Antibody data
- Antibody Data
- Antigen structure
- References [46]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Other assay [35]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-183 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Actin Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Purifed from natural sources
- Description
- By Western blot, PA1-183 detects a prominent ~42kD protein in human, mouse, non-human primate, and rat whole cell lysates. For visualization of actin filaments by immunofluorescence, fixation and permeabilization with methanol is required.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references DNMT1-Mediated DNA Methylation Targets CDKN2B to Promote the Repair of Retinal Ganglion Cells in Streptozotocin-Induced Mongolian Gerbils during Diabetic Retinopathy.
KISS1R and ANKRD31 Cooperate to Enhance Leydig Cell Gene Expression via the Cytoskeletal-Nucleoskeletal Pathway.
Downregulation of miR-375 contributes to ERBB2-mediated VEGFA overexpression in esophageal cancer.
Metallothionein 3-Zinc Axis Suppresses Caspase-11 Inflammasome Activation and Impairs Antibacterial Immunity.
Leflunomide Induces Dose-Dependent Lung Injury in Mice via Stimulating Vimentin and NLRP3 Inflammasome Production.
microRNA-196b promotes esophageal squamous cell carcinogenesis and chemoradioresistance by inhibiting EPHA7, thereby restoring EPHA2 activity.
Surface activation with oxygen plasma promotes osteogenesis with enhanced extracellular matrix formation in three-dimensional microporous scaffolds.
Social isolation-related depression accelerates ethanol intake via microglia-derived neuroinflammation.
Impact of the molar activity and PSMA expression level on [(18)F]AlF-PSMA-11 uptake in prostate cancer.
Microtubule-sliding modules based on kinesins EG5 and PRC1-dependent KIF4A drive human spindle elongation.
Quantitative Proteomic Approach Reveals Altered Metabolic Pathways in Response to the Inhibition of Lysine Deacetylases in A549 Cells under Normoxia and Hypoxia.
Utilizing an Animal Model to Identify Brain Neurodegeneration-Related Biomarkers in Aging.
IL-9 Abrogates the Metastatic Potential of Breast Cancer by Controlling Extracellular Matrix Remodeling and Cellular Contractility.
Regulation of tumor immune suppression and cancer cell survival by CXCL1/2 elevation in glioblastoma multiforme.
Coumestrol mitigates retinal cell inflammation, apoptosis, and oxidative stress in a rat model of diabetic retinopathy via activation of SIRT1.
Bcl2l10 induces metabolic alterations in ovarian cancer cells by regulating the TCA cycle enzymes SDHD and IDH1.
Metformin Protects From Rotenone-Induced Nigrostriatal Neuronal Death in Adult Mice by Activating AMPK-FOXO3 Signaling and Mitigation of Angiogenesis.
Enteropathogenic Escherichia coli Infection Induces Diarrhea, Intestinal Damage, Metabolic Alterations, and Increased Intestinal Permeability in a Murine Model.
miR-4463 regulates aromatase expression and activity for 17β-estradiol synthesis in response to follicle-stimulating hormone.
Activated ROCK/Akt/eNOS and ET-1/ERK pathways in 5-fluorouracil-induced cardiotoxicity: modulation by simvastatin.
Neuroprotective Fragment C of Tetanus Toxin Modulates IL-6 in an ALS Mouse Model.
DAPK3 participates in the mRNA processing of immediate early genes in chronic lymphocytic leukaemia.
Influences of circulatory factors on intervertebral disc aging phenotype.
Bcl2l10 mediates the proliferation, invasion and migration of ovarian cancer cells.
Systematic Development and Optimization of Inhalable Pirfenidone Liposomes for Non-Small Cell Lung Cancer Treatment.
Effects of suppressing bioavailability of insulin-like growth factor on age-associated intervertebral disc degeneration.
A point mutation decouples the lipid transfer activities of microsomal triglyceride transfer protein.
Actively Targeted Nanodelivery of Echinomycin Induces Autophagy-Mediated Death in Chemoresistant Pancreatic Cancer In Vivo.
Silymarin, boswellic acid and curcumin enriched dietetic formulation reduces the growth of inherited intestinal polyps in an animal model.
Pirfenidone suppresses the abnormal activation of human Müller cells after platelet-derived growth factor-BB stimulation.
Chenodeoxycholic Acid Ameliorates AlCl(3)-Induced Alzheimer's Disease Neurotoxicity and Cognitive Deterioration via Enhanced Insulin Signaling in Rats.
Role of HDAC1 in the progression of gastric cancer and the correlation with lncRNAs.
Systemic clearance of p16(INK4a) -positive senescent cells mitigates age-associated intervertebral disc degeneration.
Contractile acto-myosin network on nuclear envelope remnants positions human chromosomes for mitosis.
KDEL receptor regulates secretion by lysosome relocation- and autophagy-dependent modulation of lipid-droplet turnover.
Chronic high-fat diet decreased detrusor mitochondrial respiration and increased nerve-mediated contractions.
Downregulation of ROS1 enhances the therapeutic efficacy of arsenic trioxide in acute myeloid leukemia cell lines.
Blockade of soluble epoxide hydrolase attenuates post-ischemic neuronal hyperexcitation and confers resilience against stroke with TrkB activation.
Grifolic acid induces mitochondrial membrane potential loss and cell death of RAW264.7 macrophages.
Fenofibrate Administration Reduces Alcohol and Saccharin Intake in Rats: Possible Effects at Peripheral and Central Levels.
Red-shifted luciferase-luciferin pairs for enhanced bioluminescence imaging.
1,25-(OH)(2)D(3) and its analogue BXL-628 inhibit high glucose-induced activation of RhoA/ROCK pathway in HK-2 cells.
Unlocking Aspirin's Chemopreventive Activity: Role of Irreversibly Inhibiting Platelet Cyclooxygenase-1.
Crocin Inhibits Oxidative Stress and Pro-inflammatory Response of Microglial Cells Associated with Diabetic Retinopathy Through the Activation of PI3K/Akt Signaling Pathway.
Study of the Effects of Betaine and/or C-Phycocyanin on the Growth of Lung Cancer A549 Cells In Vitro and In Vivo.
Overexpression of the pituitary tumor transforming gene upregulates metastasis in malignant neoplasms of the human salivary glands.
Wang X, Zhang J, Liao Y, Jin Y, Yu X, Li H, Yang Q, Li X, Chen R, Wu D, Zhu H
Computational and mathematical methods in medicine 2022;2022:9212116
Computational and mathematical methods in medicine 2022;2022:9212116
KISS1R and ANKRD31 Cooperate to Enhance Leydig Cell Gene Expression via the Cytoskeletal-Nucleoskeletal Pathway.
Ricci G, Guillou F, Catizone A, Mele VG, Moggio M, Chioccarelli T, Diano N, Meccariello R, Pierantoni R, Fasano S, Cobellis G, Chianese R, Manfrevola F
Frontiers in cell and developmental biology 2022;10:877270
Frontiers in cell and developmental biology 2022;10:877270
Downregulation of miR-375 contributes to ERBB2-mediated VEGFA overexpression in esophageal cancer.
Ren S, Tan X, Fu MZ, Ren S, Wu X, Chen T, Latham PS, Lin P, Man YG, Fu SW
Journal of Cancer 2021;12(23):7138-7146
Journal of Cancer 2021;12(23):7138-7146
Metallothionein 3-Zinc Axis Suppresses Caspase-11 Inflammasome Activation and Impairs Antibacterial Immunity.
Chowdhury D, Gardner JC, Satpati A, Nookala S, Mukundan S, Porollo A, Landero Figueroa JA, Subramanian Vignesh K
Frontiers in immunology 2021;12:755961
Frontiers in immunology 2021;12:755961
Leflunomide Induces Dose-Dependent Lung Injury in Mice via Stimulating Vimentin and NLRP3 Inflammasome Production.
El-Sherbiny M, Atef H, Eladl MA, Mohamed AS, El-Shafey M, Ali HS, Zaitone SA, Alomar SY, Alqahtani SAM, Aloyouni SY, Attia MA
Frontiers in pharmacology 2021;12:631216
Frontiers in pharmacology 2021;12:631216
microRNA-196b promotes esophageal squamous cell carcinogenesis and chemoradioresistance by inhibiting EPHA7, thereby restoring EPHA2 activity.
Tan X, Ren S, Fu MZ, Ren S, Yang C, Wu X, Chen T, Latham PS, Meltzer SJ, Fu SW
American journal of cancer research 2021;11(7):3594-3610
American journal of cancer research 2021;11(7):3594-3610
Surface activation with oxygen plasma promotes osteogenesis with enhanced extracellular matrix formation in three-dimensional microporous scaffolds.
Yamada S, Yassin MA, Weigel T, Schmitz T, Hansmann J, Mustafa K
Journal of biomedical materials research. Part A 2021 Sep;109(9):1560-1574
Journal of biomedical materials research. Part A 2021 Sep;109(9):1560-1574
Social isolation-related depression accelerates ethanol intake via microglia-derived neuroinflammation.
Lee JS, Lee SB, Kim DW, Shin N, Jeong SJ, Yang CH, Son CG
Science advances 2021 Nov 5;7(45):eabj3400
Science advances 2021 Nov 5;7(45):eabj3400
Impact of the molar activity and PSMA expression level on [(18)F]AlF-PSMA-11 uptake in prostate cancer.
Piron S, Verhoeven J, De Coster E, Descamps B, Kersemans K, Pieters L, Vral A, Vanhove C, De Vos F
Scientific reports 2021 Nov 19;11(1):22623
Scientific reports 2021 Nov 19;11(1):22623
Microtubule-sliding modules based on kinesins EG5 and PRC1-dependent KIF4A drive human spindle elongation.
Vukušić K, Ponjavić I, Buđa R, Risteski P, Tolić IM
Developmental cell 2021 May 3;56(9):1253-1267.e10
Developmental cell 2021 May 3;56(9):1253-1267.e10
Quantitative Proteomic Approach Reveals Altered Metabolic Pathways in Response to the Inhibition of Lysine Deacetylases in A549 Cells under Normoxia and Hypoxia.
Martín-Bernabé A, Tarragó-Celada J, Cunin V, Michelland S, Cortés R, Poignant J, Boyault C, Rachidi W, Bourgoin-Voillard S, Cascante M, Seve M
International journal of molecular sciences 2021 Mar 25;22(7)
International journal of molecular sciences 2021 Mar 25;22(7)
Utilizing an Animal Model to Identify Brain Neurodegeneration-Related Biomarkers in Aging.
Yang MH, Chen YA, Tu SC, Chi PL, Chuang KP, Chang CC, Lee CH, Chen YL, Lee CH, Yuan CH, Tyan YC
International journal of molecular sciences 2021 Mar 23;22(6)
International journal of molecular sciences 2021 Mar 23;22(6)
IL-9 Abrogates the Metastatic Potential of Breast Cancer by Controlling Extracellular Matrix Remodeling and Cellular Contractility.
Das S, Surve V, Marathe S, Wad S, Karulkar A, Srinivasan S, Dwivedi A, Barthel SR, Purwar R
Journal of immunology (Baltimore, Md. : 1950) 2021 Jun 1;206(11):2740-2752
Journal of immunology (Baltimore, Md. : 1950) 2021 Jun 1;206(11):2740-2752
Regulation of tumor immune suppression and cancer cell survival by CXCL1/2 elevation in glioblastoma multiforme.
Hu J, Zhao Q, Kong LY, Wang J, Yan J, Xia X, Jia Z, Heimberger AB, Li S
Science advances 2021 Jan;7(5)
Science advances 2021 Jan;7(5)
Coumestrol mitigates retinal cell inflammation, apoptosis, and oxidative stress in a rat model of diabetic retinopathy via activation of SIRT1.
Xu Y, Zhang Y, Liang H, Liu X
Aging 2021 Feb 1;13(4):5342-5357
Aging 2021 Feb 1;13(4):5342-5357
Bcl2l10 induces metabolic alterations in ovarian cancer cells by regulating the TCA cycle enzymes SDHD and IDH1.
Lee SY, Kwon J, Lee KA
Oncology reports 2021 Apr;45(4)
Oncology reports 2021 Apr;45(4)
Metformin Protects From Rotenone-Induced Nigrostriatal Neuronal Death in Adult Mice by Activating AMPK-FOXO3 Signaling and Mitigation of Angiogenesis.
El-Ghaiesh SH, Bahr HI, Ibrahiem AT, Ghorab D, Alomar SY, Farag NE, Zaitone SA
Frontiers in molecular neuroscience 2020;13:84
Frontiers in molecular neuroscience 2020;13:84
Enteropathogenic Escherichia coli Infection Induces Diarrhea, Intestinal Damage, Metabolic Alterations, and Increased Intestinal Permeability in a Murine Model.
Ledwaba SE, Costa DVS, Bolick DT, Giallourou N, Medeiros PHQS, Swann JR, Traore AN, Potgieter N, Nataro JP, Guerrant RL
Frontiers in cellular and infection microbiology 2020;10:595266
Frontiers in cellular and infection microbiology 2020;10:595266
miR-4463 regulates aromatase expression and activity for 17β-estradiol synthesis in response to follicle-stimulating hormone.
Lee SY, Kang YJ, Kwon J, Nishi Y, Yanase T, Lee KA, Koong MK
Clinical and experimental reproductive medicine 2020 Sep;47(3):194-206
Clinical and experimental reproductive medicine 2020 Sep;47(3):194-206
Activated ROCK/Akt/eNOS and ET-1/ERK pathways in 5-fluorouracil-induced cardiotoxicity: modulation by simvastatin.
Muhammad RN, Sallam N, El-Abhar HS
Scientific reports 2020 Sep 7;10(1):14693
Scientific reports 2020 Sep 7;10(1):14693
Neuroprotective Fragment C of Tetanus Toxin Modulates IL-6 in an ALS Mouse Model.
Moreno-Martinez L, de la Torre M, Muñoz MJ, Zaragoza P, Aguilera J, Calvo AC, Osta R
Toxins 2020 May 17;12(5)
Toxins 2020 May 17;12(5)
DAPK3 participates in the mRNA processing of immediate early genes in chronic lymphocytic leukaemia.
Thomas F, Holmes KB, Kreuz S, Hillmen P, Lefevre PF
Molecular oncology 2020 Jun;14(6):1268-1281
Molecular oncology 2020 Jun;14(6):1268-1281
Influences of circulatory factors on intervertebral disc aging phenotype.
Lei C, Colangelo D, Patil P, Li V, Ngo K, Wang D, Dong Q, Yousefzadeh MJ, Lin H, Lee J, Kang J, Sowa G, Wyss-Coray T, Niedernhofer LJ, Robbins PD, Huffman DM, Vo N
Aging 2020 Jun 11;12(12):12285-12304
Aging 2020 Jun 11;12(12):12285-12304
Bcl2l10 mediates the proliferation, invasion and migration of ovarian cancer cells.
Lee SY, Kwon J, Woo JH, Kim KH, Lee KA
International journal of oncology 2020 Feb;56(2):618-629
International journal of oncology 2020 Feb;56(2):618-629
Systematic Development and Optimization of Inhalable Pirfenidone Liposomes for Non-Small Cell Lung Cancer Treatment.
Parvathaneni V, Kulkarni NS, Shukla SK, Farrales PT, Kunda NK, Muth A, Gupta V
Pharmaceutics 2020 Feb 28;12(3)
Pharmaceutics 2020 Feb 28;12(3)
Effects of suppressing bioavailability of insulin-like growth factor on age-associated intervertebral disc degeneration.
Kritschil R, Zhang Z, Lei C, Zhong J, Dong Q, Lee J, Conover CA, Sowa G, Vallejo AN, Vo N
JOR spine 2020 Dec;3(4):e1112
JOR spine 2020 Dec;3(4):e1112
A point mutation decouples the lipid transfer activities of microsomal triglyceride transfer protein.
Wilson MH, Rajan S, Danoff A, White RJ, Hensley MR, Quinlivan VH, Recacha R, Thierer JH, Tan FJ, Busch-Nentwich EM, Ruddock L, Hussain MM, Farber SA
PLoS genetics 2020 Aug;16(8):e1008941
PLoS genetics 2020 Aug;16(8):e1008941
Actively Targeted Nanodelivery of Echinomycin Induces Autophagy-Mediated Death in Chemoresistant Pancreatic Cancer In Vivo.
Thomas A, Samykutty A, Gomez-Gutierrez JG, Yin W, Egger ME, McNally M, Chuong P, MacCuaig WM, Albeituni S, Zeiderman M, Li M, Edil BH, Grizzle WE, McMasters KM, McNally LR
Cancers 2020 Aug 14;12(8)
Cancers 2020 Aug 14;12(8)
Silymarin, boswellic acid and curcumin enriched dietetic formulation reduces the growth of inherited intestinal polyps in an animal model.
Girardi B, Pricci M, Giorgio F, Piazzolla M, Iannone A, Losurdo G, Principi M, Barone M, Ierardi E, Di Leo A
World journal of gastroenterology 2020 Apr 14;26(14):1601-1612
World journal of gastroenterology 2020 Apr 14;26(14):1601-1612
Pirfenidone suppresses the abnormal activation of human Müller cells after platelet-derived growth factor-BB stimulation.
Tao YJ, Chen Q, Wang L, Yang X, Cun Q, Yang WY, Zhong H
International journal of ophthalmology 2019;12(7):1075-1082
International journal of ophthalmology 2019;12(7):1075-1082
Chenodeoxycholic Acid Ameliorates AlCl(3)-Induced Alzheimer's Disease Neurotoxicity and Cognitive Deterioration via Enhanced Insulin Signaling in Rats.
Bazzari FH, Abdallah DM, El-Abhar HS
Molecules (Basel, Switzerland) 2019 May 24;24(10)
Molecules (Basel, Switzerland) 2019 May 24;24(10)
Role of HDAC1 in the progression of gastric cancer and the correlation with lncRNAs.
Yu Z, Zeng J, Liu H, Wang T, Yu Z, Chen J
Oncology letters 2019 Mar;17(3):3296-3304
Oncology letters 2019 Mar;17(3):3296-3304
Systemic clearance of p16(INK4a) -positive senescent cells mitigates age-associated intervertebral disc degeneration.
Patil P, Dong Q, Wang D, Chang J, Wiley C, Demaria M, Lee J, Kang J, Niedernhofer LJ, Robbins PD, Sowa G, Campisi J, Zhou D, Vo N
Aging cell 2019 Jun;18(3):e12927
Aging cell 2019 Jun;18(3):e12927
Contractile acto-myosin network on nuclear envelope remnants positions human chromosomes for mitosis.
Booth AJR, Yue Z, Eykelenboom JK, Stiff T, Luxton GWG, Hochegger H, Tanaka TU
eLife 2019 Jul 3;8
eLife 2019 Jul 3;8
KDEL receptor regulates secretion by lysosome relocation- and autophagy-dependent modulation of lipid-droplet turnover.
Tapia D, Jiménez T, Zamora C, Espinoza J, Rizzo R, González-Cárdenas A, Fuentes D, Hernández S, Cavieres VA, Soza A, Guzmán F, Arriagada G, Yuseff MI, Mardones GA, Burgos PV, Luini A, González A, Cancino J
Nature communications 2019 Feb 13;10(1):735
Nature communications 2019 Feb 13;10(1):735
Chronic high-fat diet decreased detrusor mitochondrial respiration and increased nerve-mediated contractions.
Powers SA, Ryan TE, Pak ES, Fraser MO, McClung JM, Hannan JL
Neurourology and urodynamics 2019 Aug;38(6):1524-1532
Neurourology and urodynamics 2019 Aug;38(6):1524-1532
Downregulation of ROS1 enhances the therapeutic efficacy of arsenic trioxide in acute myeloid leukemia cell lines.
Li J
Oncology letters 2018 Jun;15(6):9392-9396
Oncology letters 2018 Jun;15(6):9392-9396
Blockade of soluble epoxide hydrolase attenuates post-ischemic neuronal hyperexcitation and confers resilience against stroke with TrkB activation.
Chang LH, Lin HC, Huang SS, Chen IC, Chu KW, Chih CL, Liang YW, Lee YC, Chen YY, Lee YH, Lee IH
Scientific reports 2018 Jan 8;8(1):118
Scientific reports 2018 Jan 8;8(1):118
Grifolic acid induces mitochondrial membrane potential loss and cell death of RAW264.7 macrophages.
Zhao Y, Zhang H, Yan A, Zhu J, Liu K, Chen D, Xie R, Xu X, Su X
Molecular medicine reports 2018 Feb;17(2):3281-3287
Molecular medicine reports 2018 Feb;17(2):3281-3287
Fenofibrate Administration Reduces Alcohol and Saccharin Intake in Rats: Possible Effects at Peripheral and Central Levels.
Rivera-Meza M, Muñoz D, Jerez E, Quintanilla ME, Salinas-Luypaert C, Fernandez K, Karahanian E
Frontiers in behavioral neuroscience 2017;11:133
Frontiers in behavioral neuroscience 2017;11:133
Red-shifted luciferase-luciferin pairs for enhanced bioluminescence imaging.
Yeh HW, Karmach O, Ji A, Carter D, Martins-Green MM, Ai HW
Nature methods 2017 Oct;14(10):971-974
Nature methods 2017 Oct;14(10):971-974
1,25-(OH)(2)D(3) and its analogue BXL-628 inhibit high glucose-induced activation of RhoA/ROCK pathway in HK-2 cells.
Zhang W, Yi B, Zhang K, Li A, Yang S, Huang J, Liu J, Zhang H
Experimental and therapeutic medicine 2017 May;13(5):1969-1976
Experimental and therapeutic medicine 2017 May;13(5):1969-1976
Unlocking Aspirin's Chemopreventive Activity: Role of Irreversibly Inhibiting Platelet Cyclooxygenase-1.
Lichtenberger LM, Fang D, Bick RJ, Poindexter BJ, Phan T, Bergeron AL, Pradhan S, Dial EJ, Vijayan KV
Cancer prevention research (Philadelphia, Pa.) 2017 Feb;10(2):142-152
Cancer prevention research (Philadelphia, Pa.) 2017 Feb;10(2):142-152
Crocin Inhibits Oxidative Stress and Pro-inflammatory Response of Microglial Cells Associated with Diabetic Retinopathy Through the Activation of PI3K/Akt Signaling Pathway.
Yang X, Huo F, Liu B, Liu J, Chen T, Li J, Zhu Z, Lv B
Journal of molecular neuroscience : MN 2017 Apr;61(4):581-589
Journal of molecular neuroscience : MN 2017 Apr;61(4):581-589
Study of the Effects of Betaine and/or C-Phycocyanin on the Growth of Lung Cancer A549 Cells In Vitro and In Vivo.
Bingula R, Dupuis C, Pichon C, Berthon JY, Filaire M, Pigeon L, Filaire E
Journal of oncology 2016;2016:8162952
Journal of oncology 2016;2016:8162952
Overexpression of the pituitary tumor transforming gene upregulates metastasis in malignant neoplasms of the human salivary glands.
Liu J, Wang Y, He H, Jin W, Zheng R
Experimental and therapeutic medicine 2015 Aug;10(2):763-768
Experimental and therapeutic medicine 2015 Aug;10(2):763-768
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
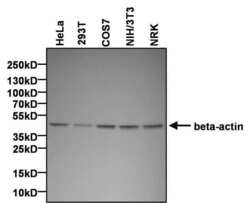
- Experimental details
- Western blot analysis of beta-actin was performed by loading 25 µg of various whole cell lysates and 10 µL of PageRuler Plus Prestained Protein Ladder (Product # 26619) per well onto a 4-20% Tris-HCl polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked with 5% BSA/TBST for at least 1 hour. The membrane was probed with a beta-actin polyclonal antibody (Product # PA1-183) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBS-0.1%Tween-20, and probed with a goat anti-rabbit IgG-HRP secondary antibody (Product # 31460) at a dilution of 1:40,000 for at least 1 hour. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western Blot was performed using Anti-beta Actin Polyclonal Antibody (Product # PA1-183) and a ~40 kDa band corresponding to Actin, cytoplasmic 1 was observed across cell lines and tissues tested. Whole cell extracts (30 µg lysate) of HeLa (Lane 1), A549 (Lane 2), A-431 (Lane 3), U-2 OS (Lane 4), Jurkat (Lane 5), PC-12 (Lane 6), NIH/3T3 (Lane 7), Mouse Spleen (Lane 8), Mouse Liver (Lane 9), Rat Spleen (Lane 10), Rat Liver (Lane 11) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # LC2001) by iBlot® 2 Dry Blotting System (Product # IB21001). The Blot was probed with the primary antibody (1:2000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). doi:10.1038/bcj.2014.18
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of beta-actin (green) in HeLa cells. Cells were fixed with 4% paraformaldehyde (PFA) for 15 minutes at room temperature and permeabilized with 0.5% Triton X-100 in PBS for 15 minutes at room temperature (left panel) or fixed and permeabilized with ice cold methanol for 10 minutes at room temperature (middle panel). Cells were blocked with 0.3% BSA in PBS for 15 minutes at room temperature, and probed with a beta-actin polyclonal antibody (Product # PA1-183) at a dilution of 1:100 for at least 1 hour at room temperature. Cells were then washed with PBS, and incubated with a DyLight 488 goat anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:500 for 30 minutes at room temperature. As a positive control, PFA-fixed cells were stained with DyLight 488-conjugated Phalloidin (Product # 21833) at a dilution of 1:1000 (right panel). Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of beta-actin was performed on HeLa cells. Antigen-antibody complexes were formed by incubating 750 µg of HeLa whole cell lysates with 2 µg of a beta-actin polyclonal antibody (Product # PA1-183) overnight on a rocking platform at 4øC. The immune complexes were captured on 50 µL Protein A/G Agarose (Product # 20421), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). Samples were resolved on a 4-20% Tris-HCl polyacrylamide gel, transferred to a PVDF membrane, and blocked with 5% BSA/TBS-0.1%Tween-20 for at least 1 hour. The membrane was probed with a beta-actin polyclonal antibody (Product # PA1-183) at a dilution of 1:1000 overnight rotating at 4øC, washed in TBST, and probed with Clean-blot IP detection reagent (Product # 21230) at a dilution of 1:250 for at least 1 hour. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Activation of p38 MAPK pathway on A549 after betaine and/or phycocyanin treatment. Cells were treated with either 4% betaine, 20 mu g*L -1 of C-PC, or a combination of both molecules for 24 and 48 h. (a) Expression of p38 MAPK in different treatments was monitored by western blot. beta -actin was used as a loading-control protein. (b) p38 MAPK along with beta -actin band signals were quantified with the ImageJ software. After normalization with beta -actin, p38 MAPK values were plotted on the graph as relative values compared to their respective controls. Bars are presented as the mean +- standard deviation. * p < 0.05. Statistical significance between groups was assessed using repeated measures one-way ANOVA followed by Tukey's posttest. UT stands for untreated condition, B stands for betaine, and PC stands for C-PC.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 2 FIGURE Assessment of catabolic activity in disc tissue of 23-month-old PAPPA -/- mice compared to 23-month-old Wt mice. Western blotting and densitometry measurements were performed as described in Section 2. A, A schematic of the mouse aggrecan core protein covalently linked to the sulphate-rich GAG and noncovalently bound to a hyaluronan chain via the link protein. The cleavage sites between G1 and G2 interglobular domains by ADAMTS (G1-NVTEGE392) and MMP (G1VDIPEN360) proteases are indicated. B, Western blot of 23-month-old PAPPA -/- and 23-month-old Wt disc tissues probed for aggrecan fragments. Six-month-old Wt tissues serve as young controls. Arrows indicate two specific cleavage products corresponding to ADAMTS (~65 kD) and MMP (~55 kD). Three replicates per condition. C, Quantification of aggrecan fragment western blot data for 6-month-old Wt, 23-month-old Wt, and 23-month-old PAPPA -/- tissues. Bars represent mean values of three different mouse tissues; error bars indicate SD. All values were normalized to beta-actin control. *, P < .05. Abbreviations: disc, intervertebral disc; MMP, matrix metalloproteinase; ADAMTS, a disintegrin and metalloproteinase with thrombospondin motifs; Wt, wild-type
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 3 FIGURE Assessment of catabolic protease expression in disc tissue of 23-month-old PAPPA -/- mice compared to 23-month-old Wt mice. Data shown are representative immunoblots and densitometric quantification of MMP-3 (A) and ADAMTS-4 (B) protein expression. Bars represent mean values of three different mouse tissues; error bars indicate SD, n = 3. All values were normalized to beta-actin control. *, P < .05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 4 FIGURE Assessment of disc cellular senescence in disc tissue of 23-month-old PAPPA -/- mice compared to 23-month-old Wt mice. Immunohistochemistry slides were prepared as described in Section 2. Representative examples are shown. A, Lamin B1 staining for qualitative assessment of senescent disc cells. There was higher intensity of lamin B1 staining in PAPPA -/- mice as indicated by the black arrows. No detectable lamin B1 staining was seen in 23-month-old Wt mice samples. B, Western blotting probed for p53 protein expression. Three replicates per condition. C, Densitometric quantification of p53 immunoblots of PAPPA -/- mice showed a decrease in p53 expression, albeit not statistically significant ( P = .12), compared to 23-month-old Wt mice. Bars represent mean values of three different mouse tissues; error bars indicate SD. All values were normalized to beta-actin control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
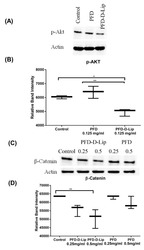
- Experimental details
- Figure 12 ( A ) Western blot representing inhibitory effect of treatments on expression of p-AKT protein in A549 cells. ( B) PFD-D-Lip significantly suppresses p-AKT expression in A549 cells. Cell lysates were prepared following 24-hour treatment of A549 cells with control, PFD and PFD-D-Lip (0.125 mg/mL) and were subjected to western blot analyses using antibodies against p-AKT and actin as a loading control, as indicated. ( C ) Western blot representing inhibitory effect of treatments on expression of beta-catenin protein in A549 cells. ( D ) PFD-D-Lip suppresses beta-catenin expression in A549 cells. Cell lysates were prepared following 72-hour treatment of A549 cells with control, PFD and PFD-D-Lip (0.25 and 0.5 mg/mL) and were subjected to western blot analyses using antibodies against beta-catenin and actin as a loading control, as indicated. The western blot densities were determined and plotted as relative band intensity against treatments. Western blot bands represent representative image of n = 3. Data represent mean +- SD ( n = 3). * p (alpha < 0.05), ** p (alpha < 0.01).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 EPEC infection model increases intestinal permeability and decreases claudin-1 expression in colon of mice. (A) Intestinal permeability was determined in the plasma of control and EPEC-infected mice at day 7 p.i. using FITC dextran assay. Each point represents a mouse. Lines represent mean +- SEM (n=8). * p < 0.006 using Student's t-test . (B) Significant positive correlation between diarrhea score and INF-gamma levels in colon tissues of EPEC-infected mice at day 7 p.i (Spearman rank test). (C) . Representative western blots of levels of claudin-1 and beta-actin in colonic tissues of control and EPEC-infected mice at day 7 p.i. (D) Quantification of western blot bands of claudin-1 and beta-actin. Bars represent mean +- SEM (n=3). # p < 0.03 using Student's t-test . (E) Representative western blots of levels of claudin-2 and beta-actin in colonic tissues of control and EPEC-infected mice at day 7 p.i. (F) Quantification of western blot bands of Claudin-2 and beta-actin. Bars represent mean +- SEM (n=3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3. Bcl2l10-knockdown significantly decreases the protein expression levels of SDHD and IDH1. (A) SKOV3 and A2780 cells were transfected with siNC or siBcl2l10 and harvested 48 h after transfection. The protein expression levels of BCL2L10, SDHD, IDH1 and FH were measured by western blot analysis using beta-actin as an internal control gene. (B) Protein signals of BCL2L10, SDHD, IDH1 and FH were measured and normalized to the corresponding beta-actin signals. The relative expression levels of proteins were calculated by comparing all the normalized signals to that of the negative control group. All the experiments were repeated at least three times, and the data are expressed as the mean +- SEM. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. Bcl2l10-knockdown significantly increases TPM4 expression. (A) SKOV3 and A2780 cells were transfected with siNC or siBcl2l10 and harvested 48 h following transfection. mRNA expression levels of TPM4 were calculated from CT values obtained by reverse transcription-quantitative PCR and were normalized to the expression levels of GAPDH. (B) Protein levels of TPM4 was measured by western blot analysis. beta-actin was used as an internal control gene. Signals of TPM4 were measured and normalized to corresponding beta-actin signals. Relative expression levels of TPM4 were calculated by comparing all the normalized signals to that of the negative control group. All experiments were repeated at least 3 times, and data are expressed as the mean +- SEM. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 SIRT1 was down-regulated in the STZ-induced DR rat model. STZ-treated rats were further treated with DMSO, 10 mg/kg CMS, 50 mg/kg CMS, and 100 mg/kg CMS. n = 15 per treatment. ( A ) Representative images of retinal tissues observed by HE-staining (x 400) (scale bar = 25 mum) and quantitative analysis of thickness. * p < 0.05 compared to sham-operated rats and # p < 0.05 compared to STZ-treated rats. ( B ) Ultrastructure of rat retinal tissues observed under a TEM (x 10000) (scale bar = 1 mum). The arrow represents the capillary BMT, which is used to measure basement membrane width. ( C ) Expression pattern of SIRT1 measured by RT-qPCR in rat retinal tissues, normalized to beta-actin. ( D ) Representative Western blots of SIRT1 protein and its quantitation in rat retinal tissues, normalized to beta-actin. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to the sham-operated rats, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to the rats injected with STZ and treated with DMSO. The results were measurement data and expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test. (n = 15). DR, diabetic retinopathy; STZ, streptozotocin; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; SIRT1, Sirtuin 1; TEM, transmission electron microscopy; ANOVA, analysis of variance; n, number; GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; ONL, outer nu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 CMS reduced oxidative stress, inflammation, and apoptosis in DR rats. STZ-treated rats were treated with DMSO, 10 mg/kg CMS, 50 mg/kg CMS, and 100 mg/kg CMS. n = 15 per treatment. ( A ) Expression pattern of iNOS as determined by RT-qPCR in rat retinal tissues, normalized to beta-actin. ( B ) Representative Western blots of iNOS protein and its quantitation in rat retinal tissues, normalized to beta-actin. ( C ) Expression of NO measured by nitrite test in rat retinal tissues. ( D - F ) Expression patterns of IL-6 ( D ), TNF-alpha ( E ), and CRP ( F ) measured by ELISA in the cell supernatant. ( G , H ) Representative images of apoptotic cells (x 400) (scale bar = 25 mum) ( G ) and cell apoptosis in rat retinal tissues ( H ) by TUNEL. ( I ) Representative Western blots of cleaved caspase-3 protein and its quantitation in rat retinal tissues, normalized to beta-actin. ( J , K ) Representative Western blots of Cyt-C protein and its quantitation in the cytoplasm and mitochondria, normalized to beta-actin. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to the sham-operated rats, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to the rats injected with STZ and treated with DMSO. The results were measurement data, which were expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test (n = 15). iNOS, inducible nitric oxide synthase; CMS, coumestrol, STZ, streptozotocin; DMSO, dimethyl sulfoxi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 CMS activated the expression of SIRT1 in HG-treated hRMECs. hRMECs were transfected with sh-SIRT1-1, sh-SIRT1-2, or sh-SIRT1-3, and HG-exposed hRMECs were treated with DMSO, 1-CMS, 2-CMS, or 3-CMS, respectively. ( A ) Cell viability assessed by CCK-8 assay. ( B ) SIRT1 expression pattern determined by RT-qPCR in hRMECs, normalized to beta-actin. ( C ) Representative Western blots of SIRT1 protein and its quantitation in hRMECs, normalized to beta-actin. ( D , E ) Representative images (x 400) (scale bar = 25 mum) ( D ) as well as SIRT1 activity and nuclear accumulation in hRMECs ( E ) detected by immunofluorescence staining. ( F ) Expression pattern of SIRT1 as determined by RT-qPCR in hRMECs, normalized to beta-actin. ( G ) Representative Western blots of VEGF protein and its quantitation in hRMECs, normalized to beta-actin. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to the control cells, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to cells stimulated with NC or HG + DMSO. The results were measurement data and expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test. The cell experiments were repeated three times independently. NC, negative control; CMS, coumestrol, HG, high glucose; DMSO, dimethyl sulfoxide; SIRT1, sirtuin 1; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; VEGF, vascular endothelial growth factor; ANOVA, analysis of variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 CMS suppressed HG-induced oxidative stress and inflammation in hRMECs through activating SIRT1. hRMECs were treated with sh-NC, sh-SIRT1, sh-SIRT1 + DMSO and sh-SIRT1 + CMS, respectively. ( A ) Expression pattern of iNOS as determined by RT-qPCR in hRMECs, normalized to beta-actin. ( B ) Representative Western blots of iNOS protein and its quantitation in hRMECs, normalized to beta-actin. ( C ), Expression pattern of NO as determined by nitrite test in hRMECs. ( D - F ), Expression patterns of IL-6 ( D ), TNF-alpha ( E ), and CRP ( F ) as measured by ELISA in the cell supernatant. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to sh-NC-treated cells, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to cells stimulated with sh-SIRT1 and treated with DMSO. The results were measurement data and expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test. The cell experiments were repeated three times independently. NC, negative control; CMS, coumestrol, HG, high glucose; hRMECs, human retinal microvascular endothelial cells; SIRT1, sirtuin 1; DMSO, dimethyl sulfoxide; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; IL-6, interleukin-6; TNF-alpha, tumor necrosis factor alpha; CRP, C-reactive protein; ELISA, Enzyme linked immunosorbent assay; ANOVA, analysis of variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 CMS suppressed the apoptosis of HG-treated hRMECs . hRMECs were treated with sh-NC, sh-SIRT1, sh-SIRT1 + DMSO and sh-SIRT1 + CMS, respectively. ( A ) Cell cycle distribution as determined by flow cytometry analysis. ( B ) Cell apoptosis as determined by flow cytometry analysis. ( C ) Representative Western blots of cleaved caspase-3 protein and its quantitation in hRMECs, normalized to beta-actin. ( D , E ) Western blots of Cyt-C protein ( D ) and its quantitation ( E ) in the cytoplasm and mitochondria, normalized to beta-actin. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to sh-NC-treated cells, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to cells stimulated with sh-SIRT1 + DMSO. The results were the measurement data and expressed as mean +- standard deviation. Comparisons between multiple groups should be analyzed by one-way ANOVA with Tukey''s p ost hoc test. The cell experiments were repeated three times independently. CMS, coumestrol; HG, high glucose; hRMECs, human retinal microvascular endothelial cells; NC, negative control; DMSO, dimethyl sulfoxide; SIRT1, sirtuin 1; NO, nitric oxide; Cyt-C, cytochrome c; ANOVA, analysis of variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 ( A ) Brain lysates from WY, WO, GY, and GO mice were collected, and levels of SMP30 were analyzed by Western blotting. ( B ) Beta-actin was used to normalize Western blot data ( n = 8). Abbreviations: young wild-type mice (WY); old wild-type mice (WO); young GNMT -/- mice (GY); old GNMT -/- mice (GO).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 ( A ) Brain lysates from WY, WO, GY, and GO mice were collected, and levels of ADNP were analyzed by Western blotting. ( B ) Beta-actin was used to normalize Western blot data ( n = 8, *** p < 0.001). Abbreviations: young wild-type mice (WY); old wild-type mice (WO); young GNMT -/- mice (GY); old GNMT -/- mice (GO).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 ( A ) Brain lysates from Wt, GY, and GO mice were collected, and levels of RIPK1 were analyzed by Western blotting. The RIPK1 expression was down-regulated or non-detectable in some WY and WO mice; thus, the WY and WO mice were combined into one group as Wt. ( B ) Beta-actin was used to normalize Western blot data ( n = 8, * p < 0.05 and ** p < 0.005). Abbreviations: wild-type mice (Wt); young GNMT -/- mice (GY); old GNMT -/- mice (GO).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 9 ( A ) Brain lysates from WY, WO, GY, and GO mice were collected, and levels of Caspase 3 were analyzed by Western blotting. ( B ) Beta-actin was used to normalize Western blot data ( n = 8). Abbreviations: young wild-type mice (WY); old wild-type mice (WO); young GNMT -/- mice (GY); old GNMT -/- mice (GO).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Effect of KDAC inhibition on enzyme activities in A549 cells under normoxia and hypoxia. ( A , B ) The ATP- dependent 6-phosphofructokinase (PFK1) ( A ) and lactate dehydrogenase (LDH) ( B ) enzymatic activities were measured after 24 h of incubation, and activities were normalized to intracellular protein content in each condition. A549 cells were treated with 1 muM of TSA, 20 mM of NAM, and both 1 muM TSA and 20 mM NAM for 24 h of incubation under normoxia and hypoxia. Cells incubated in medium without KDACIs served as control. Bars represent the means +- standard error of the mean of three independent experiments. The asterisks above bars indicate statistically significant differences compared to normoxic control cells. The asterisks above curly brackets indicate statistically significant differences between hypoxic and normoxic treatments and between hypoxic treatments and hypoxic control cells. Statistical significance was assessed by a two-tailed Student''s t -test. *, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Degenerative changes in intervertebral discs of naturally aged mice. (a) Expression of selected senescent markers, p16 Ink4a (a1), p53 (a2) and p21 (a3) in disc tissue from young (6 month) and old (22 month) mice was determined by Western blotting and qRT-PCR. Graphs on the right of the Western images are quantification of results (a2 and a3) whereby the volume of respective protein band divided by volume of beta-actin band. (b) Increased disc aggrecan proteolysis with age. (b1) A schematic of the mouse aggrecan core protein covalently linked to the sulphate-rich glycosaminoglycan (GAG) and noncovalently bound to a hyaluronic acid (HA) chain via the link protein. The cleavage sites between G1 and G2 interglobular domains by ADAMTS (G1-NVTEGE 392 ) and MMP (G1VDIPEN 360 ) proteases are indicated. (b2) Western blot analysis of aggrecan fragments generated by ADAMTS and MMP proteases with graphs showing quantification results. Representative Western blots for three young and three old mice were shown (panels a2, a3, b2). (c) Proteoglycan synthesis as measured by 35 S-sulphate incorporation using whole disc organ cultures. Student's t test was used for parametric data (graphs a1, a2, b2), and the Mann-Whitney test was used for two-variable nonparametric data (graphs a3, c). Data are means +- SD of four independent experiments (4 mice) for graphs a1, a3, c and three mice for graphs a2, b2. * p < 0.05. Y = young and O = old. Representative Western images of three different
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Effects of DAPK3 inhibition on CLL and lymphoid cell proliferation and cellular function. (A) Representative western blots displaying levels of EGR1 and DAPK3-T265-P in CLL cells pretreated with 0-120 mu m DAPKi for 1 h and with or without anti-IgM stimulation for 1 h. beta-actin was used as a loading control. Blots are normalised to unstimulated sample (unstimulated = 1.0) with image lab software (below). Error bars represent the SD of three independent experiments. Significant differences calculated using two-way ANOVA followed by Dunnett's multiple comparison test with anti-IgM stimulated 0 u m sample as control. P values = 0.0076, 0.0256 and 0.0087 for unstimulated 0 and 80 and 120 u m samples, respectively. (B) Flow cytometric analysis of CLL cells cultured on a CD40L-expressing cell feeder layer. CLL cells were pretreated with either 1 u m ibrutinib (green), increasing concentrations (5-80 u m ) of DAPKi (red), both inhibitors (green/red), or DMSO (white; negative control) for 1 h as indicated. Proliferation was assessed via the percentage of cells positive for Ki-67. Data shown are representative of n = 3 CLL patients. (C) Flow cytometric analysis of CLL cells cultured on a CD40L-expressing cell feeder layer. CLL cells were pretreated with either 1 u m ibrutinib, increasing concentrations (5-80 u m ) of DAPKi or DMSO (negative control) for 1 h. Apoptosis was assessed using the Annexin V Apoptosis Detection Kit, as indicated. Proliferation was assessed via the pe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Mechanistic evaluation of SDC1-Lip echinomycin-induced autophagic (non-apoptotic) cell death using Western blot. As observed in Figure 2 A,B, SDC1-Lip echinomycin induced cell death. To evaluate potential mechanisms of cell death, S2VP10, S2CP9, and Miapaca2 cells were treated with PBS, Gemzar, echinomycin alone, or SDC1-Lip echinomycin. Cell lysates were evaluated for signaling molecules important in autophagy (LC3-I, -II, p62, ATG7, and ATG12) and apoptosis (pro-caspase-3, cleaved caspase-3, pro-caspase-9, and cleaved caspase-9). ( A - F ) Echinomycin alone and ( G - L ) SDC1-Lip echinomycin treatment of S2VP10 and S2CP9 showed increases in the LC3-II/I ratio, indicating macroautophagy and lack of cleaved caspases-3 and -9, thus no apoptosis. Miapaca2 cells did not demonstrate activation of autophagy in response to either echinomycin or SDC1-Lip echinomycin. Control apoptosis treatment, Gemzar, resulted in some activation of apoptosis. Quantification comparisons are shown in B-F and H-L as well as Figure S6A-D . * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 miR-375 regulates ERBB2 and VEGFA in EC . A. Expression of miR-375 after transfection. B. Forced expression of miR-375 in EC cell lines resulted in decreased ERBB2 and VEGFA expression. C. Map of the plasmids pEZX-MT04 containing miR-375, and pEZX-MT05 containing 3'-UTR of ERBB2 to illustrate the binding site of miR-375 at the 3'-UTR of ERBB2, and its mutant control sequence. D. Dual luciferase reporter assay. Co-transfection with pEZX-miR-375 and pEZX-ERBB2 3' UTR wild type significantly decreased the luciferase activities compared to that with pEZX-miR-375 and ERBB2 3' UTR mutant/miR-375 scrambled control and ERBB2 3' UTR wild type sequence in ESCC cell lines. The data were reported as mean +- S.D. from three independent experiments (** p < 0.01, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Representative western blotting showing estrogen receptor alpha, estrogen receptor beta, cyclin D1, caspase 3 (cleaved form) in Apc Min/+ mice assuming standard or enriched diet evaluated in normal and polypoid tissues; beta actin was used as loading control (panel shows representative bolts of three different experiments performed with similar results).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. miR-4463 targets cytochrome P450 family 19 subfamily A member 1 ( CYP19A1 ) and estrogen receptor alpha ( ESR1 ) genes and inhibits steroidogenesis in KGN cells. After KGN cells were transfected with a miR-4463 mimic, miR-4463 inhibitor, and miR-4463 mimic with folliclestimulating hormone (FSH), the expression levels of CYP19A1, estrogen receptor alpha (ERalpha), and synthesis of 17beta-estradiol were analyzed. (A) Expression of miR-4463 increased by knockdown of follicle-stimulating hormone receptor ( FSHR ) in KGN cells. (B) The putative sites in the 3'-untranslated region (UTR) of CYP19A1 and ESR1 mRNA contain the seed sequence for hsa-miR-4463 binding. The nucleotides in bold letters indicate the seed region of miR-4463. (C, D) Protein expression levels were measured by Western blot analysis, and beta-actin was used as an internal control. The relative protein expression levels were calculated by measuring the density and area of the bands. (E) 17beta-estradiol levels were measured by enzyme-linked immunosorbent assay using cell culture supernatant after transfection with miR-4463 mimic, miR-4463 inhibitor, and miR-4463 mimic with FSH. The experiments were repeated at least three times, and the data are presented as the mean +- standard error of the mean. siRNA, small interfering RNA. a) p < 0.05: indicate statistical significance. The p -values were obtained by one-way analysis of variance and the Student-Newman-Keuls test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. Treatment of follicle-stimulating hormone receptor ( FSHR ) small interfering RNA (siRNA) decreased CYP19A1 expression. (A) The protein expression levels were measured by Western blot analysis, and beta-actin was used as an internal control gene. (B) The relative protein expression levels were calculated by measuring the density and area of the bands. The experiments were repeated at least three times, and the data are presented as the mean +- standard error of the mean. NC, negative control group; siFSHR, FSHR siRNA-treated group. a) p < 0.001: indicates statistical significance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4. Microglia synaptic pruning and neuroinflammation. After IS with or without ethanol exposure for 28 days, mice were euthanized. Raphe nuclei homogenates were used to analyze protein and cytokine expression. The protein levels of Iba-1, PSD95, synaptophysin, cleaved caspase 3, and C1q were determined by Western blotting ( A ), and their intensities were semiquantified ( B ). Levels of the cytokines TNF-alpha ( C ) and IL-10 ( D ) in the raphe nuclei were determined by enzyme immunoassay. The data are expressed as the means +- SD ( n = 5). * P < 0.05 and ** P < 0.01 compared to the unstressed mice, and # P < 0.05 and ## P < 0.01 compared to the mice not exposed to ethanol. Gene expression levels of TNF-alpha ( E ) and IL-1beta ( F ) were determined by real-time PCR analysis. The data are expressed as the means +- SD ( n = 3). * P < 0.05 and ** P < 0.01 compared to the vehicle-treated cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Analysis of PSMA expression levels. WB of PCa tumor tissues and a positive control, all samples were run on the same blot. The non-relevant bands were cropped from the image, the full-length blot is presented in Supplementary Fig. 1 ( a ). Semi-quantification of WB, the relative density of PSMA was normalized using beta-actin as loading control. Quantification was only performed using bands from the same blot ( b ). Data for PC-3, 22Rv1 and C4-2 (n = 5) is presented as mean +- SD. IHC of PSMA expression of PC-3 ( c ), 22Rv1 ( d ) and C4-2 ( e ) tumor tissue.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Actin accumulates outside of the NE in prophase, and its network shrinks after NEBD. ( A ) Actin accumulates on the NE around NEBD. Images (z-projections) show a representative cell expressing GFP-tubulin and mCherry-Lifeact (that fluorescently marks F-actin). Time is relative to NEBD. Timing of NEBD was determined by the influx of cytoplasmic GFP-tubulin into the nucleus. Scale bars, 10 um. Graph shows mean Lifeact fluorescence intensity (per pixel) around the nucleus over time (n = 8; error bars, s.e.m). ( B ) Actin localizes outside of the NE. Images show single super-resolution z-sections of cells, which were fixed and stained for actin with phalloidin (actin dye; red). Cells were also immunostained (green) with anti-Lamin B1 (top) or anti-actin antibody (bottom), respectively. The same secondary antibody was used for the two immunostainings. These cells were undergoing chromosome compaction (confirmed by DNA staining). Insets show magnification of the regions in yellow boxes. Scale bar, 10 um. Graphs show intensity of actin dye (red) and immunostaining (green) along the dashed yellow lines in left images (middle parts are omitted). The peaks in graphs, marked with asterisks (top) and daggers (bottom), locate at the regions in yellow boxes in left images. a.u, arbitrary unit. ( C ) The LINC complex is required for accumulation of the actin network. Images show single z-sections of representative cells, expressing either an RFP-tagged LINC-DN construct (SR-KASH)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Catalase expression in liver and the brain of fenofibrate-treated animals. Catalase levels were determined by western blot in brain and liver of ethanol or saccharin drinking rats (treated with fenofibrate or vehicle; n = 7 for each group). Catalase levels were normalized against beta-actin. Bars represent the normalized densitometric quantification of the bands. Catalase expression of the saccharin group without fenofibrate administration was used as control (100%). ** p < 0.01 with respect to control; n.s. = difference statistically non-significant.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoprecipitation
Immunoprecipitation