Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-1299-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Myeloperoxidase (MPO) Monoclonal Antibody (MPO455-8E6), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody MPO455-8E6 recognizes myeloperoxidase (MPO), a protein within the azurophilic granules of myeloid cells. MPO is a multimeric protein comprised of two 55 kDa subunits and two 15 kDa subunits. The larger subunits associate with a heme protein resulting in a greenish color. As an enzyme, MPO breaks down hydrogen peroxide and oxidizes tyrosine. The products of this reaction, hypochlorous acid and tyrosyl radical, causethe cytotoxic and killing effects characteristic of neutrophils. Myeloperoxidase is important in the diagnosis of some cancers and increases in serum levels have been shown to correlate with cardiac events. Applications Reported: This MPO455-8E6 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This MPO455-8E6 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of normal human peripheral blood cells using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). For best results, whole blood should first be stained with antibodies to surface antigens then treated with 1X RBC lysis buffer (Product # 00-4333-57) to lyse erythrocytes. Then, for intracellular staining for MPO, cells should be fixed with IC Fixation Buffer (Product # 00-8222-49) washed two times with Permeabilization Buffer (Product # 00-8333-56) and then incubated with MPO455-8E6 for 30-60 minutes. After washing, cells may be analyzed on a flow cytometer. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 520 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- MPO455-8E6
- Vial size
- 25 Tests
- Concentration
- 5 μL/Test
- Storage
- 4°C, store in dark, DO NOT FREEZE!
Submitted references Generation of Human Neutrophils from Induced Pluripotent Stem Cells in Chemically Defined Conditions Using ETV2 Modified mRNA.
Loss of testosterone impairs anti-tumor neutrophil function.
The role of platelets in mediating a response to human influenza infection.
Targeting of Neutrophil Lewis X Blocks Transepithelial Migration and Increases Phagocytosis and Degranulation.
Majumder A, Suknuntha K, Bennin D, Klemm L, Brok-Volchanskaya VS, Huttenlocher A, Slukvin I
STAR protocols 2020 Sep 18;1(2)
STAR protocols 2020 Sep 18;1(2)
Loss of testosterone impairs anti-tumor neutrophil function.
Markman JL, Porritt RA, Wakita D, Lane ME, Martinon D, Noval Rivas M, Luu M, Posadas EM, Crother TR, Arditi M
Nature communications 2020 Mar 31;11(1):1613
Nature communications 2020 Mar 31;11(1):1613
The role of platelets in mediating a response to human influenza infection.
Koupenova M, Corkrey HA, Vitseva O, Manni G, Pang CJ, Clancy L, Yao C, Rade J, Levy D, Wang JP, Finberg RW, Kurt-Jones EA, Freedman JE
Nature communications 2019 Apr 16;10(1):1780
Nature communications 2019 Apr 16;10(1):1780
Targeting of Neutrophil Lewis X Blocks Transepithelial Migration and Increases Phagocytosis and Degranulation.
Brazil JC, Sumagin R, Cummings RD, Louis NA, Parkos CA
The American journal of pathology 2016 Feb;186(2):297-311
The American journal of pathology 2016 Feb;186(2):297-311
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Intracellular staining of normal human peripheral blood cells with Anti-Human CD3 APC (Product # 17-0036-42) and Mouse IgG1 K Isotype Control FITC (Product # 11-4714-42) (left) or Anti-Human Myeloperoxidase (MPO) FITC (right) using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Total cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
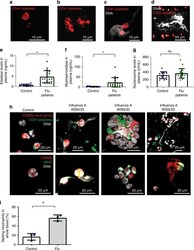
- Fig. 1 Characterization of human blood from influenza-infected patients. a - d Blood from influenza-infected patients was fixed after intravenous collection and stained as described in Methods. In all cases DNA was assessed by DAPI. a Many platelets appeared similar to those observed in control blood from healthy donors with a size of 2-5 um. b Some platelets from influenza-infected patients had undergone spreading with a distinctive distribution of CD41. c Platelets and platelet microparticles associated with DNA (arrow). d Spread platelets were found surrounding released DNA with distinct DNA content in their center. Of note, blood from influenza-infected patients and controls in ( a - d ) was not permeabilized. Since formaldehyde can cause certain levels of permeabilization as a function of cross-linking positive staining in control samples for H4 and MPO do not necessarily indicate activation. e - g Levels of proteins related to DNA release in the plasma of influenza-infected patients assessed by ELISA. Source data are provided as a Source Data file. e neutrophil elastase, f myeloperoxidase (MPO), and g histone nucleosome core. The graphs represent the average +- SD of healthy donors ( n = 15) and influenza-infected patients ( n = 18); significance for ( e - g ) was assessed by Mann-Whitney U test, star symbol (*) indicates p < 0.0001. h , i To synchronize time of influenza presence as a function of infection and quantify the released DNA, we
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
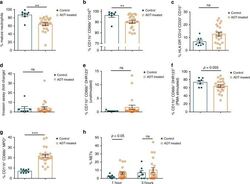
- Fig. 5 Analysis of neutrophils from control or androgen deprivation therapy (ADT) treated patients. a Percent of circulating neutrophils from control or ADT treated prostate cancer patients that have a mature, segmented appearance morphologically. b Percent Cd11b + CD66b + CD16 lo neutrophils. c Percent myeloid derived suppressor cell (MDSC)-like neutrophils defined as HLA-DR - CD14 - CD33 + CD15 + . d Percent fold change in migration (percent crystal violet positive area of transwell stimulated with a chemoattractant divided by percent area without chemoattractant). e Percent of unstimulated neutrophils producing DHR123. f Percent of neutrophils producing DHR123 following PMA stimulation. g Percent myeloperoxidase + (MPO + ) neutrophils. h Percent of plated netosising neutrophils at 1 and 3 h following purification. Data in a - h are mean +- s.e.m., * p < 0.05, ** p < 0.01, *** p < 0.001 by multivariable linear regression using ADT as its main predictor and adjusting for presence of prostatectomy, age, race, and current status; control prostate cancer patients ( n = 8 in a , c , g and n = 7 in b , d , e , f , h ) and prostate cancer pati e nts receiving ADT ( n = 22 in a , c , g and n = 21 in b , d , e , f , h ).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
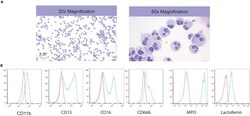
- Figure 6 Induction of Neutrophil Formation from Myeloid Progenitors (A) Representative images of Wright stained cytospins showing the morphology of neutrophils. (B) Flow cytometric analysis of CD11b, CD15, CD16, CD66b, MPO and lactoferrin expression in generated neutrophils.
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry