Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [2]
- Immunohistochemistry [3]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-950 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PARP1 Monoclonal Antibody (C-2-10)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- This antibody does not detect PARP from chicken tissues. By Western blot, this antibody recognizes a 116 kDa protein which corresponds to PARP and the 85 kDa apoptosis-induced cleavage product of prICE and CPP32 from rat thymus extract. This antibody recognizes an epitope found between amino acids 216-375 in the DNA-binding domain of PARP.
- Reactivity
- Human, Mouse, Rat, Bovine, Hamster
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- C-2-10
- Vial size
- 50 µL
- Concentration
- Conc. Not Determined
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references LRP1 Regulates Retinal Angiogenesis by Inhibiting PARP-1 Activity and Endothelial Cell Proliferation.
New insights into PARP inhibitors' effect on cell cycle and homology-directed DNA damage repair.
Identification of TSG101 functional domains and p21 loci required for TSG101-mediated p21 gene regulation.
Decreased parkin solubility is associated with impairment of autophagy in the nigrostriatum of sporadic Parkinson's disease.
ADP ribosylation by PARP-1 suppresses HOXB7 transcriptional activity.
Role of human transcription elongation factor DSIF in the suppression of senescence and apoptosis.
Cellular dynamics of the negative transcription elongation factor NELF.
Apoptosis-inducing factor is a major contributor to neuronal loss induced by neonatal cerebral hypoxia-ischemia.
Mechanisms of oncogenic KIT signal transduction in primary gastrointestinal stromal tumors (GISTs).
Mao H, Lockyer P, Townley-Tilson WH, Xie L, Pi X
Arteriosclerosis, thrombosis, and vascular biology 2016 Feb;36(2):350-60
Arteriosclerosis, thrombosis, and vascular biology 2016 Feb;36(2):350-60
New insights into PARP inhibitors' effect on cell cycle and homology-directed DNA damage repair.
Jelinic P, Levine DA
Molecular cancer therapeutics 2014 Jun;13(6):1645-54
Molecular cancer therapeutics 2014 Jun;13(6):1645-54
Identification of TSG101 functional domains and p21 loci required for TSG101-mediated p21 gene regulation.
Lin YS, Chen YJ, Cohen SN, Cheng TH
PloS one 2013;8(11):e79674
PloS one 2013;8(11):e79674
Decreased parkin solubility is associated with impairment of autophagy in the nigrostriatum of sporadic Parkinson's disease.
Lonskaya I, Hebron ML, Algarzae NK, Desforges N, Moussa CE
Neuroscience 2013 Mar 1;232:90-105
Neuroscience 2013 Mar 1;232:90-105
ADP ribosylation by PARP-1 suppresses HOXB7 transcriptional activity.
Wu X, Ellmann S, Rubin E, Gil M, Jin K, Han L, Chen H, Kwon EM, Guo J, Ha HC, Sukumar S
PloS one 2012;7(7):e40644
PloS one 2012;7(7):e40644
Role of human transcription elongation factor DSIF in the suppression of senescence and apoptosis.
Komori T, Inukai N, Yamada T, Yamaguchi Y, Handa H
Genes to cells : devoted to molecular & cellular mechanisms 2009 Mar;14(3):343-54
Genes to cells : devoted to molecular & cellular mechanisms 2009 Mar;14(3):343-54
Cellular dynamics of the negative transcription elongation factor NELF.
Yung TM, Narita T, Komori T, Yamaguchi Y, Handa H
Experimental cell research 2009 Jun 10;315(10):1693-705
Experimental cell research 2009 Jun 10;315(10):1693-705
Apoptosis-inducing factor is a major contributor to neuronal loss induced by neonatal cerebral hypoxia-ischemia.
Zhu C, Wang X, Huang Z, Qiu L, Xu F, Vahsen N, Nilsson M, Eriksson PS, Hagberg H, Culmsee C, Plesnila N, Kroemer G, Blomgren K
Cell death and differentiation 2007 Apr;14(4):775-84
Cell death and differentiation 2007 Apr;14(4):775-84
Mechanisms of oncogenic KIT signal transduction in primary gastrointestinal stromal tumors (GISTs).
Duensing A, Medeiros F, McConarty B, Joseph NE, Panigrahy D, Singer S, Fletcher CD, Demetri GD, Fletcher JA
Oncogene 2004 May 13;23(22):3999-4006
Oncogene 2004 May 13;23(22):3999-4006
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
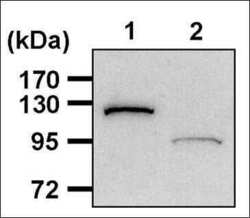
- Experimental details
- Western blot analysis of PARP was performed by loading 20 µg of THP-1 cell lysates, from cells either left untreated (Lane 1) or treated with 1µM Staurosporine to induce apoptosis (Lane 2) for 5 hours, per well onto an SDS-PAGE gel. Proteins were transferred to a PVDF membrane and blocked with 5% non-fat milk in TBST for 1 hour at room temperature. The membrane was probed with a PARP monoclonal antibody (Product # MA3-950) at a dilution of 1:2000 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG secondary antibody at a dilution of 1:40,000 for 1 hour at room temperature. Chemiluminescent detection was performed using ECL substrate. Data courtesy of the Innovators Program.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on modified whole cell extracts (1%SDS) (30ug lysate) of HeLa (Lane 1), HeLa treated with Staurosporine (1uM for 12 hours) (Lane 2), Jurkat (Lane 3), Jurkat treated with Staurosporine (1 uM for 12 hours) (Lane 4), MCF7 (Lane 5), A549 (Lane 6) and SH-SY5Y (Lane 7). The blot was probed with Anti-PARP Monoclonal Antibody (C-2-10) (Product # MA3-950, 1:500 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25 µg/ml, 1:4000 dilution). A 113 kDa band corresponding to full length PARP and 86 kDa band corresponding to cleaved PARP was observed upon cell treatment in HeLa, Jurkat and was also observed in total cell lines like MCF7, A549 and SH-SY5Y.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CRISPR-Cas9 mediated genome editing ofPARP1 (as confirmed by next generation sequencing) was achieved by using LentiArray™ Lentiviral sgRNA (Product # A32042, Assay ID CRISPR978664_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of PARP1 was performed by loading 30 µg of HeLa Wild Type (Lane 1), HeLa Wild Type treated with Staurosporin (1 µM for 16 hrs) (Lane 2), HeLa Cas9 (Lane 3), HeLa Cas9 treated with Staurosporin (1 µM for 16 hrs) (Lane 4), HeLa Cas9 cells transduced with PARP1 Lentiviral sgRNA (Lane 5) and HeLa Cas9 cells transduced with PARP1 Lentiviral sgRNA treated with Staurosporin (1 µM for 16 hrs) (Lane 6) whole cell extracts. The samples were electrophoresed using NuPAGE™ Novex™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with Anti-PARP1 Monoclonal Antibody (C-2-10) (Product # MA3-950) using 1:500 dilution and Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177 1:4,000 dilution). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Even though NGS analysis determine the clone as partial KO, there was complete loss of signal in sgRNA transduced cells using the LentiArray™ CRISPR product line confirming
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
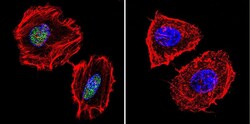
- Experimental details
- Immunofluorescent analysis of PARP in HeLa cells. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with a PARP monoclonal antibody (Product # MA3-950) at a dilution of 1:200 overnight at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503). PARP staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of PARP in HT-29 cells. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with a PARP monoclonal antibody (Product # MA3-950) at a dilution of 1:200 overnight at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503). PARP staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Images were taken at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
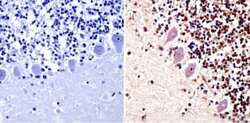
- Experimental details
- Immunohistochemistry analysis of PARP showing staining in the cytoplasm and nucleus of paraffin-embedded human cerebellum tissue (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a PARP monoclonal antibody (Product # MA3-950) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
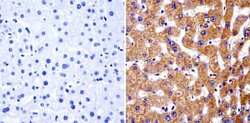
- Experimental details
- Immunohistochemistry analysis of PARP showing staining in the cytoplasm of paraffin-embedded human liver tissue (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a PARP monoclonal antibody (Product # MA3-950) diluted in 3% BSA-PBS at a dilution of 1:50 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
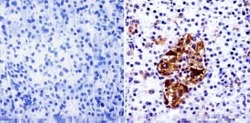
- Experimental details
- Immunohistochemistry analysis of PARP showing staining in the cytoplasm and nucleus of paraffin-embedded human pancreas tissue (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a PARP monoclonal antibody (Product # MA3-950) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA