MA5-13917
antibody from Invitrogen Antibodies
Targeting: EPCAM
17-1A, 323/A3, CD326, CO-17A, EGP-2, EGP34, EGP40, Ep-CAM, ESA, GA733-2, HEA125, KS1/4, KSA, Ly74, M4S1, MH99, MIC18, MK-1, MOC31, TACST-1, TACSTD1, TROP1
Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [3]
- Immunohistochemistry [2]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-13917 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- EpCAM Monoclonal Antibody (AUA1)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA5-13917 targets Epithelial Specific Antigen in IHC (P), ICC/IF, FACS and WB applications and shows reactivity with mouse and human samples.
- Antibody clone number
- AUA1
- Concentration
- 0.2 mg/mL
Submitted references Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Immune Suppressive Effects of Plasma-Derived Exosome Populations in Head and Neck Cancer.
Constitutive expression of RbAp46 induces epithelial-mesenchymal transition in mammary epithelial cells.
Schroeder JC, Puntigam L, Hofmann L, Jeske SS, Beccard IJ, Doescher J, Laban S, Hoffmann TK, Brunner C, Theodoraki MN, Schuler PJ
Cancers 2020 Jul 29;12(8)
Cancers 2020 Jul 29;12(8)
Immune Suppressive Effects of Plasma-Derived Exosome Populations in Head and Neck Cancer.
Beccard IJ, Hofmann L, Schroeder JC, Ludwig S, Laban S, Brunner C, Lotfi R, Hoffmann TK, Jackson EK, Schuler PJ, Theodoraki MN
Cancers 2020 Jul 21;12(7)
Cancers 2020 Jul 21;12(7)
Constitutive expression of RbAp46 induces epithelial-mesenchymal transition in mammary epithelial cells.
Li GC, Wang ZY
Anticancer research 2006 Sep-Oct;26(5A):3555-60
Anticancer research 2006 Sep-Oct;26(5A):3555-60
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of EpCAM was performed by loading 25 µg of HT29 (lane 1), MCF-7 (lane 2), A431 (lane 3) and NIH-3T3 (lane 4) onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with an EpCAM monoclonal antibody (Product # MA5-13917) at a dilution of 1:200 (HT29 and MCF-7) and 1:50 (A431 and NIH-3T3) overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~40 kDa in human cell lines, and no reactivity with mouse.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of EpCAM (green) showing staining in the membrane of A431 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with an EpCAM monoclonal antibody (Product # MA5-13917) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Nuclei were stained with Hoechst or DAPI (blue). Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of EpCAM (green) showing staining in the membrane of HT-29 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with an EpCAM monoclonal antibody (Product # MA5-13917) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Nuclei were stained with Hoechst or DAPI (blue). Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of EpCAM was performed using HT-29 and BJ cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with EpCAM Mouse Monoclonal Antibody (Product # MA5-13917) at 1:20 dilution in 0.1% BSA and incubated overnight at 4 degree and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green) in HT-29 cells. Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image of HT-29 cells, which is a positive model for EpCAM expression showing a plasma membrane localization. Panel e represents the merged image of BJ cells, that are null for EpCAM protein expression. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of CD326 (EpCAM) was performed using formalin-fixed paraffin-embedded human colon adenocarcinoma tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 3% H2O2 for 1 h at room temperature followed by 2% normal goat serum in 1X PBS for 45 minutes at room temperature. The sections were then probed with or without EpCAM Monoclonal Antibody (AUA1) (Product # MA5-13917) at a concentration of 0.4 µg/mL in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Alexa Fluor™ 647 Tyramide SuperBoost™ Kit, goat anti-mouse IgG (Product # B40916). Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification and externally deconvoluted.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Formalin-fixed, paraffin-embedded human breast carcinoma stained with ESA antibody using peroxidase-conjugate and DAB chromogen. Note cell membrane staining of tumor cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
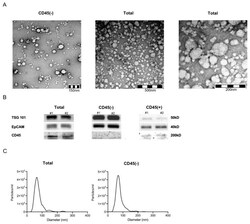
- Experimental details
- Figure 1 Exosome characterization. ( A ) Representative transmission electron microscopy pictures of CD45(-) (left) and total (middle and right) exosomes. ( B ) Western blot for detection of TSG 101, EpCAM and CD45 of total, CD45(-) and (+) exosomes from plasma of head and neck squamous cell carcinoma (HNSCC) patients (#1&2 high stage). Note that no CD45 signal was detected when using CD45(-) exosomes. Uncropped Western blots can be found at Figure S1 . ( C ) Nanoparticle tracking analysis with ZetaView of total (left) and CD45(-) exosomes (right). Vesicles between 30 and 200 nm diameter were detected.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
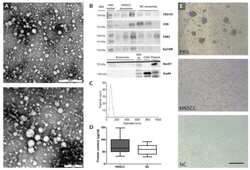
- Experimental details
- Figure 2 Successful isolation of exosomes from blood plasma was verified by Transmission Electron Microscopy (TEM), Western Blot, and Nanoparticle tracking. ( A ) Two representative TEM graphs showing negatively stained exosomes isolated from an HNSCC patient. As indicated by the size bars, exosomes vary in diameter between 30 and 150 nm and have round to oval shapes. Size bar on the top TEM graph = 500 nm, size bar on the bottom TEM graph = 200 nm. ( B ) Western Blot analysis of exosomes was performed to confirm the expression of exosomal markers TSG101, CD9 and CD63 and the expression of epithelial cell marker EpCAM (upper frame). Exosomes were also analyzed for negative markers ApoA1 and Grp94 along with plasma (diluted 50x in PBS) and cell lysate samples as positive controls. MW marker, positive control molecular weight marker. ( C ) Size distribution of exosomes was measured by nanoparticle tracking. The mean diameter was 86.8 nm. The maximal and minimal diameters were 257.5 nm and 22.5 nm, respectively. The 90th and 10th percentile were at 121.2 and 53.6 nm, respectively. ( D ) Protein content of exosomes was determined by Bicinchoninic Acid (BCA) Assay. Average protein content: 80.9 g/mL (HNSCC exosomes), 69.2 g/mL (healthy volunteer exosomes). n = 23 (HNSCC), n = 10 (NC). HNSCC, exosomes from blood plasma of HNSCC patients. NC = no cancer, exosomes from blood plasma of healthy volunteers. ( E ) B cells that were not co-cultured with exosomes exhibited colony formation
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot