi G, Cottrell E, Amar L, Wu X, O'Toole S, Goodchild E, Marker A, Senanayake R, Garg S, Åkerström T, Backman S, Jordan S, Polubothu S, Berney DM, Gluck A, Lines KE, Thakker RV, Tuthill A, Joyce C, Kaski JP, Karet Frankl FE, Metherell LA, Teo AED, Gurnell M, Parvanta L, Drake WM, Wozniak E, Klinzing D, Kuan JL, Tiang Z, Gomez Sanchez CE, Hellman P, Foo RSY, Mein CA, Kinsler VA, Björklund P, Storr HL, Zennaro MC, Brown MJ
Nature genetics 2021 Sep;53(9):1360-1372
Nature genetics 2021 Sep;53(9):1360-1372
Behind Brain Metastases Formation: Cellular and Molecular Alterations and Blood-Brain Barrier Disruption.
Godinho-Pereira J, Garcia AR, Figueira I, Malhó R, Brito MA
International journal of molecular sciences 2021 Jun 30;22(13)
International journal of molecular sciences 2021 Jun 30;22(13)
Pinhead antagonizes Admp to promote notochord formation.
Itoh K, Ossipova O, Sokol SY
iScience 2021 Jun 25;24(6):102520
iScience 2021 Jun 25;24(6):102520
Picturing Breast Cancer Brain Metastasis Development to Unravel Molecular Players and Cellular Crosstalk.
Figueira I, Galego S, Custódio-Santos T, Vicente R, Molnár K, Haskó J, Malhó R, Videira M, Wilhelm I, Krizbai I, Brito MA
Cancers 2021 Feb 22;13(4)
Cancers 2021 Feb 22;13(4)
A β-catenin-driven switch in TCF/LEF transcription factor binding to DNA target sites promotes commitment of mammalian nephron progenitor cells.
Guo Q, Kim A, Li B, Ransick A, Bugacov H, Chen X, Lindström N, Brown A, Oxburgh L, Ren B, McMahon AP
eLife 2021 Feb 15;10
eLife 2021 Feb 15;10
Myocyte Enhancer Factor 2C as a New Player in Human Breast Cancer Brain Metastases.
Galego S, Kauppila LA, Malhó R, Pimentel J, Brito MA
Cells 2021 Feb 12;10(2)
Cells 2021 Feb 12;10(2)
IL-20 Cytokines Are Involved in Epithelial Lesions Associated with Virus-Induced COPD Exacerbation in Mice.
Le Roux M, Ollivier A, Kervoaze G, Beke T, Gillet L, Pichavant M, Gosset P
Biomedicines 2021 Dec 5;9(12)
Biomedicines 2021 Dec 5;9(12)
Ctdnep1 and Eps8L2 regulate dorsal actin cables for nuclear positioning during cell migration.
Calero-Cuenca FJ, Osorio DS, Carvalho-Marques S, Sridhara SC, Oliveira LM, Jiao Y, Diaz J, Janota CS, Cadot B, Gomes ER
Current biology : CB 2021 Apr 12;31(7):1521-1530.e8
Current biology : CB 2021 Apr 12;31(7):1521-1530.e8
3,4,5-Tri-O-Caffeoylquinic Acid Promoted Hair Pigmentation Through β-Catenin and Its Target Genes.
Bejaoui M, Villareal MO, Isoda H
Frontiers in cell and developmental biology 2020;8:175
Frontiers in cell and developmental biology 2020;8:175
Evaluation of morphokinetic characteristics of zona pellucida free mouse pre-implantation embryos using time-lapse monitoring system.
Kim J, Lee J, Choi YJ, Kwon O, Lee TB, Jun JH
The International journal of developmental biology 2020;64(7-8-9):415-422
The International journal of developmental biology 2020;64(7-8-9):415-422
Sox17 and β-catenin co-occupy Wnt-responsive enhancers to govern the endoderm gene regulatory network.
Mukherjee S, Chaturvedi P, Rankin SA, Fish MB, Wlizla M, Paraiso KD, MacDonald M, Chen X, Weirauch MT, Blitz IL, Cho KW, Zorn AM
eLife 2020 Sep 7;9
eLife 2020 Sep 7;9
Endogenously produced LG3/4/5-peptide protects testes against toxicant-induced injury.
Li L, Mao B, Wu S, Li H, Lv L, Ge R, Cheng CY
Cell death & disease 2020 Jun 8;11(6):436
Cell death & disease 2020 Jun 8;11(6):436
Sevoflurane inhibits the proliferation and invasion of hepatocellular carcinoma cells through regulating the PTEN/Akt/GSK‑3β/β‑catenin signaling pathway by downregulating miR‑25‑3p.
Cao Y, Lv W, Ding W, Li J
International journal of molecular medicine 2020 Jul;46(1):97-106
International journal of molecular medicine 2020 Jul;46(1):97-106
Differential expression of cell-cell junction proteins in the testis, epididymis, and ductus deferens of domestic turkeys (Meleagris gallopavo) with white and yellow semen.
Pardyak L, Kaminska A, Brzoskwinia M, Hejmej A, Kotula-Balak M, Jankowski J, Ciereszko A, Bilinska B
Poultry science 2020 Jan;99(1):555-566
Poultry science 2020 Jan;99(1):555-566
Astrocytes deliver CK1 to neurons via extracellular vesicles in response to inflammation promoting the translation and amyloidogenic processing of APP.
Li Z, Moniruzzaman M, Dastgheyb RM, Yoo SW, Wang M, Hao H, Liu J, Casaccia P, Nogueras-Ortiz C, Kapogiannis D, Slusher BS, Haughey NJ
Journal of extracellular vesicles 2020 Dec;10(2):e12035
Journal of extracellular vesicles 2020 Dec;10(2):e12035
miR-187-3p increases gemcitabine sensitivity in breast cancer cells by targeting FGF9 expression.
Wu Y, Tao L, Liang J, Qiao Y, Liu W, Yu H, Yu X, Liu L
Experimental and therapeutic medicine 2020 Aug;20(2):952-960
Experimental and therapeutic medicine 2020 Aug;20(2):952-960
Gfi1b regulates the level of Wnt/β-catenin signaling in hematopoietic stem cells and megakaryocytes.
Shooshtarizadeh P, Helness A, Vadnais C, Brouwer N, Beauchemin H, Chen R, Bagci H, Staal FJT, Coté JF, Möröy T
Nature communications 2019 Mar 20;10(1):1270
Nature communications 2019 Mar 20;10(1):1270
β-catenin-mediated hair growth induction effect of 3,4,5-tri-O-caffeoylquinic acid.
Bejaoui M, Villareal MO, Isoda H
Aging 2019 Jun 29;11(12):4216-4237
Aging 2019 Jun 29;11(12):4216-4237
Desmoplakin is required for epidermal integrity and morphogenesis in the Xenopus laevis embryo.
Bharathan NK, Dickinson AJG
Developmental biology 2019 Jun 15;450(2):115-131
Developmental biology 2019 Jun 15;450(2):115-131
Planar cell polarity protein Dishevelled 3 (Dvl3) regulates ectoplasmic specialization (ES) dynamics in the testis through changes in cytoskeletal organization.
Li L, Mao B, Yan M, Wu S, Ge R, Lian Q, Cheng CY
Cell death & disease 2019 Feb 26;10(3):194
Cell death & disease 2019 Feb 26;10(3):194
ERK1/2 Phosphorylation of FHOD Connects Signaling and Nuclear Positioning Alternations in Cardiac Laminopathy.
Antoku S, Wu W, Joseph LC, Morrow JP, Worman HJ, Gundersen GG
Developmental cell 2019 Dec 2;51(5):602-616.e12
Developmental cell 2019 Dec 2;51(5):602-616.e12
Fusobacterium nucleatum promotes colorectal cancer by inducing Wnt/β-catenin modulator Annexin A1.
Rubinstein MR, Baik JE, Lagana SM, Han RP, Raab WJ, Sahoo D, Dalerba P, Wang TC, Han YW
EMBO reports 2019 Apr;20(4)
EMBO reports 2019 Apr;20(4)
[Di (2-ethylhexyl) phthalate-induced hypospadias in SD rats is related with Mafb expression: a transcriptome profiling-based study].
Han X, Shao W, Yue Z, Xing L, Shen L, Long C, Zhang D, He D, Lin T, Wei G
Nan fang yi ke da xue xue bao = Journal of Southern Medical University 2019 Apr 30;39(4):456-463
Nan fang yi ke da xue xue bao = Journal of Southern Medical University 2019 Apr 30;39(4):456-463
Dynein 1 supports spermatid transport and spermiation during spermatogenesis in the rat testis.
Wen Q, Tang EI, Lui WY, Lee WM, Wong CKC, Silvestrini B, Cheng CY
American journal of physiology. Endocrinology and metabolism 2018 Nov 1;315(5):E924-E948
American journal of physiology. Endocrinology and metabolism 2018 Nov 1;315(5):E924-E948
α-Catenin-dependent cytoskeletal tension controls Yap activity in the heart.
Vite A, Zhang C, Yi R, Emms S, Radice GL
Development (Cambridge, England) 2018 Mar 8;145(5)
Development (Cambridge, England) 2018 Mar 8;145(5)
Downregulation of ROS1 enhances the therapeutic efficacy of arsenic trioxide in acute myeloid leukemia cell lines.
Li J
Oncology letters 2018 Jun;15(6):9392-9396
Oncology letters 2018 Jun;15(6):9392-9396
Development of a polarized pancreatic ductular cell epithelium for physiological studies.
O'Malley Y, Rotti PG, Thornell IM, Vanegas Calderón OG, Febres-Aldana C, Durham K, Yao J, Li X, Zhu Z, Norris AW, Zabner J, Engelhardt JF, Uc A
Journal of applied physiology (Bethesda, Md. : 1985) 2018 Jul 1;125(1):97-106
Journal of applied physiology (Bethesda, Md. : 1985) 2018 Jul 1;125(1):97-106
Actin nucleator Spire 1 is a regulator of ectoplasmic specialization in the testis.
Wen Q, Li N, Xiao X, Lui WY, Chu DS, Wong CKC, Lian Q, Ge R, Lee WM, Silvestrini B, Cheng CY
Cell death & disease 2018 Feb 12;9(2):208
Cell death & disease 2018 Feb 12;9(2):208
Secreted Giardia intestinalis cysteine proteases disrupt intestinal epithelial cell junctional complexes and degrade chemokines.
Liu J, Ma'ayeh S, Peirasmaki D, Lundström-Stadelmann B, Hellman L, Svärd SG
Virulence 2018 Dec 31;9(1):879-894
Virulence 2018 Dec 31;9(1):879-894
Loss of β-catenin in resident cardiac fibroblasts attenuates fibrosis induced by pressure overload in mice.
Xiang FL, Fang M, Yutzey KE
Nature communications 2017 Sep 28;8(1):712
Nature communications 2017 Sep 28;8(1):712
Intra-axonal Synthesis of SNAP25 Is Required for the Formation of Presynaptic Terminals.
Batista AFR, Martínez JC, Hengst U
Cell reports 2017 Sep 26;20(13):3085-3098
Cell reports 2017 Sep 26;20(13):3085-3098
Antagonistic Activities of Sox2 and Brachyury Control the Fate Choice of Neuro-Mesodermal Progenitors.
Koch F, Scholze M, Wittler L, Schifferl D, Sudheer S, Grote P, Timmermann B, Macura K, Herrmann BG
Developmental cell 2017 Sep 11;42(5):514-526.e7
Developmental cell 2017 Sep 11;42(5):514-526.e7
Bioengineered Submucosal Organoids for In Vitro Modeling of Colorectal Cancer.
Devarasetty M, Skardal A, Cowdrick K, Marini F, Soker S
Tissue engineering. Part A 2017 Oct;23(19-20):1026-1041
Tissue engineering. Part A 2017 Oct;23(19-20):1026-1041
Aroclor1254 disrupts the blood-testis barrier by promoting endocytosis and degradation of junction proteins via p38 MAPK pathway.
Jia X, Xu Y, Wu W, Fan Y, Wang G, Zhang T, Su W
Cell death & disease 2017 May 25;8(5):e2823
Cell death & disease 2017 May 25;8(5):e2823
A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion.
Labernadie A, Kato T, Brugués A, Serra-Picamal X, Derzsi S, Arwert E, Weston A, González-Tarragó V, Elosegui-Artola A, Albertazzi L, Alcaraz J, Roca-Cusachs P, Sahai E, Trepat X
Nature cell biology 2017 Mar;19(3):224-237
Nature cell biology 2017 Mar;19(3):224-237
Regulation of the blood-testis barrier by a local axis in the testis: role of laminin α2 in the basement membrane.
Gao Y, Mruk D, Chen H, Lui WY, Lee WM, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2017 Feb;31(2):584-597
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2017 Feb;31(2):584-597
DTX3L is upregulated in glioma and is associated with glioma progression.
Xu P, Tao X, Zhao C, Huang Q, Chang H, Ban N, Bei Y, Xia X, Shen C, Wang K, Xu L, Wu P, Ren J, Wang D
International journal of molecular medicine 2017 Aug;40(2):491-498
International journal of molecular medicine 2017 Aug;40(2):491-498
Perfluorooctanesulfonate (PFOS)-induced Sertoli cell injury through a disruption of F-actin and microtubule organization is mediated by Akt1/2.
Gao Y, Chen H, Xiao X, Lui WY, Lee WM, Mruk DD, Cheng CY
Scientific reports 2017 Apr 24;7(1):1110
Scientific reports 2017 Apr 24;7(1):1110
Basement Membrane Laminin α2 Regulation of BTB Dynamics via Its Effects on F-Actin and Microtubule Cytoskeletons Is Mediated Through mTORC1 Signaling.
Gao Y, Chen H, Lui WY, Lee WM, Cheng CY
Endocrinology 2017 Apr 1;158(4):963-978
Endocrinology 2017 Apr 1;158(4):963-978
Genomic integration of Wnt/β-catenin and BMP/Smad1 signaling coordinates foregut and hindgut transcriptional programs.
Stevens ML, Chaturvedi P, Rankin SA, Macdonald M, Jagannathan S, Yukawa M, Barski A, Zorn AM
Development (Cambridge, England) 2017 Apr 1;144(7):1283-1295
Development (Cambridge, England) 2017 Apr 1;144(7):1283-1295
A reductionist metastasis-on-a-chip platform for in vitro tumor progression modeling and drug screening.
Skardal A, Devarasetty M, Forsythe S, Atala A, Soker S
Biotechnology and bioengineering 2016 Sep;113(9):2020-32
Biotechnology and bioengineering 2016 Sep;113(9):2020-32
Interleukin 1alpha-induced disruption of the Sertoli cell cytoskeleton affects gap junctional communication.
Chojnacka K, Bilinska B, Mruk DD
Cellular signalling 2016 May;28(5):469-480
Cellular signalling 2016 May;28(5):469-480
Polarity protein Crumbs homolog-3 (CRB3) regulates ectoplasmic specialization dynamics through its action on F-actin organization in Sertoli cells.
Gao Y, Lui WY, Lee WM, Cheng CY
Scientific reports 2016 Jun 30;6:28589
Scientific reports 2016 Jun 30;6:28589
N-Cadherin Regulates Cell Migration Through a Rab5-Dependent Temporal Control of Macropinocytosis.
Wen MH, Wang JY, Chiu YT, Wang MP, Lee SP, Tai CY
Traffic (Copenhagen, Denmark) 2016 Jul;17(7):769-85
Traffic (Copenhagen, Denmark) 2016 Jul;17(7):769-85
Hemodynamics associated with atrial fibrillation directly alters thrombotic potential of endothelial cells.
Simmers MB, Cole BK, Ogletree ML, Chen Z, Xu Y, Kong LJ, Mackman N, Blackman BR, Wamhoff BR
Thrombosis research 2016 Jul;143:34-9
Thrombosis research 2016 Jul;143:34-9
Glucose-Induced Oxidative Stress Reduces Proliferation in Embryonic Stem Cells via FOXO3A/β-Catenin-Dependent Transcription of p21(cip1).
McClelland Descalzo DL, Satoorian TS, Walker LM, Sparks NR, Pulyanina PY, Zur Nieden NI
Stem cell reports 2016 Jul 12;7(1):55-68
Stem cell reports 2016 Jul 12;7(1):55-68
Cucurbitacin B inhibits the stemness and metastatic abilities of NSCLC via downregulation of canonical Wnt/β-catenin signaling axis.
Shukla S, Sinha S, Khan S, Kumar S, Singh K, Mitra K, Maurya R, Meeran SM
Scientific reports 2016 Feb 24;6:21860
Scientific reports 2016 Feb 24;6:21860
Overexpression of plastin 3 in Sertoli cells disrupts actin microfilament bundle homeostasis and perturbs the tight junction barrier.
Li N, Lee WM, Cheng CY
Spermatogenesis 2016 Apr;6(1):e1206353
Spermatogenesis 2016 Apr;6(1):e1206353
Actin-bundling protein plastin 3 is a regulator of ectoplasmic specialization dynamics during spermatogenesis in the rat testis.
Li N, Mruk DD, Wong CK, Lee WM, Han D, Cheng CY
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2015 Sep;29(9):3788-805
Liver-Tumor Hybrid Organoids for Modeling Tumor Growth and Drug Response In Vitro.
Skardal A, Devarasetty M, Rodman C, Atala A, Soker S
Annals of biomedical engineering 2015 Oct;43(10):2361-73
Annals of biomedical engineering 2015 Oct;43(10):2361-73
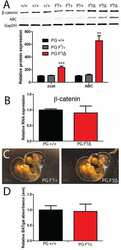
Flutamide alters β-catenin expression and distribution, and its interactions with E-cadherin in the porcine corpus luteum of mid- and late pregnancy.
Grzesiak M, Mitan A, Janik ME, Knapczyk-Stwora K, Slomczynska M
Histology and histopathology 2015 Nov;30(11):1341-52
Histology and histopathology 2015 Nov;30(11):1341-52
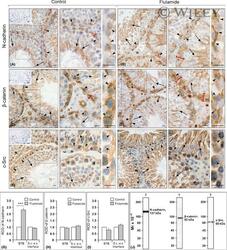
Flutamide alters the distribution of c-Src and affects the N-cadherin-β-catenin complex in the seminiferous epithelium of adult rat.
Zarzycka M, Chojnacka K, Mruk DD, Gorowska E, Hejmej A, Kotula-Balak M, Pardyak L, Bilinska B
Andrology 2015 May;3(3):569-81
Andrology 2015 May;3(3):569-81
Musashi-1 maintains blood-testis barrier structure during spermatogenesis and regulates stress granule formation upon heat stress.
ErLin S, WenJie W, LiNing W, BingXin L, MingDe L, Yan S, RuiFa H
Molecular biology of the cell 2015 May 15;26(10):1947-56
Molecular biology of the cell 2015 May 15;26(10):1947-56
rpS6 regulates blood-testis barrier dynamics through Akt-mediated effects on MMP-9.
Mok KW, Mruk DD, Cheng CY
Journal of cell science 2014 Nov 15;127(Pt 22):4870-82
Journal of cell science 2014 Nov 15;127(Pt 22):4870-82
Environmental toxicants perturb human Sertoli cell adhesive function via changes in F-actin organization mediated by actin regulatory proteins.
Xiao X, Mruk DD, Tang EI, Wong CK, Lee WM, John CM, Turek PJ, Silvestrini B, Cheng CY
Human reproduction (Oxford, England) 2014 Jun;29(6):1279-91
Human reproduction (Oxford, England) 2014 Jun;29(6):1279-91
Perfluorooctanesulfonate (PFOS) perturbs male rat Sertoli cell blood-testis barrier function by affecting F-actin organization via p-FAK-Tyr(407): an in vitro study.
Wan HT, Mruk DD, Wong CK, Cheng CY
Endocrinology 2014 Jan;155(1):249-62
Endocrinology 2014 Jan;155(1):249-62
Rai14 (retinoic acid induced protein 14) is involved in regulating f-actin dynamics at the ectoplasmic specialization in the rat testis*.
Qian X, Mruk DD, Cheng CY
PloS one 2013;8(4):e60656
PloS one 2013;8(4):e60656
Intercellular adhesion molecule-2 is involved in apical ectoplasmic specialization dynamics during spermatogenesis in the rat.
Xiao X, Cheng CY, Mruk DD
The Journal of endocrinology 2013 Jan;216(1):73-86
The Journal of endocrinology 2013 Jan;216(1):73-86
c-Yes regulates cell adhesion at the apical ectoplasmic specialization-blood-testis barrier axis via its effects on protein recruitment and distribution.
Xiao X, Mruk DD, Cheng CY
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
American journal of physiology. Endocrinology and metabolism 2013 Jan 15;304(2):E145-59
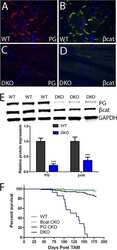
Analysis of a Jup hypomorphic allele reveals a critical threshold for postnatal viability.
Swope D, Li J, Muller EJ, Radice GL
Genesis (New York, N.Y. : 2000) 2012 Oct;50(10):717-27
Genesis (New York, N.Y. : 2000) 2012 Oct;50(10):717-27
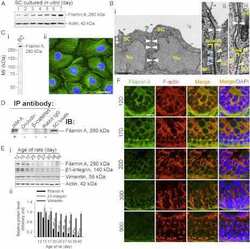
Filamin A is a regulator of blood-testis barrier assembly during postnatal development in the rat testis.
Su W, Mruk DD, Lie PP, Lui WY, Cheng CY
Endocrinology 2012 Oct;153(10):5023-35
Endocrinology 2012 Oct;153(10):5023-35
Regulation of the blood-testis barrier by coxsackievirus and adenovirus receptor.
Su L, Mruk DD, Cheng CY
American journal of physiology. Cell physiology 2012 Oct 15;303(8):C843-53
American journal of physiology. Cell physiology 2012 Oct 15;303(8):C843-53
Tumor necrosis factor α-mediated restructuring of the Sertoli cell barrier in vitro involves matrix metalloprotease 9 (MMP9), membrane-bound intercellular adhesion molecule-1 (ICAM-1) and the actin cytoskeleton.
Lydka M, Bilinska B, Cheng CY, Mruk DD
Spermatogenesis 2012 Oct 1;2(4):294-303
Spermatogenesis 2012 Oct 1;2(4):294-303
Loss of cadherin-binding proteins β-catenin and plakoglobin in the heart leads to gap junction remodeling and arrhythmogenesis.
Swope D, Cheng L, Gao E, Li J, Radice GL
Molecular and cellular biology 2012 Mar;32(6):1056-67
Molecular and cellular biology 2012 Mar;32(6):1056-67
Focal adhesion kinase-Tyr407 and -Tyr397 exhibit antagonistic effects on blood-testis barrier dynamics in the rat.
Lie PP, Mruk DD, Mok KW, Su L, Lee WM, Cheng CY
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 31;109(31):12562-7
Proceedings of the National Academy of Sciences of the United States of America 2012 Jul 31;109(31):12562-7
Microtubule affinity-regulating kinase 4 (MARK4) is a component of the ectoplasmic specialization in the rat testis.
Tang EI, Xiao X, Mruk DD, Qian XJ, Mok KW, Jenardhanan P, Lee WM, Mathur PP, Cheng CY
Spermatogenesis 2012 Apr 1;2(2):117-126
Spermatogenesis 2012 Apr 1;2(2):117-126
Drug transporters and blood--testis barrier function.
Su L, Mruk DD, Lee WM, Cheng CY
The Journal of endocrinology 2011 Jun;209(3):337-51
The Journal of endocrinology 2011 Jun;209(3):337-51
Opposing effects of Tcf3 and Tcf1 control Wnt stimulation of embryonic stem cell self-renewal.
Yi F, Pereira L, Hoffman JA, Shy BR, Yuen CM, Liu DR, Merrill BJ
Nature cell biology 2011 Jun 19;13(7):762-70
Nature cell biology 2011 Jun 19;13(7):762-70
A study to assess the assembly of a functional blood-testis barrier in developing rat testes.
Mok KW, Mruk DD, Lee WM, Cheng CY
Spermatogenesis 2011 Jul;1(3):270-280
Spermatogenesis 2011 Jul;1(3):270-280
c-Yes regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization in the seminiferous epithelium of rat testes.
Xiao X, Mruk DD, Lee WM, Cheng CY
The international journal of biochemistry & cell biology 2011 Apr;43(4):651-65
The international journal of biochemistry & cell biology 2011 Apr;43(4):651-65
Different responses in transformation of MDCK cells in 2D and 3D culture by v-Src as revealed by microarray techniques, RT-PCR and functional assays.
Töyli M, Rosberg-Kulha L, Capra J, Vuoristo J, Eskelinen S
Laboratory investigation; a journal of technical methods and pathology 2010 Jun;90(6):915-28
Laboratory investigation; a journal of technical methods and pathology 2010 Jun;90(6):915-28
Immunofluorescent analysis of connexin-43 using monoclonal antibodies to its extracellular domain.
Baklaushev VP, Gurina OI, Yusubalieva GM, Grinenko NF, Cytrin EB, Victorov IV, Chekhonin VP
Bulletin of experimental biology and medicine 2009 Oct;148(4):725-30
Bulletin of experimental biology and medicine 2009 Oct;148(4):725-30
Immunofluorescent analysis of connexin-43 using monoclonal antibodies to its extracellular domain.
Baklaushev VP, Gurina OI, Yusubalieva GM, Grinenko NF, Cytrin EB, Victorov IV, Chekhonin VP
Bulletin of experimental biology and medicine 2009 Oct;148(4):725-30
Bulletin of experimental biology and medicine 2009 Oct;148(4):725-30
Drug transporter, P-glycoprotein (MDR1), is an integrated component of the mammalian blood-testis barrier.
Su L, Cheng CY, Mruk DD
The international journal of biochemistry & cell biology 2009 Dec;41(12):2578-87
The international journal of biochemistry & cell biology 2009 Dec;41(12):2578-87
The Wnt antagonists Frzb-1 and Crescent locally regulate basement membrane dissolution in the developing primary mouth.
Dickinson AJ, Sive HL
Development (Cambridge, England) 2009 Apr;136(7):1071-81
Development (Cambridge, England) 2009 Apr;136(7):1071-81
N-cadherin haploinsufficiency affects cardiac gap junctions and arrhythmic susceptibility.
Li J, Levin MD, Xiong Y, Petrenko N, Patel VV, Radice GL
Journal of molecular and cellular cardiology 2008 Mar;44(3):597-606
Journal of molecular and cellular cardiology 2008 Mar;44(3):597-606
Rab4A GTPase catenin interactions are involved in cell junction dynamics in the testis.
Mruk DD, Lau AS, Sarkar O, Xia W
Journal of andrology 2007 Sep-Oct;28(5):742-54
Journal of andrology 2007 Sep-Oct;28(5):742-54
C-terminal truncation of connexin43 changes number, size, and localization of cardiac gap junction plaques.
Maass K, Shibayama J, Chase SE, Willecke K, Delmar M
Circulation research 2007 Dec 7;101(12):1283-91
Circulation research 2007 Dec 7;101(12):1283-91
Disruption of Sertoli-germ cell adhesion function in the seminiferous epithelium of the rat testis can be limited to adherens junctions without affecting the blood-testis barrier integrity: an in vivo study using an androgen suppression model.
Xia W, Wong CH, Lee NP, Lee WM, Cheng CY
Journal of cellular physiology 2005 Oct;205(1):141-57
Journal of cellular physiology 2005 Oct;205(1):141-57
Blood-testis barrier dynamics are regulated by {alpha}2-macroglobulin via the c-Jun N-terminal protein kinase pathway.
Wong CH, Mruk DD, Siu MK, Cheng CY
Endocrinology 2005 Apr;146(4):1893-908
Endocrinology 2005 Apr;146(4):1893-908
TGF-beta3 regulates anchoring junction dynamics in the seminiferous epithelium of the rat testis via the Ras/ERK signaling pathway: An in vivo study.
Xia W, Cheng CY
Developmental biology 2005 Apr 15;280(2):321-43
Developmental biology 2005 Apr 15;280(2):321-43
Differential expression of E-cadherin, N-cadherin and beta-catenin in proximal and distal segments of the rat nephron.
Prozialeck WC, Lamar PC, Appelt DM
BMC physiology 2004 May 17;4:10
BMC physiology 2004 May 17;4:10
Cadmium alters the localization of N-cadherin, E-cadherin, and beta-catenin in the proximal tubule epithelium.
Prozialeck WC, Lamar PC, Lynch SM
Toxicology and applied pharmacology 2003 Jun 15;189(3):180-95
Toxicology and applied pharmacology 2003 Jun 15;189(3):180-95
Is the cadherin/catenin complex a functional unit of cell-cell actin-based adherens junctions in the rat testis?
Lee NP, Mruk D, Lee WM, Cheng CY
Biology of reproduction 2003 Feb;68(2):489-508
Biology of reproduction 2003 Feb;68(2):489-508
Is the cadherin/catenin complex a functional unit of cell-cell actin-based adherens junctions in the rat testis?
Lee NP, Mruk D, Lee WM, Cheng CY
Biology of reproduction 2003 Feb;68(2):489-508
Biology of reproduction 2003 Feb;68(2):489-508
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
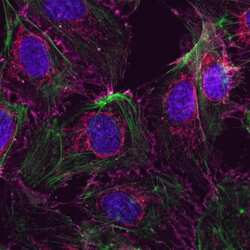
- Experimental details
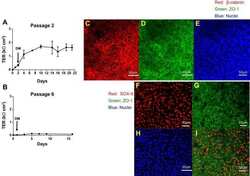
- Human Umbilical Vein Endothelial Cells (HUVEC; C0035C) were grown to confluency in Medium 200 (Product # M-200-500) plus Large Vessel Endothelial Supplement (LVES; Product # A1460801). Cells were fixed with 4% formaldehyde and permeablized with 0.25% Triton X-100 followed by staining with mouse anti-ATP synthase subunit IF-1 monoclonal antibody (Product # A-21355) and rabbit anti-ß-Catenin polyclonal antibody (Product # 71-2700). Primary antibodies were detected with Qdot® 605 Goat F (ab')2 anti-mouse IgG Conjugate (Red; Product # Q11002MP) and Qdot® 655 Goat F (ab')2 anti-rabbit IgG Conjugate (Magenta, Product # Q-11421MP), respectively. Cells were counterstained with NucBlue® Fixed Cell (Blue; Product # R37606) and ActinGreen 488 (Green, Product # R37110) ReadyProbes Reagents. Cover slips were air dried, mounted with Cytoseal 60, and imaged on a Zeiss LSM 710 confocal microscope.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
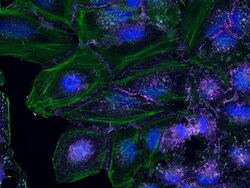
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
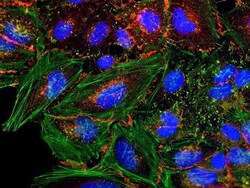
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
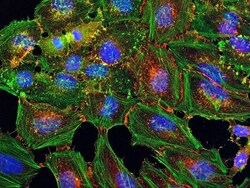
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
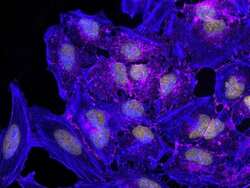
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
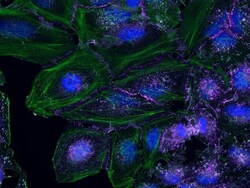
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
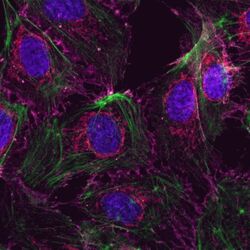
- Experimental details
- Human Umbilical Vein Endothelial Cells (HUVEC; C0035C) were grown to confluency in Medium 200 (Product # M-200-500) plus Large Vessel Endothelial Supplement (LVES; Product # A1460801). Cells were fixed with 4% formaldehyde and permeablized with 0.25% Triton X-100 followed by staining with mouse anti-ATP synthase subunit IF-1 monoclonal antibody (Product # A-21355) and rabbit anti-ß-Catenin polyclonal antibody (Product # 71-2700). Primary antibodies were detected with Qdot® 605 Goat F (ab')2 anti-mouse IgG Conjugate (Red; Product # Q11002MP) and Qdot® 655 Goat F (ab')2 anti-rabbit IgG Conjugate (Magenta, Product # Q-11421MP), respectively. Cells were counterstained with NucBlue® Fixed Cell (Blue; Product # R37606) and ActinGreen 488 (Green, Product # R37110) ReadyProbes Reagents. Cover slips were air dried, mounted with Cytoseal 60, and imaged on a Zeiss LSM 710 confocal microscope.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
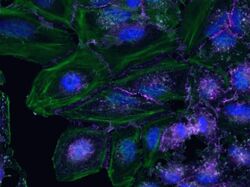
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin (Product # 71-2700) for 60 mins at RT followed by 3 wash steps in dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 (Product # Q11422MP) secondary antibody for 60 mins followed by 3 wash steps in dPBS. Cells were labeled with ReadyProbes® reagents NucBlue® Live (Product # R37605) and ActinRed 555 (Product # R37112) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X coverslip-corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
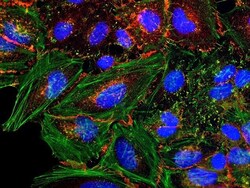
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
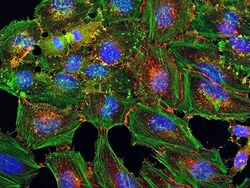
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
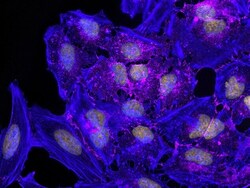
- Experimental details
- HeLa cells were plated on coverslips overnight. The next day the cells were fixed and permeabilized using the Image-iT® Fixation/Permeabilization Kit (Product # R37602) according to protocol. Cells were then incubated with 3 µg/mL anti-B-catenin antibody (Product # 71-2700) for 60 min at room temperature followed by three washes with dPBS. Cells were then incubated with a 1:1000 dilution of goat anti-rabbit Qdot® 655 secondary antibody (Product # Q-11421MP) for 60 min, followed by three washes with dPBS. Cells were labeled with NucBlue® Live cell stain (Product # R37605) and ActinGreen™ 488 ReadyProbes® reagent (Product # R37110) according to protocol. Coverslips were then mounted using ProLong® Gold Antifade Reagent (Product # P36930). Images were taken using the EVOS® FL Auto Imaging System and a 40X Coverslip corrected objective (Product # AMEP4699).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
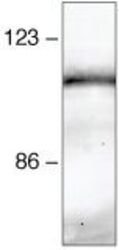
- Experimental details
- Immunoprecipitation and Western Blot Analysis: Immunoprecipitation followed by western blot analysis of HeLa cell lysates using rabbit anti-beta-catenin polyclonal antibody (Product # 71-2700).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
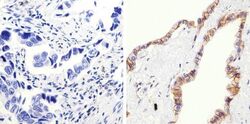
- Experimental details
- Immunohistochemistry analysis of Beta-Catenin showing staining in the membrane and cytoplasm of paraffin-embedded human lung adenocarcinoma (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Catenin Rabbit Polyclonal Antibody (Product # 71-2700) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
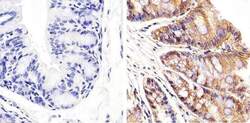
- Experimental details
- Immunohistochemistry analysis of Beta-Catenin showing membrane and weak cytoplasm staining of paraffin-embedded mouse colon tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Catenin Rabbit Polyclonal Antibody (Product # 71-2700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
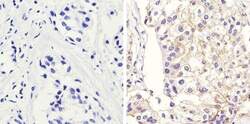
- Experimental details
- Immunohistochemistry analysis of Beta-Catenin showing staining in the membrane of paraffin-embedded human breast carcinoma (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Catenin Rabbit Polyclonal Antibody (Product # 71-2700) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation and Western Blot Analysis: Immunoprecipitation followed by western blot analysis of HeLa cell lysates using rabbit anti-beta-catenin polyclonal antibody (Product # 71-2700).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
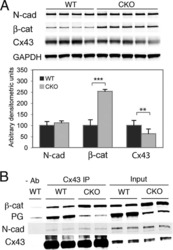
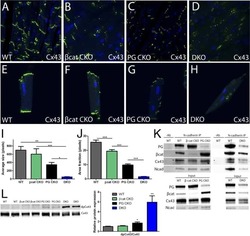
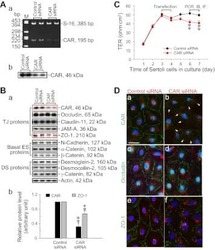
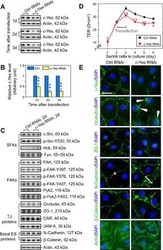
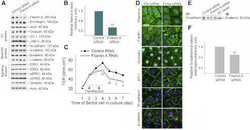
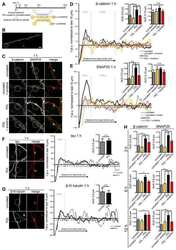
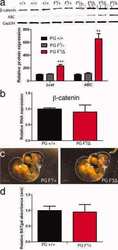
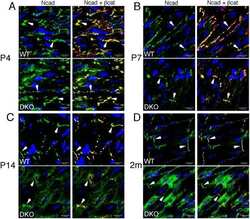
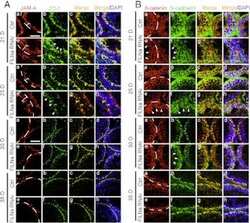
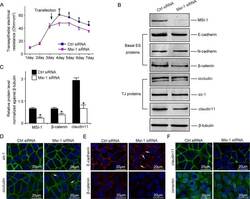
- FIGURE 2: Msi-1 knockdown by RNAi in Sertoli cells perturbs Sertoli cell-cell junctions via changes in BTB-associated proteins. (A) On day 3, Sertoli cells were transfected with Msi-1-specific siRNA duplexes and nontargeting control siRNA duplexes. The TER was detected until day 7, and each data point is the mean +- SD of n = 5 replicates from a representative experiment. *, p < 0.05. (B) Immunoblot showing the steady-state levels of Msi-1, basal ectoplasmic specialization (ES) proteins, and TJ proteins in lysates of Sertoli cells lysed 48 h after transfection; beta-tubulin served as a loading control. (C) Histogram summarizing selected immunoblotting results in B and normalized against beta-tubulin. Each bar represents the mean +- SD of n = 3 experiments. *, p < 0.05. (D-F) Changes in the localization of TJ proteins, basal ES proteins, and the framework protein vimentin at Sertoli cell-cell interfaces after Msi-1 knockdown were assessed 48 h after transfection. Sertoli cell nuclei were stained with DAPI. Note that considerably less beta-catenin and claudin-11 were detected, and the distributions of occludin and E-cadherin (white arrows) were changed at Sertoli cell-cell interfaces after Msi-1 knockdown. Scale bars: 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
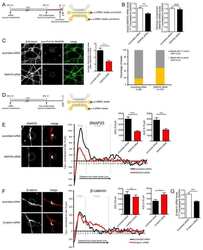
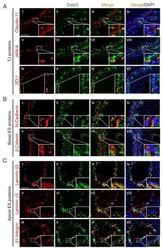
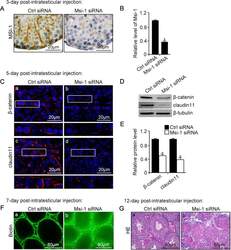
- FIGURE 3: An in vivo study assessing the role of Msi-1 in BTB function and spermatogenesis by RNAi intratesticular injection. Msi-1-targeting siRNA duplexes and nontargeting duplexes were administered to each testis in adult mice as a single treatment. The knockdown efficiency, BTB-associated protein expression, biotin tracer experiment, and hematoxylin-eosin (HE) staining at 3, 5, 9, and 12 d postintratesticular injection, respectively. (A) Considerably more intense Msi-1 staining was observed in the Sertoli cells from testes transfected with nontargeting control (a) versus testes transfected with Msi-1-specific siRNA duplexes (b). Scale bars: 20 mum. (B) Histogram summarizing immunoblotting results of Msi-1 and normalized against beta-tubulin. Each bar represents the mean +- SD of n = 3 experiments. *, p < 0.05. (C) Paraffin sections were used to study changes in the expression of beta-catenin (a and b) and claudin-11 (c and d) after Msi-1 knockdown in vivo. The boxed area was randomly selected. Scale bars: 20 mum. (D) The protein levels of beta-catenin and claudin-11 in nontargeting control and Msi-1 knockdown testes. beta-tubulin served as a loading control. (E) Histogram summarizing selected immunoblotting results in D and normalized against beta-tubulin. Each bar represents the mean +- SD of n = 3 experiments. *, p < 0.05. (F) We injected a biotin tracer into the testes of live anesthetized mice ( n = 3) and examined the subsequent changes in BTB integrity after siRNA d
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
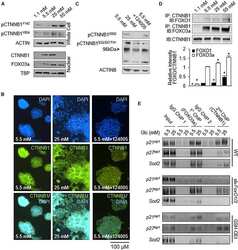
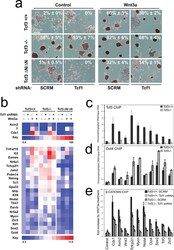
- Figure 6 Combined effect of Tcf1 and Tcf3-beta-catenin for the Wnt3a response of ES cells (a) Alkaline phosphatase staining of colonies from ES cell lines with different Tcf3 genotypes (indicated on left) and expressing an shRNA (indicated on bottom). Colonies were formed for four days in serum containing media without LIF, and with control or Wnt3a (indicated on top). Percentage of AP+ colonies (white number) represents mean +- standard deviation of biological duplicates. Wnt3a stimulated self-renewal at different levels in Tcf3+/+ , Tcf3-/- and Tcf3DeltaN/DeltaN ES cells, while knockdown of Tcf1 significantly reduced stimulations in all three ES cell lines. (b) Gene expression analysis in different Tcf3 ES cell lines +- Tcf1 shRNA and +- recombinant Wnt3a. The relative level of mRNA for each gene is compared to untreated Tcf3+/+ cells set at 1.0. Numerical values +/- standard deviations are shown in Supplementary Table S5 . (c-e) Chromatin immunoprecipitated with antibodies against Tcf3 (c), Oct4 (d), or beta-catenin (e) was measured by qPCR using primers ( Supplementary Table S5 ) specific for upstream regions of the ten genes labeled at the bottom of graph shown in (e). A representative graph from three biological replicates is shown for each experiment. (c,d) Assays were performed using chromatin from Tcf3+/+ (black) and Tcf3-/- (gray) ES cells. Values represent the mean percentage of DNA immunoprecipitated relative to input +- standard deviations of replicate measuremen
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
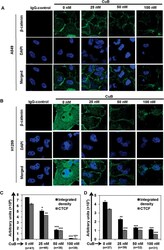
- Figure 3 CuB inhibits the expression and nuclear translocation of beta-catenin in NSCLC cells. A549 (Panel A ) and H1299 (Panel B ) cells were seeded on glass cover slips for 24 h and then treated with varying concentrations of CuB for 24 h. Endogenous cytoplasmic and nuclear beta-catenin (FITC-green) localization was visualized by immunofluorescence followed by confocal imaging. DAPI was used as nuclear stain (blue). IgG control was used as the negative control. Images are representative of three-independent experiments. The graphs (Panels C , D ) represent the raw integrated density as well as CTCF values in A549 and H1299 cells, respectively. The numbers in parenthesis are indicative of number of cells analyzed for each group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
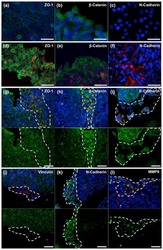
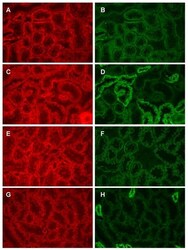
- Figure 7 beta-catenin is colocalized with both N-cadherin and E-cadherin. Samples were processed for the dual labeling of beta-catenin and either N- or E-cadherin as described in the methods and viewed using a 40x objective. Panels A and B show the dual labeling of beta-catenin (A) and N-cadherin (B) in a field from the outer cortex whereas panels C and D show the dual labeling of beta-catenin (C) and E-cadherin (D) in a similar field. Panels E-F show the same molecules in fields from the inner cortex. E and F: dual labeling of beta-catenin (E) and N-cadherin (F); G and H dual labeling of beta-catenin (G) and E-cadherin (H). Original magnification = 174x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
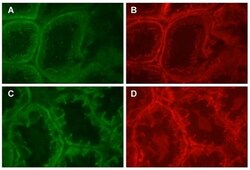
- Figure 8 Dual labeling of N-cadherin and beta-catenin in the proximal tubule. Panels A and B show that dual labeling of N-cadherin (A) and beta-catenin (B) in the S1/S2 segment of the proximal tubule, whereas panels C and D show the dual labeling of N-cadherin (C) and beta-catenin (D) in the S3 segment of the proximal tubule. Original magnification = 435x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
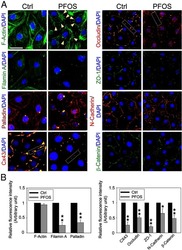
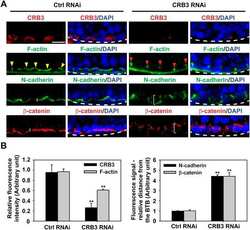
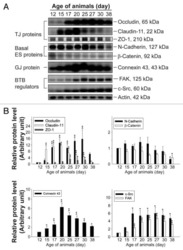
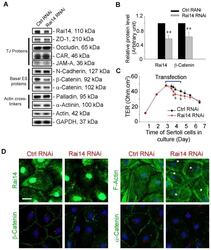
- Figure 5 Knockdown of Rai14 in the Sertoli cell epithelium with an established TJ-permeability barrier in vitro by RNAi disrupts actin filament organization and the TJ barrier. (A) Sertoli cells cultured alone on Matrigel-coated 12-well dishes for 2-day with an established TJ-permeability barrier were transfected with Rai14 siRNA duplexes (Rai14 RNAi) versus non-targeting control duplexes (Ctrl RNAi) at 100 nM using Ribojuice transfection medium for 24 hr, thereafter, cells were washed twice and cultured in F12/DMEM for 12 hr to allow recovery. Thereafter, cells were transfected again under the same conditions for another 24 hr. Thereafter, cells were rinsed with fresh F12/DMEM and cultured for an additional 12 hr before termination, and used to prepare lysates for immunoblotting using antibodies against several BTB-associated constituent or regulatory proteins. A knockdown of Rai14 by ~50% was noted in which the control was arbitrarily set at 1 against which statistical comparison was performed (B) without any apparent off-target effects (A). The findings shown herein are the results of 3 independent experiments excluding pilot experiments which were used to establish optimal experimental conditions, such as different concentrations of siRNA duplexes and Ribojuice. It was noted that we achieved only ~50-60% knockdown of Rai14 in several pilot experiments, unlike other target genes [ e.g ., Scribble, beta1-integrin, and P-glycoprotein] wherein we could silence the target gene
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
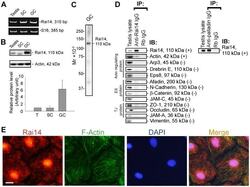
- Figure 1 Rai14 is an actin-binding protein in the rat testis. ( A ) A study by RT-PCR to confirm the expression of Rai14 in adult rat testis, Sertoli cells (SC, isolated from 20-day-old rat testes and cultured for 4-day), and germ cells (GC, isolated from adult rat testes and cultured for 16 hr). ( B ) Immunoblotting also confirmed the expression of Rai14 in the rat testis, Sertoli and germ cells, and the relative expression of Rai14 in SC vs. GC was shown in the histogram with n = 3 experiments in which the relative expression level of Rai14 in the testis was arbitrarily set at 1 so that the relative expression level between these samples can be compared. ( C ) The specificity of the anti-Rai14 antibody ( Table 1 ) was assessed by immunoblotting using lysates of GC (20 ug protein). ( D ) Using the specific anti-Rai14 antibody, Rai14 was shown to be an actin-binding protein by co-immunoprecipitation (Co-IP); however, Rai14 did not structurally interact with any of the BTB-associated proteins including several actin-binding and regulatory proteins ( e.g ., Arp3, drebrin E, Eps8) and vimentin (an intermediate filament-based constituent protein). However, Rai14 was found to structurally interact with an actin cross-linking protein palladin which is known to be involved in conferring actin filament bundles in other mammalian cells [48] . ( E ) Rai14 (red) was also shown to be an actin-binding protein by dual-labeled immunofluorescence analysis in which it co-localized with F-acti
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
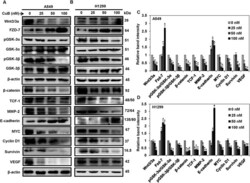
- Figure 4 CuB inhibits the Wnt/beta-catenin signaling in the NSCLC cells. A549 (Panel A ) and H1299 cells (Panel B ) were treated with 0-100 nM of CuB for 24 h and whole cell lysates were prepared. Western blot analysis was performed to analyze the expression of Wnt/beta-catenin pathway proteins. Blots are representative of three independent experiments. Graphs in (Panel C , D ) represent relative band intensities of proteins in A549 and H1299 cells normalized to beta-actin. The results were obtained from three independent experiments, mean +- SEM. *p < 0.05 versus Control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
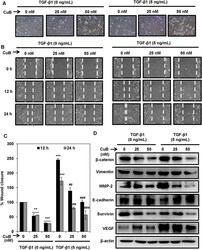
- Figure 5 CuB treatment abrogates TGbeta-1-induced EMT in A549 cells. ( A ) A549 cells were treated with TGF-beta1 (5 ng/mL) for 24 h. TGF-beta1 treated and untreated A549 cells were treated with 25 and 50 nM concentrations of CuB for 24 h. Morphological changes are shown in light microscopic images (100x magnification). ( B , C ) Wounds were created in the sub-confluent cultures of A549 cells and then cells were treated with TGF-beta1 (5 ng/mL) and 25 and 50 nM CuB. Photomicrographs were captured at 0, 12 and 24 h after treatment and wound area was measured. Data were expressed as percent wound closure in the treatment groups compared to control. The results were obtained from three independent experiments, mean +- SEM. **p < 0.01 and ***p < 0.001 compared to untreated control, ## p < 0.01 and ### p < 0.001 compared to respective TGF-beta1-treated control group. ( D ) A549 cells were treated as in (Panel A ), and whole cell lysates were prepared. Western blot analysis was performed to analyze the expression of EMT markers. Blots are representative of three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
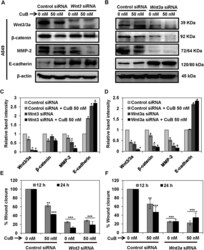
- Figure 6 Wnt/beta-catenin signaling is involved in the inhibition of metastatic progression of NSCLC A549 cells. A549 (Panels A - D ) were subjected to treatment with 40 nM of Wnt3 or Wnt3a siRNA, respectively. Control- and Wnt3/3a -siRNA transfected cells were treated with the indicated concentrations of CuB for 24 h. Cell lysates were prepared and protein expressions were analyzed by western blot. Blots are representative of three independent experiments. Graphs represent the relative band intensities normalized with beta-actin. A549 (Panels E , F ) cells transfected with Wnt3/3a -siRNA were treated with indicated concentrations of CuB for 24 h. Cellular migration was determined and plotted against percent wound closure in control group. Results were obtained from three independent experiments, mean +- SEM, Statistical significance, *p < 0.05, **p < 0.01 and ***p < 0.001 against control siRNA group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
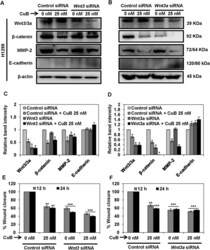
- Figure 7 Wnt/beta-catenin signaling is involved in the inhibition of metastatic progression of NSCLC H1299 cells. H1299 cells (Panels A - D ) were subjected to treatment with 40 nM of Wnt3 or Wnt3a siRNA, respectively. Control- and Wnt3/3a -siRNA transfected cells were treated with the indicated concentrations of CuB for 24 h. Cell lysates were prepared and protein expressions were analyzed by western blot. Blots are representative of three independent experiments. Graphs represent the relative band intensities normalized with beta-actin. H1299 (Panels E , F ) cells transfected with Wnt3/3a -siRNA were treated with indicated concentrations of CuB for 24 h. Cellular migration was determined and plotted against percent wound closure in control group. Results were obtained from three independent experiments, mean +- SEM, Statistical significance, *p < 0.05, **p < 0.01 and ***p < 0.001 against control siRNA group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
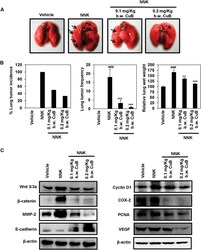
- Experimental details
- Figure 8 CuB inhibits NNK-induced lung tumorigenesis and Wnt/beta-catenin/MMP-2 signaling. ( A ) Representative images of vehicle-treated, NNK-induced CuB-untreated as well as 0.1 mg/Kg and 0.2 mg/Kg b.w. CuB-treated mice lungs. ( B ) Effects of CuB on lung tumor incidence, lung tumor frequency and relative lung wet weights in NNK-treated mice. mean +- SEM. ### p < 0.001 compared to vehicle-treated lungs, ** p < 0.01 and *** p < 0.001 compared to NNK-induced lungs. ( C ) Tissue lysates from each animal group were used to analyze the expression of different Wnt/beta-catenin signaling markers. beta-actin was used as equal loading control. Blots are representative of three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
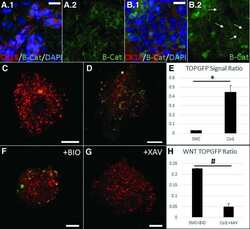
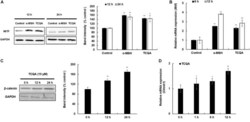
- FIGURE 4 3,4,5-Tri- O -caffeoylquinic acid (TCQA) stimulated beta-catenin and microphthalmia-associated transcription factor (MITF) expression in human epidermal melanocytes (HEMs). (A) Protein expression of MITF and beta-catenin after 0, 4, 8, and 12 h of treatment with 10 muM of TCQA. (B) Gene expression of MITF after 0, 1, 6, and 12 h of treatment with 10 muM of TCQA. (C) Immunocytochemistry of beta-catenin expression after 0, 4, 8, and 12 h of treatment with 10 muM of TCQA. Scale bar = 25 mum; magnification at 20x. (D) Gene expression of CTNNB1 after 0, 1, 6, and 12 h of treatment with 10 muM of TCQA. (E) Gene expression CTNNB1 after treatment with 10 muM of XAV939 and co-treatment of 10 muM of XAV939 and 10 muM of TCQA for 24 h. (F) MITF gene expression after treatment with 10 muM of XAV939 and co-treatment of 10 muM of XAV939 and 10 muM of TCQA for 24 h. (G) Melanin assay after 48 h of treatment with 10 muM of XAV939 and co-treatment of 10 muM of XAV939 and 10 muM of TCQA for 48 h. The band intensities were done by comparing GAPDH using LI-COR system; and the mRNA level was quantified using TaqMan real-time PCR. Results represent the mean +- SD of three independent experiments. *Statistically significant ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- FIGURE 5 3,4,5-Tri- O -caffeoylquinic acid (TCQA) enhanced microphthalmia-associated transcription factor (MITF) and beta-catenin expression in B16F10 murine melanoma cells. (A) Protein expression of MITF after 12 and 24 h of treatment with 0 and 25 muM of TCQA and 200 nM of alpha-MSH. GAPDH 24 h from Figure 3 band is reused in this figure. (B) Gene expression of MITF after 6 and 12 h of treatment with 0 and 25 muM of TCQA and 200 nM of alpha-MSH. (C) Protein expression of beta-catenin expression after 0, 12, and 24 h of treatment with 25 muM of TCQA. (D) Gene expression of Ctnnb1 after 0, 12, and 24 h of treatment with 25 muM of TCQA. The band intensities were done by comparing GAPDH using LI-COR system; and the mRNA level was quantified using TaqMan real-time PCR. Results represent the mean +- SD of three independent experiments. *Statistically significant ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
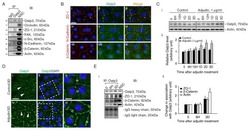
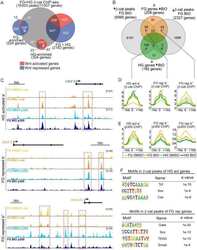
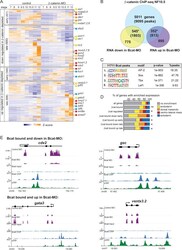
- Figure 3. The Bcat-regulated genomic program. ( A ) Time course heatmap of Bcat-regulated transcripts. Differential expressed genes from a pairwise comparison of control-MO and Bcat-MO embryos ( > 2 FC, FDR < 5%) at each stage identified 2568 Bcat-regulated transcripts; 1321 downregulated and 1247 upregulated in Bcat-MO embryos. Key genes are color coded based on regional expression from ( D ). ( B ) Venn diagram intersecting Bcat-regulated genes with Bcat-bound genes (and associated peaks) from published X. tropicalis gastrula Bcat ChIP-seq data () identified 898 putative direct targets associated with 2616 peaks. Statistically significant intersections based on hypergeometric tests* p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4. Sox17 and Bcat co-occupy enhancers to regulate the endoderm GRN. ( A ) Eighty-four genes (191 Peaks) are cobound and coregulated by Sox17 and Bcat. Venn diagram show that 415 genes are regulated by both Sox17 and Bcat (left), whereas 2411 genes (3956 peaks) are bound by both Sox17 and Bcat. Gene intersections are significant based on hypergeometric tests, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
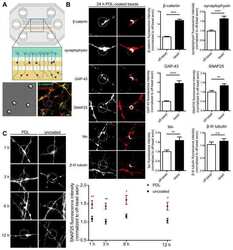
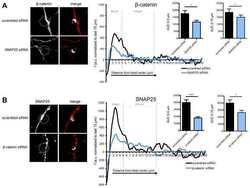
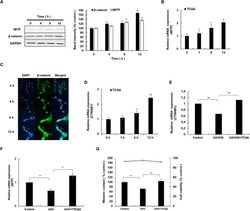
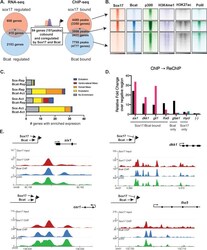
- Figure 1 Ctdnep1 and Eps8L2 are required for nuclear positioning and cell migration in fibroblasts (A) Representative images of wound-edge 3T3 fibroblasts with or without LPA stimulation in control, Ctdnep1, and Eps8L2 siRNAs stained for beta-catenin (green, cell contacts), pericentrin (red, centrosome), and DAPI (blue, nucleus). The dashed white lines mark the wound edge. (B) Average positions of the nucleus (blue) and centrosome (red) relative to the cell centroid in cells treated with control, Nesprin2G, Ctdnep1, and Eps8L2 siRNAs. (C) Percentage of oriented cells in the conditions analyzed in (B). (D) Average positions of the nucleus (blue) and centrosome (red) in cells treated with Ctdnep1 or Eps8L2 siRNAs and microinjected with KDEL-GFP, Ctdnep1-GFP, Ctdnep1_D67E-GFP (no phosphatase activity), GFP, myc-Eps8L2, or myc-Eps8L2_529-715. (E and F) Average wound closure velocity during wound closure migration (E) and instantaneous cell velocity in random migration (F) in cells treated with control, Nesprin2G, Ctdnep1, and Eps8L2 siRNAs. Data are represented as mean +- SEM. The n value means number of analyzed cells (B, D, and F) or number of experiments (C and E) with >50 cells analyzed per experiment. Statistics was performed by unpaired t test: * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001. Scale bars 10mum. See also Figure S1 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
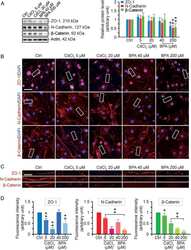
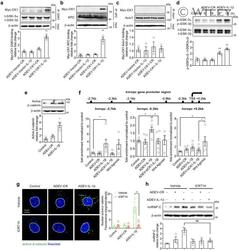
- 6 FIGURE CK1 carried in astrocyte derived EV-IL1beta directly binds to GSK3alpha/beta and APC in target neurons. Neurons were treated with astrocyte derived EV-Vector-CR (ADEV-Vector-CR), astrocyte derived EV-Vector-IL1beta (ADEV-Vector-IL1beta), astrocyte derived EV-CK1-CR (ADEV-CK1-CR), and astrocyte derived EV-CK1-IL1beta (ADEV-CK1-IL1beta) for 18 h. (a) GSK3-Myc-CK1, (b) Axin1-Myc-CK1 and (c) APC-Myc-CK1 were immunoprecipitated and the immunoblot probed for Myc tagged CK1. Myc-CK1 was quantified by band density. (d) Neurons were treated with astrocyte derived EV-CR (ADEV-CR) or astrocyte derived EV-IL1beta (ADEV-IL1beta) for 6 and 18 h, Total GSK-3alpha,beta and -GSK3alpha,beta were detected by Western blot and quantified by densitometry. (e) Neurons were treated with astrocyte derived EV-CR or astrocyte derived EV-IL-1beta for 18 h and active beta-catenin expression was measured by Western blot and quantified by densitometry. (f) Neurons were treated with astrocyte derived EV-CR (ADEV-CR), astrocyte derived EV-IL1beta (ADEV-IL-1beta), astrocyte derived EV-siCK1-IL1beta (ADEV-siCK1-IL-1beta), and Wnt agonist (20 muM) for 18 h. The binding of beta-catenin to Hnrnpc promotor sites at -2.7, -0.3 and +0.2 kb were measured by chromatin immunoprecipitation ChIP-qPCR analysis. Data are mean +- SEM of n = 3-8 independent experiments per condition. * P < 0.05, ** P < 0.01, one-way ANOVA with Tukey post hoc comparisons. (g) Neurons were treated with astrocyte derived EV-CR, astrocy
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Qualitative and quantitative analysis of immunohistochemical staining for N-cadherin (A-C, G), beta-catenin (C-D, H) and c-Src (E-F, I). Representative micrographs of control (A, C, E) and flutamide-treated rats (B, D, F) and a histogram of N-cadherin (G), beta-catenin (H) and c-Src (I) staining intensity expressed as relative optical density ( ROD ) of diaminobenzidine brown reaction products. Immunohistochemical localization of N-cadherin (A-B). Typical distribution of N-cadherin is visible in the basal compartment of the seminiferous epithelium at the blood-testis barrier ( BTB ) (arrowheads, see also the higher magnification) and at the Sertoli cell-elongated spermatid interface (arrows) in testes of control rats (A). In tubules of flutamide-treated rats, increased staining intensity at the BTB (arrowheads, see also the higher magnification) is visible. Note a diffusion of N-cadherin from the BTB site (red bracket) (B). Immunohistochemical localization of beta-catenin (C-D). Typical localization of beta-catenin is visible at the BTB site (arrowheads, see also the higher magnification) and at the Sertoli cells-elongated spermatids interface (arrows) in testes of control rats (C). In tubules of flutamide-treated rats a diffusion of beta-catenin from the BTB site is visible (red bracket) (D). Immunohistochemical localization of c-Src (E-F). Typical localization of c-Src is visible in the basal compartment of seminiferous epithelium at the BTB (arrowheads, see also the higher
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
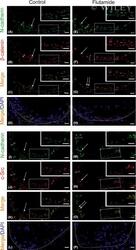
- Experimental details
- Immunofluorescent localization of N-cadherin and beta-catenin (A-H), and N-cadherin and c-Src (I-P) in testes of control (A-D and I-L) and flutamide-exposed (E-H and M-P) rats. Corresponding images were merged to show areas of colocalization of N-cadherin and beta-catenin (C, G), and N-cadherin and c-Src (K, O). Scale bars represent 15 mum. Rectangles indicate the location of the higher magnification view. In tubules of control rats, typical localizations of N-cadherin (arrows in A and I), beta-catenin (arrow in B) and c-Src (arrow in J) are visible at the BTB . Note also the colocalization of N-cadherin with beta-catenin (arrow in C) and N-cadherin with c-Src (arrow in K) at the BTB . In tubules of flutamide-treated rats, increased N-cadherin signal is observed (arrows in E and M) as well as diffusion of N-cadherin and beta-catenin signals from the BTB site (arrows and brackets in E, M, and F, respectively). Note altered distribution of c-Src (arrow in N). The colocalization of N-cadherin with beta-catenin and N-cadherin with c-Src is absent, see green and red staining (double arrows in G and O, respectively). Cell nuclei were stained with DAPI (blue; D, H, L and P). Dotted-line circles (D, H, L and P) outline the periphery of seminiferous tubules.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
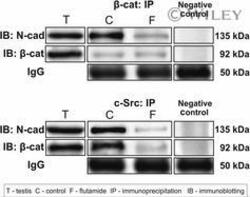
- Experimental details
- Representative immunoprecipitation results of adherens junction proteins with c-Src in testes of control and flutamide-treated rats. Lysates of testes from control and flutamide-treated rats were used for coimmunoprecipitation with antibodies against beta-catenin and c-Src. Immunocomplexes were subjected to immunoblotting and stained sequentially with anti-N-cadherin or anti-beta-catenin antibodies. Lysates without immunoprecipitation of normal rat testes (T) were used as positive controls, illustrating antibody specificity. A negative control was also included in which the precipitating antibody was substituted by IgG from the host species of the primary antibody. Experiments were repeated in triplicate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
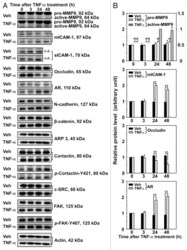
- Experimental details
- Figure 3. Effects of TNFalpha on the steady-state levels of integral membrane, scaffolding and regulatory proteins in Sertoli cells in vitro. Sertoli cells were cultured at high density on Matrigel(tm)-coated 12-well dishes as described in Materials and Methods. On day 4 (designated as 0 h in this figure), TNFalpha (25 ng/ml) was added into Sertoli cell cultures, and cells were terminated at specific time points thereafter for lysate preparation. TNFalpha was dissolved in 10 mM NaH 2 PO 4 pH 7.4 at 22degC containing 0.15 M NaCl and 0.1% BSA (wt/vol). The control consisted of culturing Sertoli cells in media containing an equivalent amount of BSA. ( A ) Immunoblots of selected proteins involved in the regulation and in the maintenance of Sertoli cell barrier function. Actin served as an indicator of equal protein loading. ( B ) Histograms summarizing results shown in ( A ) from at least three independent experiments. Histograms are not shown for proteins whose levels did not change significantly. Each data point was normalized against its corresponding actin data point and then against the protein level at 0 h, which was arbitrarily set as 1. Each bar represents the mean +- SD of n = 3-4 experiments. **p < 0.01 (ANOVA followed by Dunnett's post-hoc test). n.d., not determined.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
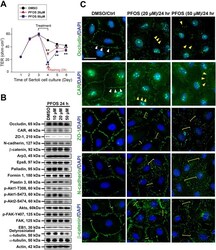
- Experimental details
- Figure 1 PFOS perturbs Sertoli cell BTB function through changes in the expression and/or localization of proteins at the immunological barrier. Sertoli cells cultured on Matrigel-coated bicameral units, dishes or coverslips were treated on day 3 with 10, 20 or 50 muM PFOS for 24 hr. Thereafter, cells were washed twice with PBS to remove residual PFOS and terminated for immunoblotting (IB) or IF on day 4. ( A ) Typical results of a TER experiment that monitored the effects of PFOS, at 20 uM and 50 uM vs . vehicle control (DMSO) on the Sertoli cell TJ-permeability barrier. Each data point is a mean +- SD of triplicate units from an experiment, which was repeated 3 times and yielded similar results. ** P < 0.01 by Student's t -test compared between PFOS-treated and control cells. ( B ) Immunoblot analysis to assess the effects of PFOS on the expression of TJ proteins: occludin, CAR and ZO-1; basal ES proteins: N-cadherin and beta-catenin; actin regulatory proteins: Arp3, Eps8, palladin, formin 1 and plastin 3; signaling molecules: activated Akts: p-Akt1-T308, p-Akt1-S473, pAkt2-S474 vs . Akts (including Akt1, 2 and 3), and activated FAK: p-FAK-Tyr407 vs . FAK; MT regulatory proteins: EB1 and detyrosinated alpha-tubulin. Actin and alpha-tubulin served as protein loading controls. PFOS was found to down-regulate p-Akt1-T308, p-Akt1-S473, p-Akt2-S474 and p-FAK-Tyr407. Composite data of immunoblottings are shown in Figure S1A ; and uncropped blots are shown in Figure S4 . ( C ) Imm
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
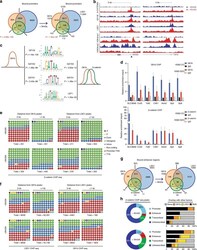
- Experimental details
- Fig. 8 GFi1b, beta-catenin and LSD1 co-occupy sites at enhancer regions and gene promoters. a Venn diagram showing GFI1B, beta-catenin and LSD1 ChIP-seq promoter binding prior to and after Wnt3A treatment of K562 cells. P indicates statistical significance of overlap between GFI1B and beta-catenin calculated using Fisher's exact test. b Example of target genes Axin2 and Yaf2 bound by GFI1B, beta-catenin and LSD1 and H3K4me1 and H3K9me2 enrichment before and after Wnt3A treatment. Arrowhead indicates the presence of Gfi1b and TCF binding motifs in the Axin 2 promoter. c Motif enrichment analysis under GFI1B peaks alone (top) and Gfi1b/beta-catenin overlapped peaks (bottom). d GFI1B (top) and beta-catenin (bottom) ChIP-PCR analysis in K562 stable clones expressing GFI1B (S23) or scramble (C5) shRNA (error bars show s.d, n = 3 technical replicates). e Distribution of beta-catenin peaks based on their distance to GFI1B peaks (left panel) and LSD1 peaks (right panel). f Distribution of LSD1 peaks based on their distance to GFI1B peaks (left panel) and reversely (right panel). g Venn diagram showing GFI1B, beta-catenin and LSD1 binding at enhancer regions prior to and after Wnt3A treatment of K562 cells. h Distribution of beta-catenin peaks (left) and their overlaps with GFI1B and/or LSD1 peaks based on the genomic feature of each bound region
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
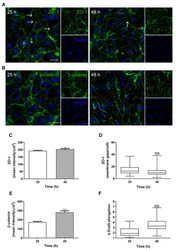
- Experimental details
- Figure 1 Shear stress (SS) promotes brain microvascular endothelial properties. Confluent monolayers of the brain endothelioma cell line b.End5 were exposed to physiological laminar non-pulsatile SS for 25 and 48 h. SS effects were evaluated by immunofluorescence analysis of the tight and adherens junction proteins ( A ) zonula occludens (ZO)-1 and ( B ) beta-catenin, respectively, which showed a delocalisation of the proteins towards the cell membrane and a decrease in membrane gaps (white arrows). Hoechst 33342 was used as counterstaining for nuclei (blue). Scale bar: 20 um. Semi-quantitative analysis of ZO-1 expression showed ( C ) a slight increase in staining intensity, from 25 to 48 h, and a ( D ) marked decrease in cell membrane gaps, while beta-catenin presented ( E ) a notorious increase in fluorescence intensity, and ( F ) a clear cellular elongation. Data are given as means +- SEM ( n = 3, 10 fields/condition). A Student's t -test for mean intensity and a Mann-Whitney test for membrane gaps and cell elongation were used to evaluate the significant differences, where * p < 0.05 and *** p < 0.001 or $$$ p < 0.001 denote differences between the indicated timepoints.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
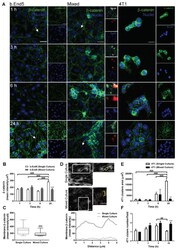
- Experimental details
- Figure 2 Adherens junctions are compromised during interaction between breast cancer cells and brain microvascular endothelial cells. Confluent monolayers of the brain endothelioma cell line b.End5 under physiological laminar non-pulsatile shear stress were exposed to 4T1 breast cancer cells (previously labelled with CellTracker(tm) DMTPX Red Dye) for 1, 3, 6 and 24 h and the expression of the adherens junction protein, beta-catenin, in single and mixed cultures was evaluated by immunofluorescence analysis. ( A ) Analysis of the expression of beta-catenin (green) revealed that the protein is present in both cell types, with a different cellular distribution in mixed cultures as compared with single ones, and further revealed a loss of elongation in b.End5 cells exposed to 4T1 cells (white arrows). Hoechst 33342 was used as counterstaining for nuclei (blue). Scale bar: 20 um. Semi-quantitative analysis revealed ( B ) a decrease in endothelial beta-catenin intensity in mixed culture at 24 h, ( C ) particularly notorious at the cell membrane, which was validated by the ( D ) plot profile analysis of the membrane region pointed out with the yellow brackets. Scale bar: 20 um. The characterisation of 4T1 cells was performed by the quantification of the ( E ) area of tumoural clusters, which increased over time in single cultures, and ( F ) number of clusters, which decreased at 24 h in single culture. Data are given as means +- SEM ( n = 3, 10 fields/condition). A one-way ANOVA was
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 LG3/4/5 protects Sertoli cells from cadmium-induced TJ-barrier disruption. a Preparation of cDNA constructs based on the 80 kDa tail of the laminin-alpha2 chain. The top panel is a schematic drawing that illustrates the functional domains of laminin-alpha2 chain. From the N-terminus, the short arm of laminin-alpha2 is comprised of three globular domains: laminin N-terminal domain (LN), laminin 4a domain (L4a) and laminin 4b domain (L4b). The long arm of laminin-alpha2 is comprised of laminin coiled coil (LCC) domain and five C-terminal laminin-type globular (LG) domains of LG1, 2, 3, 4, and 5. Proteolytic cleavage is close to the N-terminus of LG3 to generate the LG3/4/5 (i.e., the 80 kDa fragment). Six different cDNA constructs, namely LG3 (underlined in blue), LG4 (underlined in red), LG5 (underlined in green), LG3/4 (boxed in pink), LG4/5 (boxed in purple), and LG3/4/5 (boxed in black), were obtained by PCR using Sertoli cell cDNAs as the template and the corresponding specific primer pairs (Table 1 ), with the exception of the LG3/4/5 clone which was obtained by first denaturing LG3/4 and LG4/5, so that these two clones were annealed and then served as the template for PCR by using primer pairs (1) and (6). These constructs were cloned into the mammalian expression vector pCI-neo (Promega). Amino acid residues in orange represent sequences between the different LG domains and the italicized orange residues were not cloned into any of the cDNA constructs, such as LG
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
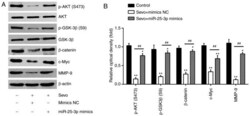
- Experimental details
- Figure 5 Sevo blocks the PTEN/Akt/GSK-3beta/beta-catenin signaling pathway by modulating the expression of miR-25-3p in HCC cells. HCCLM3 and Huh7 cells were transfected with miR-25-3p mimics or miR-25-3p inhibitor for 24 h followed by Sevo treatment for 6 h. (A) Protein levels of p-Akt, Akt, p-GSK-3beta, GSK-3beta, beta-catenin, c-Myc and MMP9 were measured using western blotting. beta-actin served as the loading control. (B) The bands were semi quantitatively analyzed using ImageJ and normalized to beta-actin density. Data are presented as the mean +- standard deviation. (n=3) of three representative experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Occludin, ZO-1, Cx43, N- and E-cadherin, and beta-catenin mRNA (A to E) and protein (A' to E') expression in yellow semen syndrome (YSS) and white normal semen (WNS) testes, epididymides, and ductuli deferentes. Representative quantitative real-time PCR analysis of occludin (A), ZO-1 (B), Cx43 (C), N- and E-cadherin (D), and beta-catenin (E) mRNA expression in YSS and WNS testes, epididymides, and ductuli deferentes. As an internal control, beta-actin mRNA level was measured. Relative quantification (RQ) is expressed as mean +- standard deviation (SD). Data were obtained from 3 separate experiments. Asterisks indicate statistically significant differences (* P < 0.05, ** P < 0.01, *** P < 0.001; Mann-Whitney U-test). YSS turkeys (n = 6) and WNS turkeys (n = 6). Representative western blots and relative expression of occludin (A'), ZO-1 (B'), Cx43 (C'), N- and E-cadherin (D'), and beta-catenin (E') proteins in YSS and WNS testes, epididymides, and ductuli deferentes. Densitometric analysis of protein content was normalized against the corresponding beta-actin. Protein levels within the WNS tissues were arbitrary set as 1. Data obtained from 3 separate analyses are expressed as mean +- SD. Asterisks indicate statistically significant differences (* P < 0.05, ** P < 0.01, *** P < 0.001; Mann-Whitney U-test). N = 5 each for YSS and WNS tissues.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
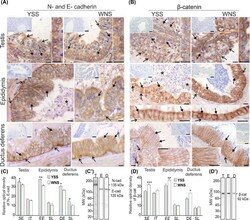
- Experimental details
- Figure 3 Qualitative (A and B) and quantitative (C and D) analysis of immunohistochemical staining for N- and E-cadherin (A) and beta-catenin (B). Representative micrographs of yellow semen syndrome (YSS) and white normal semen (WNS) testes, epididymides, and ductuli deferentes. Counterstaining was performed with Mayer's haematoxylin. Scale bars are 10 mum. Frames indicate the location of the higher magnification view. (A and B) Similar staining pattern for N- and E-cadherin (A) and beta-catenin (B) were observed in the basal compartment at the blood-testis barrier (BTB) (arrows) and in the adluminal compartment of seminiferous epithelium (open arrows) as well as in the lateral plasma membranes of adjacent epididymal principal cells (arrows) and between principal and basal cells (open arrows), and in the lateral plasma membranes of adjacent ductal columnar cells (arrows) and between columnar and basal cells (open arrows) of YSS and WNS epididymides and ductuli deferentes. Note increased N- and E-cadherin and reduced beta-catenin staining intensity at the BTB of YSS testis (see higher magnification views in A and B) and of YSS epididymis and ductus deferens sections (see higher magnification views in B). Note weaker staining for beta-catenin between epithelial principal cells (arrows) and principal and basal cells (open arrows) along the entire YSS epididymis length (see higher magnification inserts in B). Note also sloughed germ cells admixed with mature spermatozoa in YSS ep
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
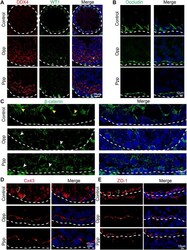
- Experimental details
- FIGURE 6 Opp and Ppp promote BTB recovery after busulfan treatment in Sertoli cells. (A) Immunofluorescence staining of control, Opp-, and Ppp-treated testes with anti-WT1 antibody (Sertoli cell, green), anti-DDX4 antibody (germ cell, red) and DAPI (blue). White dashed line depicts the basement membrane. (B) Immunofluorescence staining of control, Opp-, and Ppp-treated testes with anti-occludin antibody (BTB, green) and DAPI (blue). Yellow scale markers signify diffusively-distributed occludin signals. White scale markers depict tightly-distributed occludin signals around Sertoli and germ cells. (C) Immunofluorescence staining of control, Opp-, and Ppp-treated testes with anti-beta-catenin antibody (BTB, green) and DAPI (blue). Yellow arrow heads signify diffusively-distributed beta-catenin signals; white arrow heads signify tightly-distributed beta-catenin signals around Sertoli and germ cells. (D) Immunofluorescence staining of control, Opp-, and Ppp-treated testes with anti-Cx43 antibody (BTB, green) and DAPI (blue). Yellow scale markers depict diffusively distributed Cx43 signals; white scale markers depict tightly-distributed Cx43 signals around Sertoli and germ cells. (E) Immunofluorescence staining of control, Opp-, and Ppp-treated testes with anti-ZO-1 antibody (BTB, red) and DAPI (blue). Yellow arrow heads label diffusively-distributed ZO-1 signals. White arrow heads label tightly-distributed ZO-1 signals.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
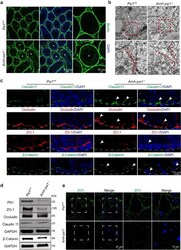
- Experimental details
- The integrity of BTB is disrupted in Amh-pa1 -/- mice. a The integrity of the BTB was observed to be destroyed in 8-week-old Amh-pa1 -/- testes. Immunofluorescence using FITC-labeled streptavidin (green) was conducted on seminiferous tubules of Pa1 F/F (upper) and Amh-pa1 -/- testes (lower) which had been injected with biotin beneath the albuginea. Nuclei were stained with DAPI (blue). Dashed boxes showed localization of the enlarged images. Asterisks indicated biotin penetrated seminiferous tubules. b Transmission electron microscope images of adult Pa1 F/F and Amh-pa1 -/- mice testes showing the cleft junctions between Sertoli cells at the basal compartment of seminiferous tubules. Red dashed boxes showed localization of the enlarged images shown in the bottom row. Red dashed lines in the enlarged images indicate the cell-cell junctions. c BTB components are shown to be disorganized in 8-week-old Amh-pa1 -/- mice testis. Immunofluorescence analysis using anti-Claudin11, anti-Occludin, anti-ZO-1, and anti-beta-catenin was conducted in the testis of Pa1 F/F mice (left) and Amh-pa1 -/- mice (right). Nuclei were stained with DAPI (blue). Arrowheads indicate the aberrant patches or diminished signals of the BTB components. Dashed curve indicated basal membrane of seminiferous tubules. d Immunoblot analysis of BTB components, including ZO-1, Occludin, Claudin11, and beta-Catenin, in the testes of adult wild-type mice and Amh-pa1 -/- mice. e Cell junction cannot be formed between
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
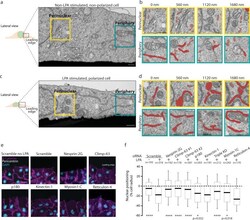
- Experimental details
- Nuclear positioning is associated with ER morphology remodeling and requires Climp-63. a FIB-SEM representative image of non-LPA stimulated, non-polarized wound-edge U2OS cell. One stack of 998 SEM images from one leading edge cell. b Representative images of perinuclear (yellow insets) and peripheral (green insets) regions. The ER is highlighted in red, light red represents tubules (1 um). Measurements represent the depth of the section. c as in a , but an LPA stimulated and polarized cell. One stack of 1798 SEM images from one leading edge cell. d as in b , but an LPA stimulated and polarized cell. e Representative widefield epifluorescence images of wound-edge NIH3T3 fibroblasts. Cells were immunostained for cell-cell contacts (purple; beta-catenin), centrosome (white; pericentrin) and DNA (DAPI, blue). f Nuclear positions relative to the cell centroid of cells represented in e . Significance (Two-tailed unpaired t-test) was calculated between experimental condition and the scramble siRNA with LPA. **** p < 0.0001 (Scramble no LPA, Nesprin-2G, Climp-63 #1, Climp-63 #2, Triple KD); * p < 0.05. Box shows first quartile, median and third quartile, and whiskers show 10-90% percentile. Experiments were repeated >=3, except ( a - d ). Scale bars: a , b , c , d 1 um; e 40 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
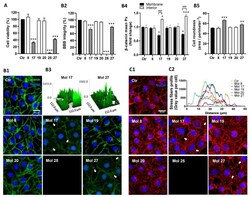
- Experimental details
- Figure 9 HBMEC treatment with selected molecules proved lack of cytotoxicity for 3 candidates. HBMEC were exposed to each of the selected molecules (8, 17, 19, 20, 25, and 27), or no addition (control; Ctr), at a concentration of 100 uM, for 24 h, for viability assays ( A ), or at U87MG EC 50 , during 9 h, for BBB integrity ( B ) or cytoskeleton ( C ) analyses. ( A ) Cell viability was assessed by MTT assay and the values are presented as a percentage relative to control. Molecules with significant cytotoxic effects are presented as pattern-filled bars, and innocuous molecules are shown as white bars. ( B ) BBB integrity was assessed by immunostaining of beta-catenin, with nuclear counterstaining by Hoechst 33342. ( B1 ) Arrows point to gaps in the HBMEC monolayer caused by incubation with toxic molecules. ( B2 ) Semi-quantitative analysis of endothelial gaps, where values are presented as percentage of control. ( B3 ) 3D representative plots of beta-catenin expression obtained by counting the number of image pixels per area in disrupted monolayers. ( B4 ) Semi-quantitative analysis of beta-catenin internalization by fold-change of mean image pixels (Px) in the membrane and interior of treated cells vs. control. ( B5 ) Morphological analysis of HBMEC roundness by cells delineation. ( C ) Cytoskeleton analysis was performed based on F-actin labeling. ( C1 ) Arrows highlight the presence of stress fibers. ( C2 ) Cytoskeleton stress fibers profile per cell representation by semi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
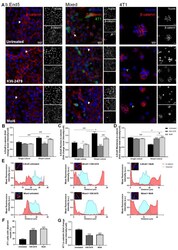
- Experimental details
- Figure 3 KW-2478 and Mol6 enhance barrier properties through increased membrane beta-catenin expression in b.End5 cells exposed to 4T1 cells. Confluent monolayers of b.End5 cells under physiological laminar non-pulsatile shear stress were exposed to 4T1 cells (previously labelled with CellTracker(tm) CMFDA Green Dye,) and treated with KW-2478 or Mol6 (1 uM), or DMEM (untreated), for 24 h and the expression of the AJs protein beta-catenin (red), in single and mixed cultures, was evaluated by immunofluorescence analysis. Hoechst 33342 was used as counterstaining for nuclei (blue). Scale bar: 20 um. ( A ) Treatment with KW-2478 and Mol6 induced an increase in beta-catenin expression in endothelial cells exposed to 4T1 cells, particularly at cell membrane (white arrows), and an increase in 4T1 cells with aberrant nuclei (white arrow heads). Semi-quantitative analysis of endothelial beta-catenin ( B ) mean intensity per cell, ( C ) nuclear intensity, and ( D ) membrane intensity confirmed the observations. ( E )The localisation of beta-catenin was studied through plot profiles of pixel intensity throughout an endothelial cell (edge to edge). Semi-quantitative analysis of tumour cell cultures confirmed ( F ) an increase in the number of aberrant nuclei upon treatment with each drug, as well as ( G ) a decrease in beta-catenin mean intensity per cell. Data are represented as mean +- SEM of three independent experiments, where 3 cells/field, 5 fields/condition were evaluated. Statist
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
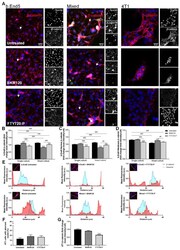
- Experimental details
- Figure 4 BKM120 induces endothelial barrier properties through the increase in membrane beta-catenin expression. Confluent monolayers of b.End5 cells under physiological laminar non-pulsatile shear stress were exposed to 4T1 cells (previously labelled with CellTracker(tm) CMFDA Green Dye) and treated with BKM120 or FTY720-P (0.1 uM), or DMEM (untreated), for 24 h and the expression of the AJs protein, beta-catenin (red), in single and mixed cultures was evaluated by immunofluorescence analysis. Hoechst 33342 was used as counterstaining for nuclei (blue). Scale bar: 20 um. ( A ) Treatment with BKM120 or FTY720-P induced an increase in beta-catenin expression in endothelial cells exposed to 4T1 cells, particularly at cell membrane (white arrows). Semi-quantitative analysis of endothelial beta-catenin ( B ) mean intensity per cell, ( C ) nuclear intensity, and especially ( D ) membrane intensity validated these observations. ( E ) The localisation of beta-catenin was studied through plot profiles of pixel intensity throughout an endothelial cell (edge to edge). Semi-quantitative analysis of tumour cell cultures revealed ( F ) no aberrant nuclei upon treatment, but ( G ) a decrease in beta-catenin mean intensity per cell after FTY720-P treatment. Data are represented as mean +- SEM of three independent experiments, where 3 cells/field, 10 fields/condition were evaluated. Statistical significances are denoted as * p < 0.05 and *** p < 0.001 vs. untreated conditions within the same
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
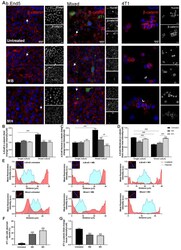
- Experimental details
- Figure 5 MH induces endothelial barrier properties through the increase in membrane beta-catenin expression. Confluent monolayers of b.End5 cells under physiological laminar non-pulsatile shear stress were exposed to 4T1 cells (previously labelled with CellTracker(tm) CMFDA Green Dye) and treated with MB or MH (1 uM), or DMEM (untreated), for 24 h and the expression of the AJs protein beta-catenin (red) in single and mixed cultures was evaluated by immunofluorescence analysis. Hoechst 33342 was used as counterstaining for nuclei (blue). Scale bar: 20 um. ( A ) Treatment with MB and MH induced an increase in beta-catenin expression in endothelial cells exposed to 4T1 cells, particularly at cell membrane (white arrows), and an increase in 4T1 cells with aberrant nuclei (white arrow heads). Semi-quantitative analysis of endothelial beta-catenin ( B ) mean intensity per cell, ( C ) nuclear intensity, and especially ( D ) membrane intensity confirmed the observations. ( E ) The localisation of beta-catenin was studied through plot profiles of pixel intensity throughout an endothelial cell (edge to edge). Semi-quantitative analysis of tumour cell cultures revealed ( F ) an increase in aberrant nuclei upon treatment with each drug, as well as ( G ) a decrease in beta-catenin intensity per cell. Data are represented as mean +- SEM of three independent experiments, where 3 cells/field, 10 fields/condition were evaluated. Statistical significances are denoted as * p < 0.05 and *** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
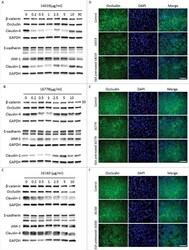
- Experimental details
- Figure 5. G. intestinalis secreted cysteine proteases have effects on epithelial barrier proteins. A, B, C. Levels of tight junction and adherence junction proteins in Caco-2 whole cell extracts were examined after 24 h of recombinant cysteine proteases treatment (A, 14019; B, 16779; C, 16160), respectively, by Western blot analysis, with GAPDH used as a loading control. Barrier proteins were detected with a mouse or rabbit anti-barrier protein antibody and then incubated with a HR- conjugated mouse or rabbit antibody. D E, F. The localization of occludin in Caco-2 cells visualized by immunofluorescence microscopy. Caco-2 monolayers were incubated with 2.5 ug/ml of 14019 (D), 16779 (E), 16160 (F) in the absence or presence of E64 (10 uM) for 24 h, followed by fixation of PFA. Occludin was detected with a mouse anti-occludin antibody and then incubated with an Alexa Fluor 488-conjugated mouse antibody. Images were taken by a Zeiss Axioplan II Imaging fluorescence microscope and analyzed by ZEN 2.1 software. Bar = 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
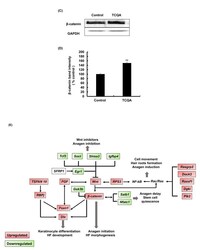
- Experimental details
- Figure 3C-E TCQA enhanced beta-catenin expression in the hair follicle. ( C ) beta-catenin protein expression was determined at the end of the treatment period. The protein was extracted from the treated area from the mice dorsal part, and western blot was carried away. ( D ) Band intensities was done assessed using LI-COR system. Results represent the mean +- SD of three independent experiments. *Statistically significant ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
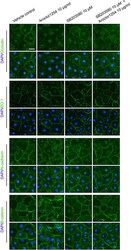
- Experimental details
- Figure 3 Aroclor1254 influenced distribution of junction proteins in primary SCs. Primarily isolated SCs were cultured on matrigel-coated coverslips at 0.05 x 10 6 /cm 2 . On day 3, Aroclor1254 (10 mu g/mL), SB203580 (10 mu m ), or the mixture of the above two was added to the medium. After 24 h, the cells were fixed with 4% paraformaldehyde. The distribution of BTB proteins at cell-cell junctions was observed by immunofluorescence using Alexa Fluor 488-conjugated secondary antibody (green) with cell nuclei stained with DAPI (blue). A reduced signal of occludin and disturbed distributions of N-cadherin and beta -catenin were detected at cell junctions of Aroclor1254-treated SCs, as compared with the sharply defined signals in the control and the Aroclor1254+SB203580 groups. Scale bar=25 mu m, which applied to all micrographs in Figure 3
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
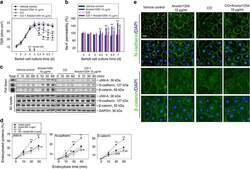
- Experimental details
- Figure 5 The effects of Aroclor1254 on the barrier function of SC epithelium after inhibiting the caveolin-dependent endocytosis. The SCs were treated with or without Aroclor1254 in the presence or absence of CO as the regimen depicted in Supplementary Figure S2a . ( a and b ) The barrier function of SC epithelium measured by recording TER ( a ) or calculating the permeability of Na-F ( b ) across the cell monolayer. The Na-F permeability in the vehicle control on day 3 was arbitrarily set at 100%. CO treatment was found to partially resist the disruption of BTB by Aroclor1254. Each bar refers to mean+-S.D. of n =3 experiments using different batches of SCs. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
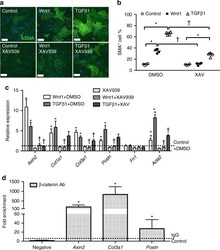
- Experimental details
- Fig. 7 Wnt/beta-catenin signaling induces cultured CF activation and ECM gene expression downstream of Tgfbeta signaling in vitro. a Representative images of alphaSMA ( green ) staining in cultured P60 CFs under indicated conditions are shown. Scale bar = 100 mum. b Wnt1 (100 ng/mL) and TGFbeta1 (10 ng/mL) induces significantly more alphaSMA + CFs relative to DMSO controls in cultured P60 CFs. Inhibition of Wnt/beta-catenin signaling by XAV939 (5 muM) abrogated Wnt1-induced alphaSMA expression in CF and also partially suppressed TGFbeta1-induced alphaSMA expression. * P < 0.05 vs. DMSO groups, + P < 0.05 vs. Control. c mRNA expression of the Wnt target gene Axin2 , and ECM genes Col1a1 , Col3a1 , Postn , and Acta2 in P60 CFs with indicated treatments was measured by quantitative PCR. * P < 0.05 vs. control, + P < 0.05 vs. DMSO. N = 4 independent cultures per group. Statistical significance was determined by Kruskal-Wallis tests followed by Mann-Whitney U tests for pairwise comparisons using Bonferonni adjustments to control for multiple testing. d ChIP assays were performed using P0 cultured CFs treated with BIO (2 muM) to determine the binding of beta-catenin in protein complexes with TCF/LEF consensus containing proximal sequences of Axin2 , Col3a1 , and Postn genes. Compared to IgG negative control, binding of beta-catenin to Col3a1 and Postn was significantly enriched. Axin2 was used as positive control. N = 4 independent cultures per group. Fold enrichment of DNA binding
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry