MA3-011
antibody from Invitrogen Antibodies
Targeting: HSP90AA1
FLJ31884, Hsp89, Hsp90, HSP90N, HSPC1, HSPCA
Antibody data
- Antibody Data
- Antigen structure
- References [64]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [4]
- Immunohistochemistry [3]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA3-011 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- HSP90 alpha Monoclonal Antibody (3G3)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA3-011 detects heat shock protein 90 kDa (HSP90) from human, mouse, rat, rainbow trout, and chicken tissues. MA3-011 has been successfully used in Western blot, immunofluorescence, immunohistochemistry, and immunoprecipitation procedures. By Western blot, this antibody detects a 90 kDa protein representing HSP90 from Hepa 1 cell extract. It has been reported that up to 90% of cytosolic HSP90 can be immunoprecipitated with this product under low ionic strength conditions. For Western blots using MA3-011, nitrocellulose is recommended. PVDF membranes typically do not yield acceptable results with this antibody. Before use, centrifuge suspension (4000 g for 15 minutes) and discard supernatant. Resuspend pellet in 200 µL of PBS, pH 7.2. If desired, 0.05% sodium azide may be added to resuspended solution. Removal of residual ammonium sulfate is not usually necessary for immunoprecipitation and Western blot procedures, however, any other uses may require its removal by dialysis or equivalent method. Stable for 6 months after PBS resuspension. The MA3-011 antigen is purified HSP90 from Hepa 1 cytosol.
- Reactivity
- Human, Mouse, Rat, Chicken/Avian
- Host
- Mouse
- Isotype
- IgM
- Antibody clone number
- 3G3
- Vial size
- 400 µL
- Concentration
- Conc. Not Determined
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references ERβ decreases breast cancer cell survival by regulating the IRE1/XBP-1 pathway.
Hsp90 regulates the phosphorylation and activity of serum- and glucocorticoid-regulated kinase-1.
Functional analysis of the LXXLL motifs of the human glucocorticoid receptor: association with altered ligand affinity.
The distinct agonistic properties of the phenylpyrazolosteroid cortivazol reveal interdomain communication within the glucocorticoid receptor.
Cytoplasmic localization of pregnane X receptor and ligand-dependent nuclear translocation in mouse liver.
Retrograde transport of the glucocorticoid receptor in neurites requires dynamic assembly of complexes with the protein chaperone hsp90 and is linked to the CHIP component of the machinery for proteasomal degradation.
Interaction of neuropeptide Y and Hsp90 through a novel peptide binding region.
Tumor-derived, chaperone-rich cell lysate activates dendritic cells and elicits potent antitumor immunity.
Modification of an essential amino group in the mineralocorticoid receptor evidences a differential conformational change of the receptor protein upon binding of antagonists, natural agonists and the synthetic agonist 11,19-oxidoprogesterone.
Modification of an essential amino group in the mineralocorticoid receptor evidences a differential conformational change of the receptor protein upon binding of antagonists, natural agonists and the synthetic agonist 11,19-oxidoprogesterone.
All of the protein interactions that link steroid receptor.hsp90.immunophilin heterocomplexes to cytoplasmic dynein are common to plant and animal cells.
All of the protein interactions that link steroid receptor.hsp90.immunophilin heterocomplexes to cytoplasmic dynein are common to plant and animal cells.
Host cell factor requirement for hepatitis C virus enzyme maturation.
Stoichiometry, abundance, and functional significance of the hsp90/hsp70-based multiprotein chaperone machinery in reticulocyte lysate.
The hsp90 chaperone complex regulates intracellular localization of the dioxin receptor.
In vitro reconstitution of a functional duck hepatitis B virus reverse transcriptase: posttranslational activation by Hsp90.
The immunophilin-like protein XAP2 regulates ubiquitination and subcellular localization of the dioxin receptor.
The dominant negative activity of the human glucocorticoid receptor beta isoform. Specificity and mechanisms of action.
KF25706, a novel oxime derivative of radicicol, exhibits in vivo antitumor activity via selective depletion of Hsp90 binding signaling molecules.
Isolation of MHC class I-restricted tumor antigen peptide and its precursors associated with heat shock proteins hsp70, hsp90, and gp96.
Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein.
Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein.
Redox-dependent regulation of nuclear import of the glucocorticoid receptor.
The benzoquinone ansamycin 17-allylamino-17-demethoxygeldanamycin binds to HSP90 and shares important biologic activities with geldanamycin.
Binding of hsp90 to the glucocorticoid receptor requires a specific 7-amino acid sequence at the amino terminus of the hormone-binding domain.
Active participation of Hsp90 in the biogenesis of the trimeric reovirus cell attachment protein sigma1.
Active participation of Hsp90 in the biogenesis of the trimeric reovirus cell attachment protein sigma1.
p50(cdc37) binds directly to the catalytic domain of Raf as well as to a site on hsp90 that is topologically adjacent to the tetratricopeptide repeat binding site.
2,3,7,8-Tetrachlorodibenzo-p-dioxin-induced activation of a protein tyrosine kinase, pp60src, in murine hepatic cytosol using a cell-free system.
Evidence that the beta-isoform of the human glucocorticoid receptor does not act as a physiologically significant repressor.
Activation of hypoxia-inducible factor 1alpha: posttranscriptional regulation and conformational change by recruitment of the Arnt transcription factor.
Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation.
Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation.
Disruption of the glucocorticoid receptor assembly with heat shock protein 90 by a peptidic antiglucocorticoid.
Protein phosphatase 5 is a major component of glucocorticoid receptor.hsp90 complexes with properties of an FK506-binding immunophilin.
Protein phosphatase 5 is a major component of glucocorticoid receptor.hsp90 complexes with properties of an FK506-binding immunophilin.
Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor.
Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor.
Compartmentation of alpha-internexin and neurofilament triplet proteins in cultured hippocampal neurons.
A model of protein targeting mediated by immunophilins and other proteins that bind to hsp90 via tetratricopeptide repeat domains.
A model of protein targeting mediated by immunophilins and other proteins that bind to hsp90 via tetratricopeptide repeat domains.
Hsp90 is required for the activity of a hepatitis B virus reverse transcriptase.
The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant.
The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant.
Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90.
Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90.
Definition of a minimal domain of the dioxin receptor that is associated with Hsp90 and maintains wild type ligand binding affinity and specificity.
Definition of a minimal domain of the dioxin receptor that is associated with Hsp90 and maintains wild type ligand binding affinity and specificity.
Identification of functional domains of the aryl hydrocarbon receptor.
Identification of functional domains of the aryl hydrocarbon receptor.
The basic helix-loop-helix/PAS factor Sim is associated with hsp90. Implications for regulation by interaction with partner factors.
The basic helix-loop-helix/PAS factor Sim is associated with hsp90. Implications for regulation by interaction with partner factors.
The 23-kDa acidic protein in reticulocyte lysate is the weakly bound component of the hsp foldosome that is required for assembly of the glucocorticoid receptor into a functional heterocomplex with hsp90.
A tyrosine kinase-dependent pathway regulates ligand-dependent activation of the dioxin receptor in human keratinocytes.
All of the factors required for assembly of the glucocorticoid receptor into a functional heterocomplex with heat shock protein 90 are preassociated in a self-sufficient protein folding structure, a "foldosome".
Comparison of tumor-specific immunogenicities of stress-induced proteins gp96, hsp90, and hsp70.
Identification of transactivation and repression functions of the dioxin receptor and its basic helix-loop-helix/PAS partner factor Arnt: inducible versus constitutive modes of regulation.
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
Characterization of the protein-protein interactions determining the heat shock protein (hsp90.hsp70.hsp56) heterocomplex.
ATP-dependent protein refolding activity in reticulocyte lysate. Evidence for the participation of different chaperone components.
ATP-dependent protein refolding activity in reticulocyte lysate. Evidence for the participation of different chaperone components.
Evidence that the 90-kDa heat shock protein (HSP90) exists in cytosol in heteromeric complexes containing HSP70 and three other proteins with Mr of 63,000, 56,000, and 50,000.
Rajapaksa G, Nikolos F, Bado I, Clarke R, Gustafsson JÅ, Thomas C
Oncogene 2015 Jul 30;34(31):4130-41
Oncogene 2015 Jul 30;34(31):4130-41
Hsp90 regulates the phosphorylation and activity of serum- and glucocorticoid-regulated kinase-1.
Belova L, Brickley DR, Ky B, Sharma SK, Conzen SD
The Journal of biological chemistry 2008 Jul 4;283(27):18821-31
The Journal of biological chemistry 2008 Jul 4;283(27):18821-31
Functional analysis of the LXXLL motifs of the human glucocorticoid receptor: association with altered ligand affinity.
Dong DD, Jewell CM, Bienstock RJ, Cidlowski JA
The Journal of steroid biochemistry and molecular biology 2006 Oct;101(2-3):106-17
The Journal of steroid biochemistry and molecular biology 2006 Oct;101(2-3):106-17
The distinct agonistic properties of the phenylpyrazolosteroid cortivazol reveal interdomain communication within the glucocorticoid receptor.
Yoshikawa N, Yamamoto K, Shimizu N, Yamada S, Morimoto C, Tanaka H
Molecular endocrinology (Baltimore, Md.) 2005 May;19(5):1110-24
Molecular endocrinology (Baltimore, Md.) 2005 May;19(5):1110-24
Cytoplasmic localization of pregnane X receptor and ligand-dependent nuclear translocation in mouse liver.
Squires EJ, Sueyoshi T, Negishi M
The Journal of biological chemistry 2004 Nov 19;279(47):49307-14
The Journal of biological chemistry 2004 Nov 19;279(47):49307-14
Retrograde transport of the glucocorticoid receptor in neurites requires dynamic assembly of complexes with the protein chaperone hsp90 and is linked to the CHIP component of the machinery for proteasomal degradation.
Galigniana MD, Harrell JM, Housley PR, Patterson C, Fisher SK, Pratt WB
Brain research. Molecular brain research 2004 Apr 7;123(1-2):27-36
Brain research. Molecular brain research 2004 Apr 7;123(1-2):27-36
Interaction of neuropeptide Y and Hsp90 through a novel peptide binding region.
Ishiwatari-Hayasaka H, Maruya M, Sreedhar AS, Nemoto TK, Csermely P, Yahara I
Biochemistry 2003 Nov 11;42(44):12972-80
Biochemistry 2003 Nov 11;42(44):12972-80
Tumor-derived, chaperone-rich cell lysate activates dendritic cells and elicits potent antitumor immunity.
Zeng Y, Feng H, Graner MW, Katsanis E
Blood 2003 Jun 1;101(11):4485-91
Blood 2003 Jun 1;101(11):4485-91
Modification of an essential amino group in the mineralocorticoid receptor evidences a differential conformational change of the receptor protein upon binding of antagonists, natural agonists and the synthetic agonist 11,19-oxidoprogesterone.
Piwien-Pilipuk G, Kanelakis KC, Ghini AA, Lantos CP, Litwack G, Burton G, Galigniana MD
Biochimica et biophysica acta 2002 Feb 13;1589(1):31-48
Biochimica et biophysica acta 2002 Feb 13;1589(1):31-48
Modification of an essential amino group in the mineralocorticoid receptor evidences a differential conformational change of the receptor protein upon binding of antagonists, natural agonists and the synthetic agonist 11,19-oxidoprogesterone.
Piwien-Pilipuk G, Kanelakis KC, Ghini AA, Lantos CP, Litwack G, Burton G, Galigniana MD
Biochimica et biophysica acta 2002 Feb 13;1589(1):31-48
Biochimica et biophysica acta 2002 Feb 13;1589(1):31-48
All of the protein interactions that link steroid receptor.hsp90.immunophilin heterocomplexes to cytoplasmic dynein are common to plant and animal cells.
Harrell JM, Kurek I, Breiman A, Radanyi C, Renoir JM, Pratt WB, Galigniana MD
Biochemistry 2002 Apr 30;41(17):5581-7
Biochemistry 2002 Apr 30;41(17):5581-7
All of the protein interactions that link steroid receptor.hsp90.immunophilin heterocomplexes to cytoplasmic dynein are common to plant and animal cells.
Harrell JM, Kurek I, Breiman A, Radanyi C, Renoir JM, Pratt WB, Galigniana MD
Biochemistry 2002 Apr 30;41(17):5581-7
Biochemistry 2002 Apr 30;41(17):5581-7
Host cell factor requirement for hepatitis C virus enzyme maturation.
Waxman L, Whitney M, Pollok BA, Kuo LC, Darke PL
Proceedings of the National Academy of Sciences of the United States of America 2001 Nov 20;98(24):13931-5
Proceedings of the National Academy of Sciences of the United States of America 2001 Nov 20;98(24):13931-5
Stoichiometry, abundance, and functional significance of the hsp90/hsp70-based multiprotein chaperone machinery in reticulocyte lysate.
Murphy PJ, Kanelakis KC, Galigniana MD, Morishima Y, Pratt WB
The Journal of biological chemistry 2001 Aug 10;276(32):30092-8
The Journal of biological chemistry 2001 Aug 10;276(32):30092-8
The hsp90 chaperone complex regulates intracellular localization of the dioxin receptor.
Kazlauskas A, Sundström S, Poellinger L, Pongratz I
Molecular and cellular biology 2001 Apr;21(7):2594-607
Molecular and cellular biology 2001 Apr;21(7):2594-607
In vitro reconstitution of a functional duck hepatitis B virus reverse transcriptase: posttranslational activation by Hsp90.
Hu J, Anselmo D
Journal of virology 2000 Dec;74(24):11447-55
Journal of virology 2000 Dec;74(24):11447-55
The immunophilin-like protein XAP2 regulates ubiquitination and subcellular localization of the dioxin receptor.
Kazlauskas A, Poellinger L, Pongratz I
The Journal of biological chemistry 2000 Dec 29;275(52):41317-24
The Journal of biological chemistry 2000 Dec 29;275(52):41317-24
The dominant negative activity of the human glucocorticoid receptor beta isoform. Specificity and mechanisms of action.
Oakley RH, Jewell CM, Yudt MR, Bofetiado DM, Cidlowski JA
The Journal of biological chemistry 1999 Sep 24;274(39):27857-66
The Journal of biological chemistry 1999 Sep 24;274(39):27857-66
KF25706, a novel oxime derivative of radicicol, exhibits in vivo antitumor activity via selective depletion of Hsp90 binding signaling molecules.
Soga S, Neckers LM, Schulte TW, Shiotsu Y, Akasaka K, Narumi H, Agatsuma T, Ikuina Y, Murakata C, Tamaoki T, Akinaga S
Cancer research 1999 Jun 15;59(12):2931-8
Cancer research 1999 Jun 15;59(12):2931-8
Isolation of MHC class I-restricted tumor antigen peptide and its precursors associated with heat shock proteins hsp70, hsp90, and gp96.
Ishii T, Udono H, Yamano T, Ohta H, Uenaka A, Ono T, Hizuta A, Tanaka N, Srivastava PK, Nakayama E
Journal of immunology (Baltimore, Md. : 1950) 1999 Feb 1;162(3):1303-9
Journal of immunology (Baltimore, Md. : 1950) 1999 Feb 1;162(3):1303-9
Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein.
Silverstein AM, Galigniana MD, Kanelakis KC, Radanyi C, Renoir JM, Pratt WB
The Journal of biological chemistry 1999 Dec 24;274(52):36980-6
The Journal of biological chemistry 1999 Dec 24;274(52):36980-6
Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein.
Silverstein AM, Galigniana MD, Kanelakis KC, Radanyi C, Renoir JM, Pratt WB
The Journal of biological chemistry 1999 Dec 24;274(52):36980-6
The Journal of biological chemistry 1999 Dec 24;274(52):36980-6
Redox-dependent regulation of nuclear import of the glucocorticoid receptor.
Okamoto K, Tanaka H, Ogawa H, Makino Y, Eguchi H, Hayashi S, Yoshikawa N, Poellinger L, Umesono K, Makino I
The Journal of biological chemistry 1999 Apr 9;274(15):10363-71
The Journal of biological chemistry 1999 Apr 9;274(15):10363-71
The benzoquinone ansamycin 17-allylamino-17-demethoxygeldanamycin binds to HSP90 and shares important biologic activities with geldanamycin.
Schulte TW, Neckers LM
Cancer chemotherapy and pharmacology 1998;42(4):273-9
Cancer chemotherapy and pharmacology 1998;42(4):273-9
Binding of hsp90 to the glucocorticoid receptor requires a specific 7-amino acid sequence at the amino terminus of the hormone-binding domain.
Xu M, Dittmar KD, Giannoukos G, Pratt WB, Simons SS Jr
The Journal of biological chemistry 1998 May 29;273(22):13918-24
The Journal of biological chemistry 1998 May 29;273(22):13918-24
Active participation of Hsp90 in the biogenesis of the trimeric reovirus cell attachment protein sigma1.
Gilmore R, Coffey MC, Lee PW
The Journal of biological chemistry 1998 Jun 12;273(24):15227-33
The Journal of biological chemistry 1998 Jun 12;273(24):15227-33
Active participation of Hsp90 in the biogenesis of the trimeric reovirus cell attachment protein sigma1.
Gilmore R, Coffey MC, Lee PW
The Journal of biological chemistry 1998 Jun 12;273(24):15227-33
The Journal of biological chemistry 1998 Jun 12;273(24):15227-33
p50(cdc37) binds directly to the catalytic domain of Raf as well as to a site on hsp90 that is topologically adjacent to the tetratricopeptide repeat binding site.
Silverstein AM, Grammatikakis N, Cochran BH, Chinkers M, Pratt WB
The Journal of biological chemistry 1998 Aug 7;273(32):20090-5
The Journal of biological chemistry 1998 Aug 7;273(32):20090-5
2,3,7,8-Tetrachlorodibenzo-p-dioxin-induced activation of a protein tyrosine kinase, pp60src, in murine hepatic cytosol using a cell-free system.
Blankenship A, Matsumura F
Molecular pharmacology 1997 Oct;52(4):667-75
Molecular pharmacology 1997 Oct;52(4):667-75
Evidence that the beta-isoform of the human glucocorticoid receptor does not act as a physiologically significant repressor.
Hecht K, Carlstedt-Duke J, Stierna P, Gustafsson J, Brönnegârd M, Wikström AC
The Journal of biological chemistry 1997 Oct 17;272(42):26659-64
The Journal of biological chemistry 1997 Oct 17;272(42):26659-64
Activation of hypoxia-inducible factor 1alpha: posttranscriptional regulation and conformational change by recruitment of the Arnt transcription factor.
Kallio PJ, Pongratz I, Gradin K, McGuire J, Poellinger L
Proceedings of the National Academy of Sciences of the United States of America 1997 May 27;94(11):5667-72
Proceedings of the National Academy of Sciences of the United States of America 1997 May 27;94(11):5667-72
Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation.
Dittmar KD, Pratt WB
The Journal of biological chemistry 1997 May 16;272(20):13047-54
The Journal of biological chemistry 1997 May 16;272(20):13047-54
Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation.
Dittmar KD, Pratt WB
The Journal of biological chemistry 1997 May 16;272(20):13047-54
The Journal of biological chemistry 1997 May 16;272(20):13047-54
Disruption of the glucocorticoid receptor assembly with heat shock protein 90 by a peptidic antiglucocorticoid.
Dao-Phan HP, Formstecher P, Lefebvre P
Molecular endocrinology (Baltimore, Md.) 1997 Jun;11(7):962-72
Molecular endocrinology (Baltimore, Md.) 1997 Jun;11(7):962-72
Protein phosphatase 5 is a major component of glucocorticoid receptor.hsp90 complexes with properties of an FK506-binding immunophilin.
Silverstein AM, Galigniana MD, Chen MS, Owens-Grillo JK, Chinkers M, Pratt WB
The Journal of biological chemistry 1997 Jun 27;272(26):16224-30
The Journal of biological chemistry 1997 Jun 27;272(26):16224-30
Protein phosphatase 5 is a major component of glucocorticoid receptor.hsp90 complexes with properties of an FK506-binding immunophilin.
Silverstein AM, Galigniana MD, Chen MS, Owens-Grillo JK, Chinkers M, Pratt WB
The Journal of biological chemistry 1997 Jun 27;272(26):16224-30
The Journal of biological chemistry 1997 Jun 27;272(26):16224-30
Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor.
Gradin K, McGuire J, Wenger RH, Kvietikova I, fhitelaw ML, Toftgård R, Tora L, Gassmann M, Poellinger L
Molecular and cellular biology 1996 Oct;16(10):5221-31
Molecular and cellular biology 1996 Oct;16(10):5221-31
Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor.
Gradin K, McGuire J, Wenger RH, Kvietikova I, fhitelaw ML, Toftgård R, Tora L, Gassmann M, Poellinger L
Molecular and cellular biology 1996 Oct;16(10):5221-31
Molecular and cellular biology 1996 Oct;16(10):5221-31
Compartmentation of alpha-internexin and neurofilament triplet proteins in cultured hippocampal neurons.
Benson DL, Mandell JW, Shaw G, Banker G
Journal of neurocytology 1996 Mar;25(3):181-96
Journal of neurocytology 1996 Mar;25(3):181-96
A model of protein targeting mediated by immunophilins and other proteins that bind to hsp90 via tetratricopeptide repeat domains.
Owens-Grillo JK, Czar MJ, Hutchison KA, Hoffmann K, Perdew GH, Pratt WB
The Journal of biological chemistry 1996 Jun 7;271(23):13468-75
The Journal of biological chemistry 1996 Jun 7;271(23):13468-75
A model of protein targeting mediated by immunophilins and other proteins that bind to hsp90 via tetratricopeptide repeat domains.
Owens-Grillo JK, Czar MJ, Hutchison KA, Hoffmann K, Perdew GH, Pratt WB
The Journal of biological chemistry 1996 Jun 7;271(23):13468-75
The Journal of biological chemistry 1996 Jun 7;271(23):13468-75
Hsp90 is required for the activity of a hepatitis B virus reverse transcriptase.
Hu J, Seeger C
Proceedings of the National Academy of Sciences of the United States of America 1996 Feb 6;93(3):1060-4
Proceedings of the National Academy of Sciences of the United States of America 1996 Feb 6;93(3):1060-4
The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant.
Chen MS, Silverstein AM, Pratt WB, Chinkers M
The Journal of biological chemistry 1996 Dec 13;271(50):32315-20
The Journal of biological chemistry 1996 Dec 13;271(50):32315-20
The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant.
Chen MS, Silverstein AM, Pratt WB, Chinkers M
The Journal of biological chemistry 1996 Dec 13;271(50):32315-20
The Journal of biological chemistry 1996 Dec 13;271(50):32315-20
Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90.
Blagosklonny MV, Toretsky J, Bohen S, Neckers L
Proceedings of the National Academy of Sciences of the United States of America 1996 Aug 6;93(16):8379-83
Proceedings of the National Academy of Sciences of the United States of America 1996 Aug 6;93(16):8379-83
Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90.
Blagosklonny MV, Toretsky J, Bohen S, Neckers L
Proceedings of the National Academy of Sciences of the United States of America 1996 Aug 6;93(16):8379-83
Proceedings of the National Academy of Sciences of the United States of America 1996 Aug 6;93(16):8379-83
Definition of a minimal domain of the dioxin receptor that is associated with Hsp90 and maintains wild type ligand binding affinity and specificity.
Coumailleau P, Poellinger L, Gustafsson JA, Whitelaw ML
The Journal of biological chemistry 1995 Oct 20;270(42):25291-300
The Journal of biological chemistry 1995 Oct 20;270(42):25291-300
Definition of a minimal domain of the dioxin receptor that is associated with Hsp90 and maintains wild type ligand binding affinity and specificity.
Coumailleau P, Poellinger L, Gustafsson JA, Whitelaw ML
The Journal of biological chemistry 1995 Oct 20;270(42):25291-300
The Journal of biological chemistry 1995 Oct 20;270(42):25291-300
Identification of functional domains of the aryl hydrocarbon receptor.
Fukunaga BN, Probst MR, Reisz-Porszasz S, Hankinson O
The Journal of biological chemistry 1995 Dec 8;270(49):29270-8
The Journal of biological chemistry 1995 Dec 8;270(49):29270-8
Identification of functional domains of the aryl hydrocarbon receptor.
Fukunaga BN, Probst MR, Reisz-Porszasz S, Hankinson O
The Journal of biological chemistry 1995 Dec 8;270(49):29270-8
The Journal of biological chemistry 1995 Dec 8;270(49):29270-8
The basic helix-loop-helix/PAS factor Sim is associated with hsp90. Implications for regulation by interaction with partner factors.
McGuire J, Coumailleau P, Whitelaw ML, Gustafsson JA, Poellinger L
The Journal of biological chemistry 1995 Dec 29;270(52):31353-7
The Journal of biological chemistry 1995 Dec 29;270(52):31353-7
The basic helix-loop-helix/PAS factor Sim is associated with hsp90. Implications for regulation by interaction with partner factors.
McGuire J, Coumailleau P, Whitelaw ML, Gustafsson JA, Poellinger L
The Journal of biological chemistry 1995 Dec 29;270(52):31353-7
The Journal of biological chemistry 1995 Dec 29;270(52):31353-7
The 23-kDa acidic protein in reticulocyte lysate is the weakly bound component of the hsp foldosome that is required for assembly of the glucocorticoid receptor into a functional heterocomplex with hsp90.
Hutchison KA, Stancato LF, Owens-Grillo JK, Johnson JL, Krishna P, Toft DO, Pratt WB
The Journal of biological chemistry 1995 Aug 11;270(32):18841-7
The Journal of biological chemistry 1995 Aug 11;270(32):18841-7
A tyrosine kinase-dependent pathway regulates ligand-dependent activation of the dioxin receptor in human keratinocytes.
Gradin K, Whitelaw ML, Toftgård R, Poellinger L, Berghard A
The Journal of biological chemistry 1994 Sep 23;269(38):23800-7
The Journal of biological chemistry 1994 Sep 23;269(38):23800-7
All of the factors required for assembly of the glucocorticoid receptor into a functional heterocomplex with heat shock protein 90 are preassociated in a self-sufficient protein folding structure, a "foldosome".
Hutchison KA, Dittmar KD, Pratt WB
The Journal of biological chemistry 1994 Nov 11;269(45):27894-9
The Journal of biological chemistry 1994 Nov 11;269(45):27894-9
Comparison of tumor-specific immunogenicities of stress-induced proteins gp96, hsp90, and hsp70.
Udono H, Srivastava PK
Journal of immunology (Baltimore, Md. : 1950) 1994 Jun 1;152(11):5398-403
Journal of immunology (Baltimore, Md. : 1950) 1994 Jun 1;152(11):5398-403
Identification of transactivation and repression functions of the dioxin receptor and its basic helix-loop-helix/PAS partner factor Arnt: inducible versus constitutive modes of regulation.
Whitelaw ML, Gustafsson JA, Poellinger L
Molecular and cellular biology 1994 Dec;14(12):8343-55
Molecular and cellular biology 1994 Dec;14(12):8343-55
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
McGuire J, Whitelaw ML, Pongratz I, Gustafsson JA, Poellinger L
Molecular and cellular biology 1994 Apr;14(4):2438-46
Molecular and cellular biology 1994 Apr;14(4):2438-46
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
McGuire J, Whitelaw ML, Pongratz I, Gustafsson JA, Poellinger L
Molecular and cellular biology 1994 Apr;14(4):2438-46
Molecular and cellular biology 1994 Apr;14(4):2438-46
A cellular factor stimulates ligand-dependent release of hsp90 from the basic helix-loop-helix dioxin receptor.
McGuire J, Whitelaw ML, Pongratz I, Gustafsson JA, Poellinger L
Molecular and cellular biology 1994 Apr;14(4):2438-46
Molecular and cellular biology 1994 Apr;14(4):2438-46
Characterization of the protein-protein interactions determining the heat shock protein (hsp90.hsp70.hsp56) heterocomplex.
Czar MJ, Owens-Grillo JK, Dittmar KD, Hutchison KA, Zacharek AM, Leach KL, Deibel MR Jr, Pratt WB
The Journal of biological chemistry 1994 Apr 15;269(15):11155-61
The Journal of biological chemistry 1994 Apr 15;269(15):11155-61
ATP-dependent protein refolding activity in reticulocyte lysate. Evidence for the participation of different chaperone components.
Nimmesgern E, Hartl FU
FEBS letters 1993 Sep 27;331(1-2):25-30
FEBS letters 1993 Sep 27;331(1-2):25-30
ATP-dependent protein refolding activity in reticulocyte lysate. Evidence for the participation of different chaperone components.
Nimmesgern E, Hartl FU
FEBS letters 1993 Sep 27;331(1-2):25-30
FEBS letters 1993 Sep 27;331(1-2):25-30
Evidence that the 90-kDa heat shock protein (HSP90) exists in cytosol in heteromeric complexes containing HSP70 and three other proteins with Mr of 63,000, 56,000, and 50,000.
Perdew GH, Whitelaw ML
The Journal of biological chemistry 1991 Apr 15;266(11):6708-13
The Journal of biological chemistry 1991 Apr 15;266(11):6708-13
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
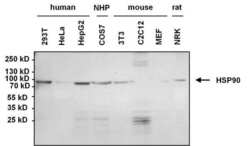
- Experimental details
- Western blot analysis of Heat Shock Protein 90 (Hsp90) was performed by loading 50 µg of various whole cell lysates and 15 µL PageRuler Prestained Protein Ladder (Product # 26616) onto a 4-20% Tris-HCl polyacrylamide gel. Proteins were transferred to a nitrocellulose membrane and blocked with 5% BSA/TBST for at least 1 hour. The membrane was probed with a Hsp90 monoclonal antibody (Product # MA3-011) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBS-0.1%Tween-20, and probed with a goat anti-mouse IgG + IgM secondary antibody (Product # 31446) at a dilution of 1:40,000 for 30 minutes. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34075).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
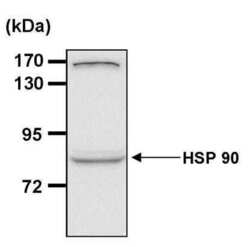
- Experimental details
- Western blot analysis of Heat Shock Protein 90 (Hsp90) was performed by loading 30 µg of HEK293T whole cell lysate per well onto an SDS-PAGE gel. Proteins were transferred to a PVDF membrane and blocked with 5% non-fat milk in TBST for 1 hour at room temperature. The membrane was probed with an Hsp90 monoclonal antibody (Product # MA3-011) at a dilution of 1:200 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgG (H + L) secondary antibody at a dilution of 1:40,000 for 1 hour at room temperature. Chemiluminescent detection was performed using ECL substrate. Data courtesy of the Innovators Program.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
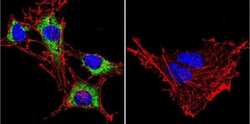
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 90 using Heat Shock Protein 90 Monoclonal antibody (3G3) (Product # MA3-011) shows staining in C6 glioma cells. Heat Shock Protein 90 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 90 (Product # MA3-011) at a dilution of 1:200 over night at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
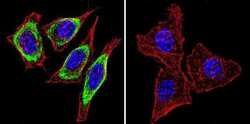
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 90 using Heat Shock Protein 90 Monoclonal antibody (3G3) (Product # MA3-011) shows staining in HeLa cells. Heat Shock Protein 90 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 90 (Product # MA3-011) at a dilution of 1:200 over night at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
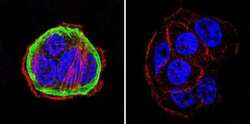
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 90 using Heat Shock Protein 90 Monoclonal antibody (3G3) (Product # MA3-011) shows staining in WiDr colon carcinoma cells. Heat Shock Protein 90 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 90 (Product # MA3-011) at a dilution of 1:200 over night at 4 C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
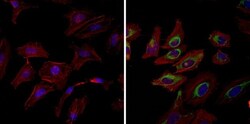
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 90 (Hsp90) (green) in HeLa cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed without (left panel) or with (right panel) a Hsp90 monoclonal antibody (Product # MA3-011), at a dilution of 1:100 for at least 1 hour at room temperature, washed with PBS, and incubated with a fluorescently-conjugated goat anti-mouse IgM secondary antibody at a dilution of 1:400 for 30 minutes at room temperature. F-Actin (red) was stained with DyLight-554 Phalloidin (Product # 21834) and nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
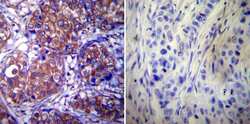
- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human breast carcinoma tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing Heat Shock Protein 90 (Product # MA3-011) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
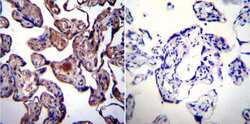
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human placenta tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing Heat Shock Protein 90 (Product # MA3-011) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
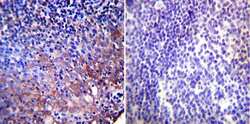
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human tonsil tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a mouse monoclonal antibody recognizing Heat Shock Protein 90 (Product # MA3-011) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
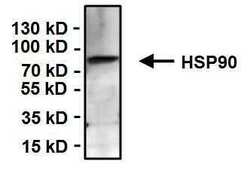
- Experimental details
- Immunoprecipitation of Heat Shock Protein 90 (Hsp90) was performed on K562 cells. Antigen-antibody complexes were formed by incubating 500 µg of whole cell lysate with 3 µg of a HSP90 monoclonal antibody (Product # MA3-011) overnight on a rocking platform at 4øC. The immune complexes were captured on 50 µL Protein A/G Agarose (Product # 20421), washed extensively, and eluted with Lane Marker Reducing Sample Buffer (Product # 39000). Samples were resolved on a 4-20% Tris-HCl polyacrylamide gel, transferred to a PVDF membrane, and blocked with 5% BSA/TBST for at least 1 hour. The membrane was probed with a Hsp90 monoclonal antibody (Product # MA3-011) at a dilution of 1:500 overnight rotating at 4øC, washed in TBST, and probed with an HRP-conjugated goat anti-mouse IgM secondary antibody (Product # 31440) at a dilution of 1:20,000 for at least 1 hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34075).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunoprecipitation
Immunoprecipitation