Antibody data
- Antibody Data
- Antigen structure
- References [76]
- Comments [0]
- Validations
- Western blot [3]
- Other assay [38]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-722G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Paxillin (Tyr118) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- The antibody has been negatively preadsorbed using i) a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated paxillin, and ii) a generic tyrosine phosphorylated peptide to remove antibody that is reactive with phosphotyrosine, irrespective of the sequence. The final product is generated by affinity chromatography using a paxillin-derived peptide that is phosphorylated at tyrosine 118.
Submitted references Nephronectin-integrin α8 signaling is required for proper migration of periocular neural crest cells during chick corneal development.
Matrix stiffness modulates tip cell formation through the p-PXN-Rac1-YAP signaling axis.
Endothelial Focal Adhesions Are Functional Obstacles for Leukocytes During Basolateral Crawling.
Cholecystokinin Receptor Antagonist Improves Efficacy of Chemotherapy in Murine Models of Pancreatic Cancer by Altering the Tumor Microenvironment.
Coordination of mitochondrial and cellular dynamics by the actin-based motor Myo19.
Integrin-linked kinase tunes cell-cell and cell-matrix adhesions to regulate the switch between apoptosis and EMT downstream of TGFβ1.
Identification of lymphocyte cell-specific protein-tyrosine kinase (LCK) as a driver for invasion and migration of oral cancer by tumor heterogeneity exploitation.
The proprotein convertase furin inhibits IL-13-induced inflammation in airway smooth muscle by regulating integrin-associated signaling complexes.
EVL regulates VEGF receptor-2 internalization and signaling in developmental angiogenesis.
Inhibiting FAK-Paxillin Interaction Reduces Migration and Invadopodia-Mediated Matrix Degradation in Metastatic Melanoma Cells.
Light-regulated allosteric switch enables temporal and subcellular control of enzyme activity.
Endogenous Cyclin D1 Promotes the Rate of Onset and Magnitude of Mitogenic Signaling via Akt1 Ser473 Phosphorylation.
S100A4 is activated by RhoA and catalyses the polymerization of non-muscle myosin, adhesion complex assembly and contraction in airway smooth muscle.
Phenotype transitions induced by mechanical stimuli in airway smooth muscle are regulated by differential interactions of parvin isoforms with paxillin and Akt.
DLC1 is a direct target of activated YAP/TAZ that drives collective migration and sprouting angiogenesis.
Common and Unique Transcription Signatures of YAP and TAZ in Gastric Cancer Cells.
c-Src controls stability of sprouting blood vessels in the developing retina independently of cell-cell adhesion through focal adhesion assembly.
Tumor Angiogenesis Is Differentially Regulated by Phosphorylation of Endothelial Cell Focal Adhesion Kinase Tyrosines-397 and -861.
Nuclear shape, protrusive behaviour and in vivo retention of human bone marrow mesenchymal stromal cells is controlled by Lamin-A/C expression.
Coupling of β(2) integrins to actin by a mechanosensitive molecular clutch drives complement receptor-mediated phagocytosis.
Vaccine against gastrin, a polyclonal antibody stimulator, decreases pancreatic cancer metastases.
Plk1 Mediates Paxillin Phosphorylation (Ser-272), Centrosome Maturation, and Airway Smooth Muscle Layer Thickening in Allergic Asthma.
A unique role for clathrin light chain A in cell spreading and migration.
Insulin Stimulates PI3K/AKT and Cell Adhesion to Promote the Survival of Individualized Human Embryonic Stem Cells.
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Cell adhesion is regulated by CDK1 during the cell cycle.
Redistribution of Adhesive Forces through Src/FAK Drives Contact Inhibition of Locomotion in Neural Crest.
Replication of biocompatible, nanotopographic surfaces.
Distinct focal adhesion protein modules control different aspects of mechanotransduction.
Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix.
Non-muscle (NM) myosin heavy chain phosphorylation regulates the formation of NM myosin filaments, adhesome assembly and smooth muscle contraction.
Cdc42 regulates Cdc42EP3 function in cancer-associated fibroblasts.
Applied stretch initiates directional invasion through the action of Rap1 GTPase as a tension sensor.
Highly Multiplexed Imaging Uncovers Changes in Compositional Noise within Assembling Focal Adhesions.
MenaINV mediates synergistic cross-talk between signaling pathways driving chemotaxis and haptotaxis.
FAK, talin and PIPKIγ regulate endocytosed integrin activation to polarize focal adhesion assembly.
Talin tension sensor reveals novel features of focal adhesion force transmission and mechanosensitivity.
The Lhx9-integrin pathway is essential for positioning of the proepicardial organ.
Src-dependent phosphorylation of caveolin-1 Tyr-14 promotes swelling and release of caveolae.
Autoantibodies against thrombospondin type 1 domain-containing 7A induce membranous nephropathy.
Kindlin-2 cooperates with talin to activate integrins and induces cell spreading by directly binding paxillin.
Modulation of FAK and Src adhesion signaling occurs independently of adhesion complex composition.
Cellular adhesome screen identifies critical modulators of focal adhesion dynamics, cellular traction forces and cell migration behaviour.
Spatiotemporal dynamics of traction forces show three contraction centers in migratory neurons.
Molecular mechanism of vinculin activation and nanoscale spatial organization in focal adhesions.
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
Definition of a consensus integrin adhesome and its dynamics during adhesion complex assembly and disassembly.
Cdc42EP3/BORG2 and Septin Network Enables Mechano-transduction and the Emergence of Cancer-Associated Fibroblasts.
Focal adhesion kinase-dependent focal adhesion recruitment of SH2 domains directs SRC into focal adhesions to regulate cell adhesion and migration.
The integrin expression profile modulates orientation and dynamics of force transmission at cell-matrix adhesions.
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Slack length reduces the contractile phenotype of the Swine carotid artery.
A non-canonical role for Rgnef in promoting integrin-stimulated focal adhesion kinase activation.
Homocysteine induces serum amyloid A3 in osteoblasts via unlocking RGD-motifs in collagen.
RhoB regulates uPAR signalling.
Specific β-containing integrins exert differential control on proliferation and two-dimensional collective cell migration in mammary epithelial cells.
Effective treatment of edema and endothelial barrier dysfunction with imatinib.
PH006, a novel and selective Src kinase inhibitor, suppresses human breast cancer growth and metastasis in vitro and in vivo.
Force- and Ca²⁺-dependent internalization of integrins in cultured endothelial cells.
Focal adhesion proteins talin-1 and vinculin negatively affect paxillin phosphorylation and limit retroviral infection.
Tissue length modulates "stimulated actin polymerization," force augmentation, and the rate of swine carotid arterial contraction.
Loss of SNAP29 impairs endocytic recycling and cell motility.
c-Jun induces mammary epithelial cellular invasion and breast cancer stem cell expansion.
Focal-adhesion targeting links caveolin-1 to a Rac1-degradation pathway.
Complete focal adhesion kinase deficiency in the mammary gland causes ductal dilation and aberrant branching morphogenesis through defects in Rho kinase-dependent cell contractility.
Paxillin-Y118 phosphorylation contributes to the control of Src-induced anchorage-independent growth by FAK and adhesion.
An adaptor role for cytoplasmic Sam68 in modulating Src activity during cell polarization.
PDK1 regulates cancer cell motility by antagonising inhibition of ROCK1 by RhoE.
Disruption of c-Jun reduces cellular migration and invasion through inhibition of c-Src and hyperactivation of ROCK II kinase.
Paxillin phosphorylation, actin polymerization, noise temperature, and the sustained phase of swine carotid artery contraction.
Inhibition of cell migration by autophosphorylated mammalian sterile 20-like kinase 3 (MST3) involves paxillin and protein-tyrosine phosphatase-PEST.
Glycogen synthase kinase 3- and extracellular signal-regulated kinase-dependent phosphorylation of paxillin regulates cytoskeletal rearrangement.
Mislocalization or reduced expression of Arf GTPase-activating protein ASAP1 inhibits cell spreading and migration by influencing Arf1 GTPase cycling.
p130cas but not paxillin is essential for Caco-2 intestinal epithelial cell spreading and migration on collagen IV.
Tyrosine phosphorylation of paxillin alpha is involved in temporospatial regulation of paxillin-containing focal adhesion formation and F-actin organization in motile cells.
Ma J, Bi L, Spurlin J, Lwigale P
eLife 2022 Mar 3;11
eLife 2022 Mar 3;11
Matrix stiffness modulates tip cell formation through the p-PXN-Rac1-YAP signaling axis.
Guo Y, Mei F, Huang Y, Ma S, Wei Y, Zhang X, Xu M, He Y, Heng BC, Chen L, Deng X
Bioactive materials 2022 Jan;7:364-376
Bioactive materials 2022 Jan;7:364-376
Endothelial Focal Adhesions Are Functional Obstacles for Leukocytes During Basolateral Crawling.
Arts JJG, Mahlandt EK, Schimmel L, Grönloh MLB, van der Niet S, Klein BJAM, Fernandez-Borja M, van Geemen D, Huveneers S, van Rijssel J, Goedhart J, van Buul JD
Frontiers in immunology 2021;12:667213
Frontiers in immunology 2021;12:667213
Cholecystokinin Receptor Antagonist Improves Efficacy of Chemotherapy in Murine Models of Pancreatic Cancer by Altering the Tumor Microenvironment.
Malchiodi ZX, Cao H, Gay MD, Safronenka A, Bansal S, Tucker RD, Weinberg BA, Cheema A, Shivapurkar N, Smith JP
Cancers 2021 Sep 30;13(19)
Cancers 2021 Sep 30;13(19)
Coordination of mitochondrial and cellular dynamics by the actin-based motor Myo19.
Majstrowicz K, Honnert U, Nikolaus P, Schwarz V, Oeding SJ, Hemkemeyer SA, Bähler M
Journal of cell science 2021 May 15;134(10)
Journal of cell science 2021 May 15;134(10)
Integrin-linked kinase tunes cell-cell and cell-matrix adhesions to regulate the switch between apoptosis and EMT downstream of TGFβ1.
Kilinc AN, Han S, Barrett LA, Anandasivam N, Nelson CM
Molecular biology of the cell 2021 Mar 1;32(5):402-412
Molecular biology of the cell 2021 Mar 1;32(5):402-412
Identification of lymphocyte cell-specific protein-tyrosine kinase (LCK) as a driver for invasion and migration of oral cancer by tumor heterogeneity exploitation.
Weiße J, Rosemann J, Müller L, Kappler M, Eckert AW, Glaß M, Misiak D, Hüttelmaier S, Ballhausen WG, Hatzfeld M, Haemmerle M, Gutschner T
Molecular cancer 2021 Jun 11;20(1):88
Molecular cancer 2021 Jun 11;20(1):88
The proprotein convertase furin inhibits IL-13-induced inflammation in airway smooth muscle by regulating integrin-associated signaling complexes.
Wu Y, Huang Y, Zhang W, Gunst SJ
American journal of physiology. Lung cellular and molecular physiology 2021 Jul 1;321(1):L102-L115
American journal of physiology. Lung cellular and molecular physiology 2021 Jul 1;321(1):L102-L115
EVL regulates VEGF receptor-2 internalization and signaling in developmental angiogenesis.
Zink J, Frye M, Frömel T, Carlantoni C, John D, Schreier D, Weigert A, Laban H, Salinas G, Stingl H, Günther L, Popp R, Hu J, Vanhollebeke B, Schmidt H, Acker-Palmer A, Renné T, Fleming I, Benz PM
EMBO reports 2021 Feb 3;22(2):e48961
EMBO reports 2021 Feb 3;22(2):e48961
Inhibiting FAK-Paxillin Interaction Reduces Migration and Invadopodia-Mediated Matrix Degradation in Metastatic Melanoma Cells.
Mousson A, Legrand M, Steffan T, Vauchelles R, Carl P, Gies JP, Lehmann M, Zuber G, De Mey J, Dujardin D, Sick E, Rondé P
Cancers 2021 Apr 14;13(8)
Cancers 2021 Apr 14;13(8)
Light-regulated allosteric switch enables temporal and subcellular control of enzyme activity.
Shaaya M, Fauser J, Zhurikhina A, Conage-Pough JE, Huyot V, Brennan M, Flower CT, Matsche J, Khan S, Natarajan V, Rehman J, Kota P, White FM, Tsygankov D, Karginov AV
eLife 2020 Sep 23;9
eLife 2020 Sep 23;9
Endogenous Cyclin D1 Promotes the Rate of Onset and Magnitude of Mitogenic Signaling via Akt1 Ser473 Phosphorylation.
Chen K, Jiao X, Di Rocco A, Shen D, Xu S, Ertel A, Yu Z, Di Sante G, Wang M, Li Z, Pestell TG, Casimiro MC, Skordalakes E, Achilefu S, Pestell RG
Cell reports 2020 Sep 15;32(11):108151
Cell reports 2020 Sep 15;32(11):108151
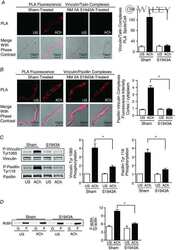
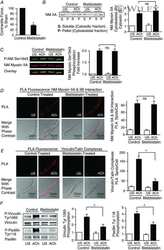
S100A4 is activated by RhoA and catalyses the polymerization of non-muscle myosin, adhesion complex assembly and contraction in airway smooth muscle.
Zhang W, Gunst SJ
The Journal of physiology 2020 Oct;598(20):4573-4590
The Journal of physiology 2020 Oct;598(20):4573-4590
Phenotype transitions induced by mechanical stimuli in airway smooth muscle are regulated by differential interactions of parvin isoforms with paxillin and Akt.
Huang Y, Gunst SJ
American journal of physiology. Lung cellular and molecular physiology 2020 May 1;318(5):L1036-L1055
American journal of physiology. Lung cellular and molecular physiology 2020 May 1;318(5):L1036-L1055
DLC1 is a direct target of activated YAP/TAZ that drives collective migration and sprouting angiogenesis.
van der Stoel M, Schimmel L, Nawaz K, van Stalborch AM, de Haan A, Klaus-Bergmann A, Valent ET, Koenis DS, van Nieuw Amerongen GP, de Vries CJ, de Waard V, Gloerich M, van Buul JD, Huveneers S
Journal of cell science 2020 Feb 12;133(3)
Journal of cell science 2020 Feb 12;133(3)
Common and Unique Transcription Signatures of YAP and TAZ in Gastric Cancer Cells.
Lee Y, Finch-Edmondson M, Cognart H, Zhu B, Song H, Low BC, Sudol M
Cancers 2020 Dec 7;12(12)
Cancers 2020 Dec 7;12(12)
c-Src controls stability of sprouting blood vessels in the developing retina independently of cell-cell adhesion through focal adhesion assembly.
Schimmel L, Fukuhara D, Richards M, Jin Y, Essebier P, Frampton E, Hedlund M, Dejana E, Claesson-Welsh L, Gordon E
Development (Cambridge, England) 2020 Apr 6;147(7)
Development (Cambridge, England) 2020 Apr 6;147(7)
Tumor Angiogenesis Is Differentially Regulated by Phosphorylation of Endothelial Cell Focal Adhesion Kinase Tyrosines-397 and -861.
Pedrosa AR, Bodrug N, Gomez-Escudero J, Carter EP, Reynolds LE, Georgiou PN, Fernandez I, Lees DM, Kostourou V, Alexopoulou AN, Batista S, Tavora B, Serrels B, Parsons M, Iskratsch T, Hodivala-Dilke KM
Cancer research 2019 Sep 1;79(17):4371-4386
Cancer research 2019 Sep 1;79(17):4371-4386
Nuclear shape, protrusive behaviour and in vivo retention of human bone marrow mesenchymal stromal cells is controlled by Lamin-A/C expression.
Dorland YL, Cornelissen AS, Kuijk C, Tol S, Hoogenboezem M, van Buul JD, Nolte MA, Voermans C, Huveneers S
Scientific reports 2019 Oct 7;9(1):14401
Scientific reports 2019 Oct 7;9(1):14401
Coupling of β(2) integrins to actin by a mechanosensitive molecular clutch drives complement receptor-mediated phagocytosis.
Jaumouillé V, Cartagena-Rivera AX, Waterman CM
Nature cell biology 2019 Nov;21(11):1357-1369
Nature cell biology 2019 Nov;21(11):1357-1369
Vaccine against gastrin, a polyclonal antibody stimulator, decreases pancreatic cancer metastases.
Osborne N, Sundseth R, Gay MD, Cao H, Tucker RD, Nadella S, Wang S, Liu X, Kroemer A, Sutton L, Cato A, Smith JP
American journal of physiology. Gastrointestinal and liver physiology 2019 Nov 1;317(5):G682-G693
American journal of physiology. Gastrointestinal and liver physiology 2019 Nov 1;317(5):G682-G693
Plk1 Mediates Paxillin Phosphorylation (Ser-272), Centrosome Maturation, and Airway Smooth Muscle Layer Thickening in Allergic Asthma.
Rezey AC, Gerlach BD, Wang R, Liao G, Tang DD
Scientific reports 2019 May 17;9(1):7555
Scientific reports 2019 May 17;9(1):7555
A unique role for clathrin light chain A in cell spreading and migration.
Tsygankova OM, Keen JH
Journal of cell science 2019 May 15;132(10)
Journal of cell science 2019 May 15;132(10)
Insulin Stimulates PI3K/AKT and Cell Adhesion to Promote the Survival of Individualized Human Embryonic Stem Cells.
Godoy-Parejo C, Deng C, Liu W, Chen G
Stem cells (Dayton, Ohio) 2019 Aug;37(8):1030-1041
Stem cells (Dayton, Ohio) 2019 Aug;37(8):1030-1041
Time-Variant SRC Kinase Activation Determines Endothelial Permeability Response.
Klomp JE, Shaaya M, Matsche J, Rebiai R, Aaron JS, Collins KB, Huyot V, Gonzalez AM, Muller WA, Chew TL, Malik AB, Karginov AV
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Cell chemical biology 2019 Aug 15;26(8):1081-1094.e6
Cell adhesion is regulated by CDK1 during the cell cycle.
Jones MC, Askari JA, Humphries JD, Humphries MJ
The Journal of cell biology 2018 Sep 3;217(9):3203-3218
The Journal of cell biology 2018 Sep 3;217(9):3203-3218
Redistribution of Adhesive Forces through Src/FAK Drives Contact Inhibition of Locomotion in Neural Crest.
Roycroft A, Szabó A, Bahm I, Daly L, Charras G, Parsons M, Mayor R
Developmental cell 2018 Jun 4;45(5):565-579.e3
Developmental cell 2018 Jun 4;45(5):565-579.e3
Replication of biocompatible, nanotopographic surfaces.
Sun X, Hourwitz MJ, Baker EM, Schmidt BUS, Losert W, Fourkas JT
Scientific reports 2018 Jan 12;8(1):564
Scientific reports 2018 Jan 12;8(1):564
Distinct focal adhesion protein modules control different aspects of mechanotransduction.
Stutchbury B, Atherton P, Tsang R, Wang DY, Ballestrem C
Journal of cell science 2017 May 1;130(9):1612-1624
Journal of cell science 2017 May 1;130(9):1612-1624
Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix.
Mekhdjian AH, Kai F, Rubashkin MG, Prahl LS, Przybyla LM, McGregor AL, Bell ES, Barnes JM, DuFort CC, Ou G, Chang AC, Cassereau L, Tan SJ, Pickup MW, Lakins JN, Ye X, Davidson MW, Lammerding J, Odde DJ, Dunn AR, Weaver VM
Molecular biology of the cell 2017 Jun 1;28(11):1467-1488
Molecular biology of the cell 2017 Jun 1;28(11):1467-1488
Non-muscle (NM) myosin heavy chain phosphorylation regulates the formation of NM myosin filaments, adhesome assembly and smooth muscle contraction.
Zhang W, Gunst SJ
The Journal of physiology 2017 Jul 1;595(13):4279-4300
The Journal of physiology 2017 Jul 1;595(13):4279-4300
Cdc42 regulates Cdc42EP3 function in cancer-associated fibroblasts.
Farrugia AJ, Calvo F
Small GTPases 2017 Jan 2;8(1):49-57
Small GTPases 2017 Jan 2;8(1):49-57
Applied stretch initiates directional invasion through the action of Rap1 GTPase as a tension sensor.
Freeman SA, Christian S, Austin P, Iu I, Graves ML, Huang L, Tang S, Coombs D, Gold MR, Roskelley CD
Journal of cell science 2017 Jan 1;130(1):152-163
Journal of cell science 2017 Jan 1;130(1):152-163
Highly Multiplexed Imaging Uncovers Changes in Compositional Noise within Assembling Focal Adhesions.
Harizanova J, Fermin Y, Malik-Sheriff RS, Wieczorek J, Ickstadt K, Grecco HE, Zamir E
PloS one 2016;11(8):e0160591
PloS one 2016;11(8):e0160591
MenaINV mediates synergistic cross-talk between signaling pathways driving chemotaxis and haptotaxis.
Oudin MJ, Miller MA, Klazen JA, Kosciuk T, Lussiez A, Hughes SK, Tadros J, Bear JE, Lauffenburger DA, Gertler FB
Molecular biology of the cell 2016 Oct 15;27(20):3085-3094
Molecular biology of the cell 2016 Oct 15;27(20):3085-3094
FAK, talin and PIPKIγ regulate endocytosed integrin activation to polarize focal adhesion assembly.
Nader GP, Ezratty EJ, Gundersen GG
Nature cell biology 2016 May;18(5):491-503
Nature cell biology 2016 May;18(5):491-503
Talin tension sensor reveals novel features of focal adhesion force transmission and mechanosensitivity.
Kumar A, Ouyang M, Van den Dries K, McGhee EJ, Tanaka K, Anderson MD, Groisman A, Goult BT, Anderson KI, Schwartz MA
The Journal of cell biology 2016 May 9;213(3):371-83
The Journal of cell biology 2016 May 9;213(3):371-83
The Lhx9-integrin pathway is essential for positioning of the proepicardial organ.
Tandon P, Wilczewski CM, Williams CE, Conlon FL
Development (Cambridge, England) 2016 Mar 1;143(5):831-40
Development (Cambridge, England) 2016 Mar 1;143(5):831-40
Src-dependent phosphorylation of caveolin-1 Tyr-14 promotes swelling and release of caveolae.
Zimnicka AM, Husain YS, Shajahan AN, Sverdlov M, Chaga O, Chen Z, Toth PT, Klomp J, Karginov AV, Tiruppathi C, Malik AB, Minshall RD
Molecular biology of the cell 2016 Jul 1;27(13):2090-106
Molecular biology of the cell 2016 Jul 1;27(13):2090-106
Autoantibodies against thrombospondin type 1 domain-containing 7A induce membranous nephropathy.
Tomas NM, Hoxha E, Reinicke AT, Fester L, Helmchen U, Gerth J, Bachmann F, Budde K, Koch-Nolte F, Zahner G, Rune G, Lambeau G, Meyer-Schwesinger C, Stahl RA
The Journal of clinical investigation 2016 Jul 1;126(7):2519-32
The Journal of clinical investigation 2016 Jul 1;126(7):2519-32
Kindlin-2 cooperates with talin to activate integrins and induces cell spreading by directly binding paxillin.
Theodosiou M, Widmaier M, Böttcher RT, Rognoni E, Veelders M, Bharadwaj M, Lambacher A, Austen K, Müller DJ, Zent R, Fässler R
eLife 2016 Jan 27;5:e10130
eLife 2016 Jan 27;5:e10130
Modulation of FAK and Src adhesion signaling occurs independently of adhesion complex composition.
Horton ER, Humphries JD, Stutchbury B, Jacquemet G, Ballestrem C, Barry ST, Humphries MJ
The Journal of cell biology 2016 Feb 1;212(3):349-64
The Journal of cell biology 2016 Feb 1;212(3):349-64
Cellular adhesome screen identifies critical modulators of focal adhesion dynamics, cellular traction forces and cell migration behaviour.
Fokkelman M, Balcıoğlu HE, Klip JE, Yan K, Verbeek FJ, Danen EH, van de Water B
Scientific reports 2016 Aug 17;6:31707
Scientific reports 2016 Aug 17;6:31707
Spatiotemporal dynamics of traction forces show three contraction centers in migratory neurons.
Jiang J, Zhang ZH, Yuan XB, Poo MM
The Journal of cell biology 2015 Jun 8;209(5):759-74
The Journal of cell biology 2015 Jun 8;209(5):759-74
Molecular mechanism of vinculin activation and nanoscale spatial organization in focal adhesions.
Case LB, Baird MA, Shtengel G, Campbell SL, Hess HF, Davidson MW, Waterman CM
Nature cell biology 2015 Jul;17(7):880-92
Nature cell biology 2015 Jul;17(7):880-92
Defining the phospho-adhesome through the phosphoproteomic analysis of integrin signalling.
Robertson J, Jacquemet G, Byron A, Jones MC, Warwood S, Selley JN, Knight D, Humphries JD, Humphries MJ
Nature communications 2015 Feb 13;6:6265
Nature communications 2015 Feb 13;6:6265
Definition of a consensus integrin adhesome and its dynamics during adhesion complex assembly and disassembly.
Horton ER, Byron A, Askari JA, Ng DHJ, Millon-Frémillon A, Robertson J, Koper EJ, Paul NR, Warwood S, Knight D, Humphries JD, Humphries MJ
Nature cell biology 2015 Dec;17(12):1577-1587
Nature cell biology 2015 Dec;17(12):1577-1587
Cdc42EP3/BORG2 and Septin Network Enables Mechano-transduction and the Emergence of Cancer-Associated Fibroblasts.
Calvo F, Ranftl R, Hooper S, Farrugia AJ, Moeendarbary E, Bruckbauer A, Batista F, Charras G, Sahai E
Cell reports 2015 Dec 29;13(12):2699-714
Cell reports 2015 Dec 29;13(12):2699-714
Focal adhesion kinase-dependent focal adhesion recruitment of SH2 domains directs SRC into focal adhesions to regulate cell adhesion and migration.
Wu JC, Chen YC, Kuo CT, Wenshin Yu H, Chen YQ, Chiou A, Kuo JC
Scientific reports 2015 Dec 18;5:18476
Scientific reports 2015 Dec 18;5:18476
The integrin expression profile modulates orientation and dynamics of force transmission at cell-matrix adhesions.
Balcioglu HE, van Hoorn H, Donato DM, Schmidt T, Danen EH
Journal of cell science 2015 Apr 1;128(7):1316-26
Journal of cell science 2015 Apr 1;128(7):1316-26
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Hoffmann JE, Fermin Y, Stricker RL, Ickstadt K, Zamir E
eLife 2014 Jun 3;3:e02257
eLife 2014 Jun 3;3:e02257
Symmetric exchange of multi-protein building blocks between stationary focal adhesions and the cytosol.
Hoffmann JE, Fermin Y, Stricker RL, Ickstadt K, Zamir E
eLife 2014 Jun 3;3:e02257
eLife 2014 Jun 3;3:e02257
Slack length reduces the contractile phenotype of the Swine carotid artery.
Rembold CM, Garvey SM, Tejani AD
Journal of vascular research 2013;50(3):221-7
Journal of vascular research 2013;50(3):221-7
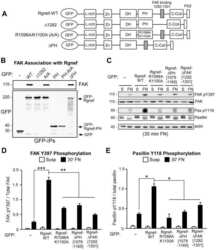
A non-canonical role for Rgnef in promoting integrin-stimulated focal adhesion kinase activation.
Miller NL, Lawson C, Kleinschmidt EG, Tancioni I, Uryu S, Schlaepfer DD
Journal of cell science 2013 Nov 1;126(Pt 21):5074-85
Journal of cell science 2013 Nov 1;126(Pt 21):5074-85
Homocysteine induces serum amyloid A3 in osteoblasts via unlocking RGD-motifs in collagen.
Thaler R, Zwerina J, Rumpler M, Spitzer S, Gamsjaeger S, Paschalis EP, Klaushofer K, Varga F
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Feb;27(2):446-63
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2013 Feb;27(2):446-63
RhoB regulates uPAR signalling.
Alfano D, Ragno P, Stoppelli MP, Ridley AJ
Journal of cell science 2012 May 15;125(Pt 10):2369-80
Journal of cell science 2012 May 15;125(Pt 10):2369-80
Specific β-containing integrins exert differential control on proliferation and two-dimensional collective cell migration in mammary epithelial cells.
Jeanes AI, Wang P, Moreno-Layseca P, Paul N, Cheung J, Tsang R, Akhtar N, Foster FM, Brennan K, Streuli CH
The Journal of biological chemistry 2012 Jul 13;287(29):24103-12
The Journal of biological chemistry 2012 Jul 13;287(29):24103-12
Effective treatment of edema and endothelial barrier dysfunction with imatinib.
Aman J, van Bezu J, Damanafshan A, Huveneers S, Eringa EC, Vogel SM, Groeneveld AB, Vonk Noordegraaf A, van Hinsbergh VW, van Nieuw Amerongen GP
Circulation 2012 Dec 4;126(23):2728-38
Circulation 2012 Dec 4;126(23):2728-38
PH006, a novel and selective Src kinase inhibitor, suppresses human breast cancer growth and metastasis in vitro and in vivo.
Ma JG, Huang H, Chen SM, Chen Y, Xin XL, Lin LP, Ding J, Liu H, Meng LH
Breast cancer research and treatment 2011 Nov;130(1):85-96
Breast cancer research and treatment 2011 Nov;130(1):85-96
Force- and Ca²⁺-dependent internalization of integrins in cultured endothelial cells.
Kiyoshima D, Kawakami K, Hayakawa K, Tatsumi H, Sokabe M
Journal of cell science 2011 Nov 15;124(Pt 22):3859-70
Journal of cell science 2011 Nov 15;124(Pt 22):3859-70
Focal adhesion proteins talin-1 and vinculin negatively affect paxillin phosphorylation and limit retroviral infection.
Brown C, Morham SG, Walsh D, Naghavi MH
Journal of molecular biology 2011 Jul 29;410(5):761-77
Journal of molecular biology 2011 Jul 29;410(5):761-77
Tissue length modulates "stimulated actin polymerization," force augmentation, and the rate of swine carotid arterial contraction.
Tejani AD, Walsh MP, Rembold CM
American journal of physiology. Cell physiology 2011 Dec;301(6):C1470-8
American journal of physiology. Cell physiology 2011 Dec;301(6):C1470-8
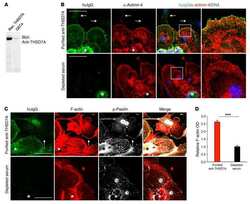
Loss of SNAP29 impairs endocytic recycling and cell motility.
Rapaport D, Lugassy Y, Sprecher E, Horowitz M
PloS one 2010 Mar 18;5(3):e9759
PloS one 2010 Mar 18;5(3):e9759
c-Jun induces mammary epithelial cellular invasion and breast cancer stem cell expansion.
Jiao X, Katiyar S, Willmarth NE, Liu M, Ma X, Flomenberg N, Lisanti MP, Pestell RG
The Journal of biological chemistry 2010 Mar 12;285(11):8218-26
The Journal of biological chemistry 2010 Mar 12;285(11):8218-26
Focal-adhesion targeting links caveolin-1 to a Rac1-degradation pathway.
Nethe M, Anthony EC, Fernandez-Borja M, Dee R, Geerts D, Hensbergen PJ, Deelder AM, Schmidt G, Hordijk PL
Journal of cell science 2010 Jun 1;123(Pt 11):1948-58
Journal of cell science 2010 Jun 1;123(Pt 11):1948-58
Complete focal adhesion kinase deficiency in the mammary gland causes ductal dilation and aberrant branching morphogenesis through defects in Rho kinase-dependent cell contractility.
van Miltenburg MH, Lalai R, de Bont H, van Waaij E, Beggs H, Danen EH, van de Water B
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2009 Oct;23(10):3482-93
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2009 Oct;23(10):3482-93
Paxillin-Y118 phosphorylation contributes to the control of Src-induced anchorage-independent growth by FAK and adhesion.
Sachdev S, Bu Y, Gelman IH
BMC cancer 2009 Jan 12;9:12
BMC cancer 2009 Jan 12;9:12
An adaptor role for cytoplasmic Sam68 in modulating Src activity during cell polarization.
Huot ME, Brown CM, Lamarche-Vane N, Richard S
Molecular and cellular biology 2009 Apr;29(7):1933-43
Molecular and cellular biology 2009 Apr;29(7):1933-43
PDK1 regulates cancer cell motility by antagonising inhibition of ROCK1 by RhoE.
Pinner S, Sahai E
Nature cell biology 2008 Feb;10(2):127-37
Nature cell biology 2008 Feb;10(2):127-37
Disruption of c-Jun reduces cellular migration and invasion through inhibition of c-Src and hyperactivation of ROCK II kinase.
Jiao X, Katiyar S, Liu M, Mueller SC, Lisanti MP, Li A, Pestell TG, Wu K, Ju X, Li Z, Wagner EF, Takeya T, Wang C, Pestell RG
Molecular biology of the cell 2008 Apr;19(4):1378-90
Molecular biology of the cell 2008 Apr;19(4):1378-90
Paxillin phosphorylation, actin polymerization, noise temperature, and the sustained phase of swine carotid artery contraction.
Rembold CM, Tejani AD, Ripley ML, Han S
American journal of physiology. Cell physiology 2007 Sep;293(3):C993-1002
American journal of physiology. Cell physiology 2007 Sep;293(3):C993-1002
Inhibition of cell migration by autophosphorylated mammalian sterile 20-like kinase 3 (MST3) involves paxillin and protein-tyrosine phosphatase-PEST.
Lu TJ, Lai WY, Huang CY, Hsieh WJ, Yu JS, Hsieh YJ, Chang WT, Leu TH, Chang WC, Chuang WJ, Tang MJ, Chen TY, Lu TL, Lai MD
The Journal of biological chemistry 2006 Dec 15;281(50):38405-17
The Journal of biological chemistry 2006 Dec 15;281(50):38405-17
Glycogen synthase kinase 3- and extracellular signal-regulated kinase-dependent phosphorylation of paxillin regulates cytoskeletal rearrangement.
Cai X, Li M, Vrana J, Schaller MD
Molecular and cellular biology 2006 Apr;26(7):2857-68
Molecular and cellular biology 2006 Apr;26(7):2857-68
Mislocalization or reduced expression of Arf GTPase-activating protein ASAP1 inhibits cell spreading and migration by influencing Arf1 GTPase cycling.
Liu Y, Yerushalmi GM, Grigera PR, Parsons JT
The Journal of biological chemistry 2005 Mar 11;280(10):8884-92
The Journal of biological chemistry 2005 Mar 11;280(10):8884-92
p130cas but not paxillin is essential for Caco-2 intestinal epithelial cell spreading and migration on collagen IV.
Sanders MA, Basson MD
The Journal of biological chemistry 2005 Jun 24;280(25):23516-22
The Journal of biological chemistry 2005 Jun 24;280(25):23516-22
Tyrosine phosphorylation of paxillin alpha is involved in temporospatial regulation of paxillin-containing focal adhesion formation and F-actin organization in motile cells.
Nakamura K, Yano H, Uchida H, Hashimoto S, Schaefer E, Sabe H
The Journal of biological chemistry 2000 Sep 1;275(35):27155-64
The Journal of biological chemistry 2000 Sep 1;275(35):27155-64
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of COS-7 (Lane 1), COS-7 treated with 100 uM Pervanadate for 1 hour (Lane 2), A431 (Lane 3), A431 treated with 100 uM Pervanadate for 1 hour (Lane 4), HeLa (Lane 5) and HeLa treated with 100 uM Pervanadate for 1 hour (Lane 6).The blots were probed with Anti-Phospho-Paxillin pTyr118 Rabbit Polyclonal Antibody (Product # 44-722G, 1:500 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Secondary Antibody, HRP conjugate (Product # G-21234, 1:5000 dilution). A 66 kDa band corresponding to Paxillin pTyr118 was enhanced across treated cell lines. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 12 % Bis-Tris gel (Product # NP0342BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western Blot analysis using Rabbit anti Paxillin (pY118) Polyclonal Antibody (Product # 44-722G)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30 µg lysate) of COS-7 (Lane 1), COS-7 treated with 100 uM Pervanadate for 1 hour (Lane 2), A431 (Lane 3), A431 treated with 100 uM Pervanadate for 1 hour (Lane 4), HeLa (Lane 5) and HeLa treated with 100 uM Pervanadate for 1 hour (Lane 6).The blots were probed with Anti-Phospho-Paxillin pTyr118 Rabbit Polyclonal Antibody (Product # 44-722G, 1:500 dilution) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Secondary Antibody, HRP conjugate (Product # G-21234, 1:5000 dilution). A 66 kDa band corresponding to Paxillin pTyr118 was enhanced across treated cell lines. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 12 % Bis-Tris gel (Product # NP0342BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Loss of SNAP29 affects the pattern of focal adhesion complexes. (A-L) Control (SV80 and F572T) and CEDNIK (F110T or F165T) fibroblasts were fixed and stained with anti-FAK (A-C), anti-p-FAK (D-F), anti-PAX (G-I) and anti-p-PAX antibodies (J-L). Nuclei were stained with Hoechst. Bar, 10 um. (M) Western blot analysis showing the expression of FAK, p-FAK, PAX and p-PAX in control (F572T) and CEDNIK (F165T, F110T) cells. Anti-ERK antibodies were used as a loading control. The blot is a representative of 5 independent experiments. (N) Bar graph illustrating the expression levels of the focal adhesion components: FAK, p-FAK, PAX and p-PAX. Densitometry was used to quantify the amount of each focal adhesion component as seen in Western blot, using the Image Densitometer 1Dprime. To normalize the results, the intensity of each band was divided by the intensity of ERK in the same lane. The value obtained for control cells (F572T) was considered as 100%. (O) Bar graph showing quantitation of the intensity of FAK, p-FAK, PAX and p-PAX staining, measured in 14-168 cells, using the ImageJ software. The value obtained for control cells (SV80) was considered as 100%. Expression pattern of FAK, phospho-FAK, paxillin and phospho-paxillin is different in CEDNIK cells in comparison to control cells. However, their total level is similar.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
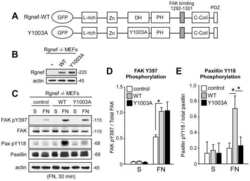
- Figure 5--figure supplement 1. FAK phosphorylation in Tln Ctr , Tln Ko , Tln Ko+T1V , Kind Ctr , Kind Ko and Kind Ko+K2GFP cells. ( A ) Densitometric quantification of western blot signals of lysates from untreated, EGF- and Mn 2+ -treated Kind Ctr , Tln Ko and Kind Ko cells seeded either on FN or PLL and probed with anti-Tyr-397 phosphorylated FAK (pY397-FAK) antibodies (n=3 independent repeats; significances are calculated with respect to PLL adherent cells; error bars indicate standard error of the mean). ( B , C ) Western blotting of indicated signaling proteins in untreated, EGF- and Mn 2+ -treated Tln Ctr , Tln Ko and Tln Ko cells re-expressing Venus-tagged talin-1 (Tln Ko+T1V ) ( B ), and Kind Ctr , Kind Ko and Kind Ko cells re-expressing GFP-tagged kindlin-2 (Kind Ko+K2GFP ) ( C ) seeded either on FN or PLL. EGF, epidermal growth factor; FAK, focal adhesion kinase; K2GFP, green fluorescent protein-tagged kindlin-2; FN, fibronectin; GFP, green fluorescent protein; PLL, poly-L-lysine; T1V, Venus-tagged full length talin-1. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Focal adhesion association of SRC controls SRC-mediated substrate phosphorylation, focal adhesion formation and cell migration. ( a ) Cell lysates from U2OS cells expressing GFP, SRC-GFP, SRCDeltaSH2-GFP, SRCY527F-GFP or SRCY527FDeltaSH2-GFP were analyzed by Western blotting for pY577-FAK, pY397-FAK, pY118-paxillin, pY31-paxillin, paxillin, GAPDH and GFP. ( b ) Cell lysates from SYF cells expressing GFP, SRC-GFP, SRCDeltaSH2-GFP, SRCY527F-GFP or SRCY527FDeltaSH2-GFP were analyzed by Western blotting for pY577-FAK, pY397-FAK, pY118-paxillin, pY31-paxillin, paxillin, GAPDH and GFP. ( c ) U2OS cells expressing GFP, SRCY527F-GFP or SRCY527FDeltaSH2-GFP (blue) were immunostained to detect F-actin (phalloidin; red) and paxillin (green), and imaged by epi-fluorescence and TIRFM, respectively. Scale bar, 10 mum. ( d ) The number of segmented paxillin-marked FAs within U2OS cells, as described in ( c ). Data are means +- s.e.m. (GFP: n = 11 cells; SRCY527F-GFP: n = 13 cells; SRCY527FDeltaSH2-GFP: n = 12 cells). ** p < 0.01; *** p < 0.001; NS, no significance. ( e ) Size distribution of segmented paxillin-marked FAs of U2OS cells, as described in ( c ). Data are means +- s.e.m. * p < 0.05; ** p < 0.01; *** p < 0.001; NS, no significance. ( f ) The migratory behavior of U2OS cells expressing GFP, SRCY527F-GFP or SRCY527FDeltaSH2-GFP for 6 hrs. Cells were plated for 16 hrs, and then monitored for 6 hrs. (right) These images delineate the trajectory of the GFP-marked cells over a
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
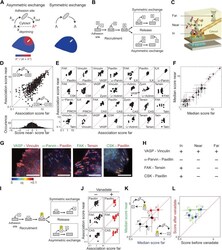
- Figure 3. Symmetric material exchange between stationary focal adhesions and cytosol. ( A ) The symmetric and asymmetric models of material exchange between adhesion sites and cytosol. In symmetric exchange a component, A , exits from adhesion site in the same state it had upon entering to it. In asymmetric exchange A exits in a different, primed state A* and relaxes back to state A in the cytosol, thereby generating a spatial gradient of the primed state emanating from adhesion sites. ( B ) Formulation of the two models for the case in which priming (of A ) is based on interaction (with protein B ). Here, asymmetric exchange would generate a spatial gradient of AB complex concentration emanating from adhesion sites. ( C - F ) Discriminating between the two models by measuring the physical associations near (= 9) ( E ) or for the median score of each pair +- MAD ( F ). Data-points far from the equality diagonals (dashed red lines) would correspond to asymmetric exchanges, as illustrated in ( D ), while data-points along the diagonal indicate symmetric exchange. Histogram in ( D ) shows the distribution of the difference in association scores near and far from focal adhesions. ( G ) FLIM images color-coding the fraction, alpha, of
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
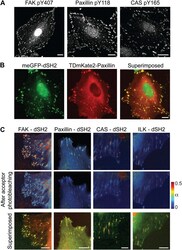
- Figure 3--figure supplement 1. FAK, paxillin, and CAS are tyrosine-phosphorylated and interact with SH2 domain in focal adhesions. ( A ) Immunofluorescence images of REF52 stained for FAK pY407, paxillin pY118, and CAS pY165 using phospho-specific antibodies. Scale bars, 15 mum. ( B ) A REF52 cell co-expressing meGFP-dSH2 and TD-mKate2-paxillin. Note the localization of dSH2 in cell-matrix adhesion sites, as previously reported ( Kirchner et al., 2003 ). Scale bar, 10 mum. ( C ) Top and middle rows, FLIM images color-coding the fraction (alpha) of the donor- (mCitrine) tagged protein (FAK, paxillin, CAS, or ILK) that FRET to the acceptor- (mCherry) tagged dSH2 before and after acceptor photobleaching. ILK is used here as a negative control for tyrosine phosphorylation. Note that low levels of alpha are observed also for ILK in focal adhesions, plausibly due to density effects. However, FAK, paxillin, and CAS exhibit higher alpha values, suggesting FRET due to direct interaction. Scale bars, 10 mum. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
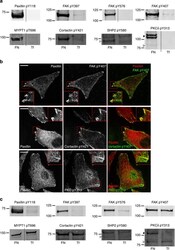
- Figure 4 Validation of adhesion complex-localized phosphorylation sites. ( a ) Immunoblot analyses of the protein complexes isolated from cells spread on FN or Tf for 120 min. Samples were probed using phosphospecific antibodies targeting: paxillin pTyr118; FAK pTyr397, pTyr576 and pTyr407; MYPT1 pThr696; cortactin pTyr421; SHP2 pTyr580; and PKCdelta pTyr313. ( b ) Immunofluorescence images of cells spread on FN for 120 min. Cells were stained using the phosphospecific antibodies FAK pTyr407, cortactin pTyr421 and PKCdelta pTyr313 (all green). In each case, cells were also stained using an antibody targeting the focal adhesion marker paxillin (red). Scale bar, 20 mum. ( c ) Immunoblot analyses of the WCLs of cells spread on FN or Tf for 120 min. Samples were probed using the same antibodies as used to probe isolated complexes in a . Dashed lines in a , c indicate where the images have been cropped for display purposes. White arrows to left of blots highlight the expected MWs of the corresponding proteins. Black arrows to the left of blots highlight a putative high MW isoform of PKCdelta.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Focal adhesion kinase activity, but not SRC activity, positively regulates the abundance of SRC_SH2 domain within focal adhesions. ( a ) Total cell lysate from untreated U2OS cells (-) or cells treated with FAKi (focal adhesion kinase inhibitor; 50 muM, 1 h), PP2 (SRC inhibitor; 10 muM, 1 h) or SU6656 (SRC inhibitor; 5 muM, 1 h) were analyzed by Western blotting to detect pY118-paxillin, pY31-paxillin, SRC, FAK, paxillin and GAPDH. ( b ) U2OS cells transfected with pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) were untreated (-) or treated with FAKi or PP2, and immunostained to detect paxillin. TIRFM images of GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) (green) and paxillin (red). Bar, 10 mum. The 20 mum x 20 mum areas indicated in the upper images are magnified in the images below. Values indicate Pearson's correlation coefficients (PC) of images of paxillin-Cy5 and GFP, GFP-SRC(SH2) or GFP-SHP2(NSH2) in U2OS cells untreated (-) or treated with FAKi or PP2. Data are means +- s.e.m. (GFP: n = 3 cells (-), n = 4 cells (FAKi), n = 6 cells (PP2); GFP-SRC(SH2): n = 7 cells (-), n = 6 cells (FAKi), n = 7 cells (PP2); GFP-SHP2(NSH2): n = 5 cells (-), n = 5 cells (FAKi), n = 4 cells (PP2)). ** p < 0.01; NS, no significance. ( c ) Time-lapse TIRFM images of U2OS cells co-transfected with mApple-paxillin (red) and pGFP-C1, pGFP-SRC(SH2) or pGFP-SHP2(NSH2) (green) during FAKi or PP2 treatment. The time is shown in hour. Bar, 10 mum. ( d ) The ratio of the average density (intensity p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
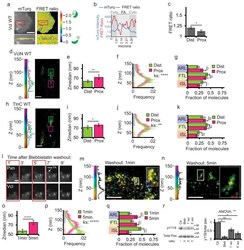
- Figure 8 Vinculin activation and nano-scale localization are spatiotemporally regulated (a) Top: mTurquoise (mTurq) and processed mTurquoise/NeonGreen FRET ratio images of an HFF cell expressing the vinculin FRET biosensor. Bottom: Zoom of outlined area. FA mask (grey lines) created from the mTurq image and superimposed onto FRET ratio image. (b) mTurquoise intensity and FRET ratio along the 5um long line in the zoom in (a). Dark shaded areas are cytosolic (cyto) regions adjacent to the FA (light shaded area) (c) Mean FRET ratio quantification of the distal (Dist, facing cell edge) 1/3 and proximal (Prox, facing cell center) 1/3 of FA linescans. (d, h) Representative iPALM renderings of HFFs expressing vinculin-N-tdEos (d) or talin-C-tdEos (h). Histograms of the Z-position of molecules within Dist and Prox thirds displayed next to the colourscale. (e, i) Mean of Z-median measurements from Dist or Prox thirds (f, j) Averaged Z-position frequency histograms of molecules. Solid line, mean frequency; Shaded region, bootstrapped 95% confidence intervals. (g, k) Mean fraction of molecules localized to the three FA layers. (l) Time-lapse TIRFM images of FAs during blebbistatin washout. HFFs expressing paxillin-mCherry (top) and vinculin-EGFP (bottom) treated with 50uM blebbistatin for 2 hours. Blebbistatin washout at t=0 min, Arrow: newly formed FA. Boxed areas: time points for iPALM analysis. Scale bar= 2mum. (m, n) Representative iPALM renderings of vinculin-N-tdEos at 1min (m) an
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. The role of substrate adhesion in neuronal migration and force generation. (a) Pulling assay. Pulling the LP of GCs at the middle point. Broken lines show the location of soma and LP growth cone before and after pulling. Bars, 10 um. (b) Summary of results of the pulling experiments. Under migratory or stationary conditions, the number of GCs that showed the first detachment site at the soma or at the growth cone of LP after pulling is shown. (c) Immunostaining of markers of adhesion complexes (FAK, paxillin, vinculin). (d) FAK inhibition with Y11 (~50-200 uM) enhanced the strain energy of GCs ( n = 7). Broken lines show the normalized strain energy (1 unit) before drug application.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Automated image analysis of focal adhesion morphology allows quantitative assessment of different dynamic states of FAs. ( a ) Automated image analysis identifies individual focal adhesions based on pTyr118-paxillin or vinculin staining. ( b ) Image analysis and quantification of focal adhesion sizes in a set of representative images of the nocodazole assay. Focal adhesion size distribution of nocodazole-induced FA assembly and microtubule-induced FA disassembly were compared using a Kolmogorov-Smirnov (KS) test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
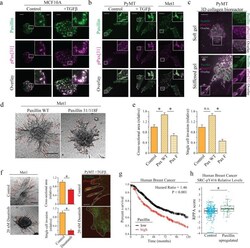
- FIGURE 9: Enhanced paxillin phosphorylation permits invasion in cells with a basal phenotype. (a) Paxillin localization to the basal surface in MCF10A cells with and without TGFbeta treatment visualized with immunofluorescence staining and TIRF imaging of paxillin and phosphorylated paxillin, pPax[31]. Scale bar, 10 um (main image), 1 um (inset). (b) Paxillin expression in PyMT, Met1, and PyMT + TGFbeta visualized with immunofluorescence staining and confocal imaging of paxillin and phosphorylated paxillin pPax[31]. Scale bar, 10 um (main image), 1 um (inset). (c) Paxillin expression of PyMT cell spheroids visualized in soft and stiffened 3D collagen gel. Leader cells in stiffened gel have higher levels of phosphorylated paxillin indicated by pPax[31] immunostaining. Scale bar, 20 um (main image), 10 um (inset). (d) A 3D collagen invasion assay of multicellular spheroids composed of Met1 cells overexpressing either paxillin WT or paxillin Y31/118F in 2.5 mg/ml 3D collagen gels for 72 h. Scale bar, 100 um. (e) Quantification of invasion. Paxillin WT overexpression significantly increased invasion compared with control or paxillin Y31/118F overexpression, as determined by cross-sectional area of spheroid and protrusions and single-cell invasion (* p < 0.05; >20 spheroids). (f) A 3D collagen invasion assay of multicellular spheroids composed of metastatic Met1 cells treated with dimethyl sulfoxide or 20 nM dasatinib, a Src/Abl inhibitor, in 2.5 mg/ml 3D collagen gels for 72 h. D
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Supplementary Figure 7
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. Effects of FAK, Src and combined FAK and Src inhibition on IAC proteins and phosphorylation . (A-I) HFF cells spread on FN for 1 h were treated with DMSO, FAK [i], Src [i], or combined FAK [i] + Src [i] for 1 h. IACs were visualized by staining for pY (green) and alpha5 integrin (red; A-C), FAK Y397 (green) and vinculin (red; D-F) or paxillin Y118 (green) and paxillin (red; G-I). Bars: (main) 20 um; (ROI) 5 um. Graphs show quantification of the cell area covered by positive staining of the indicated protein (mean +- SEM, n = 10 cells). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001; ns, not significant; Kruskal-Wallis test with Dunn's post hoc correction (in C, F, and I, comparisons with cells treated with DMSO are shown). Representative images are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
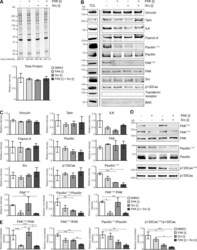
- Figure 5. Effects of FAK, Src, and combined FAK and Src inhibition on IAC composition. (A and B) HFF cells spread on FN for 1 h were treated with DMSO, FAK [i], Src [i], or combined FAK [i] + Src [i] for 1 h. IACs were isolated using the workflow in Fig. 2 A and were analyzed by SDS-PAGE (A) and immunoblotting (B). Representative total protein intensity values for each lane are indicated. Graph shows intensity values normalized to the DMSO condition (mean +- SEM, n = 3). Cell lysates from cells spread on FN (TCL) were used as a positive control. Molecular mass values (kD) are displayed. (C) For quantification of immunoblotted membranes in B, the band intensity values for each protein are shown relative to the DMSO condition (mean +- SEM, n = 3). Quantification was not performed for Tf receptor or BAK. (D) Total cell lysates collected from HFF cells spread on FN for 1 h and treated with DMSO or inhibitors for 1 h were analyzed by immunoblotting. Molecular mass values (kD) are displayed. (E) For quantification of immunoblotted membranes in D, phosphorylation values normalized to the corresponding total protein values are shown relative to the DMSO condition (mean +- SEM; n = 4, FAK Y397 , FAK Y576 , and paxillin Y118 ; n = 3, p130Cas Y249 ). t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Representative images are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
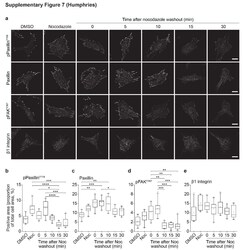
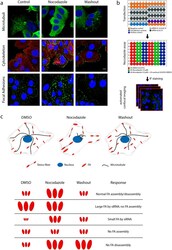
- Figure 1 A nocodazole-based microtubule depletion assay to study focal adhesion dynamics in high-throughput fashion. ( a ) Treatment of MCF7 WT cells with nocodazole (10 muM, 4 h) depolymerized microtubules (MT), leading to activation of the actin cytoskeleton and focal adhesion assembly. Washout of nocodazole allows MT regrowth, resulting in focal adhesion disassembly. MCF7 WT cells were stained for vinculin and phosphorylated paxillin to visualize focal adhesions. ( b ) Schematic representation of screen setup. Transfection was performed in 96-well plates and controls were included in each plate. MCF7 WT cells were exposed as indicated, to induce FA assembly and disassembly. Plates were stained for vinculin and phosphorylated paxillin and imaged with automated confocal microscopy. ( c ) Schematic representation of cellular and FA organization in the nocodazole assay, with possible FA phenotypes caused by siRNA knockdown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 3 Figure S100A4 depletion inhibits the membrane localization and activation of vinculin and paxillin, and S100A4 depletion inhibits actin polymerization but does not affect myosin RLC phosphorylation A and B , in situ PLA fluorescence and PLA fluorescence merged with phase contrast from unstimulated (US) or ACh stimulated cells freshly dissociated from sham-treated or S100A4-depleted tissues. A , paxillin-vinculin complexes are distributed throughout the cytoplasm of unstimulated cells from both sham-treated and S100A4-depleted tissues. ACh stimulation resulted the localization of paxillin-vinculin complexes to the membrane of sham-treated cells (US, n = 13; ACh, n = 15, p = 0.0001), whereas paxillin-vinculin complexes remained distributed throughout the cytoplasm of S100A4-depleted cells (US, n = 12; ACh, n = 14, p = 0.0001). B , ACh stimulation resulted in a marked increase in the number of talin-vinculin complexes at the cell membrane in sham-treated cells (US, n = 21; ACh, n = 29, p = 0.0001). S100A4 depletion inhibited the ACh-induced increase of the interaction between talin and vinculin at the cell membrane (US, n = 26; ACh, n = 30, p = 0.0681). Few spots were observed in S100A4-depleted treated cells with or without stimulation. C , representative immunoblots from extracts of sham-treated muscle tissues and S100A4 shRNA-treated tissues stimulated with ACh or not stimulated (US). The increases in vinculin Tyr1065 phosphorylation and paxillin Tyr118 phosphorylation in r
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 6 Figure Depletion of rhotekin inhibits NM myosin II polymerization and membrane recruitment and activation of vinculin and paxillin in response to ACh A , immunoblot of NM myosin II in cytoskeletal (P) and soluble (S) fractions from extracts of tracheal SM tissues treated with RTKN siRNA or untreated (Sham) and stimulated for 5 min with 10 -5 M ACh or unstimulated (US). RTKN depletion significantly inhibited the ACh-induced increase in the ratio of NM myosin II in the cytoskeletal (P) vs . soluble (S) fractions ( n = 6, p = 0.0001). B , in situ PLA fluorescence in freshly dissociated US or ACh-stimulated SM cells from sham-or siRNA-treated tracheal SM tissues. ACh stimulation of sham-treated tissues resulted in a significant increase in the number of NM myosin IIA and IIB complexes at the cell cortex ( n = 20 cells/group). RTKN depletion inhibited the ACh-induced increase in the interaction between NM myosin IIA and IIB ( n = 20 cells/group, P = 0.0001). C , RTKN depletion significantly inhibited the increase in NM myosin II Ser1943 phosphorylation in tracheal muscle tissues in response to 5 min stimulation with 10 -5 M ACh ( n = 11, P = 0.0001). D , in situ PLA fluorescence shows interactions between talin and vinculin in US or ACh-stimulated freshly dissociated SM cells from sham-treated or RTKN-depleted tissues. ACh stimulation resulted in a significant increase in the number of talin-vinculin complexes at the cell membrane (US cells, n = 19; ACh, n = 25 cells, p = 0.0001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 4: Depletion of ILK decreases the activation and assembly of focal adhesion proteins. Immunoblotting analysis for GAPDH and (A) total FAK, pY397-FAK (pFAK), (B) total paxillin (Pax), and pY118-paxillin (pPax) in shcntl and shILK cells cultured on soft or stiff substrata. Quantification shows mean +- SD for three independent experiments; immunoblotting replicates and statistical analysis are shown in Supplemental Figure S4. Immunofluorescence analysis for F-actin (green), nuclei (blue), and (C) FAK (magenta), (D) pFAK (magenta), (E) Pax (magenta), or (F) pPax (magenta) in shcntl and shILK cells cultured on plastic. Scale bars, 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Cell migration and focal adhesion formation in MSCs compared to other primary mesodermal-derived adherent cells. ( A ) Average migration velocity of ABMSC compared to FBMSC, HUVEC and fibroblasts, measured by live cell phase contrast microscopy. Images were obtained every 30 minutes for a total of 12 hours at 37 degC, 5% CO 2 and migration was tracked using ImageJ. Median +- range. n = 4 independent experiments including 30 cells each. ns = non-significant, *p < 0.05, ***p < 0.001 (Kruskal-Wallis, multiple comparisons uncorrected Dunn's test). ( B ) Maximum absolute travel distance from point origin of ABMSC, FBMSC, HUVEC and fibroblasts within 12 hours. Median +- range. n = 4 independent experiments, including 30 cells each. ns = non-significant, **p < 0.01 (Kruskal-Wallis, multiple comparisons uncorrected Dunn's test). ( C ) Graph shows ABMSC migration velocity on varying concentrations of human-derived fibronectin (hFN) and bovine-derived fibronectin (bFN). Images were obtained every 30 minutes for a total of 12 hours at 37 degC, 5% CO 2 and migration was tracked using ImageJ. Median +- range. n = 4 independent experiments comprising cells from 4 different donors, analysis includes 15 cells per condition. Spearman correlation test. ( D ) Average migration velocity of ABMSCs cultured in either Dulbecco's Modified Eagle's Medium (DMEM) with 10% fetal calf serum (FCS) or with 5% Platelet lysate (PL) and cultured on 20 ug/ml hFN or bFN. n = 4 independent experiments c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
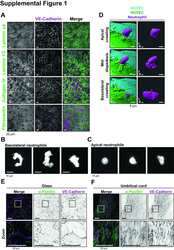
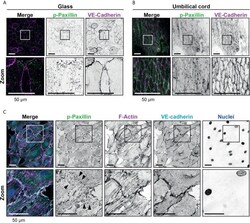
- Figure 2 Presence of focal adhesions at endothelium from the umbilical cord. (A) Immunofluorescent staining for phospho-paxillin (green) and VE-cadherin (magenta) of HUVECs grown on glass substrate. ROI is indicated in the merge. Images are inverted for clarification. Lower panel shows zoom from ROI. Scale bar, 50 um. (B) Immunofluorescent staining for phospho-paxillin (green) and VE-cadherin (magenta) of HUVECs on an umbilical cord. ROI is indicated in the merge. Images are inverted for clarification. Lower panel shows zoom from ROI. Scale bar, 50 um (C) Immunofluorescent staining for phospho-paxillin (green), F-actin (magenta), VE-cadherin (cyan) and nuclei (blue) of BOEC seeded on umbilical cord, treated with TNF-alpha. ROI is indicated in the merge. Images are inverted for clarification. Lower panel shows zoom from ROI. Black arrowheads indicate focal adhesions. Scale bar, 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
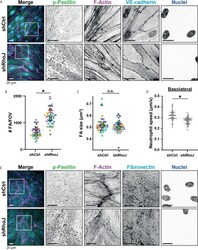
- Figure 5 Focal adhesions upon RhoJ silencing. (A) Immunofluorescent staining for phospho-paxillin (green), F-actin (magenta), VE-cadherin (Turquoise) and DNA (blue) on HUVECs stimulated for 20h with TNFalpha. ROI is indicated in the merge. Images are inverted for clarification. Upper panel are control cells (shCtrl), lower panel are cells with RhoJ depletion (shRhoJ). Scale bar, 20 um. (B) Quantification of the number of focal adhesions (FA) per field of view (FOV) for control (shCtrl) and RhoJ knockdown (shRhoJ) conditions. Superplot in which small dots are individual datapoints, large dots represent means from 5 independent experiments (five colors). Mean and standard deviation are indicated. Welch's T-test *p=0.000218. (C) Quantification of the size of focal adhesions (FA) for control (shCtrl) and RhoJ knockdown (shRhoJ) conditions. Superplot in which small dots are individual datapoints, large dots represent means from 5 independent experiments (five colors). Mean and standard deviation are indicated. Welch's T-test p=0.2712415, not significant (n.s.). (D) Quantification of the speed of basolateral neutrophils under control (shCtrl) and RhoJ knockdown (shRhoJ) endothelial cells. Violin plot in which small dots are individual datapoints. Mann-Whitney U-test: p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
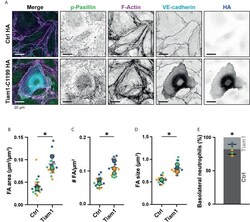
- Figure 7 Focal adhesions upon Tiam1-C1199 expression. (A) Immunofluorescent staining for phospho-paxillin (green), F-actin (magenta), VE-cadherin (Turquoise) and HA (blue) on HUVECs stimulated for 20h with TNFalpha. Images are inverted for clarification. Upper panel are control cells (Ctrl HA), lower panel are cells expressing Tiam1-C1199 (Tiam1-C1199). Scale bar, 20 um. (B) Quantification of the area focal adhesions (FA) cover or control (Ctrl) and Tiam1-C1199-expressing cells (Tiam1-C1199). Superplot in which small dots are individual datapoints, large dots represent means from 3 independent experiments (three colors). Mean and standard deviation are indicated. Welch's T-test *p=0.02411431. (C) Quantification of the number of focal adhesions (FA) per field of view (FOV) for control (Ctrl) and Tiam1-C1199-expressing cells (Tiam1-C1199). Superplot in which small dots are individual datapoints, large dots represent means from 3 independent experiments (three colors). Mean and standard deviation are indicated. Welch's T-test *p=0.04694375. (D) Quantification of the size of focal adhesions (FA) in control (Ctrl) and Tiam1-C1199-expressing cells (Tiam1-C1199). Superplot in which small dots are individual datapoints, large dots represent means from 3 independent experiments (three colors). Mean and standard deviation are indicated. Welch's T-test *p=0.02819648 (E) Quantification of the percentage of TEM events in which a neutrophil migrates at a control-Tiam1-C1199 junction and mi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
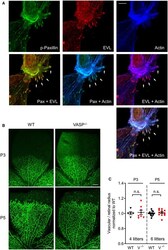
- Figure EV3 Subcellular localization of EVL and postnatal angiogenesis in VASP -/- mice A EVL localizes to focal adhesions in endothelial cells. MLEC from wild-type mice were stained for phospho-paxilin (green) as a marker for focal adhesions, EVL (red) and actin (blue). White arrows indicate integrin-based focal adhesions at the tips of actin stress fibers. Representative images from three independent experiments are shown. Scale bar, 10 um. B, C Postnatal retinal angiogenesis in VASP -/- mice. (B) Isolectin B4-stained vasculature in whole mount retinas of wild-type (WT) and global VASP -/- mice on postnatal days 3 and 5 (P3, P5) assessed by confocal microscopy. Scale bars 200 um. (C) Analysis of the radial vascular outgrowth relative to retinal radius and normalized to wild-type littermates. Error bars represent SEM; no significant difference was observed between the two genotypes at P3 ( P > 0.999) or P5 ( P > 0.999) (one-way ANOVA with Bonferroni's multi-comparison test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
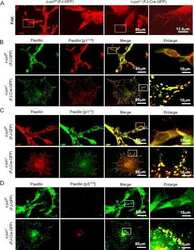
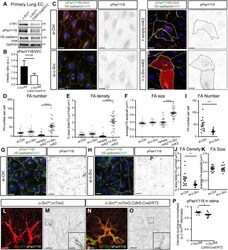
- Fig. 7. c-Src mediates paxillin phosphorylation and subsequent focal adhesion formation. (A) Endothelial cells were isolated from lungs of c-Src flox/flox and c-Src flox/flox ; Cdh5-CreERT2 mice, and protein expression was assessed by western blot. (B) A decrease in phospho-paxillin at Y118 was observed ( n >5 mice). (C) HUVECs transfected with siRNA for control or c-Src, or with overexpression of empty-mKate2 or c-Src-mKate2 cultured on fibronectin-coated coverslips were immunostained for p-paxillin-Y118 (pPaxY118), c-Src (for siRNA-treated cells), VE-cadherin and DAPI. c-Src-mKate2 is visualized with mKate2 signal. Single channel images of pPaxY118 are shown on the right; mKate2-positive cells are outlined with dashed lines. Scale bars: 20 um. (D-F) Focal adhesion number, density and size based on pPaxY118 staining was significantly increased upon c-Src-mKate2 overexpression ( n >30 cells in total from three independent experiments). (G,H) HUVECs transfected with siRNA for control or c-Src cultured on vitronectin-coated coverslips were stained for p-paxillin-Y118 (pPaxY118), VE-cadherin and DAPI. Single channel images of pPaxY118 are shown on the right. Scale bars: 20 um. (I-K) Focal adhesion number and density based on pPaxY118 staining was significantly decreased upon c-Src depletion, while the size was not significantly changed. n >15 cells in total from three independent experiments. (L-O) c-Src flox/flox ; mTmG and c-Src flox/flox ; mTmG; Cdh5-CreERT2 retinas were immu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
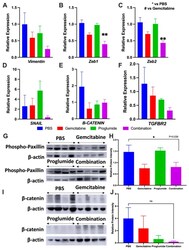
- Figure 4 Combination therapy with gemcitabine and proglumide decreases metastases by preventing epithelial to mesenchymal transition (EMT). Quantitative PCR analysis for mRNA expression of transcription factors and proteins from PDAC tumors ( n = 4 per group) with relative gene expression (2 -DeltaDeltaCt +- SEM) for: ( A ) Vimentin , ( B ) Zeb1 , ( C ) Zeb2 , ( D ) SNAIL1 , ( E ) beta-CATENIN , and ( F ) TBFbetaR2 . Unpaired t-test was used for analysis (* p = 0.05; ** p < 0.01) and GAPDH was used as the loading control. ( G ) Western blot for phospho-paxillin for tumor protein extracts of each group normalized with beta -actin. (The whole western blot is shown in the Figure S4 ). ( H ) Analysis of bands by densitometry show significant differences in the groups * p < 0.05. ( I ) Western blot of PANC-1 tumor protein extracts reacted with an antibody for beta-catenin normalized with beta -actin. (The whole western blot is shown in the Figure S4 ). ( J ) Analysis of the bands by densitometry show a decrease in the intensity of the bands in the proglumide and combination groups although this did not quite reach statistical significance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
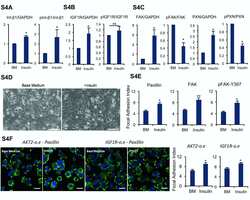
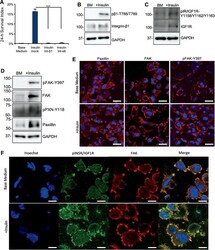
- Figure 4 Insulin promotes focal adhesion formation in dissociated cells. (A): Plots showing 24-hour cell survival of insulin with integrin beta1 and alpha6 blocking antibodies compared with Insulin+TNNT2 antibody (mock) on matrigel (***, p < .001, n = 3). (B): Western blot showing the level of integrin beta1 and phosphorylated integrin beta1-788-789 2 hours after plating on matrigel-coated surface in base medium with or without insulin. Quantification is shown in Supporting Information Figure S4A . (C): Western blot showing the protein expression of IGF1R and phospho-IGF1R 2 hours after plating on matrigel in base medium with or without insulin. Quantification is shown in Supporting Information Figure S4B . (D): Western blot showing the protein expression of focal adhesion kinase (FAK), phospho-FAK-Y397, Paxillin, and phospho-Paxillin-Y118 2 hours after plating on matrigel in base medium with or without insulin. Quantification is shown in Supporting Information Figure S4C . (E): Immunofluorescent image showing Paxillin, FAK, and phospho-FAK-Y397 expression in focal adhesions 2 hours after plating on matrigel-coated surface with or without insulin. Scale bar = 20 mum. Quantification is shown in Supporting Information Figure S4E . (F): Immunostaining showing FAK (red) and phospho-INSR/IGF1R-Y1158-Y1162-Y1163 (green) expression in focal adhesions 2 hours after plating on matrigel-coated surface with or without insulin. Nuclei were stained with Hoechst (blue). Scale bar = 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Phosphorylated paxillin (Ser-272) is localized in spindle poles during mitosis. ( A ) Mitotic cells were plated on coverslips and were stained with antibodies against phospho-paxillin (pS272-paxillin, pY31-paxillin or pY118-paxillin) and alpha-tublin. Nuclei were stained using DAPI. pS272-paxillin, but not pY31-paxillin and pY118-paxillin, is localized in the spindle poles. Scale bar: 10 mum. Arrows point to spindle poles. ( B ) Z stack images of cells showing connection of microtubule spindle with pS272-paxillin in the poles. Scale bar, 5 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
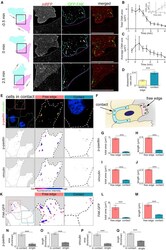
- Figure 2 Cell-Matrix Adhesions Disassemble near the Contact upon Collision (A) Outline of colliding cells showed in zoom of contact area between two cells. Cells outlined in cyan and magenta. The cell labeled in cyan is expressing membrane RFP (mRFP) (red) and GFP-FAK (green). (B and C) Total area (B) and average size (C) of CMAs near the contact upon a collision. 0 min = first frame in contact. n = 15 cells. (B insert) Total area of CMAs near the contact as cells separate. 0 min = last frame in contact n = 3 cells. (D) Disassembly rate of CMAs as indicated. Leading edge, n = 24; contact, n = 27. (E and K) Immunocytochemistry on cells in contact against p-paxillin and vinculin (E) or cells expressing FAK-GFP (K) as indicated. Bottom: color indicating fluorescence intensity. (F) Schematic illustrating how regions of interest were defined for analysis. Contact region illustrated in blue and free edge in pink. (G-J, L, and M) Total area and length of CMAs using the markers indicated. n = 80 cells for (G) and (H); n = 76 cells for (I) and (J); n = 16 cells for (L) and (M). (N-Q) CMA polarity determined by ratio of total area (or length) of p-paxillin (N and O) or vinculin (P and Q) labeled CMAs in free edge over contact or leading edge over trailing edge. Scale bars, 20 mum. Line graphs and box graphs show mean, error: +- SEM. *** p
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot