Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Flow cytometry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 47-0209-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-CD20 Monoclonal Antibody (2H7), APC-eFluor 780, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 2H7 monoclonal antibody reacts with human CD20, a 33-36 kDa transmembrane protein. CD20 is expressed by developing B cells as well as mature B cells but not plasma cells. CD20 has been detected at low levels on a small subset of mature T cells. It is suggested that CD20 plays a role in B-cell activation. Applications Reported: This 2H7 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 2H7 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blod cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. APC-eFluor 780 emits at 780 nm and is excited with the Red laser (633 nm). Please make sure that your instrument is capable of detecting this fluorochome. Light sensitivity: This tandem is sensitive to photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (cat. 00-8222) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (cat. 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 633-647 nm; Emission: 780 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2H7
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Evidence for persistence of the SHIV reservoir early after MHC haploidentical hematopoietic stem cell transplantation.
Peripherally derived T regulatory and γδ T cells have opposing roles in the pathogenesis of intractable pediatric epilepsy.
Tumor-associated B-cells induce tumor heterogeneity and therapy resistance.
Automation of cellular therapy product manufacturing: results of a split validation comparing CD34 selection of peripheral blood stem cell apheresis product with a semi-manual vs. an automatic procedure.
Automated CD34+ cell isolation of peripheral blood stem cell apheresis product.
Phase I study of a bispecific ligand-directed toxin targeting CD22 and CD19 (DT2219) for refractory B-cell malignancies.
Transcriptional profiling of mRNAs and microRNAs in human bone marrow precursor B cells identifies subset- and age-specific variations.
An efficient low cost method for gene transfer to T lymphocytes.
Alterations in peripheral blood B cell subsets and dynamics of B cell responses during human schistosomiasis.
Characterization of proposed human B-1 cells reveals pre-plasmablast phenotype.
Schistosomes induce regulatory features in human and mouse CD1d(hi) B cells: inhibition of allergic inflammation by IL-10 and regulatory T cells.
Colonna L, Peterson CW, Schell JB, Carlson JM, Tkachev V, Brown M, Yu A, Reddy S, Obenza WM, Nelson V, Polacino PS, Mack H, Hu SL, Zeleski K, Hoffman M, Olvera J, Furlan SN, Zheng H, Taraseviciute A, Hunt DJ, Betz K, Lane JF, Vogel K, Hotchkiss CE, Moats C, Baldessari A, Murnane RD, English C, Astley CA, Wangari S, Agricola B, Ahrens J, Iwayama N, May A, Stensland L, Huang MW, Jerome KR, Kiem HP, Kean LS
Nature communications 2018 Oct 25;9(1):4438
Nature communications 2018 Oct 25;9(1):4438
Peripherally derived T regulatory and γδ T cells have opposing roles in the pathogenesis of intractable pediatric epilepsy.
Xu D, Robinson AP, Ishii T, Duncan DS, Alden TD, Goings GE, Ifergan I, Podojil JR, Penaloza-MacMaster P, Kearney JA, Swanson GT, Miller SD, Koh S
The Journal of experimental medicine 2018 Apr 2;215(4):1169-1186
The Journal of experimental medicine 2018 Apr 2;215(4):1169-1186
Tumor-associated B-cells induce tumor heterogeneity and therapy resistance.
Somasundaram R, Zhang G, Fukunaga-Kalabis M, Perego M, Krepler C, Xu X, Wagner C, Hristova D, Zhang J, Tian T, Wei Z, Liu Q, Garg K, Griss J, Hards R, Maurer M, Hafner C, Mayerhöfer M, Karanikas G, Jalili A, Bauer-Pohl V, Weihsengruber F, Rappersberger K, Koller J, Lang R, Hudgens C, Chen G, Tetzlaff M, Wu L, Frederick DT, Scolyer RA, Long GV, Damle M, Ellingsworth C, Grinman L, Choi H, Gavin BJ, Dunagin M, Raj A, Scholler N, Gross L, Beqiri M, Bennett K, Watson I, Schaider H, Davies MA, Wargo J, Czerniecki BJ, Schuchter L, Herlyn D, Flaherty K, Herlyn M, Wagner SN
Nature communications 2017 Sep 19;8(1):607
Nature communications 2017 Sep 19;8(1):607
Automation of cellular therapy product manufacturing: results of a split validation comparing CD34 selection of peripheral blood stem cell apheresis product with a semi-manual vs. an automatic procedure.
Hümmer C, Poppe C, Bunos M, Stock B, Wingenfeld E, Huppert V, Stuth J, Reck K, Essl M, Seifried E, Bonig H
Journal of translational medicine 2016 Mar 16;14:76
Journal of translational medicine 2016 Mar 16;14:76
Automated CD34+ cell isolation of peripheral blood stem cell apheresis product.
Spohn G, Wiercinska E, Karpova D, Bunos M, Hümmer C, Wingenfeld E, Sorg N, Poppe C, Huppert V, Stuth J, Reck K, Essl M, Seifried E, Bönig H
Cytotherapy 2015 Oct;17(10):1465-71
Cytotherapy 2015 Oct;17(10):1465-71
Phase I study of a bispecific ligand-directed toxin targeting CD22 and CD19 (DT2219) for refractory B-cell malignancies.
Bachanova V, Frankel AE, Cao Q, Lewis D, Grzywacz B, Verneris MR, Ustun C, Lazaryan A, McClune B, Warlick ED, Kantarjian H, Weisdorf DJ, Miller JS, Vallera DA
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Mar 15;21(6):1267-72
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Mar 15;21(6):1267-72
Transcriptional profiling of mRNAs and microRNAs in human bone marrow precursor B cells identifies subset- and age-specific variations.
Jensen K, Brusletto BS, Aass HC, Olstad OK, Kierulf P, Gautvik KM
PloS one 2013;8(7):e70721
PloS one 2013;8(7):e70721
An efficient low cost method for gene transfer to T lymphocytes.
Chicaybam L, Sodre AL, Curzio BA, Bonamino MH
PloS one 2013;8(3):e60298
PloS one 2013;8(3):e60298
Alterations in peripheral blood B cell subsets and dynamics of B cell responses during human schistosomiasis.
Labuda LA, Ateba-Ngoa U, Feugap EN, Heeringa JJ, van der Vlugt LE, Pires RB, Mewono L, Kremsner PG, van Zelm MC, Adegnika AA, Yazdanbakhsh M, Smits HH
PLoS neglected tropical diseases 2013;7(3):e2094
PLoS neglected tropical diseases 2013;7(3):e2094
Characterization of proposed human B-1 cells reveals pre-plasmablast phenotype.
Covens K, Verbinnen B, Geukens N, Meyts I, Schuit F, Van Lommel L, Jacquemin M, Bossuyt X
Blood 2013 Jun 27;121(26):5176-83
Blood 2013 Jun 27;121(26):5176-83
Schistosomes induce regulatory features in human and mouse CD1d(hi) B cells: inhibition of allergic inflammation by IL-10 and regulatory T cells.
van der Vlugt LE, Labuda LA, Ozir-Fazalalikhan A, Lievers E, Gloudemans AK, Liu KY, Barr TA, Sparwasser T, Boon L, Ngoa UA, Feugap EN, Adegnika AA, Kremsner PG, Gray D, Yazdanbakhsh M, Smits HH
PloS one 2012;7(2):e30883
PloS one 2012;7(2):e30883
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
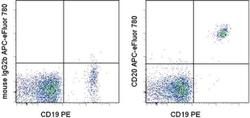
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 PE (Product # 12-0199-80) and Mouse IgG2b K Isotype Control APC-eFluor® 780 (Product # 47-4732-80) (left) or Anti-Human CD20 APC-eFluor® 780 (right). Cells in the lymphocyte gate were used for analysis.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry