Antibody data
- Antibody Data
- Antigen structure
- References [39]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Other assay [30]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 71-6900 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-STAT5 alpha (Tyr694) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Bovine
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 50 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Occludin is a target of Src kinase and promotes lipid secretion by binding to BTN1a1 and XOR.
STAT5 is Expressed in CD34(+)/CD38(-) Stem Cells and Serves as a Potential Molecular Target in Ph-Negative Myeloproliferative Neoplasms.
Brain leptin reduces liver lipids by increasing hepatic triglyceride secretion and lowering lipogenesis.
STAT5BN642H is a driver mutation for T cell neoplasia.
Direct targets of pSTAT5 signalling in erythropoiesis.
Elevated collagen-I augments tumor progressive signals, intravasation and metastasis of prolactin-induced estrogen receptor alpha positive mammary tumor cells.
Histone H1 and Chromosomal Protein HMGN2 Regulate Prolactin-induced STAT5 Transcription Factor Recruitment and Function in Breast Cancer Cells.
Mycotoxin ochratoxin A disrupts renal development via a miR-731/prolactin receptor axis in zebrafish.
Prolactin signaling through focal adhesion complexes is amplified by stiff extracellular matrices in breast cancer cells.
Identification of STAT3 and STAT5 proteins in the rat suprachiasmatic nucleus and the Day/Night difference in astrocytic STAT3 phosphorylation in response to lipopolysaccharide.
Common γ-chain cytokine signaling is required for macroautophagy induction during CD4+ T-cell activation.
Cysteine induces longitudinal bone growth in mice by upregulating IGF-I.
Reliable quantification of protein expression and cellular localization in histological sections.
Miz1 deficiency in the mammary gland causes a lactation defect by attenuated Stat5 expression and phosphorylation.
Reciprocal occupancy of BCL6 and STAT5 on Growth Hormone target genes: contrasting transcriptional outcomes and promoter-specific roles of p300 and HDAC3.
Role of STAT5 and epigenetics in lactation-associated upregulation of multidrug transporter ABCG2 in the mammary gland.
Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells.
Induction of multidrug resistance transporter ABCG2 by prolactin in human breast cancer cells.
A humanized pattern of aromatase expression is associated with mammary hyperplasia in mice.
Impairment of hepatic growth hormone and glucocorticoid receptor signaling causes steatosis and hepatocellular carcinoma in mice.
Vascular smooth muscle Jak2 deletion prevents angiotensin II-mediated neointima formation following injury in mice.
Stat5a serine 725 and 779 phosphorylation is a prerequisite for hematopoietic transformation.
Disruption of the growth hormone--signal transducer and activator of transcription 5--insulinlike growth factor 1 axis severely aggravates liver fibrosis in a mouse model of cholestasis.
JAK2, but not Src family kinases, is required for STAT, ERK, and Akt signaling in response to growth hormone in preadipocytes and hepatoma cells.
Mammary epithelial-specific deletion of the focal adhesion kinase gene leads to severe lobulo-alveolar hypoplasia and secretory immaturity of the murine mammary gland.
Direct glucocorticoid receptor-Stat5 interaction in hepatocytes controls body size and maturation-related gene expression.
STAT3 signaling is activated in human skeletal muscle following acute resistance exercise.
Stat5 tetramer formation is associated with leukemogenesis.
Growth hormone receptor expression and function in pituitary adenomas.
Caveolar and lipid raft localization of the growth hormone receptor and its signaling elements: impact on growth hormone signaling.
Glucocorticoid receptor function in hepatocytes is essential to promote postnatal body growth.
Stat5a is tyrosine phosphorylated and nuclear localized in a high proportion of human breast cancers.
Dendritic cell maturation requires STAT1 and is under feedback regulation by suppressors of cytokine signaling.
Distinct pathways involving the FK506-binding proteins 12 and 12.6 underlie IL-2-versus IL-15-mediated proliferation of T cells.
Essential functions of p21-activated kinase 1 in morphogenesis and differentiation of mammary glands.
Inhibition of prolactin (PRL)-induced proliferative signals in breast cancer cells by a molecular mimic of phosphorylated PRL, S179D-PRL.
Dysregulation of mammary Stats 1,3 and 5 and PRL receptors by overexpression of TGFalpha.
Activation of Stat3 in v-Src-transformed fibroblasts requires cooperation of Jak1 kinase activity.
Disulfide linkage of growth hormone (GH) receptors (GHR) reflects GH-induced GHR dimerization. Association of JAK2 with the GHR is enhanced by receptor dimerization.
Lu Y, Zhou T, Xu C, Wang R, Feng D, Li J, Wang X, Kong Y, Hu G, Kong X, Lu P
PLoS biology 2022 Jan;20(1):e3001518
PLoS biology 2022 Jan;20(1):e3001518
STAT5 is Expressed in CD34(+)/CD38(-) Stem Cells and Serves as a Potential Molecular Target in Ph-Negative Myeloproliferative Neoplasms.
Hadzijusufovic E, Keller A, Berger D, Greiner G, Wingelhofer B, Witzeneder N, Ivanov D, Pecnard E, Nivarthi H, Schur FKM, Filik Y, Kornauth C, Neubauer HA, Müllauer L, Tin G, Park J, de Araujo ED, Gunning PT, Hoermann G, Gouilleux F, Kralovics R, Moriggl R, Valent P
Cancers 2020 Apr 21;12(4)
Cancers 2020 Apr 21;12(4)
Brain leptin reduces liver lipids by increasing hepatic triglyceride secretion and lowering lipogenesis.
Hackl MT, Fürnsinn C, Schuh CM, Krssak M, Carli F, Guerra S, Freudenthaler A, Baumgartner-Parzer S, Helbich TH, Luger A, Zeyda M, Gastaldelli A, Buettner C, Scherer T
Nature communications 2019 Jun 20;10(1):2717
Nature communications 2019 Jun 20;10(1):2717
STAT5BN642H is a driver mutation for T cell neoplasia.
Pham HTT, Maurer B, Prchal-Murphy M, Grausenburger R, Grundschober E, Javaheri T, Nivarthi H, Boersma A, Kolbe T, Elabd M, Halbritter F, Pencik J, Kazemi Z, Grebien F, Hengstschläger M, Kenner L, Kubicek S, Farlik M, Bock C, Valent P, Müller M, Rülicke T, Sexl V, Moriggl R
The Journal of clinical investigation 2018 Jan 2;128(1):387-401
The Journal of clinical investigation 2018 Jan 2;128(1):387-401
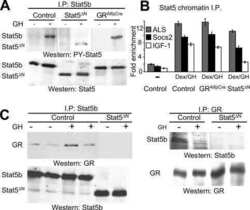
Direct targets of pSTAT5 signalling in erythropoiesis.
Gillinder KR, Tuckey H, Bell CC, Magor GW, Huang S, Ilsley MD, Perkins AC
PloS one 2017;12(7):e0180922
PloS one 2017;12(7):e0180922
Elevated collagen-I augments tumor progressive signals, intravasation and metastasis of prolactin-induced estrogen receptor alpha positive mammary tumor cells.
Barcus CE, O'Leary KA, Brockman JL, Rugowski DE, Liu Y, Garcia N, Yu M, Keely PJ, Eliceiri KW, Schuler LA
Breast cancer research : BCR 2017 Jan 19;19(1):9
Breast cancer research : BCR 2017 Jan 19;19(1):9
Histone H1 and Chromosomal Protein HMGN2 Regulate Prolactin-induced STAT5 Transcription Factor Recruitment and Function in Breast Cancer Cells.
Schauwecker SM, Kim JJ, Licht JD, Clevenger CV
The Journal of biological chemistry 2017 Feb 10;292(6):2237-2254
The Journal of biological chemistry 2017 Feb 10;292(6):2237-2254
Mycotoxin ochratoxin A disrupts renal development via a miR-731/prolactin receptor axis in zebrafish.
Wu TS, Yang JJ, Wang YW, Yu FY, Liu BH
Toxicology research 2016 Mar 1;5(2):519-529
Toxicology research 2016 Mar 1;5(2):519-529
Prolactin signaling through focal adhesion complexes is amplified by stiff extracellular matrices in breast cancer cells.
Barcus CE, Keely PJ, Eliceiri KW, Schuler LA
Oncotarget 2016 Jul 26;7(30):48093-48106
Oncotarget 2016 Jul 26;7(30):48093-48106
Identification of STAT3 and STAT5 proteins in the rat suprachiasmatic nucleus and the Day/Night difference in astrocytic STAT3 phosphorylation in response to lipopolysaccharide.
Moravcová S, Červená K, Pačesová D, Bendová Z
Journal of neuroscience research 2016 Jan;94(1):99-108
Journal of neuroscience research 2016 Jan;94(1):99-108
Common γ-chain cytokine signaling is required for macroautophagy induction during CD4+ T-cell activation.
Botbol Y, Patel B, Macian F
Autophagy 2015;11(10):1864-77
Autophagy 2015;11(10):1864-77
Cysteine induces longitudinal bone growth in mice by upregulating IGF-I.
Moon PD, Kim MH, Oh HA, Nam SY, Han NR, Jeong HJ, Kim HM
International journal of molecular medicine 2015 Aug;36(2):571-6
International journal of molecular medicine 2015 Aug;36(2):571-6
Reliable quantification of protein expression and cellular localization in histological sections.
Schlederer M, Mueller KM, Haybaeck J, Heider S, Huttary N, Rosner M, Hengstschläger M, Moriggl R, Dolznig H, Kenner L
PloS one 2014;9(7):e100822
PloS one 2014;9(7):e100822
Miz1 deficiency in the mammary gland causes a lactation defect by attenuated Stat5 expression and phosphorylation.
Sanz-Moreno A, Fuhrmann D, Wolf E, von Eyss B, Eilers M, Elsässer HP
PloS one 2014;9(2):e89187
PloS one 2014;9(2):e89187
Reciprocal occupancy of BCL6 and STAT5 on Growth Hormone target genes: contrasting transcriptional outcomes and promoter-specific roles of p300 and HDAC3.
Lin G, LaPensee CR, Qin ZS, Schwartz J
Molecular and cellular endocrinology 2014 Sep;395(1-2):19-31
Molecular and cellular endocrinology 2014 Sep;395(1-2):19-31
Role of STAT5 and epigenetics in lactation-associated upregulation of multidrug transporter ABCG2 in the mammary gland.
Wu AM, Yang M, Dalvi P, Turinsky AL, Wang W, Butcher D, Egan SE, Weksberg R, Harper PA, Ito S
American journal of physiology. Endocrinology and metabolism 2014 Oct 1;307(7):E596-610
American journal of physiology. Endocrinology and metabolism 2014 Oct 1;307(7):E596-610
Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells.
Barcus CE, Keely PJ, Eliceiri KW, Schuler LA
The Journal of biological chemistry 2013 May 3;288(18):12722-32
The Journal of biological chemistry 2013 May 3;288(18):12722-32
Induction of multidrug resistance transporter ABCG2 by prolactin in human breast cancer cells.
Wu AM, Dalvi P, Lu X, Yang M, Riddick DS, Matthews J, Clevenger CV, Ross DD, Harper PA, Ito S
Molecular pharmacology 2013 Feb;83(2):377-88
Molecular pharmacology 2013 Feb;83(2):377-88
A humanized pattern of aromatase expression is associated with mammary hyperplasia in mice.
Zhao H, Pearson EK, Brooks DC, Coon JS 5th, Chen D, Demura M, Zhang M, Clevenger CV, Xu X, Veenstra TD, Chatterton RT, DeMayo FJ, Bulun SE
Endocrinology 2012 Jun;153(6):2701-13
Endocrinology 2012 Jun;153(6):2701-13
Impairment of hepatic growth hormone and glucocorticoid receptor signaling causes steatosis and hepatocellular carcinoma in mice.
Mueller KM, Kornfeld JW, Friedbichler K, Blaas L, Egger G, Esterbauer H, Hasselblatt P, Schlederer M, Haindl S, Wagner KU, Engblom D, Haemmerle G, Kratky D, Sexl V, Kenner L, Kozlov AV, Terracciano L, Zechner R, Schuetz G, Casanova E, Pospisilik JA, Heim MH, Moriggl R
Hepatology (Baltimore, Md.) 2011 Oct;54(4):1398-409
Hepatology (Baltimore, Md.) 2011 Oct;54(4):1398-409
Vascular smooth muscle Jak2 deletion prevents angiotensin II-mediated neointima formation following injury in mice.
Kirabo A, Oh SP, Kasahara H, Wagner KU, Sayeski PP
Journal of molecular and cellular cardiology 2011 Jun;50(6):1026-34
Journal of molecular and cellular cardiology 2011 Jun;50(6):1026-34
Stat5a serine 725 and 779 phosphorylation is a prerequisite for hematopoietic transformation.
Friedbichler K, Kerenyi MA, Kovacic B, Li G, Hoelbl A, Yahiaoui S, Sexl V, Müllner EW, Fajmann S, Cerny-Reiterer S, Valent P, Beug H, Gouilleux F, Bunting KD, Moriggl R
Blood 2010 Sep 2;116(9):1548-58
Blood 2010 Sep 2;116(9):1548-58
Disruption of the growth hormone--signal transducer and activator of transcription 5--insulinlike growth factor 1 axis severely aggravates liver fibrosis in a mouse model of cholestasis.
Blaas L, Kornfeld JW, Schramek D, Musteanu M, Zollner G, Gumhold J, van Zijl F, Schneller D, Esterbauer H, Egger G, Mair M, Kenner L, Mikulits W, Eferl R, Moriggl R, Penninger J, Trauner M, Casanova E
Hepatology (Baltimore, Md.) 2010 Apr;51(4):1319-26
Hepatology (Baltimore, Md.) 2010 Apr;51(4):1319-26
JAK2, but not Src family kinases, is required for STAT, ERK, and Akt signaling in response to growth hormone in preadipocytes and hepatoma cells.
Jin H, Lanning NJ, Carter-Su C
Molecular endocrinology (Baltimore, Md.) 2008 Aug;22(8):1825-41
Molecular endocrinology (Baltimore, Md.) 2008 Aug;22(8):1825-41
Mammary epithelial-specific deletion of the focal adhesion kinase gene leads to severe lobulo-alveolar hypoplasia and secretory immaturity of the murine mammary gland.
Nagy T, Wei H, Shen TL, Peng X, Liang CC, Gan B, Guan JL
The Journal of biological chemistry 2007 Oct 26;282(43):31766-76
The Journal of biological chemistry 2007 Oct 26;282(43):31766-76
Direct glucocorticoid receptor-Stat5 interaction in hepatocytes controls body size and maturation-related gene expression.
Engblom D, Kornfeld JW, Schwake L, Tronche F, Reimann A, Beug H, Hennighausen L, Moriggl R, Schütz G
Genes & development 2007 May 15;21(10):1157-62
Genes & development 2007 May 15;21(10):1157-62
STAT3 signaling is activated in human skeletal muscle following acute resistance exercise.
Trenerry MK, Carey KA, Ward AC, Cameron-Smith D
Journal of applied physiology (Bethesda, Md. : 1985) 2007 Apr;102(4):1483-9
Journal of applied physiology (Bethesda, Md. : 1985) 2007 Apr;102(4):1483-9
Stat5 tetramer formation is associated with leukemogenesis.
Moriggl R, Sexl V, Kenner L, Duntsch C, Stangl K, Gingras S, Hoffmeyer A, Bauer A, Piekorz R, Wang D, Bunting KD, Wagner EF, Sonneck K, Valent P, Ihle JN, Beug H
Cancer cell 2005 Jan;7(1):87-99
Cancer cell 2005 Jan;7(1):87-99
Growth hormone receptor expression and function in pituitary adenomas.
Clausen LR, Kristiansen MT, Rasmussen LM, Billestrup N, Blaabjerg O, Ledet T, Jørgensen JO
Clinical endocrinology 2004 May;60(5):576-83
Clinical endocrinology 2004 May;60(5):576-83
Caveolar and lipid raft localization of the growth hormone receptor and its signaling elements: impact on growth hormone signaling.
Yang N, Huang Y, Jiang J, Frank SJ
The Journal of biological chemistry 2004 May 14;279(20):20898-905
The Journal of biological chemistry 2004 May 14;279(20):20898-905
Glucocorticoid receptor function in hepatocytes is essential to promote postnatal body growth.
Tronche F, Opherk C, Moriggl R, Kellendonk C, Reimann A, Schwake L, Reichardt HM, Stangl K, Gau D, Hoeflich A, Beug H, Schmid W, Schütz G
Genes & development 2004 Mar 1;18(5):492-7
Genes & development 2004 Mar 1;18(5):492-7
Stat5a is tyrosine phosphorylated and nuclear localized in a high proportion of human breast cancers.
Cotarla I, Ren S, Zhang Y, Gehan E, Singh B, Furth PA
International journal of cancer 2004 Feb 20;108(5):665-71
International journal of cancer 2004 Feb 20;108(5):665-71
Dendritic cell maturation requires STAT1 and is under feedback regulation by suppressors of cytokine signaling.
Jackson SH, Yu CR, Mahdi RM, Ebong S, Egwuagu CE
Journal of immunology (Baltimore, Md. : 1950) 2004 Feb 15;172(4):2307-15
Journal of immunology (Baltimore, Md. : 1950) 2004 Feb 15;172(4):2307-15
Distinct pathways involving the FK506-binding proteins 12 and 12.6 underlie IL-2-versus IL-15-mediated proliferation of T cells.
Dubois S, Shou W, Haneline LS, Fleischer S, Waldmann TA, Müller JR
Proceedings of the National Academy of Sciences of the United States of America 2003 Nov 25;100(24):14169-74
Proceedings of the National Academy of Sciences of the United States of America 2003 Nov 25;100(24):14169-74
Essential functions of p21-activated kinase 1 in morphogenesis and differentiation of mammary glands.
Wang RA, Vadlamudi RK, Bagheri-Yarmand R, Beuvink I, Hynes NE, Kumar R
The Journal of cell biology 2003 May 12;161(3):583-92
The Journal of cell biology 2003 May 12;161(3):583-92
Inhibition of prolactin (PRL)-induced proliferative signals in breast cancer cells by a molecular mimic of phosphorylated PRL, S179D-PRL.
Schroeder MD, Brockman JL, Walker AM, Schuler LA
Endocrinology 2003 Dec;144(12):5300-7
Endocrinology 2003 Dec;144(12):5300-7
Dysregulation of mammary Stats 1,3 and 5 and PRL receptors by overexpression of TGFalpha.
Schroeder MD, Rose-Hellekant TA, Sandgren EP, Schuler LA
Molecular and cellular endocrinology 2001 Apr 25;175(1-2):173-83
Molecular and cellular endocrinology 2001 Apr 25;175(1-2):173-83
Activation of Stat3 in v-Src-transformed fibroblasts requires cooperation of Jak1 kinase activity.
Zhang Y, Turkson J, Carter-Su C, Smithgall T, Levitzki A, Kraker A, Krolewski JJ, Medveczky P, Jove R
The Journal of biological chemistry 2000 Aug 11;275(32):24935-44
The Journal of biological chemistry 2000 Aug 11;275(32):24935-44
Disulfide linkage of growth hormone (GH) receptors (GHR) reflects GH-induced GHR dimerization. Association of JAK2 with the GHR is enhanced by receptor dimerization.
Zhang Y, Jiang J, Kopchick JJ, Frank SJ
The Journal of biological chemistry 1999 Nov 12;274(46):33072-84
The Journal of biological chemistry 1999 Nov 12;274(46):33072-84
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of STAT5 (pY694) was performed by loading 20 µg of HeLa (lane1), HeLa treated for 60 minutes with 100 ng/mL of IFN alpha (lane2) and HeLa treated for 60 minutes with 100 ng/mL of IFN gamma (lane3) cell lysates using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800). Proteins were transferred to a PVDF membrane and blocked with 5 % skim milk for 1 hour at room temperature. STAT5 (pY641) was detected at ~90 kDa using STAT5 (pY694) Rabbit polyclonal Antibody (Product # 71-6900) at 0.5 µg - 1 µg/mL in 2.5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of STAT5 (pY694) was done on 70% confluent log phase HeLa cells; untreated & treated (serum starved for 16 hours followed by treatment with 20 ng/mL IFN alpha for 1 hour). The cells were fixed with 4% paraformaldehyde for 15 minutes; permeabilized with 0.25% Triton™ X-100 for 10 minutes followed by blocking with 5% BSA for 1 hour at room temperature. The cells were incubated with STAT5 (pY694) Rabbit Polyclonal Antibody (Product # 71-6900) at 2 µg - 4 µg in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor® 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 594 Phalloidin (Product # A12381). Panel d is a merged image showing translocated STAT5 (pY694) in nucleus. Panel e shows no primary antibody. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
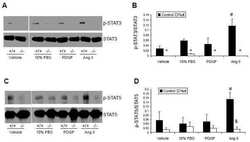
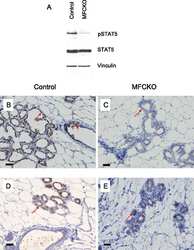
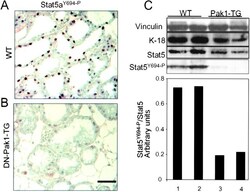
- Figure 7. DN-Pak1-TG mammary glands show decreased Stat5 tyrosine phosphorylation and expression. (A and B) Immunohistochemical staining of phospho-Stat5a in mammary glands obtained from wild-type and Pak1-TG mice on lactation day 5. (C) Effect of Pak1 inactivation on the levels of Stat5a and phosphorylated Stat5a by immunoblot analysis. Bar: (A and B) 20 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
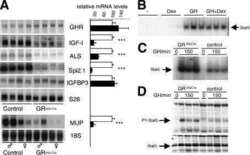
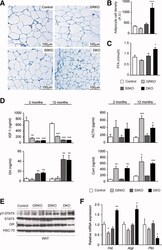
- Fig. 2 Combination of hepatic GH resistance and hypercortisolism causes peripheral lipodystrophy in DKO animals. (A) Histology of WAT. Epigonadal WAT of 12-month-old mice was stained with hematoxylin and eosin. (B) Quantification of WAT cell density. Hematoxylin and eosin-stained sections were used to analyze cell density using HistoQuest image analysis (TissueGnostics GmbH, Vienna, Austria). (C) Levels of FFA were determined in 2-month-old mice using colorimetric assays. (D) Levels of GH and IGF-1 were determined by ELISA. Levels of corticosterone (Cort) and ACTH were determined by radioimmunoassay (n = 8/genotype/time point). (E) Representative western blotting analysis of WAT homogenates from 2-month-old mice. Expression levels of STAT5 and GR proteins were determined using specific antibodies. HSC-70 served as the loading control. (F) Relative mRNA levels of Hsl , Atgl , and Plin were quantified by qRT-PCR in WAT from 6-month-old mice. Ct values were normalized to GAPDH (DeltaCt method; n = 4/genotype). * P < 0.05; ** P < 0.01; *** P < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
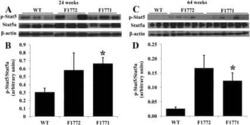
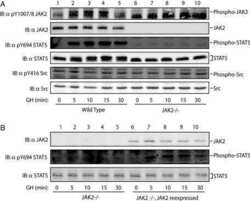
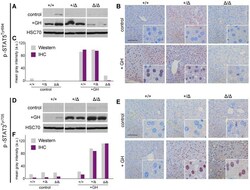
- Figure 2 p-STAT5 Tyr694 and p-STAT3 Tyr705 expression upon GH treatment compared to control mice. A Western Blot of p-STAT5 Tyr694 expression in Stat5ab +/+ (n = 2), Stat5ab +/Delta (n = 2) and Stat5ab Delta/Delta (n = 2) livers of GH treated and control mice. HSC70 is shown as loading control. B IHC of p-STAT5 Tyr694 in sections of the livers (n = 3) used in A. Red, AEC; blue, hematoxylin. Size bar: 100 um. High magnifications (60x) are shown in insets. C Quantification of p-STAT5 Tyr694 levels from Western blots (grey) by densitometric scanning and IHC (magenta) using the HistoQuest software ( Stat5ab +/+ , control; number of cells n = 3276, GH; number of cells n = 2286; Stat5ab +/Delta control; number of cells n = 2577, GH; number of cells n = 3549; Stat5ab Delta/Delta control; number of cells n = 3299, GH; number of cells n = 2200). The highest arbitrary expression values from A and B were set to 100 for direct comparison. D Western blot of p-STAT3 Tyr705 expression in Stat5ab +/+ (n = 2), Stat5ab +/Delta (n = 2) and Stat5ab Delta/Delta (n = 2) livers of GH treated and control mice. HSC70 is shown as loading control. E IHC of p-STAT3 Tyr705 in sections of the livers (n = 3) used in D. Red, AEC; blue, hematoxylin. Size bar: 100 um. High magnifications are shown in insets (60x). F Quantification of p-STAT3 Tyr705 levels from Western blots (grey) by densitometric scanning and IHC (magenta) using the HistoQuest software ( Stat5ab +/+ , control; number of cells n = 2634, GH; n
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
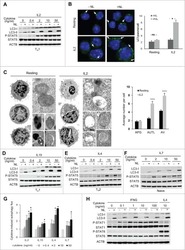
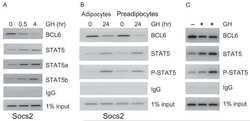
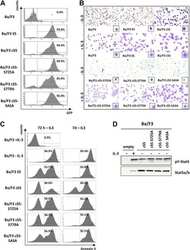
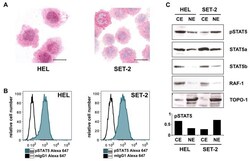
- Figure 3 ( A ) Cytospin preparations of HEL cells and SET-2 cells were stained overnight with an anti-pSTAT5 antibody using immunocytochemistry. Scale bar: 20 uM. ( B ) HEL cells and SET-2 cells were stained with an anti-pSTAT5 Alexa-647 antibody for 30 min at room temperature and intracellular expression levels were analyzed by flow cytometry. ( C ) Nuclear (NE) and cytoplasmic (CE) fractions of HEL or SET-2 cells were analyzed for expression of phosphorylated STAT5 (pSTAT5), total STAT5a and total STAT5b by Western blotting. Antibodies against RAF-1 (cytoplasm) and TOPO-1 (nucleus) were used as fraction controls. The columns show the densitometry for pSTAT5. Uncropped blots are shown in Figure S1 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
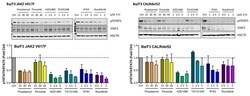
- Figure 4 Ba/F3 cells modified to express JAK2 V617F or CALRdel52 were incubated with various JAK2 or STAT5 inhibitors (as indicated) for four hours at 37 degC. Thereafter, the expression of activated STAT5 (pSTAT5), total STAT5 (STAT5) and heat shock cognate 70 (HSC70), which was used as loading control, was analyzed using Western blotting. The results were quantified using densitometry and are expressed as the ratio of activated and total STAT5 normalized to the results obtained with control cells (Ctrl). Uncropped blots are shown in Figure S2 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
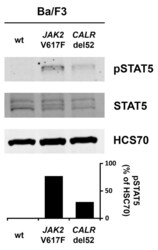
- Figure A3 Wild type (wt) Ba/F3 cells or Ba/F3 cells transfected with either JAK2 V617F or CALRdel52 were incubated overnight in medium without IL-3. After, cell lysates were subjected to Western blotting using antibodies against pSTAT5, STAT5 and HSC70. The columns show the levels of pSTAT5 in percent of HSC70 as analyzed by densitometry. Uncropped blots are shown in Figure S3 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
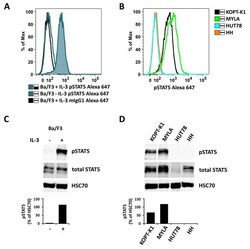
- Figure A5 Ba/F3 cells were kept in medium without IL-3 overnight and split in the morning. One half was incubated with 10 ng/mL IL-3 for 40 min (Ba/F3 + IL-3), while the other half was kept in medium without IL-3 (Ba/F3 - IL-3). Thereafter, cells were analyzed for expression of pSTAT5 either by flow cytometry ( A ) or Western blotting ( C ). In ( A ), an Alexa-647-labeled anti-pSTAT5 antibody was used (with IL-3: blue histogram; without IL-3: open blue histogram). mIgG1 Alexa-647 was used as an isotype control (open black histogram) in Ba/F3 + IL-3 cells ( A ). Human lymphoma cell lines with detectable pSTAT5 levels (KOPT-K1 and MYLA) or no detectable pSTAT5 (HUT78 and HH) were also analyzed for pSTAT5 expression using flow cytometry ( B ) and Western blotting ( D ). In Western blot experiments, HSC70 served as a loading control. The columns show the levels of pSTAT5 in percent of HSC70 as analyzed by densitometry. Uncropped blots are shown in Figure S4 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
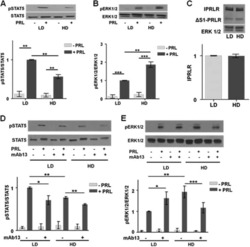
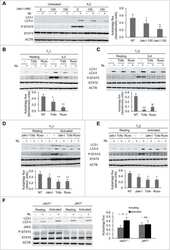
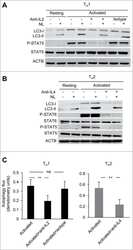
- Figure 2 Collagen ligand density does not modulate PRL signals to ERK1/2 or FAK A-C. T47D were cells plated on 25 kPa polyacrylamide gels coated with either 50, 200, or 800 mug/ml collagen-I, serum starved for 24h, then treated +- PRL (4 nM) for 15 min. Cell lysates were immunoblotted with the indicated antibodies. Top panels: Representative immunoblots. Bottom panels: Quantification of immunoblots by densitometry. Means +- S.E.M. n = 3. Different letters represent significant differences between treatments, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
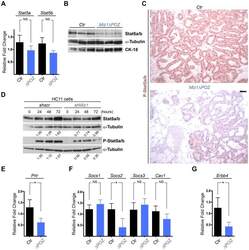
- Figure 5 Stat5 phosphorylation is reduced in Miz1 Delta POZ mammary glands and in shMiz1 HC11 cells. ( A ) Analysis of Stat5a/b mRNA expression in Miz1 Delta POZ (DeltaPOZ) and control mammary gland tissue ( Ctr ). ( B ) Immunoblot analysis of Stat5 in wildtype and Miz1 Delta POZ mammary glands. ( C ) Immunohistochemical staining of pStat5 in the mammary gland tissue from wildtype and Miz1 Delta POZ animals. ( D ) Western blots from HC11 cells stably transfected with a scrambled short hairpin (sh) RNA (shscr) or a Miz1 shRNA (see also Fig. 4 E and F ). Numbers indicated are fold changes of band intensities obtained by densitometry (see Materials and Methods). Quantitative RT-PCR for the prolactin receptor ( E ; Prlr ), the Supressors of cytokine signalling ( Socs ) 1, 2 and 3 and caveolin-1 ( F ; Cav1 ), and for ErbB4 ( G ). Lactation day 6 samples were used in all in vivo experiments. Scale bar in C: 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
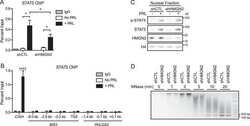
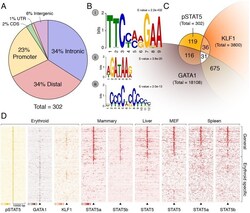
- Fig 1 pSTAT5 binds promoters and enhancers of key erythroid genes in concert with GATA1 and KLF1. (A) 302 peaks were annotated with respect to the nearest TSS as defined by RefSeq. Only ~23% of peaks reside within gene promoters (1 kb upstream or 100 nt downstream of a TSS). The majority are intronic or distal (within 50 kb of a gene body) consistent with enhancer occupancy. (B) De novo motif discovery on all 302 peaks using MEME (see Methods ) identified highly significant enrichment of a (i) palindromic GAS element (TTCYMRGAA), a (ii) GATA binding site (WGATAR), and a (iii) KLF binding site (CCMCRCCCN). No other significantly enriched motifs were found. (C) Venn diagram and of erythroid co-occupancy between pSTAT5, KLF1 and GATA1 at key erythroid enhancers and promoters. KLF1 ChIP-seq was generated in K1-ER cells and GATA1 ChIP-seq was generated in G1-ER4 cells after induction with tamoxifen. Co-occupancy defined as ChIP summits within 500 bp. (D) Density heat-map of ChIP signal centred on pSTAT5 peak summits from GATA1 and KLF1 as above (C) and additional STAT5 ChIP in other murine tissues. The Y-axis represents individual peak regions, and the X-axis represents 10 kb surrounding the summit. Read intensities were normalized to number of reads in dataset and hierarchically clustered according to intensity within 500 bp of peak centre.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
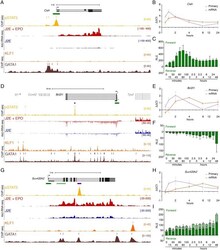
- Fig 3 Immediate direct transcriptional targets of EPOR-pSTAT5 signalling. (A) ChIP-seq and 4sU-RNA-seq in erythroid cells following 30 mins of EPO induction across the Cish gene. Read density per million mapped reads (y-axis) for pSTAT5 (ochre), KLF1 (orange) and GATA1 (brown) ChIP. Coloured bars above each track represent peak summits as called by MACS2. Read density profiles (y-axis) for 4sU-RNA-seq from J2E cells (blue) and J2E cells post EPO-induction (red) are displayed for both forward (+ values) and reverse (- values) strands. A schematic of gene structure and alternative transcripts from RefSeq is shown in black with scale bar. The green bar represents the region selected for CAGE tag counts shown in (C). (B) qRT-PCR for Cish primary pre-spliced transcripts and processed mRNA over a 24-hour time course following EPO stimulation. (C) CAGE tag counts expressed as relative log expression (RLE) over the indicated pSTAT5-occupied promoter (green bar in panel A) from 0 to 24 hours post EPO stimulation. ChIP-seq and 4sU-RNA-seq across the Bcl2l1 (D) and Suv420h2 (G) genes. Overall design and colour coding of tracks is the same as for (A). Purple bars indicate pSTAT5-occupied intronic enhancers. Dynamic EPO-induced CAGE tags over this region are shown in S4 Fig . qRT-PCR for Bcl2l1 (E) and Suv420h2 (H) primary pre-spliced transcripts and processed mRNA over a 24-hour time course following EPO stimulation as in (B). CAGE tag counts for Bcl2l1 (E) and Suv420h2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
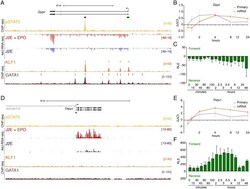
- Fig 4 Delayed pSTAT5-dependent and independent EPO-induced gene expression. ChIP-seq and 4sU-RNA-seq in erythroid cells following 30 mins of EPO induction across the Gypc (A) and Thbs1 (D) genes. The overall design and colour coding of tracks is the same as for Fig 3 . qRT-PCR and CAGE tag counts as in Fig 3B and 3C for Gypc and Fig 3E and 3F for Thbs1 respectively. Bidirectional CAGE tags over the intronic enhancer (purple bar in panel A) are shown in S4C Fig . Gypc provides an example of delayed transcriptional activation by pSTAT5, whereas Thbs1 appears to be activated independently of pSTAT5.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA