Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-0209-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD20 Monoclonal Antibody (2H7), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 2H7 monoclonal antibody reacts with human CD20, a 33-36 kDa transmembrane protein. CD20 is expressed by developing B cells as well as mature B cells but not plasma cells. CD20 has been detected at low levels on a small subset of mature T cells. It is suggested that CD20 plays a role in B-cell activation. Applications Reported: This 2H7 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 2H7 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Light sensitivity: This tandem dye is sensitive photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-822-49) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333-54) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2H7
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references TRPA1 triggers hyperalgesia and inflammation after tooth bleaching.
Early Transcriptional Changes within Liver, Adrenal Gland, and Lymphoid Tissues Significantly Contribute to Ebola Virus Pathogenesis in Cynomolgus Macaques.
Genetic modification of primary human B cells to model high-grade lymphoma.
Rapid Germinal Center and Antibody Responses in Non-human Primates after a Single Nanoparticle Vaccine Immunization.
Relative safety of various spermatogenic stem cell purification methods for application in spermatogenic stem cell transplantation.
Evidence for persistence of the SHIV reservoir early after MHC haploidentical hematopoietic stem cell transplantation.
Tumor-associated B-cells induce tumor heterogeneity and therapy resistance.
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Sialylation of IgG Fc domain impairs complement-dependent cytotoxicity.
Phase I study of a bispecific ligand-directed toxin targeting CD22 and CD19 (DT2219) for refractory B-cell malignancies.
Characterization of simian immunodeficiency virus (SIV) that induces SIV encephalitis in rhesus macaques with high frequency: role of TRIM5 and major histocompatibility complex genotypes and early entry to the brain.
Transcriptional profiling of mRNAs and microRNAs in human bone marrow precursor B cells identifies subset- and age-specific variations.
An efficient low cost method for gene transfer to T lymphocytes.
Development of neurological disease is associated with increased immune activation in simian immunodeficiency virus-infected macaques.
Chen C, Huang X, Zhu W, Ding C, Huang P, Li R
Scientific reports 2021 Aug 31;11(1):17418
Scientific reports 2021 Aug 31;11(1):17418
Early Transcriptional Changes within Liver, Adrenal Gland, and Lymphoid Tissues Significantly Contribute to Ebola Virus Pathogenesis in Cynomolgus Macaques.
Jankeel A, Menicucci AR, Woolsey C, Fenton KA, Mendoza N, Versteeg K, Cross RW, Geisbert TW, Messaoudi I
Journal of virology 2020 May 18;94(11)
Journal of virology 2020 May 18;94(11)
Genetic modification of primary human B cells to model high-grade lymphoma.
Caeser R, Di Re M, Krupka JA, Gao J, Lara-Chica M, Dias JML, Cooke SL, Fenner R, Usheva Z, Runge HFP, Beer PA, Eldaly H, Pak HK, Park CS, Vassiliou GS, Huntly BJP, Mupo A, Bashford-Rogers RJM, Hodson DJ
Nature communications 2019 Oct 4;10(1):4543
Nature communications 2019 Oct 4;10(1):4543
Rapid Germinal Center and Antibody Responses in Non-human Primates after a Single Nanoparticle Vaccine Immunization.
Havenar-Daughton C, Carnathan DG, Boopathy AV, Upadhyay AA, Murrell B, Reiss SM, Enemuo CA, Gebru EH, Choe Y, Dhadvai P, Viviano F, Kaushik K, Bhiman JN, Briney B, Burton DR, Bosinger SE, Schief WR, Irvine DJ, Silvestri G, Crotty S
Cell reports 2019 Nov 12;29(7):1756-1766.e8
Cell reports 2019 Nov 12;29(7):1756-1766.e8
Relative safety of various spermatogenic stem cell purification methods for application in spermatogenic stem cell transplantation.
Tian J, Ma K, Pei CB, Zhang SH, Li X, Zhou Y, Yan B, Wang HY, Ma LH
Stem cell research & therapy 2019 Dec 16;10(1):382
Stem cell research & therapy 2019 Dec 16;10(1):382
Evidence for persistence of the SHIV reservoir early after MHC haploidentical hematopoietic stem cell transplantation.
Colonna L, Peterson CW, Schell JB, Carlson JM, Tkachev V, Brown M, Yu A, Reddy S, Obenza WM, Nelson V, Polacino PS, Mack H, Hu SL, Zeleski K, Hoffman M, Olvera J, Furlan SN, Zheng H, Taraseviciute A, Hunt DJ, Betz K, Lane JF, Vogel K, Hotchkiss CE, Moats C, Baldessari A, Murnane RD, English C, Astley CA, Wangari S, Agricola B, Ahrens J, Iwayama N, May A, Stensland L, Huang MW, Jerome KR, Kiem HP, Kean LS
Nature communications 2018 Oct 25;9(1):4438
Nature communications 2018 Oct 25;9(1):4438
Tumor-associated B-cells induce tumor heterogeneity and therapy resistance.
Somasundaram R, Zhang G, Fukunaga-Kalabis M, Perego M, Krepler C, Xu X, Wagner C, Hristova D, Zhang J, Tian T, Wei Z, Liu Q, Garg K, Griss J, Hards R, Maurer M, Hafner C, Mayerhöfer M, Karanikas G, Jalili A, Bauer-Pohl V, Weihsengruber F, Rappersberger K, Koller J, Lang R, Hudgens C, Chen G, Tetzlaff M, Wu L, Frederick DT, Scolyer RA, Long GV, Damle M, Ellingsworth C, Grinman L, Choi H, Gavin BJ, Dunagin M, Raj A, Scholler N, Gross L, Beqiri M, Bennett K, Watson I, Schaider H, Davies MA, Wargo J, Czerniecki BJ, Schuchter L, Herlyn D, Flaherty K, Herlyn M, Wagner SN
Nature communications 2017 Sep 19;8(1):607
Nature communications 2017 Sep 19;8(1):607
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Zhu H, Hu F, Sun X, Zhang X, Zhu L, Liu X, Li X, Xu L, Shi L, Gan Y, Su Y
Frontiers in immunology 2016;7:512
Frontiers in immunology 2016;7:512
Sialylation of IgG Fc domain impairs complement-dependent cytotoxicity.
Quast I, Keller CW, Maurer MA, Giddens JP, Tackenberg B, Wang LX, Münz C, Nimmerjahn F, Dalakas MC, Lünemann JD
The Journal of clinical investigation 2015 Nov 2;125(11):4160-70
The Journal of clinical investigation 2015 Nov 2;125(11):4160-70
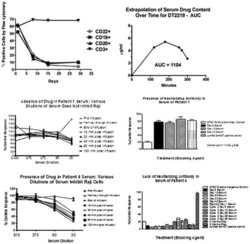
Phase I study of a bispecific ligand-directed toxin targeting CD22 and CD19 (DT2219) for refractory B-cell malignancies.
Bachanova V, Frankel AE, Cao Q, Lewis D, Grzywacz B, Verneris MR, Ustun C, Lazaryan A, McClune B, Warlick ED, Kantarjian H, Weisdorf DJ, Miller JS, Vallera DA
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Mar 15;21(6):1267-72
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Mar 15;21(6):1267-72
Characterization of simian immunodeficiency virus (SIV) that induces SIV encephalitis in rhesus macaques with high frequency: role of TRIM5 and major histocompatibility complex genotypes and early entry to the brain.
Matsuda K, Dang Q, Brown CR, Keele BF, Wu F, Ourmanov I, Goeken R, Whitted S, Riddick NE, Buckler-White A, Hirsch VM
Journal of virology 2014 Nov;88(22):13201-11
Journal of virology 2014 Nov;88(22):13201-11
Transcriptional profiling of mRNAs and microRNAs in human bone marrow precursor B cells identifies subset- and age-specific variations.
Jensen K, Brusletto BS, Aass HC, Olstad OK, Kierulf P, Gautvik KM
PloS one 2013;8(7):e70721
PloS one 2013;8(7):e70721
An efficient low cost method for gene transfer to T lymphocytes.
Chicaybam L, Sodre AL, Curzio BA, Bonamino MH
PloS one 2013;8(3):e60298
PloS one 2013;8(3):e60298
Development of neurological disease is associated with increased immune activation in simian immunodeficiency virus-infected macaques.
Dang Q, Whitted S, Goeken RM, Brenchley JM, Matsuda K, Brown CR, Lafont BA, Starost MF, Iyengar R, Plishka RJ, Buckler-White A, Hirsch VM
Journal of virology 2012 Dec;86(24):13795-9
Journal of virology 2012 Dec;86(24):13795-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of human peripheral blood mononuclear cells with CD45 Pacific Blue, CD19 FITC, CD3 APC, and CD20 PE-Cyanine7. As expected based on known relative expression patterns, CD20 clone 2H7 stains a subset of lymphocytes (pink), but not monocytes (orange) and granulocytes (blue).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
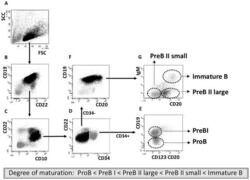
- Figure 1 Cell sorting of precursor B cells subsets from CD10 positively selected cells. Immunomagnetic selection and subsequent FACS were used to isolate the five populations from pediatric and adult human BM. Shown are the FACS dot plots with sorting gates to obtain CD34 + CD19 - ProB cells, CD34 + CD19 + PreBI cells, CD34 - CD19 + CD20 dim PreBII large cells, CD34 - CD19 + CD20 - PreBII small cells, and CD34 - CD19 + CD20 high IgM + Immature B cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
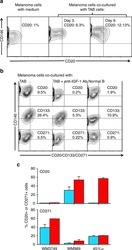
- Fig. 4 IGF-1-dependent induction of cancer stem cell markers CD20, CD133, and CD271 (NGFR) on melanoma cells. a Melanoma cells (WM3749) co-cultured with TAB cells (days 3 and 9) show high expression of CD20 ( middle and right panels ) compared with the control culture ( left panel ) as determined by FACS analysis. Melanoma cells were co-stained with anti-CD146 (MCAM, PE-conjugated) and anti-CD20 (FITC-conjugated) antibodies to distinguish them from B cells, which are CD146-negative;percentages indicate co-expression of both markers on the malignant cells. b Melanoma cells (WM3749) co-cultured with TAB cells (day 6) show high expression of CD20, CD133 and CD271 ( left panel ) compared with minimal or low expression of those markers when tumor cells are co-cultured with NB cells ( right panel ). Co-culture of melanoma cells with TAB cells did not modulate the expression of CD144 (vascular-endothelial cadherin marker) that are normally expressed by aggressive melanomas (data not shown). Induction of CD20, CD133, and CD271 was blocked when anti-IGF-1 neutralizing antibody (10 mug/ml) was used in the co-culture ( middle panel ). Anti-IL-1, anti-PDGF or anti-VEGF antibodies had no effect on CD marker expression (data not shown). Percentages indicate co-expression of CD20, CD133, or CD271on CD146 + melanoma cells. Results are representative of two independent experiments. c Melanoma cells(WM3749, WM989 and 451Lu) cultured in the presence of recombinant IGF-1 (25 ng/ml) for 5 days sh
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
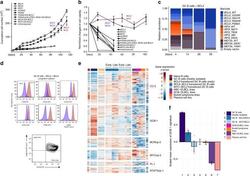
- Fig. 2 Long-term expansion of human germinal center B cells ex vivo. a Primary human GC B cells were transduced with the indicated oncogenes and oncogene combinations and cultured separately for up to 120 days. Graph shows calculated theoretical absolute cell numbers (+-s.e.m., n = 3). Viable cells were assessed by trypan blue exclusion. Source data are provided as a Source Data file. b Primary human GC B cells were transduced with different oncogenes and oncogene combinations and monitored by flow cytometry. Graph shows the change in cell viability assessed by scatter characteristic by flow cytometry ( +- s.e.m., n = 3). Source data are provided as a Source Data file. c Primary human GC B cells were transduced with BCL2 in combination with other transcription factors in a pooled, competitive culture. Graph shows relative abundance of transcription factors or their mutant versions over four different timepoints ( n = 3). d Primary human GC B cells were transduced with the oncogenic cocktail BCL2 and BCL6 and cultured to day 73. Representative flow cytometry analysis ( n = 3) for the expression of the GC B cell markers CD38, CD20, CD19, CD80, CD22, CD95, CXCR4, and CD86. Red histograms show GC B cells compared to primary human naive B cells (blue). e Heat map of gene expression of freshly isolated GC B cells ( n = 3), transduced GC B cells ( BCL2-BCL6, BCL2-MYC ) cultured ex vivo for 5 or 73 days ( n = 3), plasma cell line ( n = 1), naive B cells ( n = 1), and lymphoma cell li
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
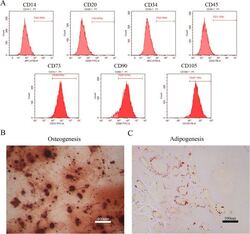
- Experimental details
- Figure 1 Identification of dental pulp stem cells (DPSCs). Human DPSCs were positive for the cell surface antigens CD73, CD90, and CD105, as well as negative for CD14, CD20, CD34, and CD45 demonstrated by flow cytometry ( A ). DPSCs were cultured under osteogenic ( B , 14 days) or adipogenic ( C , 21 days) conditions, and showed mineralized nodules and lipid clusters as revealed by alizarin red and oil red staining, respectively. Scale bar = 400 ( B ) or 100 ( C ) mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
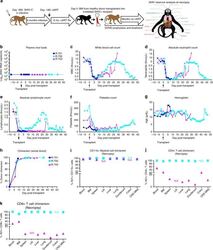
- Experimental details
- Fig. 3 Allogeneic HCT in SHIV-C Infected, cART-treated RM. a Schematic of SHIV-C infection and allogeneic bone marrow transplantation strategy in SHIV-C infected, cART-treated RM. b Plasma Viral Loads (PVL) were measured longitudinally following transplant as specified in Fig. 1a . c White blood count (WBC) (x10 3 /uL). d Absolute Neutrophil Count (ANC) (x10 3 /uL). e Absolute Lymphocyte Count (ALC) (x10 3 /uL). f Platelet Count (PLT) (x10 3 /uL). g Hemoglobin (Hgb) concentration (g/dL). h Percent whole blood donor chimerism, measured by microsatellite analysis. i Percent donor myeloid (CD11b + CD3-) chimerism, measured flow cytometrically. j Percent donor CD4 + T cell (CD3+ CD4+ CD8- CD20- CD11b- lymphocytes) chimerism, measured flow cytometrically. k Percent donor CD8 + T cell (CD3+ CD8+ CD4- CD20- CD11b- lymphocytes) chimerism, measured by flow cytometry
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Longitudinal eOD-GT8 60-mer-Specific B GC and Non-B GC Cell Responses in Draining LNs after a Single IM or s.c. Immunization (A) Flow cytometry identification of total eOD-GT8 60-mer-specific LN Bcells before and after immunization (top and bottom samples pairs are fromindividual RMs). Gated on CD20 + B cells. Full gating in Figure S2 . (B) Frequency of total eOD-GT8 60-mer-specific B cells after IM or s.c.immunization (Mann-Whitney, one tailed). (C) Flow cytometry identification of eOD-GT8 60-mer-specific plasmablast(KI67 + BCL6 - ) or GC(KI67 + BCL6 + ) B cells in the LN. (D) Frequency of KI67 + BCL6 - plasmablastsamong eOD-GT8 60-mer-specific B cells in the draining axillary LN over time. (E) Frequency of B GC cells among total eOD-GT860-mer-specific B cells in the draining axillary LN over time (Mann-Whitney, twotailed). (F) Representative flow cytometry gating of eOD-GT8 60-mer-specific Bcells among B GC cells. (G) Frequency of eOD-GT8 60-mer-specific B GC cells in thedraining axillary LN over time (Mann-Whitney, two tailed). (H) Total number of eOD-GT8 60-mer-specific B GC cells in theLN FNA sample of the draining axillary LN. (I) Total number of eOD-GT8 60-mer-specific B cells in the LN FNA sampleof the draining axillary LN. *p < 0.05. Each point represents an individual LN FNA sample. n =8, four LN FNAs per immunization condition at each time point. See also FiguresS2 and S3 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
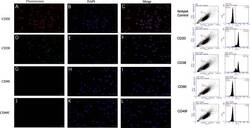
- Experimental details
- Fig. 1 Marker verification and validation of independent markers in BALL cells by immunofluorescence and flow cytometry. The expression of CD20 ( a - c ) and CD38 ( d - f ), were positive on BALL cell membranes. CD90f ( g - i ) and CD49f ( j , k ) were not expressed in BALL cells. DAPI indicates the cell nucleus. This finding was confirmed via the conducted flow cytometry analysis. Scale bar = 100 mum
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry