Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-0439-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD43 Monoclonal Antibody (eBio84-3C1 (84-3C1)), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody ebio84-3C1 reacts with CD43, which is also known as leukosialin, galactoglycoprotein and sialophorin. CD43 is a sialomucin which like many mucins can have both adhesive and anti-adhesive functions. Expression of CD43 is found on most leukocytes except resting B lymphocytes. Proteolytic processing upon activation decreases surface expression. CD43 is involved in activation of T cells, B cells, NK cells, and monocytes. The counter-receptor for CD43 is CD169/SIGLEC-1, which is expressed on macrophages. Applications Reported: This eBio84-3C1 (84-3C1) antibody has been reported for use in flow cytometric analysis, western blotting, and immunohistochemical staining of formalin-fixed paraffin embedded tissue sections. Applications Tested: This eBio84-3C1 (84-3C1) antibody has been tested by flow cytometric analysis of human peripheral blood cells and by immunohistochemistry of formalin-fixed paraffin embedded human spleen using low pH antigen retreival at less than or equal to 20 µg/mL. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- eBio84-3C1 (84-3C1)
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references In vitro differentiation of human embryonic stem cells to hemogenic endothelium and blood progenitors via embryoid body formation.
Systems-Level Immunomonitoring from Acute to Recovery Phase of Severe COVID-19.
Verification and rectification of cell type-specific splicing of a Seckel syndrome-associated ATR mutation using iPS cell model.
The Generation of Human γδT Cell-Derived Induced Pluripotent Stem Cells from Whole Peripheral Blood Mononuclear Cell Culture.
Aberrant expression of interleukin-10 and activation-induced cytidine deaminase in B cells from patients with Behçet's disease.
Activation of KLF1 Enhances the Differentiation and Maturation of Red Blood Cells from Human Pluripotent Stem Cells.
Enforced Expression of HOXB4 in Human Embryonic Stem Cells Enhances the Production of Hematopoietic Progenitors but Has No Effect on the Maturation of Red Blood Cells.
Protective Role for B-1b B Cells and IgM in Obesity-Associated Inflammation, Glucose Intolerance, and Insulin Resistance.
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
Reprogramming human endothelial cells to haematopoietic cells requires vascular induction.
CD43 monoclonal antibodies recognize the large sialoglycoprotein of human leukocytes.
Garcia-Alegria E, Potts B, Menegatti S, Kouskoff V
STAR protocols 2021 Mar 19;2(1):100367
STAR protocols 2021 Mar 19;2(1):100367
Systems-Level Immunomonitoring from Acute to Recovery Phase of Severe COVID-19.
Rodriguez L, Pekkarinen PT, Lakshmikanth T, Tan Z, Consiglio CR, Pou C, Chen Y, Mugabo CH, Nguyen NA, Nowlan K, Strandin T, Levanov L, Mikes J, Wang J, Kantele A, Hepojoki J, Vapalahti O, Heinonen S, Kekäläinen E, Brodin P
Cell reports. Medicine 2020 Aug 25;1(5):100078
Cell reports. Medicine 2020 Aug 25;1(5):100078
Verification and rectification of cell type-specific splicing of a Seckel syndrome-associated ATR mutation using iPS cell model.
Ichisima J, Suzuki NM, Samata B, Awaya T, Takahashi J, Hagiwara M, Nakahata T, Saito MK
Journal of human genetics 2019 May;64(5):445-458
Journal of human genetics 2019 May;64(5):445-458
The Generation of Human γδT Cell-Derived Induced Pluripotent Stem Cells from Whole Peripheral Blood Mononuclear Cell Culture.
Watanabe D, Koyanagi-Aoi M, Taniguchi-Ikeda M, Yoshida Y, Azuma T, Aoi T
Stem cells translational medicine 2018 Jan;7(1):34-44
Stem cells translational medicine 2018 Jan;7(1):34-44
Aberrant expression of interleukin-10 and activation-induced cytidine deaminase in B cells from patients with Behçet's disease.
Yoon JY, Lee Y, Yu SL, Yoon HK, Park HY, Joung CI, Park SR, Kwon M, Kang J
Biomedical reports 2017 Dec;7(6):520-526
Biomedical reports 2017 Dec;7(6):520-526
Activation of KLF1 Enhances the Differentiation and Maturation of Red Blood Cells from Human Pluripotent Stem Cells.
Yang CT, Ma R, Axton RA, Jackson M, Taylor AH, Fidanza A, Marenah L, Frayne J, Mountford JC, Forrester LM
Stem cells (Dayton, Ohio) 2017 Apr;35(4):886-897
Stem cells (Dayton, Ohio) 2017 Apr;35(4):886-897
Enforced Expression of HOXB4 in Human Embryonic Stem Cells Enhances the Production of Hematopoietic Progenitors but Has No Effect on the Maturation of Red Blood Cells.
Jackson M, Ma R, Taylor AH, Axton RA, Easterbrook J, Kydonaki M, Olivier E, Marenah L, Stanley EG, Elefanty AG, Mountford JC, Forrester LM
Stem cells translational medicine 2016 Aug;5(8):981-90
Stem cells translational medicine 2016 Aug;5(8):981-90
Protective Role for B-1b B Cells and IgM in Obesity-Associated Inflammation, Glucose Intolerance, and Insulin Resistance.
Harmon DB, Srikakulapu P, Kaplan JL, Oldham SN, McSkimming C, Garmey JC, Perry HM, Kirby JL, Prohaska TA, Gonen A, Hallowell P, Schirmer B, Tsimikas S, Taylor AM, Witztum JL, McNamara CA
Arteriosclerosis, thrombosis, and vascular biology 2016 Apr;36(4):682-91
Arteriosclerosis, thrombosis, and vascular biology 2016 Apr;36(4):682-91
Mitochondrial ATP transporter Ant2 depletion impairs erythropoiesis and B lymphopoiesis.
Cho J, Seo J, Lim CH, Yang L, Shiratsuchi T, Lee MH, Chowdhury RR, Kasahara H, Kim JS, Oh SP, Lee YJ, Terada N
Cell death and differentiation 2015 Sep;22(9):1437-50
Cell death and differentiation 2015 Sep;22(9):1437-50
Reprogramming human endothelial cells to haematopoietic cells requires vascular induction.
Sandler VM, Lis R, Liu Y, Kedem A, James D, Elemento O, Butler JM, Scandura JM, Rafii S
Nature 2014 Jul 17;511(7509):312-8
Nature 2014 Jul 17;511(7509):312-8
CD43 monoclonal antibodies recognize the large sialoglycoprotein of human leukocytes.
Borche L, Lozano F, Vilella R, Vives J
European journal of immunology 1987 Oct;17(10):1523-6
European journal of immunology 1987 Oct;17(10):1523-6
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-CD43 Monoclonal Antibody (Product # 14-0439-82) and bands around 90-130 kDa corresponding to CD43 were observed in the cell lines tested except HT-29 which is reported to be negative. Membrane enriched extracts (30 µg lysate) of K-562 (Lane 1), MOLT-4 (Lane 2), HEL 92.1.7 (Lane 3), U-937 (Lane 4) and HT-29 (Lane 5) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # LC2001) by XCell SureLock™ Mini-Cell and XCell II™ Blot Module (Product # EI0002). The blot was probed with the primary antibody (1:500 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using SuperSignal™ West Dura Extended Duration Substrate (Product # 34076).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
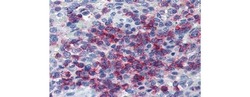
- Experimental details
- Immunohistochemistry of formalin-fixed paraffin embedded human spleen stained with 20 µg/mL Anti-Human CD43 Purified followed by Anti-Mouse IgG Biotin, Streptavidin Alkaline Phosphatase, and Fast Red. Nuclei are counterstained with hematoxylin.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
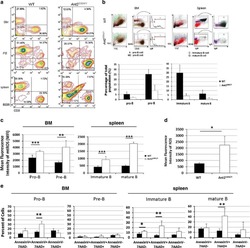
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Extended Data Figure 2 FGRS-transduction and vascular-induction reprogram HUVECs, but not hES-ECs, to proliferating functional rEC-MPPs A. Multi-colony niche-like structure that physically separates developing hematopoietic colonies from surrounding E4EC vascular niche . The emerging multi-colony sinusoidal-like structures create a unique cellular interface between E4EC monolayers and transduced ECs giving rise to hematopoietic clusters (n=4, scale bar is 1000 um.) B. Expansion potential of reprogrammed CD45 + hematopoietic cells . CD45 + (12x103) and CD45- (60 x10 3 ) cells were sorted into separate wells and expanded for two days. We observed 5-fold expansion of CD45 + cells (56.6 x10 3 +-7.9 x10 3 ; n=3) and dramatic reduction of CD45 - cells number (4.6x10 3 +-1.0 x10 3 ; n=3). C . Clonal expansion of CD45 + cells. CD45 + cells were FACS sorted into 96-well plates at the density of 1 and 2 cells/well. After seven days of culture, we observed CD45 + cell expansion in 6.3+-2.1 wells (93.1+-14.5 cells/well) of 1-cell sort and 29.0+-4.3 wells (112.1+-21.2 cells/well) of the 2-cell sort (n=3). The difference between cell number in 1 and 2-cell sort was statistically not significant (p=0.78) suggesting that the difference in the number of wells with detected cell expansion was rather due to survival of sorted cells than a reflection of the number of cells sorted into a well. D. FGRS-induced generation of hematopoietic cells by hES-derived endothelium (hES-EC) . Representative e
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
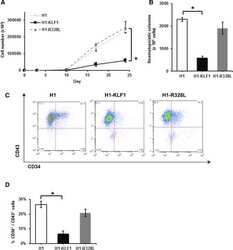
- Figure 1 Constitutive KLF1 expression in human embryonic stem cells (hESCs) results in reduced proliferation and hematopoietic progenitor cell production. ( A) : Cell counts throughout the erythroid differentiation protocol of control H1 hESCs (H1) and H1 hESCs transfected with a vector containing either wild type KLF1 (H1-KLF1) or the mutant form of KLF1 (H1-R328L). (B) : Total number of CFU-Cs generated from differentiating H1, H1-KLF1, and H1-R328L hESCs at day 10 of the differentiation protocol. (C) : Flow cytometry analysis of differentiating H1, H1-EKLF, and H1-R328L hESC at day 10 of the differentiation protocol using antibodies against CD34 and CD43 to mark hematopoietic progenitor cells (HPCs). (D) : Quantification flow cytometry data showing the %CD34 + /CD43 + HPCs at day 10 of the differentiation protocol. All data represents the mean of at least three independent experiments with error bars representing SEM. p values were calculated using two-way ANOVA followed by Tukey's multiple comparisons test (A) or one-way ANOVA followed by Holm-Sidak's multiple comparison test (B and D) (* p < .05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
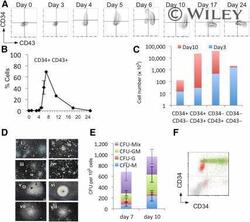
- Monitoring hematopoietic progenitor production throughout the differentiation protocol. Representative flow cytometry analysis of cells from days 0 to 24 of the differentiation protocol using antibodies against CD34 and CD43 (A) and the quantification of the percentage positive (B) was performed from at least three independent experiments. Absolute cell numbers of the differentiation represented in (A) are shown for days 3 and 10 (C) . Representative colonies generated from day 7 (Di-Div) and day 10 (Dv-Dviii) cells are shown, CFU-C activity having been assessed by methylcellulose assays on days 7 and 10 (E) . These data represent three independent experiments, and bars indicate the SEM. Flow cytometry analysis to assess the expression of Runx1C-GFP in the CD34/CD43 double-positive cell population; Runx1C-GFP + cells are shown in green and Runx1C-GFP - cells in red (F) . Original magnification x60. Abbreviations: CFU, colony-forming unit; G, granulocyte; GFP, green fluorescent protein; GM, granulocyte macrophage; M, macrophage; Q, quartile.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
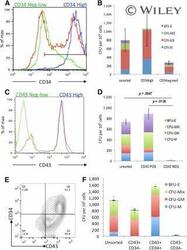
- CFU-C activity was enriched within the CD34 hi CD43 + cell population. CD34 + cells from day 7 cultures (A) and CD43 + cells from day 10 cultures (C) were sorted using magnetically activated cell sorting enrichment and reanalyzed by flow cytometry to assess purity (unsorted [red], enriched positive cells [blue], and flow-through low/negative cells [green]). Hematopoietic activity was quantified by plating these cell populations in CFU-C assays (B, D) . Day 10 cultures were stained for both CD34 and CD43 (E) and CD43 + /CD34 - , CD43 + /CD34 + double-positive, and CD43 - CD34 - double-negative cells were plated in CFU-C assays to quantify their hematopoietic activity (F) . Abbreviations: BFU-E, burst-forming unit erythroid; CFU, colony-forming unit; GM, granulocyte macrophage; M, monocyte; Max, maximum; MIX, mixed; NEG, negative; POS, positive; Q, quartile.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
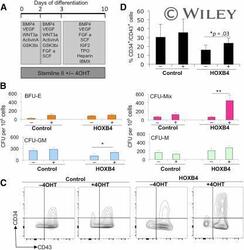
- Activation of HOXB4 enhanced the production of hematopoietic progenitors. Tamoxifen (labeled + or -) was added to cultures to activate HOXB4 in H1 human embryonic stem cells (hESCs) expressing HOXB4-ER T2 (labeled HOXB4) or parental H1 hESCs (labeled control) between days 0 and 10 (A) and then assessed for CFU-C activity (B) and expression of hematopoietic progenitor markers, CD34 and CD43, by flow cytometry (C, D) . Data were generated from four independent experiments, with error bars representing SEM. *, p = .03; **, p = .04. Abbreviations: 4OHT, tamoxifen; BFU-E, burst-forming unit erythroid; BMP4, bone morphogenetic protein 4; C, cell; CFU, colony-forming unit; FGF a, fibroblast growth factor-alpha; GM, granulocyte macrophage; GSK, glycogen synthase kinase; IBMX, isobutylmethylxanthine; IGF2, insulin-like growth factor 2; M, macrophage; Q, quartile; SCF, stem cell factor; TPO, thrombopoietin; VEGF, vascular endothelial growth factor.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
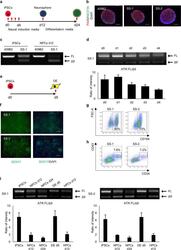
- Fig. 2 ATR c.2101 A>G mutation demonstrates cell type splicing. a Scheme of neural differentiation culture. Red arrows indicate the timing of splicing validation. b Immunostaining of SS-neurospheres at day 12. PAX6, red; NESTIN, green. 409B2 was used as the control iPSC line. c Splicing pattern validation of SS-iPSC-derived cells with RT-PCR products performed with primers spanning exon 9 ( n = 3, independent experiments). FL, full length isoform; Delta9, exon 9 skipped isoform. d Evaluation of ATR splicing patterns during early stage neural differentiation (day 0 to day 4). Quantification analysis was performed with the intensity ratio of the FL band and Delta9 band. Data represent the mean +- SEM ( n = 3, independent experiments). e Scheme of definitive endoderm (DE) differentiation. The red arrow indicates the time of splicing validation. f Immunostaining of SS-iPSC-derived DE cells at day 5. g Flow cytometric analysis of endodermal marker CXCR4 on SS-iPSC-derived DE cells. h Flow cytometric analysis of SS-iPSC-derived HPCs. i ATR RT-PCR analysis of different SS-iPSC-derived cell types with primers spanning exon 9. Quantification analysis was performed with the intensity ratio of the FL band and Delta9 band. Data represent the mean +- SEM ( n = 3, independent experiments). d, days
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 10 Hematopoiesis analysis Flow cytometry analysis of the indicated cell surface markers at day 4 of hemogenic endothelium culture.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
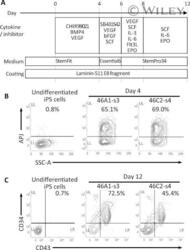
- The redifferentiation of gammadeltaT-iPSCs into hematopoietic lineage. (A) : A schematic diagram showing the redifferentation protocol for deriving hematopoietic progenitor cells from gammadeltaT-iPSCs. (B) : The flow cytometric analysis for a hematovascular precursor marker (APJ) in gammadeltaT-iPSC-derived cells. The percentages of APJ-positive cells are shown. (C) : The flow cytometric analysis for hematopoietic lineage markers (CD34 and CD43) in gammadeltaT-iPSC-derived cells. The percentages of CD34/CD43 double-positive cells are shown. Abbreviations: iPSCs, induced pluripotent stem cells.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry