Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Immunohistochemistry [1]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-19469 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Catenin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Heat mediated antigen retrieval recommended prior to tissue staining.
- Concentration
- 1 mg/mL
Submitted references Reduced Graphene Oxide Modulates the FAK-Dependent Signaling Pathway in Glioblastoma Multiforme Cells In Vitro.
R-Spondin 2 and WNT/CTNNB1 Signaling Pathways Are Required for Porcine Follicle Development and In Vitro Maturation.
Differential activation of Gsk-3β in the cortex and the hippocampus induces cognitive and behavioural impairments in middle-aged ovariectomized rat.
Marine Collagen Substrates for 2D and 3D Ovarian Cancer Cell Systems.
Mechano-signalling, induced by fullerene C(60) nanofilms, arrests the cell cycle in the G2/M phase and decreases proliferation of liver cancer cells.
Diamond, graphite, and graphene oxide nanoparticles decrease migration and invasiveness in glioblastoma cell lines by impairing extracellular adhesion.
Intestinal knockout of Nedd4 enhances growth of Apc(min) tumors.
Vesicular Galectin-3 levels decrease with donor age and contribute to the reduced osteo-inductive potential of human plasma derived extracellular vesicles.
Szczepaniak J, Sosnowska M, Wierzbicki M, Witkowska-Pilaszewicz O, Strojny-Cieslak B, Jagiello J, Fraczek W, Kusmierz M, Grodzik M
Materials (Basel, Switzerland) 2022 Aug 24;15(17)
Materials (Basel, Switzerland) 2022 Aug 24;15(17)
R-Spondin 2 and WNT/CTNNB1 Signaling Pathways Are Required for Porcine Follicle Development and In Vitro Maturation.
Hwang SU, Yoon JD, Kim M, Cai L, Choi H, Oh D, Kim E, Hyun SH
Animals : an open access journal from MDPI 2021 Mar 5;11(3)
Animals : an open access journal from MDPI 2021 Mar 5;11(3)
Differential activation of Gsk-3β in the cortex and the hippocampus induces cognitive and behavioural impairments in middle-aged ovariectomized rat.
Rana AK, Sharma S, Singh D
Comprehensive psychoneuroendocrinology 2020 Nov;4:100019
Comprehensive psychoneuroendocrinology 2020 Nov;4:100019
Marine Collagen Substrates for 2D and 3D Ovarian Cancer Cell Systems.
Paradiso F, Fitzgerald J, Yao S, Barry F, Taraballi F, Gonzalez D, Conlan RS, Francis L
Frontiers in bioengineering and biotechnology 2019;7:343
Frontiers in bioengineering and biotechnology 2019;7:343
Mechano-signalling, induced by fullerene C(60) nanofilms, arrests the cell cycle in the G2/M phase and decreases proliferation of liver cancer cells.
Sosnowska M, Kutwin M, Jaworski S, Strojny B, Wierzbicki M, Szczepaniak J, Łojkowski M, Święszkowski W, Bałaban J, Chwalibog A, Sawosz E
International journal of nanomedicine 2019;14:6197-6215
International journal of nanomedicine 2019;14:6197-6215
Diamond, graphite, and graphene oxide nanoparticles decrease migration and invasiveness in glioblastoma cell lines by impairing extracellular adhesion.
Wierzbicki M, Jaworski S, Kutwin M, Grodzik M, Strojny B, Kurantowicz N, Zdunek K, Chodun R, Chwalibog A, Sawosz E
International journal of nanomedicine 2017;12:7241-7254
International journal of nanomedicine 2017;12:7241-7254
Intestinal knockout of Nedd4 enhances growth of Apc(min) tumors.
Lu C, Thoeni C, Connor A, Kawabe H, Gallinger S, Rotin D
Oncogene 2016 Nov 10;35(45):5839-5849
Oncogene 2016 Nov 10;35(45):5839-5849
Vesicular Galectin-3 levels decrease with donor age and contribute to the reduced osteo-inductive potential of human plasma derived extracellular vesicles.
Weilner S, Keider V, Winter M, Harreither E, Salzer B, Weiss F, Schraml E, Messner P, Pietschmann P, Hildner F, Gabriel C, Redl H, Grillari-Voglauer R, Grillari J
Aging 2016 Jan;8(1):16-33
Aging 2016 Jan;8(1):16-33
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of HeLa Whole Cell Lysate using Product # PA5-19469, beta Catenin primary antibody at a dilution of 1 µg/mL (lane 1). Staining of NIH 3T3 Whole Cell Lysate at a dilution of 1 µg/mL (lane 2). Blot treated with a secondary HRP-conjugated Goat polyclonal anti-Rabbit antibody was used at a dilution of 1:3000.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-beta Catenin Polyclonal Antibody (Product # PA5-19469) and a 85 kDa band corresponding to Catenin beta-1 was observed across the cell lines and tissues tested except Hep G2 which contain an exon 3-4 truncation mutation in the CTNNB1 gene corresponding to deletion of amino acids 25-140 in the ß-catenin protein. Whole cell extracts (30 µg lysate) of Hep G2 (Lane 1), SK-MEL-31 (Lane 2), MCF7 (Lane 3), MCF7 treated with 10 ng/mL of TGF-beta1 for 24 hours (Lane 4), A-431 (Lane 5), HeLa (Lane 6) and Mouse Lung (Lane 7) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # LC2002) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody ( at a concentration of 0.5 µg/mL) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
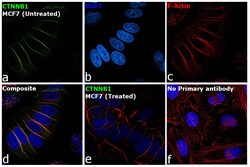
- Experimental details
- Immunofluorescence analysis of Catenin beta-1 was performed using 70% confluent log phase MCF7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with beta Catenin Polyclonal Antibody (Product # PA5-19469) at a concentration of 1 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing membrane localization. Panel e represents decreased expression of Beta catenin -1 in MCF7 cells treated with 10 ng/mL of TGF-beta1 for 24 hours. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical (formalin-fixed, paraffin-embedded) staining of Human Liver tissue using Product # PA5-19469, anti-Beta Catenin antibody. Primary antibody was used at a concentration of 1 µg/mL and exposed for 15 mins at room temp. The sample was pretreated using heat mediated antigen retrieval with Sodium Citrate Buffer (pH6/20 mins). The detection method was a HRP conjugated polymer, DAB chromogen and the sample was counterstained with haematoxylin and mounted with DPX.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
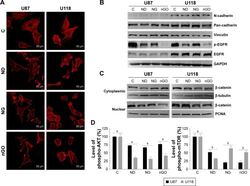
- Experimental details
- Figure 5 EGFR/AKT/mTOR and beta-catenin signaling in glioblastoma cells after treated with nanoparticles. Notes: ( A ) Confocal microscope images of U87 and U118 cells actin cytoskeleton. Cells were grown on extracellular matrix for 24 h and treated with diamond nanoparticles, graphite nanoparticles, or graphene oxide nanoparticles at a concentration of 20 mug/mL and incubated for 24 h. F-Actin was stained with phalloidin conjugated with Atto 633. ( B ) Western blot analysis of N-cadherin, pan-cadherin, vinculin, p-EGFR, and EGFR. GAPDH was used as a loading control. ( C ) Western blot analysis of nuclear and cytoplasmic protein fractions used for determination of beta-catenin protein level. PCNA and beta-tubulin were used as loading controls for nuclear and cytoplasmic fractions, respectively. ( D ) ELISA analysis of AKT and mTOR phosphorylation in comparison to control. Treatment with nanoparticles significantly reduced phospho-AKT ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Galectin-3s molecular way of action ( A ) Relative fold change of Runx-2 mRNA levels during osteogenic differentiation over a time course of 18 days. Runx-2 mRNA levels of Galectin-3 overexpressing (Gal3) (indicated in grey squares) or empty vector control transfected cells (C) (displayed by black dots) were evaluated by qPCR and normalized to GAPDH. Runx-2 mRNA transcription was significantly increased at day 4 of differentiation in Galectin-3 overexpressing ASCs (Gal3) compared to empty vector control transfected cells (C) Levels of empty vector control transfected cells are displayed as black dots and data obtained from Galectin-3 overexpressing cells as grey dots. ( B-C ) Spearman correlation of beta-Catenin protein levels before induction of osteogenic differentiation to ( B ) intracellular Galectin-3 levels or (C) corresponding mineralization capacity of ASCs. ( D ) Overexpression of Galectin-3 wild type (Gal3) and Serine-96 to Alanine (A) mutant compared to empty vector control transfected ASCs (C) was confirmed by Western blot. Galectin-3 as well as beta-Catenin protein levels have been normalized to GAPDH ( E-F ) ASCs overexpressing the Galectin-3 mutant (A) showed a significant reduction in their osteogenic differentiation capacity compared to Galectin-3 wild type (Gal3) overexpressing cells as analysed by ( E ) Alizarin Red S staining ( F ) ALP activity assay and ( G ) qPCR on Osteonectin. ( H-J ) ASCs were either untreated (untreated) or exposed to cell p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Identification and localization of roof plate-specific spondin2 (RSPO2) related factors in porcine ovarian tissues according to follicle size by immunofluorescence. Each ovary with small (1-2 mm), medium (3-6 mm), and large (7-9 mm) follicles. ( A ) RSPO2 and Catenin beta-1 (CTNNB1), ( B ) G-protein coupled receptor 4 (LGR4) and G-protein coupled receptor 5 (LGR5). theca interna (T.I. ), theca externa (T.E. ), granulosa cells (GC), and ooplasm (*).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of integrin alpha5, integrin beta1, beta-catenin, PAN-cadherin, and FAK after GN and rGO treatment at 25 ug/mL concentration for 24 h in U87 and Hs5 cells. GAPDH was used as a loading control. Abbreviations: rGO, reduced graphene oxide; GN, graphene; C, control group (untreated group); ExF, exfoliation; term, thermal; ATS, ammonium thiosulfate; TUD, thiourea dioxide; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
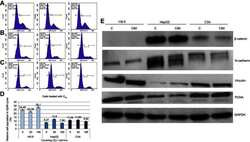
- Experimental details
- Figure 3 Propidium iodide (PI) 488 assay analysis. Notes: Effect of C 60 on the number (percentage) of ( A ) HS-5, ( B ) HepG2 or ( C ) C3A cells in the cycle phases. ( D ) Relative HS-5, HepG2 and C3A cell population in G2/M cycle (%). ( E ) Western blot analysis of beta-catenin, N-cadherin, vinculin and PCNA. GAPDH was used as a loading control. Abbreviations: C 60 , fullerenes; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; PCNA, proliferating cell nuclear antigen.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
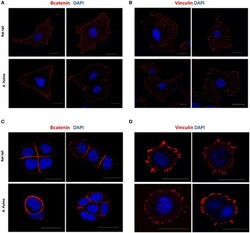
- Experimental details
- Figure 5 Adherents junctions and focal adhesion assembly in OvCa cells grown on R. pulmo and rat tail collagen substrate. SKOV.3 immunostaining of (A) B-catenin and (B) vinculin cells plated onto rat tail type I collagen, and R. pulmo jellyfish collagen. OVCAR.3 immunostaining of (C) B-catenin and (D) vinculin cells plated onto rat tail type I collagen, and R. pulmo jellyfish collagen. Scale bars represent 20 um.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot