Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [2]
- Immunohistochemistry [1]
- Flow cytometry [2]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-14494 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CDX2 Monoclonal Antibody (EPR2764Y)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- MA5-14494 targets CDX2 in WB, IHC (P), ICC/IF and FACS applications and shows reactivity with Human and Mouse samples.
- Antibody clone number
- EPR2764Y
- Concentration
- Conc. Not Determined
Submitted references A Point Mutation R122C in RUNX3 Promotes the Expansion of Isthmus Stem Cells and Inhibits Their Differentiation in the Stomach.
DMRT1-mediated reprogramming drives development of cancer resembling human germ cell tumors with features of totipotency.
The relationship among PDX1, CDX2, and mucin profiles in gastric carcinomas; correlations with clinicopathologic parameters.
Douchi D, Yamamura A, Matsuo J, Lee JW, Nuttonmanit N, Melissa Lim YH, Suda K, Shimura M, Chen S, Pang S, Kohu K, Kaneko M, Kiyonari H, Kaneda A, Yoshida H, Taniuchi I, Osato M, Yang H, Unno M, Bok-Yan So J, Yeoh KG, Chuang LSH, Bae SC, Ito Y
Cellular and molecular gastroenterology and hepatology 2022;13(5):1317-1345
Cellular and molecular gastroenterology and hepatology 2022;13(5):1317-1345
DMRT1-mediated reprogramming drives development of cancer resembling human germ cell tumors with features of totipotency.
Taguchi J, Shibata H, Kabata M, Kato M, Fukuda K, Tanaka A, Ohta S, Ukai T, Mitsunaga K, Yamada Y, Nagaoka SI, Yamazawa S, Ohnishi K, Woltjen K, Ushiku T, Ozawa M, Saitou M, Shinkai Y, Yamamoto T, Yamada Y
Nature communications 2021 Aug 19;12(1):5041
Nature communications 2021 Aug 19;12(1):5041
The relationship among PDX1, CDX2, and mucin profiles in gastric carcinomas; correlations with clinicopathologic parameters.
Oz Puyan F, Can N, Ozyilmaz F, Usta U, Sut N, Tastekin E, Altaner S
Journal of cancer research and clinical oncology 2011 Dec;137(12):1749-62
Journal of cancer research and clinical oncology 2011 Dec;137(12):1749-62
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
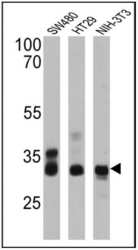
- Experimental details
- Western blot analysis of CDX2 was performed by loading 25 µg of SW480 (lane 1), HT29 (lane 2) and NIH-3T3 (lane 3) cell lysates onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a CDX2 monoclonal antibody (Product # MA5-14494) at a dilution of 1:200 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~33 kDa.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using CDX2 Monoclonal Antibody (EPR2764Y) (Product # MA5-14494) and a 40 kDa band corresponding to CDX2 was observed across all the cell lines tested. Whole cell extracts (30 µg lysate) of CaCO2 (Lane 1), SW480 (Lane 2), HCT116 (Lane 3), HeLa (Lane 4) and MCF7 (Lane 5) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0322BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
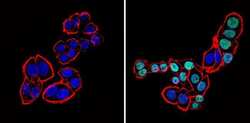
- Experimental details
- Immunofluorescent analysis of CDX2 (green) showing staining in the nucleus of SW480 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a CDX2 monoclonal antibody (Product # MA5-14494) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
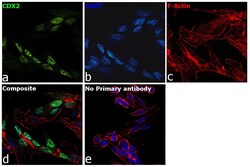
- Experimental details
- Immunofluorescence analysis of CDX2 was performed using SW480 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with CDX2 Monoclonal Antibody (EPR2764Y) (Product # MA5-14494) at 1:100 dilution in 0.1% BSA and incubated overnight at 4 degree and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the composite image showing nuclear localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
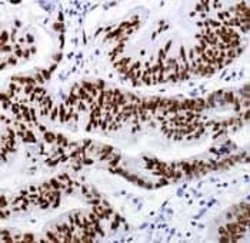
- Experimental details
- Formalin-fixed, paraffin-embedded colon carcinoma stained with rabbit monoclonal anti-CDX2 antibody using peroxidase conjugate and DAB chromogen. Note nuclear staining.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
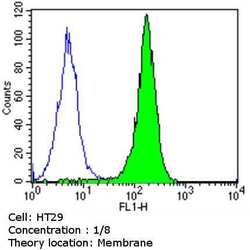
- Experimental details
- Flow cytometry analysis of CDX2 in HT29 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CDX2 monoclonal antibody (Product # MA5-14494) at a dilution of 1:10 for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
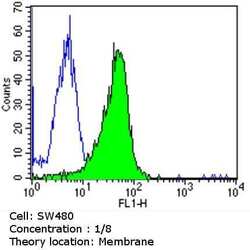
- Experimental details
- Flow cytometry analysis of CDX2 in SW480 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CDX2 monoclonal antibody (Product # MA5-14494) at a dilution of 1:10 for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated goat anti-mouse IgG (H+L) secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
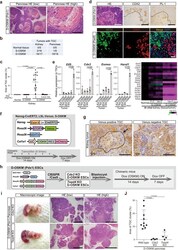
- Experimental details
- Fig. 3 Trophoblastic differentiation of D-OSKM tumor cells. a Representative histological images of D-OSKM pancreatic tumors containing trophoblast giant cell (TGC) clusters. Dotted lines indicate TGC clusters (>3 cells/high-power field [x400]). Scale bars: 1000 mum (left), 200 mum (right). b Incidence of tumors containing trophoblast giant cells in S-/D-OSKM chimeric mice. c Quantification of TGC cluster area in S-/D-OSKM kidney tumors (normal kidney, n = 5; S-OSKM kidney tumor, n = 16; D-OSKM kidney tumor following Dox treatment at various doses [2.0 mg/mL, n = 17; 0.4 mg/mL, n = 4; 0.25 mg/mL, n = 7; 0.13 mg/mL, n = 4]). Data are presented as means +- SD of biologically independent samples (Kruskal-Wallis test and Dunn's multiple-comparison test, two-sided). d Representative histological images and immunostaining for CDX2 and PL-1 of D-OSKM pancreatic tumors (upper panels). Dotted lines indicate TGC clusters. Scale bars: 200 mum. Immunofluorescence staining for OCT4 and CDX2 of D-OSKM kidney tumors (lower panels). Scale bars: 50 mum. e (Left) qRT-PCR analyses of trophoblast-related gene expression in S-/D-OSKM kidney tumors. Data are presented as means +- SD of biological triplicates. Relative expression levels to TSCs are shown (one-way ANOVA and Dunnett's multiple-comparison test, two-sided) (Right) Heatmap showing relative expression of trophoblast-related genes in S-/D-OSKM kidney tumors. Color range is shown using a log 2 scale. f Lineage-tracing system for Nanog -exp
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
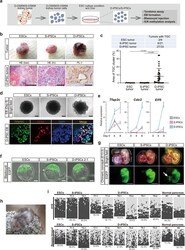
- Experimental details
- Fig. 4 Derivation of D-OSKM tumor-derived PSCs with the potential to differentiate toward the extraembryonic lineage. a Schematic illustration of iPSC derivation from S-/D-OSKM kidney tumor cells (S-/D-iPSCs). A total of nine S-/D-iPSC clones were established from three independent kidney tumors. b Representative macroscopic (upper) and histological (lower) images of ESC-, S-iPSC-, and D-iPSC-derived subcutaneous tumors in immunocompromised mice (upper panels). Immunostaining for PL-1 is shown in the lower panel. Scale bars: 500 mum (left), 200 mum (middle and right). c Incidence of tumors containing TGCs (upper) and quantification of TGC cluster area in ESC ( n = 9), S-iPSC ( n = 21), and D-iPSC tumors ( n = 33). Nine independent S-/D-iPSC clones were used. Data are presented as means +- SD (Kruskal-Wallis test and Dunn's multiple-comparison test, two-sided). d Representative images of ESC-, S-iPSC-, and D-iPSC-derived EBs (upper panels). Lower panels show immunofluorescence staining for TFAP2C and CDX2 of D-iPSC EB at differentiation Day 8. Scale bars: 200 mum (upper), 50 mum (lower). e qRT-PCR analyses for expression of trophoblast-related genes during EB formation. Data are presented as means +- SD of biological triplicates. The mean expression level at differentiation Day 0 ESCs was defined as 1. f Representative images of blastocysts injected with GFP-labeled ESCs, S-iPSCs, or D-iPSCs. Scale bars: 20 mum. g Representative images of E13.5 chimeric concepti. The chimeric
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
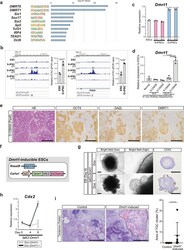
- Fig. 5 Elevated DMRT1 chromatin accessibility is associated with the extended differentiation potential of PSCs. a Motif enrichment analysis of D-iPSC-specific enriched peaks in ATAC-seq ( n = 270), performed using HOMER (Fisher's exact test, two-sided). b Representative specific peaks in D-iPSCs and ATAC-qPCR analysis. Genomic regions containing DMRT1/6-binding motifs , and qPCR products are shown. Data are presented as means +- SD of biological duplicates. The mean ATAC-qPCR value of ESCs was defined as 1 ( t -test, two-sided). c qRT-PCR analysis of Dmrt1 expression in ESCs, S-iPSCs, and D-iPSCs. Data are presented as means +- SD of technical triplicates. The mean expression level of ESCs was defined as 1 (one-way ANOVA, two-sided). d qRT-PCR analysis for expression of Dmrt1 in S-/D-OSKM kidney tumors. Data are presented as means +- SD of biological triplicates. Relative expression levels to ESCs are shown (one-way ANOVA and Dunnett's multiple-comparison test, two-sided). e Representative histological images and immunostaining for OCT4, DAZL, and DMRT1 of D-OSKM kidney tumors. Scale bars: 200 mum. f Schematic illustration of the genomic construct of Dmrt1 -inducible ESCs. g Representative images and immunostaining for CDX2 in Dmrt1 -inducible EBs on differentiation Day 8. Scale bars: 200 mum (left), 500 mum (right). h qRT-PCR analysis of Cdx2 expression during EB differentiation. Data are presented as means +- SD of biological triplicates. The mean expression level in ESCs
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
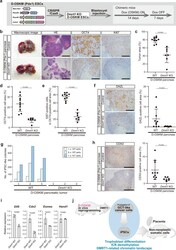
- Fig. 6 DMRT1 drives in vivo reprogramming and propagation of GCT-like tumor cells. a Schematic illustration of the experimental protocol for in vivo D-OSKM reprogramming of somatic cells deficient for Dmrt1 . b Representative macroscopic and histological images of pancreatic tumors in Dmrt1 -KO D-OSKM chimeric mice. Immunostaining for OCT4 and Ki67 in pancreatic tumors is shown in the right panels. Scale bars: 1000 mum (left), 200 mum (middle and right). c - e Quantification of tumor area (c), OCT4-positive cell area in the tumor (d), and Ki67-positive cell ratio in the OCT4-positive cell area (e) in the pancreas. D-OSKM mice that developed pancreatic tumors were evaluated in this study (control, n = 10; Dmrt1 KO18, n = 2; Dmrt1 KO21, n = 5). Data are presented as means +- SD of biologically independent samples (Mann-Whitney test, two-sided). f Immunostaining for DAZL and quantification of the DAZL-positive cell area in pancreatic tumors (control, n = 10; Dmrt1 KO18, n = 2; Dmrt1 KO21, n = 5). Data are presented as means +- SD of biologically independent samples (Mann-Whitney test, two-sided) scale bars: 200 mum. g Derivation efficiency of iPSC-like colonies from pancreatic tumors (control, n = 3; Dmrt1 KO21, n = 3). Colony counting was performed at Day 7. h Immunostaining for CDX2 and quantification of the CDX2-positive cell area in pancreatic tumors (control, n = 10; Dmrt1 KO18, n = 2; Dmrt1 KO21, n = 5). Data are presented as means +- SD of biologically independent samples
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
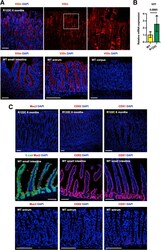
- Characterization of metaplastic glands by using intestinal markers. ( A ) IF staining for villin in the corpus of 6-month-old RUNX3 R122C/R122C mice, and in the small intestine, antrum, and corpus of WT mice. ( B ) qPCR for Vil1 expression level in the corpus tissues of 6-month-old WT and RUNX3 R122C/R122C mice ( n = 3 mice each). ( C ) IF staining for Muc2, CDX2, and CDX1 in the corpus of 6-month-old RUNX3 R122C/R122C mice, and in the small intestine and antrum of WT mice. Scale bars : 100 mum. Data are presented as the means +- SEM and were analyzed by the Student t test. Box indicates enlarged region. DAPI, 4',6-diamidino-2-phenylindole.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot