Antibody data
- Antibody Data
- Antigen structure
- References [26]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [8]
- Immunohistochemistry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-14516 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- p53 Monoclonal Antibody (SP5)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- MA5-14516 targets p53 in IHC (P), ICC/IF, and WB applications and shows reactivity with human samples. This antibody does not cross-react with mouse in immunofluorescent applications. The MA5-14516 immunogen is recombinant human full-length wild type p53 protein. This antibody was orginally validated as part of a Thermo Scientific Cellomics High Content Screening Kit. The antibody sold separately may have slightly different performance and may need to be further optimized for the best results.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- SP5
- Vial size
- 500 µL
- Concentration
- 0.004 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references AntiGan: An Epinutraceutical Bioproduct with Antitumor Properties in Cultured Cell Lines.
Circular RNA circLDB2 functions as a competing endogenous RNA to suppress development and promote cisplatin sensitivity in non-squamous non-small cell lung cancer.
PLA2G16 is a mutant p53/KLF5 transcriptional target and promotes glycolysis of pancreatic cancer.
Defective base excision repair in the response to DNA damaging agents in triple negative breast cancer.
MDM2 Derived from Dedifferentiated Liposarcoma Extracellular Vesicles Induces MMP2 Production from Preadipocytes.
Nucleolar aggresomes mediate release of pericentric heterochromatin and nuclear destruction of genotoxically treated cancer cells.
Simple Prediction Model of Axillary Lymph Node Positivity After Analyzing Molecular and Clinical Factors in Early Breast Cancer.
Preclinical Efficacy of the MDM2 Inhibitor RG7112 in MDM2-Amplified and TP53 Wild-type Glioblastomas.
Tissue biomarkers in prognostication of serous ovarian cancer following neoadjuvant chemotherapy.
Quantitative analysis of γ-H2AX and p53 nuclear expression levels in ovarian and fallopian tube epithelium from risk-reducing salpingo-oophorectomies in BRCA1 and BRCA2 mutation carriers.
Immunohistochemistry with apoptotic-antiapoptotic proteins (p53, p21, bax, bcl-2), c-kit, telomerase, and metallothionein as a diagnostic aid in benign, borderline, and malignant serous and mucinous ovarian tumors.
Genetic characterization of mesenchymal, clear cell, and dedifferentiated chondrosarcoma.
Proliferation and maturation of microvessels in arteriovenous malformations--expression patterns of angiogenic and cell cycle-dependent factors.
Evaluation of immunohistochemical expression of p53, p21, p27, cyclin D1, and Ki67 in oral and oropharyngeal squamous cell carcinoma.
The cooperating mutation or "second hit" determines the immunologic visibility toward MYC-induced murine lymphomas.
Clinical implication of p53 overexpression in breast cancer patients younger than 50 years with a triple-negative subtype who undergo a modified radical mastectomy.
Correlation of immunohistochemical expression of p53 with unamplified chromosome 17 polysomy in invasive breast carcinoma.
Extent and patterns of MGMT promoter methylation in glioblastoma- and respective glioblastoma-derived spheres.
Osteosarcoma arising in a long-standing uterine leiomyoma: a case report and literature review.
Recurrence of benign meningiomas: predictive value of proliferative index, BCL2, p53, hormonal receptors and HER2 expression.
Role of loss of o⁶-methylguanine dna methyltransferase (MGMT) expression in non-small cell lung carcinomas (NSCLCs): with reference to the relationship with p53 overexpression.
Immunohistochemical expression of SPARC is correlated with recurrence, survival and malignant potential in meningiomas.
The comparative effects of pneumoperitoneum on apoptosis and p53 expression in gastrointestinal organs.
Expression of senescence-related genes in human corneal endothelial cells.
Prognostic significance of cell proliferation and apoptosis-regulating proteins in Epstein-Barr virus positive and negative pediatric Hodgkin lymphoma.
Inhibition of p38alpha MAPK enhances proteasome inhibitor-induced apoptosis of myeloma cells by modulating Hsp27, Bcl-X(L), Mcl-1 and p53 levels in vitro and inhibits tumor growth in vivo.
Martínez-Iglesias O, Carrera I, Naidoo V, Cacabelos R
Life (Basel, Switzerland) 2022 Jan 10;12(1)
Life (Basel, Switzerland) 2022 Jan 10;12(1)
Circular RNA circLDB2 functions as a competing endogenous RNA to suppress development and promote cisplatin sensitivity in non-squamous non-small cell lung cancer.
Wang Y, Li L, Zhang W, Zhang G
Thoracic cancer 2021 Jul;12(13):1959-1972
Thoracic cancer 2021 Jul;12(13):1959-1972
PLA2G16 is a mutant p53/KLF5 transcriptional target and promotes glycolysis of pancreatic cancer.
Xia W, Bai H, Deng Y, Yang Y
Journal of cellular and molecular medicine 2020 Nov;24(21):12642-12655
Journal of cellular and molecular medicine 2020 Nov;24(21):12642-12655
Defective base excision repair in the response to DNA damaging agents in triple negative breast cancer.
Lee KJ, Piett CG, Andrews JF, Mann E, Nagel ZD, Gassman NR
PloS one 2019;14(10):e0223725
PloS one 2019;14(10):e0223725
MDM2 Derived from Dedifferentiated Liposarcoma Extracellular Vesicles Induces MMP2 Production from Preadipocytes.
Casadei L, Calore F, Braggio DA, Zewdu A, Deshmukh AA, Fadda P, Lopez G, Wabitsch M, Song C, Leight JL, Grignol VP, Lev D, Croce CM, Pollock RE
Cancer research 2019 Oct 1;79(19):4911-4922
Cancer research 2019 Oct 1;79(19):4911-4922
Nucleolar aggresomes mediate release of pericentric heterochromatin and nuclear destruction of genotoxically treated cancer cells.
Salmina K, Huna A, Inashkina I, Belyayev A, Krigerts J, Pastova L, Vazquez-Martin A, Erenpreisa J
Nucleus (Austin, Tex.) 2017 Mar 4;8(2):205-221
Nucleus (Austin, Tex.) 2017 Mar 4;8(2):205-221
Simple Prediction Model of Axillary Lymph Node Positivity After Analyzing Molecular and Clinical Factors in Early Breast Cancer.
Chung MJ, Lee JH, Kim SH, Suh YJ, Choi HJ
Medicine 2016 May;95(20):e3689
Medicine 2016 May;95(20):e3689
Preclinical Efficacy of the MDM2 Inhibitor RG7112 in MDM2-Amplified and TP53 Wild-type Glioblastomas.
Verreault M, Schmitt C, Goldwirt L, Pelton K, Haidar S, Levasseur C, Guehennec J, Knoff D, Labussière M, Marie Y, Ligon AH, Mokhtari K, Hoang-Xuan K, Sanson M, Alexander BM, Wen PY, Delattre JY, Ligon KL, Idbaih A
Clinical cancer research : an official journal of the American Association for Cancer Research 2016 Mar 1;22(5):1185-96
Clinical cancer research : an official journal of the American Association for Cancer Research 2016 Mar 1;22(5):1185-96
Tissue biomarkers in prognostication of serous ovarian cancer following neoadjuvant chemotherapy.
Khandakar B, Mathur SR, Kumar L, Kumar S, Datta Gupta S, Iyer VK, Kalaivani M
BioMed research international 2014;2014:401245
BioMed research international 2014;2014:401245
Quantitative analysis of γ-H2AX and p53 nuclear expression levels in ovarian and fallopian tube epithelium from risk-reducing salpingo-oophorectomies in BRCA1 and BRCA2 mutation carriers.
Staff S, Tolonen T, Laasanen SL, Mecklin JP, Isola J, Mäenpää J
International journal of gynecological pathology : official journal of the International Society of Gynecological Pathologists 2014 May;33(3):309-16
International journal of gynecological pathology : official journal of the International Society of Gynecological Pathologists 2014 May;33(3):309-16
Immunohistochemistry with apoptotic-antiapoptotic proteins (p53, p21, bax, bcl-2), c-kit, telomerase, and metallothionein as a diagnostic aid in benign, borderline, and malignant serous and mucinous ovarian tumors.
Ozer H, Yenicesu G, Arici S, Cetin M, Tuncer E, Cetin A
Diagnostic pathology 2012 Sep 20;7:124
Diagnostic pathology 2012 Sep 20;7:124
Genetic characterization of mesenchymal, clear cell, and dedifferentiated chondrosarcoma.
Meijer D, de Jong D, Pansuriya TC, van den Akker BE, Picci P, Szuhai K, Bovée JV
Genes, chromosomes & cancer 2012 Oct;51(10):899-909
Genes, chromosomes & cancer 2012 Oct;51(10):899-909
Proliferation and maturation of microvessels in arteriovenous malformations--expression patterns of angiogenic and cell cycle-dependent factors.
Meijer-Jorna LB, van der Loos CM, Teeling P, de Boer OJ, Florquin S, van der Horst CM, van der Wal AC
Journal of cutaneous pathology 2012 Jun;39(6):610-20
Journal of cutaneous pathology 2012 Jun;39(6):610-20
Evaluation of immunohistochemical expression of p53, p21, p27, cyclin D1, and Ki67 in oral and oropharyngeal squamous cell carcinoma.
Perisanidis C, Perisanidis B, Wrba F, Brandstetter A, El Gazzar S, Papadogeorgakis N, Seemann R, Ewers R, Kyzas PA, Filipits M
Journal of oral pathology & medicine : official publication of the International Association of Oral Pathologists and the American Academy of Oral Pathology 2012 Jan;41(1):40-6
Journal of oral pathology & medicine : official publication of the International Association of Oral Pathologists and the American Academy of Oral Pathology 2012 Jan;41(1):40-6
The cooperating mutation or "second hit" determines the immunologic visibility toward MYC-induced murine lymphomas.
Schuster C, Berger A, Hoelzl MA, Putz EM, Frenzel A, Simma O, Moritz N, Hoelbl A, Kovacic B, Freissmuth M, Müller M, Villunger A, Müllauer L, Schmatz AI, Streubel B, Porpaczy E, Jäger U, Stoiber D, Sexl V
Blood 2011 Oct 27;118(17):4635-45
Blood 2011 Oct 27;118(17):4635-45
Clinical implication of p53 overexpression in breast cancer patients younger than 50 years with a triple-negative subtype who undergo a modified radical mastectomy.
Lee DS, Kim SH, Suh YJ, Kim S, Kim HK, Shim BY
Japanese journal of clinical oncology 2011 Jul;41(7):854-66
Japanese journal of clinical oncology 2011 Jul;41(7):854-66
Correlation of immunohistochemical expression of p53 with unamplified chromosome 17 polysomy in invasive breast carcinoma.
Krishnamurti U, Zarineh A, Atem FD, Silverman JF
Applied immunohistochemistry & molecular morphology : AIMM 2011 Jan;19(1):28-32
Applied immunohistochemistry & molecular morphology : AIMM 2011 Jan;19(1):28-32
Extent and patterns of MGMT promoter methylation in glioblastoma- and respective glioblastoma-derived spheres.
Sciuscio D, Diserens AC, van Dommelen K, Martinet D, Jones G, Janzer RC, Pollo C, Hamou MF, Kaina B, Stupp R, Levivier M, Hegi ME
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Jan 15;17(2):255-66
Clinical cancer research : an official journal of the American Association for Cancer Research 2011 Jan 15;17(2):255-66
Osteosarcoma arising in a long-standing uterine leiomyoma: a case report and literature review.
Wang RC, Wen MC, Wang J, Ho SC, Jan YJ
International journal of surgical pathology 2011 Feb;19(1):99-103
International journal of surgical pathology 2011 Feb;19(1):99-103
Recurrence of benign meningiomas: predictive value of proliferative index, BCL2, p53, hormonal receptors and HER2 expression.
Abdelzaher E, El-Gendi SM, Yehya A, Gowil AG
British journal of neurosurgery 2011 Dec;25(6):707-13
British journal of neurosurgery 2011 Dec;25(6):707-13
Role of loss of o⁶-methylguanine dna methyltransferase (MGMT) expression in non-small cell lung carcinomas (NSCLCs): with reference to the relationship with p53 overexpression.
Myong NH
Cancer research and treatment 2010 Jun;42(2):95-100
Cancer research and treatment 2010 Jun;42(2):95-100
Immunohistochemical expression of SPARC is correlated with recurrence, survival and malignant potential in meningiomas.
Bozkurt SU, Ayan E, Bolukbasi F, Elmaci I, Pamir N, Sav A
APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 2009 Sep;117(9):651-9
APMIS : acta pathologica, microbiologica, et immunologica Scandinavica 2009 Sep;117(9):651-9
The comparative effects of pneumoperitoneum on apoptosis and p53 expression in gastrointestinal organs.
Arikan Y, Tosun M, Yilmaz S, Saykol V, Söylemez Z
Journal of laparoendoscopic & advanced surgical techniques. Part A 2008 Jun;18(3):365-71
Journal of laparoendoscopic & advanced surgical techniques. Part A 2008 Jun;18(3):365-71
Expression of senescence-related genes in human corneal endothelial cells.
Song Z, Wang Y, Xie L, Zang X, Yin H
Molecular vision 2008 Jan 29;14:161-70
Molecular vision 2008 Jan 29;14:161-70
Prognostic significance of cell proliferation and apoptosis-regulating proteins in Epstein-Barr virus positive and negative pediatric Hodgkin lymphoma.
Aktaş S, Kargi A, Olgun N, Diniz G, Erbay A, Vergin C
Lymphatic research and biology 2007;5(3):175-82
Lymphatic research and biology 2007;5(3):175-82
Inhibition of p38alpha MAPK enhances proteasome inhibitor-induced apoptosis of myeloma cells by modulating Hsp27, Bcl-X(L), Mcl-1 and p53 levels in vitro and inhibits tumor growth in vivo.
Navas TA, Nguyen AN, Hideshima T, Reddy M, Ma JY, Haghnazari E, Henson M, Stebbins EG, Kerr I, O'Young G, Kapoun AM, Chakravarty S, Mavunkel B, Perumattam J, Luedtke G, Dugar S, Medicherla S, Protter AA, Schreiner GF, Anderson KC, Higgins LS
Leukemia 2006 Jun;20(6):1017-27
Leukemia 2006 Jun;20(6):1017-27
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
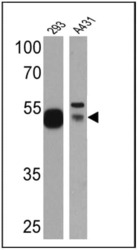
- Experimental details
- Western blot analysis of p53 was performed by loading 25 µg of HEK293 (Lane 1) and A431 (Lane 2)cell lysates onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a p53 monoclonal antibody (Product # MA5-14516) at a dilution of 1:500 (HEK293) and 1:20 (A431) overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at approx. 53 kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
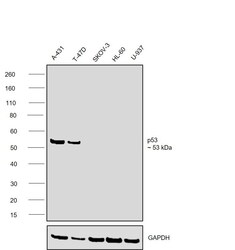
- Experimental details
- Western blot was performed using Anti-p53 Monoclonal Antibody (SP5) (Product # MA5-14516) and a ~53 kDa band corresponding to TP53 was observed across cell lines tested . Whole cell extracts (30 µg lysate) of A-431 (Lane 1), T-47D (Lane 2), SK-O-V3 (Lane 3), HL-60 (Lane 4), U-937 (Lane 5) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:500 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:20000) using the iBright™ FL1500 Imaging System (Product # A44115). Chemiluminescent detection was performed using SuperSignal™ West Atto Ultimate Sensitivity Substrate (Product # A38556).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence staining of p21 and p53 in A549 cells treated with media only (non-treated) or with 5 µM camptothecin for 20 hours.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence staining of p21 and p53 in A549 cells treated with media only (non-treated) or with 5 µM camptothecin for 20 hours.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of p53 (green) showing staining in the nucleus of HT29 cells. Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a p53 monoclonal antibody (Product # MA5-14516) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence staining of p21 and p53 in A549 cells treated with media only (non-treated) or with 5 µM camptothecin for 20 hours.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence staining of p21 and p53 in A549 cells treated with media only (non-treated) or with 5 µM camptothecin for 20 hours.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of p53 (green) showing staining in the nucleus of A431 cells. Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a p53 monoclonal antibody (Product # MA5-14516) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of TP53 was performed using 70% confluent log phase T-47D cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with p53 Monoclonal Antibody (SP5) (Product # MA5-14516) at 1:100 in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing nuclear localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 40X. magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of p53 (green) in HeLa cells either left untreated (left panel) or treated with 5uM Camptothecin (right panel) for 20 hours. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a p53 monoclonal antibody (Product # MA5-14516) at a dilution of 1:50 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:400 for 30 minutes at room temperature. F-Actin (red) was stained with DyLight 554 Phalloidin (Product # 21834) and nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan or ToxInsight Instrument at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
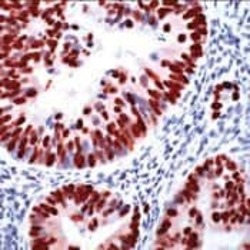
- Experimental details
- Immunohistochemical analysis of p53 using a monoclonal antibody (Product # MA5-14516).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
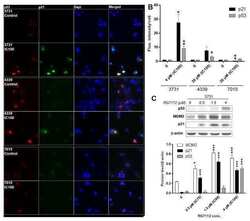
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 CircLDB2 regulated non-squamous NSCLC development and cisplatin sensitivity in vitro by miR-346. Vector-transduced or circLDB2-infected A549 and H460 cells were transfected with miR-NC mimic or miR-346 mimic. (a) and (b) CCK-8 assay of cell proliferation. (c) Representative images depicting a colony formation assay. (d) Representative images showing a cell apoptosis assay and flow cytometry for cell apoptosis. (e) Wound-healing assay of cell migration. (f) Cell invasion by transwell assay. (g) and (h) transfected cells were exposed to various concentrations of cisplatin, followed by the evaluation of cell viability by CCK-8 assay. (i) and (j) Western blot showing the levels of p53 and PUMA in transfected cells. * p < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 7 LIMCH1 was a functional target of miR-346. (a) and (b) Western blot analysis of LIMCH1 protein level in cells transfected with sh-NC or sh-LIMCH1. A549 and H460 cells were transfected with anti-NC + sh-NC, anti-miR-346 + sh-NC or anti-miR-346 + sh-LIMCH1. (c) and (d) CCK-8 assay of proliferation of transfected cells. (e) Representative images depicting a colony formation assay. (f) Representative images depicting a cell apoptosis assay and cell apoptosis by flow cytometry. (g) Wound-healing assay of cell migration. (h) Transwell assay of cell invasion. (i) and (j) CCK-8 assay for viability of transfected cells under cisplatin exposure. (k) and (l) Western blot showing the levels of p53 and PUMA in transfected cells. * p < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 8 Overexpression of circLDB2 promoted the antitumor effect of cisplatin in vivo. The xenograft tumors were generated by subcutaneous injection of vector-transduced or circLDB2-transduced A549 cells (2 x 10 6 per mouse, n = 5 per group), and cisplatin treatment was performed twice a week by intraperitoneal injection. Mice were terminated at day 28 after cell implantation. (a) Representative images of the xenograft tumors. (b) Growth curves of the excised tumors. (c) Tumor average weight of the xenograft tumors. (d) Western blot showing the levels of LIMCH1, p53, and PUMA in the excised tumors. * p < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 3 CircLDB2 overexpression regulated cell proliferation, apoptosis, migration, invasion, and cisplatin sensitivity in vitro. A549 and H460 cells were transduced with negative control lentivirus (vector) or circLDB2 overexpression lentivirus (circLDB2). (a) and (b) CCK-8 assay of proliferation of transduced cells. (c) Representative images depicting a colony formation assay and colony formation assay of colony formation ability. (d) Representative images depicting a cell apoptosis assay and cell apoptosis by flow cytometry. (e) Representative images showing a cell migration assay and cell migration by wound-healing assay. (f) Representative images showing a cell invasion assay and cell invasion by transwell assay. (g) and (h) CCK-8 assay of viability of transduced cells under cisplatin (various doses) exposure. (i) and (j) representative images depicting a western blot assay and western blot showing the levels of p53 and PUMA in transduced cells. * p < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
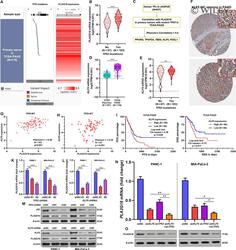
- 4 FIGURE Both mutant p53 and KLF5 increase PLA2G16 expression in pancreatic cancer. A, A heatmap showing TP53 mutation and PLA2G16 expression in pancreatic adenocarcinoma (PAAD) cases in The Cancer Genome Atlas (TCGA). B, A violin plot chart comparing PLA2G16 expression between PAAD cases with or without TP53 mutations. C, Flow chart showing the screening process to identify PLA2G16 correlated TFs in TP53 mutant PAAD cases. D and E, Comparison of KLF5 mRNA expression between normal pancreas in GTEx and PAAD in TCGA (D) and between PAAD cases with or without TP53 mutations (E). F, Representative images of KLF5 protein expression in PAAD tissue. Image credit: Human Protein Atlas, from: https://www.proteinatlas.org/ENSG00000102554-KLF5/pathology/pancreatic+cancer . G and H, Plot charts showing the correlation between PLA2G16 and KLF5 mRNA expression in TP53 mutant (G) and TP53 wild-type (H) PAAD cases. I and J, K-M survival analysis of PFS (I) and DSS (J) in PAAD cases in TCGA Pan-Cancer, by median KLF5 expression separation. K and L, Quantitative real-time PCR (qRT-PCR) analysis of PLA2G16 mRNA expression in PANC-1 and MIA-PaCa-2 cells 48 h after lentiviral-mediated TP53 (K) or KLF5 (L) inhibition. M, Western blot analysis of p53, KLF5 and PLA2G16 protein expression 72 h after lentiviral-mediated TP53 (up) or KLF5 (down) inhibition. N and O, qRT-PCR (M) and Western blot assay (O) of PLA2G16 expression in PANC-1 and MIA-PaCa-2 cells with TP53 and KLF5 inhibition (sh TP53 #2 and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 5 FIGURE Mutant p53 enhances KLF5-induced PLA2G16 transcriptional activation. A, Predicted binding sites of KLF5 in the promoter region of PLA2G16 . B, The promoter activity of the PLA2G16 gene was measured using a dual-luciferase reporter assay. PANC-1 and MIA-PaCa-2 cells were transfected with pGL3-basic or reporter constructs carrying different lengths of the 5'-flanking region of the PLA2G16 promoter as indicated. C-E, KLF5 depletion reduced the activity of the PLA2G16 promoter. PANC-1 and MIA-PaCa-2 cells with or without lentiviral-mediated KLF5 inhibition were transfected reporter constructs carrying pGL3-(-1303/+239) (C), pGL3-(-400/+239) (D) and pGL3-(-100/+239) (E). 24 h later, luciferase activity was determined. F, Western blot assay of KLF5 expression 48 h after lentiviral-mediated overexpression. G-J, 24 h after infection with lenti-KLF5 or vector, PANC-1 and MIA-PaCa-2 cells were transfected reporter constructs carrying pGL3-(-1303/+239) (G-H) or mutant-pGL3-(-1303/+239) with mutant sequences (C to A) of the four binding sites (I, J). 24 h later, luciferase activity was determined. K, Schematic image showing the location of the designed primer sets for ChIP-quantitative polymerase chain reaction (qPCR) assay, by anti-KLF5 immunoprecipitation. L-O, ChIP-qPCR assays were performed using anti-KLF5 (L, M) or anti-TP53 (N, O) and control IgG antibodies in PANC-1 (L, N) and MIA-PaCa-2 (M, O) cells. Fold enrichment of the indicated PLA2G16 promoter segments was calculat
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 4 Nuclear intensity of key BER proteins. A) Quantification of mean nuclear fluorescence intensity is shown for PARP1 (**** P < 0.0001, MDA-231 to MDA-157, HCC1806, and MDA-468), PAR (**** P < 0.0001, MDA-468 to MDA-157, MDA-231, and HCC1806), and p53 (**** P < 0.0001, MDA-157 to MDA-231, MDA-157 to MDA-468, MDA-157 to HCC1806, MDA-231 to MDA-468, MDA-231 to HCC1806, and MDA-468 to HCC1806). B) Representative fluorescent images used for quantification in A for PARP1, PAR, and p53 for all cell lines tested. Scale bar = 20 mum.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry