Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [1]
- Immunohistochemistry [2]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-32465 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Cytokeratin 5 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Heat-mediated antigen retrieval is recommended prior to staining, using a 10mM citrate buffer, pH 6.0, for 10 minutes followed by cooling at room temperature for 20 min. Following antigen retrieval, incubate samples with primary antibody for 30 min at room temperature. A suggested positive control is skin, breast, prostate or mesothelioma.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 500 µL
- Concentration
- Conc. Not Determined
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references Assessment of Beta-2 Microglobulin Gene Edited Airway Epithelial Stem Cells as a treatment for Sulfur Mustard Inhalation.
The NOTCH3 Downstream Target HEYL Is Required for Efficient Human Airway Basal Cell Differentiation.
Cigarette Smoke Activates NOTCH3 to Promote Goblet Cell Differentiation in Human Airway Epithelial Cells.
ΔN-Tp63 Mediates Wnt/β-Catenin-Induced Inhibition of Differentiation in Basal Stem Cells of Mucociliary Epithelia.
Role of KRAS in regulating normal human airway basal cell differentiation.
Characterization of an immortalized human small airway basal stem/progenitor cell line with airway region-specific differentiation capacity.
Na+/H+ Exchangers Are Required for the Development and Function of Vertebrate Mucociliary Epithelia.
Mandatory role of HMGA1 in human airway epithelial normal differentiation and post-injury regeneration.
Endothelial Cell Mediated Promotion of Ciliated Cell Differentiation of Human Airway Basal Cells via Insulin and Insulin-Like Growth Factor 1 Receptor Mediated Signaling.
JAG1-Mediated Notch Signaling Regulates Secretory Cell Differentiation of the Human Airway Epithelium.
Activation of NOTCH1 or NOTCH3 signaling skews human airway basal cell differentiation toward a secretory pathway.
Endothelial MMP14 is required for endothelial-dependent growth support of human airway basal cells.
Activation of soluble guanylyl cyclase prevents foam cell formation and atherosclerosis.
Generation of a human airway epithelium derived basal cell line with multipotent differentiation capacity.
Naeimi Kararoudi M, Alsudayri A, Hill CL, Elmas E, Sezgin Y, Thakkar A, Hester ME, Malleske DT, Lee DA, Neal ML, Perry MR, Harvilchuck JA, Reynolds SD
Frontiers in genome editing 2022;4:781531
Frontiers in genome editing 2022;4:781531
The NOTCH3 Downstream Target HEYL Is Required for Efficient Human Airway Basal Cell Differentiation.
Bodas M, Subramaniyan B, Moore AR, Metcalf JP, Ocañas SR, Freeman WM, Georgescu C, Wren JD, Walters MS
Cells 2021 Nov 18;10(11)
Cells 2021 Nov 18;10(11)
Cigarette Smoke Activates NOTCH3 to Promote Goblet Cell Differentiation in Human Airway Epithelial Cells.
Bodas M, Moore AR, Subramaniyan B, Georgescu C, Wren JD, Freeman WM, Brown BR, Metcalf JP, Walters MS
American journal of respiratory cell and molecular biology 2021 Apr;64(4):426-440
American journal of respiratory cell and molecular biology 2021 Apr;64(4):426-440
ΔN-Tp63 Mediates Wnt/β-Catenin-Induced Inhibition of Differentiation in Basal Stem Cells of Mucociliary Epithelia.
Haas M, Gómez Vázquez JL, Sun DI, Tran HT, Brislinger M, Tasca A, Shomroni O, Vleminckx K, Walentek P
Cell reports 2019 Sep 24;28(13):3338-3352.e6
Cell reports 2019 Sep 24;28(13):3338-3352.e6
Role of KRAS in regulating normal human airway basal cell differentiation.
Ogawa F, Walters MS, Shafquat A, O'Beirne SL, Kaner RJ, Mezey JG, Zhang H, Leopold PL, Crystal RG
Respiratory research 2019 Aug 9;20(1):181
Respiratory research 2019 Aug 9;20(1):181
Characterization of an immortalized human small airway basal stem/progenitor cell line with airway region-specific differentiation capacity.
Wang G, Lou HH, Salit J, Leopold PL, Driscoll S, Schymeinsky J, Quast K, Visvanathan S, Fine JS, Thomas MJ, Crystal RG
Respiratory research 2019 Aug 23;20(1):196
Respiratory research 2019 Aug 23;20(1):196
Na+/H+ Exchangers Are Required for the Development and Function of Vertebrate Mucociliary Epithelia.
Sun DI, Tasca A, Haas M, Baltazar G, Harland RM, Finkbeiner WE, Walentek P
Cells, tissues, organs 2018;205(5-6):279-292
Cells, tissues, organs 2018;205(5-6):279-292
Mandatory role of HMGA1 in human airway epithelial normal differentiation and post-injury regeneration.
Zhang H, Yang J, Walters MS, Staudt MR, Strulovici-Barel Y, Salit J, Mezey JG, Leopold PL, Crystal RG
Oncotarget 2018 Mar 6;9(18):14324-14337
Oncotarget 2018 Mar 6;9(18):14324-14337
Endothelial Cell Mediated Promotion of Ciliated Cell Differentiation of Human Airway Basal Cells via Insulin and Insulin-Like Growth Factor 1 Receptor Mediated Signaling.
Gomi K, Tang Y, Arbelaez V, Crystal RG, Walters MS
Stem cell reviews and reports 2017 Apr;13(2):309-317
Stem cell reviews and reports 2017 Apr;13(2):309-317
JAG1-Mediated Notch Signaling Regulates Secretory Cell Differentiation of the Human Airway Epithelium.
Gomi K, Staudt MR, Salit J, Kaner RJ, Heldrich J, Rogalski AM, Arbelaez V, Crystal RG, Walters MS
Stem cell reviews and reports 2016 Aug;12(4):454-63
Stem cell reviews and reports 2016 Aug;12(4):454-63
Activation of NOTCH1 or NOTCH3 signaling skews human airway basal cell differentiation toward a secretory pathway.
Gomi K, Arbelaez V, Crystal RG, Walters MS
PloS one 2015;10(2):e0116507
PloS one 2015;10(2):e0116507
Endothelial MMP14 is required for endothelial-dependent growth support of human airway basal cells.
Ding BS, Gomi K, Rafii S, Crystal RG, Walters MS
Journal of cell science 2015 Aug 15;128(16):2983-8
Journal of cell science 2015 Aug 15;128(16):2983-8
Activation of soluble guanylyl cyclase prevents foam cell formation and atherosclerosis.
Tsou CY, Chen CY, Zhao JF, Su KH, Lee HT, Lin SJ, Shyue SK, Hsiao SH, Lee TS
Acta physiologica (Oxford, England) 2014 Apr;210(4):799-810
Acta physiologica (Oxford, England) 2014 Apr;210(4):799-810
Generation of a human airway epithelium derived basal cell line with multipotent differentiation capacity.
Walters MS, Gomi K, Ashbridge B, Moore MA, Arbelaez V, Heldrich J, Ding BS, Rafii S, Staudt MR, Crystal RG
Respiratory research 2013 Dec 3;14(1):135
Respiratory research 2013 Dec 3;14(1):135
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Cytokeratin 5 Polyclonal Antibody (Product # PA5-32465) and a 60 kDa band corresponding to KRT5 was observed across tissues and cell lines tested. Whole cell extracts (30 µg lysate) of Mouse Brain (Lane 1), Rat Brain (Lane 2), Mouse Tongue (Lane 3), A-431 (Lane 4) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:4000 dilution) using the iBright™ FL1500 Imaging System (Product # A44115). Chemiluminescent detection was performed using SuperSignal™ West Pico PLUS Chemiluminescent Substrate (Product # 34580).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockdown of Cytokeratin 5 was achieved by transfecting A-431 with Cytokeratin 5 specific siRNAs (Silencer® select Product # s7956, S7957). Western blot analysis (Fig. a) was performed using Whole cell extracts from the Cytokeratin 5 knockdown cells (lane 3), non-targeting scrambled siRNA transfected cells (lane 2) and untransfected cells (lane 1). The blot was probed with Cytokeratin 5 Polyclonal Antibody (Product # PA5-32465, 1:1000 dilution) and Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Decrease in signal upon siRNA mediated knock down confirms that antibody is specific to cytokeratin 5.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Cytokeratin 5 Polyclonal Antibody(Product # PA5-32465) and a 60kDa band corresponding to Cytokeratin 5 was observed across the cell lines tested. Whole cell extracts (30 µg lysate) of MCF 10A (Lane 1), MCF7 (Lane 2), A-431 (Lane 3), HaCaT (Lane 4), IMR-32 (Lane 5), SH-SY5Y (Lane 6) and U-937 (Lane 7) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # LC2001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Cytokeratin 5 was performed using 70% confluent log phase A-431 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Cytokeratin 5 Polyclonal Antibody (Product # PA5-32465) at 1:100 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034, 1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoskeletal localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
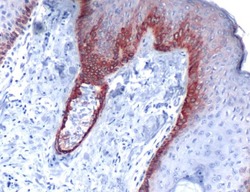
- Experimental details
- Immunohistochemical analysis of Keratin 5 using anti-Keratin 5 Polyclonal Antibody (Product # PA5-32465) in Skin Cancer Tissue. The recommened dilution for this antibody in immunohistochemistry applications is 1:100.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of Cytokeratin 5 using a polyclonal antibody (Product # PA1-37974).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Isolation of BCi-NS1.1 clone of the BCi-NS1 immortalized human airway basal cell line. A . Morphology of BCi-NS1.1 clone at passage 18 and passage 44. B . Comparison of the parental and clonal immortalized basal cells by Western analysis of cell type specific markers. Lane 1 - Primary basal cells; lane 2 - BCi-NS1 parental immortalized basal cells, passage 24; and lane 3 - BCi-NS1.1 clonal immortalized basal cells, passage 3. For all cells, shown is the expression of the basal cell markers KRT5 and TP63. GAPDH was used as a loading control. C . Immunohistochemical characterization of cytopreps of BCi-NS1.1 cells (passage 22) with cell-type specific markers: KRT5 (basal cell); TP63 (basal cell); CD151 (basal cell); beta-tubulin IV (ciliated cell); MUC5AC (secretory cell); TFF3 (goblet cell); CC10 (Clara cell); chromogranin A (neuroendocrine cell); N-cadherin (mesenchymal cell) and isotype control. Bar = 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8 Quantification of IL-13 modulation of the differentiation of BCi-NS1.1 immortalized airway basal cells. BCi-NS1.1 cells were cultured on air-liquid interface (ALI) in the absence and presence of IL-13. A . TaqMan analysis of cell type specific mRNA markers at ALI day 28, including basal cell markers (KRT5, TP63); secretory cell markers (MUC5AC, MUC5B); goblet cell marker (TFF3); Clara cell marker (CC10); and ciliated cell markers (DNAI1, FOXJ1). Data shown is the average +- the standard error of n = 5 independent experiments (BCi-NS1.1, passage 6-15). Statistics were calculated by 2-tailed Student's t test. U = untreated; IL-13 = treated with IL-13. B . Western analysis of untreated and IL-13 treated cells of cell type specific markers at ALI day 28. Lane 1 - untreated, lane 2 - IL-13 treated. Shown is the expression of a basal cell marker (TP63); Clara cell marker (CC10) and ciliated cell markers (DNAI1, FOXJ1). GAPDH was used as a loading control. C . Immunofluorescent staining of ALI day 28 membranes for untreated and IL-13 treated cells for MUC5AC (secretory cell, green) and nuclei (blue). Bar = 20 mum. For B and C , all data shown is representative of n = 3 independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Effect of silencing KRAS expression on regulation of basal cell (BC) differentiation into a mucociliated epithelium. Primary human airway BC were untreated or transfected with either control, or KRAS specific siRNA and cultured on ALI for 14 days to assess the impact of KRAS on BC differentiation into a mucociliated epithelium. a Immunofluorescence staining of KRT5 + BC. Sections of cells on ALI day 14 stained for KRT5 (red) and DAPI (nuclei, blue). b Alcian blue staining of secretory cells. Sections of cells on ALI day 14 stained with Alcian blue (blue). c Immunofluorescence staining of MUC5B + secretory cells. Sections of cells on ALI day 14 stained for MUC5B (red) and DAPI (nuclei, blue). d Immunofluorescence staining of SCGB1A1 + secretory cells. Sections of cells on ALI day 14 stained for SCGB1A1 (red) and DAPI (nuclei, blue). e Immunofluorescence staining of beta-tubulin IV + ciliated cells. Sections of cells on ALI day 14 stained for beta-tubulin IV (green) and DAPI (nuclei, blue). f Immunofluorescence staining of IVL + squamous cells. Sections of cells on ALI day 14 stained for IVL (red). The data for a - f are the mean percentage of positively stained cells for n = 3 independent experiments performed with independent donors of BC. Error error bars indicate standard error of the mean. ANOVA was used to determine the statistical significance among groups as described in the methods section. Scale bar 20 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Consequences of constitutive KRAS activity on promotion of basal cell (BC) differentiation into secretory and ciliated cells Primary human airway BC were infected with control lentivirus or lentivirus over-expressing wild-type (WT) KRAS or the constitutively active G12 V mutant (activated) and cultured on ALI for 28 days to assess the impact of KRAS on BC differentiation into a mucociliated epithelium. a Immunofluorescence staining of KRT5 + BC. Sections of cells on ALI day 28 membranes were stained for KRT5 (red) and DAPI (nuclei, blue). b Alcian blue staining of secretory cells. Sections of cells on ALI day 28 membranes were stained for Alcian blue (blue). c Immunofluorescence staining of MUC5B + secretory cells. Sections of cells on ALI day 28 membranes were stained for MUC5B (red) and DAPI (nuclei, blue). d Immunofluorescence staining of SCGB1A1 + secretory cells. Sections of cells on ALI day 28 membranes were stained for SCGB1A1 (red) and DAPI (nuclei, blue). e Immunofluorescence staining of beta-tubulin IV + ciliated cells. Sections of cells on ALI day 28 membranes were stained for beta-tubulin IV (green) and DAPI (nuclei, blue). f Immunohistochemical staining of IVL + squamous cells. Sections of cells on ALI day 28 membranes were stained for IVL (red) and DAPI (nuclei, blue). The data for a - f are the mean for n = 3 independent experiments performed with independent donors of BC. The bars indicate the mean percentage of positively stained cells for n = 3 indepe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5. Pharmacological inhibition of NHE function impairs MCC cilia formation and regeneration in human airway epithelial cells (HAECs). HAECs were grown in air-liquid interface (ALI) culture and treated either with vehicle (DMSO controls) or 25 muM of the NHE inhibitor EIPA for 7 days. A Top view on cultured cells at ALI day 28, after 7 days of treatment during week 4 of regeneration. Immunofluorescence staining for MCC cilia (Ac.-alpha-Tub., magenta), F-actin (green), and nuclei (DAPI, blue) revealed defective MCC ciliation in EIPA-treated specimens. Scale bars indicate magnification. B Immunofluorescence staining for MCC cilia (Ac.-alpha-Tub., green), Keratin 5-positive basal cells (KRT5, magenta), and nuclei (DAPI, blue) revealed ciliation defects in EIPA-treated specimens, but no loss of basal cells. Scale bars indicate magnification. C Morphology of control (DMSO)- and EIPA-treated HAECs at ALI day 28, after treatment during the indicated week of regeneration, revealed successful epithelialization of HAECs in controls and EIPA-treated specimens. Fixation directly after EIPA treatment (week 4) further revealed a thinning of the epithelium. Apical surface up. Scale bars indicate magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. Wnt/beta-Catenin Signaling Inhibits Differentiation and Promotes Stemness in Human BCs (A-F) Human immortalized BC (BCIs) kept in air-liquid interface (ALI) culture for up to 4 weeks. Human recombinant R-spondin2 (RSPO2) was used to activate Wnt/beta-catenin signaling starting at ALI day 7 (D7). n = 3 cultures per time point and treatment. (A) Confocal imaging of specimens stained for Ac.-alpha-tubulin (MCCs, blue), CC10 (Club cells, green), and Actin (cell membranes, magenta) show reduced MCC and Club cell numbers in RSPO2-treated cultures. (B) RSPO2 does not reduce the number of DeltaN-TP63+ (green) cells. Nuclei (DAPI, blue). (C) Quantification from (A) and (B). Mann Whitney test, not significant, ns (p > 0.05); *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. Inhibition of Wnt/beta-Catenin Signaling Transiently Reduces Stemness and Promotes Differentiation in Human BCs (A-F) BCIs in ALI culture for up to 4 weeks. Human recombinant DKK1 (DKK1) was used to inhibit Wnt/beta-catenin signaling starting at ALI day 7 (D7). (A) Confocal imaging of specimens stained for Ac.-alpha-tubulin (MCCs, blue), CC10 (Club cells, green), and Actin (cell membranes, magenta) shows moderately increased MCC and Club cell numbers after DKK1 treatment. (B) DKK1 leads to a transient decrease in BCs but does not lead to loss of DeltaN-TP63+ (green) cells. Nuclei (DAPI, blue). (C) Quantification from (A) and (B), respectively. Mann Whitney test, not significant, ns (p > 0.05); ***p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7. Wnt/beta-Catenin-Induced Increase in BCs and Loss of Epithelial Differentiation Are Reversible (A and B) In Xenopus , BIO treatments from st. 8-17 or st. 8-30 inhibit differentiation as compared to DMSO treated controls, but the epithelium can regenerate after removal of BIO and recovery until st. 33 (A) or st. 25 (B). (A) BIO treatment reduces MCC (Ac.-alpha-tubulin, green), Ionocyte (no staining, black), and SSC (large vesicles, PNA staining, magenta) numbers in confocal micrographs at st. 28, which recover after regeneration until st. 33. Actin staining (green). (B) ISH shows reduction in foxj1 expressing cells and an increase in DeltaN-tp63 expression in BIO-treated whole mounts (upper row) and transversal sections (bottom row) at st. 17, which both recover after regeneration until st. 25. DMSO (n = 5); BIO st. 17 (n = 5); DMSO st. 25 (n = 5); BIO+recovery st. 25 (n = 5). (C) Confocal imaging of specimens stained for Ac.-alpha-tubulin (MCCs, blue), CC10 (Club cells, green), and Actin (cell membranes, magenta) show reduced MCC and Club cell numbers in RSPO2-treated cultures from ALI D7 to D21 but regeneration of MCCs and Club cells at ALI D28. n = 3 cultures per time point and treatment (upper panels). No loss of DeltaN-TP63+ (green) BCs is observed in these experiments (bottom panels). Nuclei (DAPI, blue). (D and E) Optical orthogonal sections of confocal images. RSPO2-treated cultures display normalized epithelial thickness and staining for BC markers DeltaN-TP
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot