Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [2]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 701241 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- APOE Recombinant Rabbit Monoclonal Antibody (16H22L18)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- 16H22L18
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Monomeric C-reactive protein via endothelial CD31 for neurovascular inflammation in an ApoE genotype-dependent pattern: A risk factor for Alzheimer's disease?
Monocyte-derived alveolar macrophage apolipoprotein E participates in pulmonary fibrosis resolution.
Differential Signaling Mediated by ApoE2, ApoE3, and ApoE4 in Human Neurons Parallels Alzheimer's Disease Risk.
Toxic amyloid-β oligomers induced self-replication in astrocytes triggering neuronal injury.
Zhang Z, Na H, Gan Q, Tao Q, Alekseyev Y, Hu J, Yan Z, Yang JB, Tian H, Zhu S, Li Q, Rajab IM, Blusztajn JK, Wolozin B, Emili A, Zhang X, Stein T, Potempa LA, Qiu WQ
Aging cell 2021 Nov;20(11):e13501
Aging cell 2021 Nov;20(11):e13501
Monocyte-derived alveolar macrophage apolipoprotein E participates in pulmonary fibrosis resolution.
Cui H, Jiang D, Banerjee S, Xie N, Kulkarni T, Liu RM, Duncan SR, Liu G
JCI insight 2020 Mar 12;5(5)
JCI insight 2020 Mar 12;5(5)
Differential Signaling Mediated by ApoE2, ApoE3, and ApoE4 in Human Neurons Parallels Alzheimer's Disease Risk.
Huang YA, Zhou B, Nabet AM, Wernig M, Südhof TC
The Journal of neuroscience : the official journal of the Society for Neuroscience 2019 Sep 11;39(37):7408-7427
The Journal of neuroscience : the official journal of the Society for Neuroscience 2019 Sep 11;39(37):7408-7427
Toxic amyloid-β oligomers induced self-replication in astrocytes triggering neuronal injury.
Wang W, Hou TT, Jia LF, Wu QQ, Quan MN, Jia JP
EBioMedicine 2019 Apr;42:174-187
EBioMedicine 2019 Apr;42:174-187
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
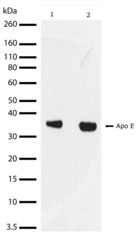
- Experimental details
- Western blot analysis of ApoE in mouse brain (lane 1) and rat liver lysate (lane 2) using an ApoE recombinant rabbit monoclonal antibody (Product # 701241) at a dilution of 2 µg/mL. Samples were detected using chemiluminescence (ECL). Results show a band at ~34kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
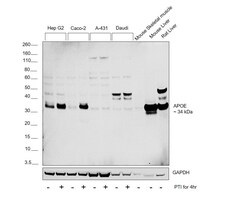
- Experimental details
- Western blot was performed using Anti-APOE Recombinant Rabbit Monoclonal Antibody (16H22L18) (Product # 701241) and a 34 kDa band corresponding to APOE was observed across the cell lines and tissues tested. Whole cell extracts (30 µg lysate) of Hep G2 (Lane 1), Hep G2 treated with Protein Transport Inhibitor (PTI; 1x for 4 hours) (Lane 2), Caco-2 (Lane 3), Caco-2 treated with Protein Transport Inhibitor (PTI; 1x for 4 hours) (Lane 4), A-431 (Lane 5), A-431 treated with Protein Transport Inhibitor (PTI; 1x for 4 hours) (Lane 6), Daudi (Lane 7), Daudi treated with Protein Transport Inhibitor (PTI; 1x for 4 hours) (Lane 8), Mouse Skeletal Muscle (Lane 9), Mouse Liver (Lane 10), Rat Liver (Lane 11) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (2 ug/ml) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036,1:10000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on Fig 1: Human plasma (Lane 1); Fig 2: Whole cell extracts (30 µg lysate) of Hep G2 (Lane 1) and Caco-2 (Lane 2). The blots were probed with Anti-ApoE Rabbit Monoclonal Antibody (Product # 701241, 2 µg/mL) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal Secondary Antibody, HRP conjugate (Product # A27036, 0.4 µg/mL, 1:2500 dilution). In Fig 1, a 36 kDa band corresponding to ApoE was observed. In Fig 2, a 34 kDa band was observed across both cell lines, and, an additional 36 kDa band corresponding to an isoform of ApoE was observed in Lane 1. Known quantity of protein samples were electrophoresed using Novex®NuPAGE®12 % Bis-Tris gel (Product # NP0342BOX), XCell SureLock Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
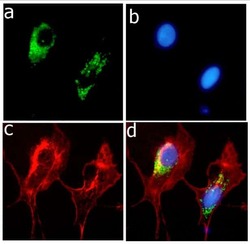
- Experimental details
- Immunofluorescent analysis of ApoE in HepG2 cells using an ApoE recombinant rabbit monoclonal antibody (Product # 701241) followed by detection using an Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody (green) (Image A). Nuclei were stained using DAPI (Image B) and actin stained with Alexa Fluor 594 phalloidin (red) (image C). Image D is a composite image showing cytoplasmic localization of ApoE.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
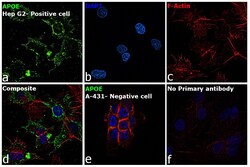
- Experimental details
- Immunofluorescence analysis of APOE was performed using 70% confluent log phase Hep G2 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with APOE Recombinant Rabbit Monoclonal Antibody (16H22L18) (Product # 701241) at 1:100 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034), (1:2000), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Cytoplasmic localization. Panel e represents A-431 showing no expression of APOE. Panel f represents control Hep G2 cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
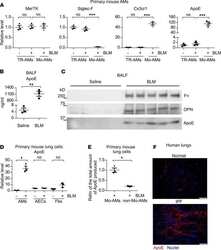
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
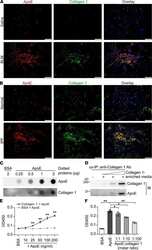
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
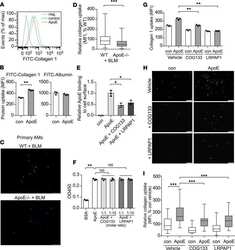
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
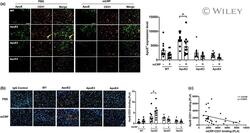
- Experimental details
- 6 FIGURE The binding of ApoE protein to CD31 + endothelial cells across ApoE genotype mice. (a) Because the endothelial CD31-mCRP bindings were different depending on ApoE genotypes, we hypothesized the association between ApoE protein and CD31. Representative images of double immunostaining of ApoE (green) and CD31 (red) and the merged images (yellow) of the cortex sections of WT vs. different ApoE knock-in mice are shown. The ApoE + fluorescence intensity was compared between PBS and mCRP-treated mice and reached a significant difference only in the ApoE2 mice ( p = 0.02). n = 11-14 mice in each group. (b) Further, PLA was applied to detect the binding of ApoE and CD31 on cortex sections from different ApoE genotype mice treated with PBS versus mCRP. Positive PLA fluorescence signals are shown in orange, and nuclei are stained with DAPI (blue). Quantifications of orange fluorescence intensity of ApoE-CD31 binding in the cortex are shown and again reached a significant difference only in ApoE2 mice by mCRP treatment ( p = 0.04). (c) We found that PLA (mCRP-CD31 binding) levels were negatively correlated with PLA (ApoE-CD31 binding) in the brain ( r = -0.51, p = 0.005). n = 8 in each group. Data are expressed as the mean +- SEM. Student's t -test and two-way ANOVA with Tukey's post hoc test and Pearson or Spearman correlation test were applied. * p < 0.05, ** p < 0.01, *** p < 0.001. The scale bars are 50, 20, and 10 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- 7 FIGURE Characterization of mCRP in blood-facing endothelial of cerebrovasculature of AD brains. The human temporal cortex of healthy controls ( n = 8) and AD patients ( n = 10) were used for immunostaining. (a) Representative images of CD31 + capillaries (micorvessels; red), mCRP (green) and the merged images (yellow) in the temporal cortex are shown and quantified. AD brains had higher mCRP signal in microvessels (yellow; p = 0.01) and tended to have lower levels of microvessel CD31 expression (red; p = 0.09) than control brains. AD brains tended to have higher levels of binding between mCRP and CD31 detected by using PLA ( p = 0.09) than control brains. (b) Western blots for pCD31 and CD31 expression in the temporal cortex were performed and quantified after normalization against beta-actin and compared between AD and controls. AD brains had higher levels of pCD31 ( p = 0.02), tended to have lower levels of total CD31 ( p = 0.07), and had a higher pCD31/CD31 ratio ( p = 0.03) than control brains. (c) Double immunostaining of temporal cortex sections with CD31 (red), pCD31 (green) and the merged images (yellow) were conducted to examine CD31 phosphorylation and microvessel integrity. AD brains had higher brain pCD31 levels ( p = 0.05), pCD31/CD31 ratio ( p = 0.03), and microvessel pCD31 ( p = 0.01). Measurement of the lengths of CD31 + microvessels from 3D images revealed shorter lengths in AD brains than in controls ( p < 0.001). (d) To further confirm that low expression
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 4 Natural AbetaOs induced amyloidogenic processing in astrocytes. Western blots and quantitative analysis showing BACE1, sAPPbeta and apoE levels in primary astrocyte (a) and neuron (e) cultures. Bar graphs illustrating the protein expression levels of BACE1, sAPPbeta and apoE in primary astrocyte (b-d) and neuron (f-h) (one-way ANOVA, b: F = 52.03, p < .0001, c: F = 69.79, p < .0001, d: F = 46.81, p < .0001, f: F = 0.436, p < .729, g: F = 0.143, p = .933, h: F = 0.126, p = .943, n = 6/group). The levels of Abeta40 and Abeta42 in the medium obtained from astrocytes or neurons (i-l) (one-way ANOVA, i: F = 310, p < .0001, j: F = 53.06, p < .0001, k: F = 2.73, p = .051, l: F = 2.11, p = .132, n = 6/group). The data are shown as the mean +- SEM; * p < .05, ** p < .01. Fig. 4
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 5 Natural AbetaOs injection induced behavioral and biochemical changes in SD rats. (a) MWM escape latency during training (one-way ANOVA, F = 29.34, p < .0001; n = 10/group). (b) Time in the target quadrant (one-way ANOVA, F = 14.79, p < .0001, n = 10/group). (c) The number of crossings (one-way ANOVA, F = 9.81, p = .0006, n = 10/group). (d) Western blot showing dynamic changes in Abeta assembly (4G8) in the brains of SD rats (time indicated above the lanes). The right lanes indicate the respective migration positions of monomers, trimers, nonamers, dodecamers and sAPP. (e-h) Bar graphs illustrating the quantitative analysis of monomers, trimers, nonamers and dodecamers (one-way ANOVA, e: F = 108.3, p < .0001; f: F = 164.9, p < .0001; g: F = 45.89, p < .0001; h: F = 61.43, p < .0001; n = 6/group). (i) Western blots showing the expression of GFAP, BACE1, sAPPbeta and apoE in rat brains. (j-m) Bar graphs illustrating the quantitative analysis of the Western blot results (one-way ANOVA, j: F = 61.83, p < .0001; k: F = 31.09, p < .0001; l: F = 41.37, p < .0001; m: F = 72.38, p < .0001; n = 6/group). The data are shown as the mean +- SEM; * p < .05, ** p < .01. Fig. 5
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot