MA1-064
antibody from Invitrogen Antibodies
Targeting: AP2A1
ADTAA, CLAPA1
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [54]
- Comments [0]
- Validations
- Immunocytochemistry [8]
- Other assay [11]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-064 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- alpha Adaptin Monoclonal Antibody (AP6)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA1-064 detects assembly polypeptide 2 (AP2) from a wide variety of mammalian sources, including human, hamster, monkey, bovine, rat, and mouse. It recognizes the products of both alpha-adaptin genes, alpha A and alpha C as well as an alternatively spliced isoform of alpha adaptin found in neurons. MA1-064 has been successfully used in Western blot, immunofluorescence, immunocytochemistry, and immunoprecipitation procedures. By Western blot, this antibody detects an ~100 kDa doublet representing the two isoforms of alpha adaptin in rat brain (alpha C is the dominant form seen) Microinjection of cells with MA1-064 results in variable blockade of endocytosis. The MA1-064 immunogen is purified adaptors.
- Reactivity
- Human, Mouse, Rat, Bovine, Hamster
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- AP6
- Vial size
- 100 μL
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references Endocytosis in the axon initial segment maintains neuronal polarity.
Proteomics analysis identifies PEA-15 as an endosomal phosphoprotein that regulates α5β1 integrin endocytosis.
Quantitative proteomics of synaptosome S-nitrosylation in Alzheimer's disease.
Sequential screening nominates the Parkinson's disease associated kinase LRRK2 as a regulator of Clathrin-mediated endocytosis.
Loss of huntingtin function slows synaptic vesicle endocytosis in striatal neurons from the htt(Q140/Q140) mouse model of Huntington's disease.
A DNM2 Centronuclear Myopathy Mutation Reveals a Link between Recycling Endosome Scission and Autophagy.
Neuron/Glial Antigen 2-Type VI Collagen Interactions During Murine Temporomandibular Joint Osteoarthritis.
Coupling of microtubule motors with AP-3 generated organelles in axons by NEEP21 family member calcyon.
An Aberrant Phosphorylation of Amyloid Precursor Protein Tyrosine Regulates Its Trafficking and the Binding to the Clathrin Endocytic Complex in Neural Stem Cells of Alzheimer's Disease Patients.
Proteome profiling of s-nitrosylated synaptosomal proteins by isobaric mass tags.
Endocytic activity of HIV-1 Vpu: Phosphoserine-dependent interactions with clathrin adaptors.
αTAT1 downregulation induces mitotic catastrophe in HeLa and A549 cells.
Synaptotagmin-11 inhibits clathrin-mediated and bulk endocytosis.
Selective integrin endocytosis is driven by interactions between the integrin α-chain and AP2.
The clathrin-binding and J-domains of GAK support the uncoating and chaperoning of clathrin by Hsc70 in the brain.
Endophilin marks and controls a clathrin-independent endocytic pathway.
Endophilin, Lamellipodin, and Mena cooperate to regulate F-actin-dependent EGF-receptor endocytosis.
Dynamin triple knockout cells reveal off target effects of commonly used dynamin inhibitors.
Releasable SNAP-tag probes for studying endocytosis and recycling.
EHD2 regulates caveolar dynamics via ATP-driven targeting and oligomerization.
Disabled-2 protein facilitates assembly polypeptide-2-independent recruitment of cystic fibrosis transmembrane conductance regulator to endocytic vesicles in polarized human airway epithelial cells.
Mistargeting of SH3TC2 away from the recycling endosome causes Charcot-Marie-Tooth disease type 4C.
Overexpression of myosin VI in prostate cancer cells enhances PSA and VEGF secretion, but has no effect on endocytosis.
Regulation of endosomal membrane traffic by a Gadkin/AP-1/kinesin KIF5 complex.
Functional equivalence of the clathrin heavy chains CHC17 and CHC22 in endocytosis and mitosis.
Immunocell-array for molecular dissection of multiple signaling pathways in mammalian cells.
Internalization of large double-membrane intercellular vesicles by a clathrin-dependent endocytic process.
Defective CFTR apical endocytosis and enterocyte brush border in myosin VI-deficient mice.
Recruitment of clathrin onto endosomes by the Tom1-Tollip complex.
Rapid turnover of stereocilia membrane proteins: evidence from the trafficking and mobility of plasma membrane Ca(2+)-ATPase 2.
Death-receptor activation halts clathrin-dependent endocytosis.
Expression of the adaptor protein m-Numb in mouse male germ cells.
SPG3A protein atlastin-1 is enriched in growth cones and promotes axon elongation during neuronal development.
Role of the endocytic machinery in the sorting of lysosome-associated membrane proteins.
Exchange of clathrin, AP2 and epsin on clathrin-coated pits in permeabilized tissue culture cells.
The DnaJ-domain protein RME-8 functions in endosomal trafficking.
Highly cooperative control of endocytosis by clathrin.
In vitro formation of recycling vesicles from endosomes requires adaptor protein-1/clathrin and is regulated by rab4 and the connector rabaptin-5.
MUC1 membrane trafficking is modulated by multiple interactions.
SHIP2 is recruited to the cell membrane upon macrophage colony-stimulating factor (M-CSF) stimulation and regulates M-CSF-induced signaling.
RNAi-mediated Hip1R silencing results in stable association between the endocytic machinery and the actin assembly machinery.
The adaptor protein ARH escorts megalin to and through endosomes.
Imaging actin and dynamin recruitment during invagination of single clathrin-coated pits.
The beta-appendages of the four adaptor-protein (AP) complexes: structure and binding properties, and identification of sorting nexin 9 as an accessory protein to AP-2.
The actin-binding protein Hip1R associates with clathrin during early stages of endocytosis and promotes clathrin assembly in vitro.
The tripartite motif family identifies cell compartments.
Nucleocytoplasmic shuttling of endocytic proteins.
Tandem arrangement of the clathrin and AP-2 binding domains in amphiphysin 1 and disruption of clathrin coat function by amphiphysin fragments comprising these sites.
ADP-Ribosylation factor 1 (ARF1) regulates recruitment of the AP-3 adaptor complex to membranes.
Adaptor self-aggregation, adaptor-receptor recognition and binding of alpha-adaptin subunits to the plasma membrane contribute to recruitment of adaptor (AP2) components of clathrin-coated pits.
Clathrin assembly protein AP-2 induces aggregation of membrane vesicles: a possible role for AP-2 in endosome formation.
100-kD proteins of Golgi- and trans-Golgi network-associated coated vesicles have related but distinct membrane binding properties.
100-kD proteins of Golgi- and trans-Golgi network-associated coated vesicles have related but distinct membrane binding properties.
100-kDa polypeptides in peripheral clathrin-coated vesicles are required for receptor-mediated endocytosis.
Eichel K, Uenaka T, Belapurkar V, Lu R, Cheng S, Pak JS, Taylor CA, Südhof TC, Malenka R, Wernig M, Özkan E, Perrais D, Shen K
Nature 2022 Sep;609(7925):128-135
Nature 2022 Sep;609(7925):128-135
Proteomics analysis identifies PEA-15 as an endosomal phosphoprotein that regulates α5β1 integrin endocytosis.
Caliva MJ, Yang WS, Young-Robbins S, Zhou M, Yoon H, Matter ML, Grimes ML, Conrads T, Ramos JW
Scientific reports 2021 Oct 6;11(1):19830
Scientific reports 2021 Oct 6;11(1):19830
Quantitative proteomics of synaptosome S-nitrosylation in Alzheimer's disease.
Wijasa TS, Sylvester M, Brocke-Ahmadinejad N, Schwartz S, Santarelli F, Gieselmann V, Klockgether T, Brosseron F, Heneka MT
Journal of neurochemistry 2020 Mar;152(6):710-726
Journal of neurochemistry 2020 Mar;152(6):710-726
Sequential screening nominates the Parkinson's disease associated kinase LRRK2 as a regulator of Clathrin-mediated endocytosis.
Heaton GR, Landeck N, Mamais A, Nalls MA, Nixon-Abell J, Kumaran R, Beilina A, Pellegrini L, Li Y, International Parkinson Disease Genomics Consortium (IPDGC), Harvey K, Cookson MR
Neurobiology of disease 2020 Jul;141:104948
Neurobiology of disease 2020 Jul;141:104948
Loss of huntingtin function slows synaptic vesicle endocytosis in striatal neurons from the htt(Q140/Q140) mouse model of Huntington's disease.
McAdam RL, Morton A, Gordon SL, Alterman JF, Khvorova A, Cousin MA, Smillie KJ
Neurobiology of disease 2020 Feb;134:104637
Neurobiology of disease 2020 Feb;134:104637
A DNM2 Centronuclear Myopathy Mutation Reveals a Link between Recycling Endosome Scission and Autophagy.
Puri C, Manni MM, Vicinanza M, Hilcenko C, Zhu Y, Runwal G, Stamatakou E, Menzies FM, Mamchaoui K, Bitoun M, Rubinsztein DC
Developmental cell 2020 Apr 20;53(2):154-168.e6
Developmental cell 2020 Apr 20;53(2):154-168.e6
Neuron/Glial Antigen 2-Type VI Collagen Interactions During Murine Temporomandibular Joint Osteoarthritis.
Yotsuya M, Bertagna AE, Hasan N, Bicknell S, Sato T, Reed DA
Scientific reports 2019 Jan 11;9(1):56
Scientific reports 2019 Jan 11;9(1):56
Coupling of microtubule motors with AP-3 generated organelles in axons by NEEP21 family member calcyon.
Shi L, Hines T, Bergson C, Smith D
Molecular biology of the cell 2018 Aug 15;29(17):2055-2068
Molecular biology of the cell 2018 Aug 15;29(17):2055-2068
An Aberrant Phosphorylation of Amyloid Precursor Protein Tyrosine Regulates Its Trafficking and the Binding to the Clathrin Endocytic Complex in Neural Stem Cells of Alzheimer's Disease Patients.
Poulsen ET, Iannuzzi F, Rasmussen HF, Maier TJ, Enghild JJ, Jørgensen AL, Matrone C
Frontiers in molecular neuroscience 2017;10:59
Frontiers in molecular neuroscience 2017;10:59
Proteome profiling of s-nitrosylated synaptosomal proteins by isobaric mass tags.
Wijasa TS, Sylvester M, Brocke-Ahmadinejad N, Kummer MP, Brosseron F, Gieselmann V, Heneka MT
Journal of neuroscience methods 2017 Nov 1;291:95-100
Journal of neuroscience methods 2017 Nov 1;291:95-100
Endocytic activity of HIV-1 Vpu: Phosphoserine-dependent interactions with clathrin adaptors.
Stoneham CA, Singh R, Jia X, Xiong Y, Guatelli J
Traffic (Copenhagen, Denmark) 2017 Aug;18(8):545-561
Traffic (Copenhagen, Denmark) 2017 Aug;18(8):545-561
αTAT1 downregulation induces mitotic catastrophe in HeLa and A549 cells.
Chien JY, Tsen SD, Chien CC, Liu HW, Tung CY, Lin CH
Cell death discovery 2016;2:16006
Cell death discovery 2016;2:16006
Synaptotagmin-11 inhibits clathrin-mediated and bulk endocytosis.
Wang C, Wang Y, Hu M, Chai Z, Wu Q, Huang R, Han W, Zhang CX, Zhou Z
EMBO reports 2016 Jan;17(1):47-63
EMBO reports 2016 Jan;17(1):47-63
Selective integrin endocytosis is driven by interactions between the integrin α-chain and AP2.
De Franceschi N, Arjonen A, Elkhatib N, Denessiouk K, Wrobel AG, Wilson TA, Pouwels J, Montagnac G, Owen DJ, Ivaska J
Nature structural & molecular biology 2016 Feb;23(2):172-9
Nature structural & molecular biology 2016 Feb;23(2):172-9
The clathrin-binding and J-domains of GAK support the uncoating and chaperoning of clathrin by Hsc70 in the brain.
Park BC, Yim YI, Zhao X, Olszewski MB, Eisenberg E, Greene LE
Journal of cell science 2015 Oct 15;128(20):3811-21
Journal of cell science 2015 Oct 15;128(20):3811-21
Endophilin marks and controls a clathrin-independent endocytic pathway.
Boucrot E, Ferreira AP, Almeida-Souza L, Debard S, Vallis Y, Howard G, Bertot L, Sauvonnet N, McMahon HT
Nature 2015 Jan 22;517(7535):460-5
Nature 2015 Jan 22;517(7535):460-5
Endophilin, Lamellipodin, and Mena cooperate to regulate F-actin-dependent EGF-receptor endocytosis.
Vehlow A, Soong D, Vizcay-Barrena G, Bodo C, Law AL, Perera U, Krause M
The EMBO journal 2013 Oct 16;32(20):2722-34
The EMBO journal 2013 Oct 16;32(20):2722-34
Dynamin triple knockout cells reveal off target effects of commonly used dynamin inhibitors.
Park RJ, Shen H, Liu L, Liu X, Ferguson SM, De Camilli P
Journal of cell science 2013 Nov 15;126(Pt 22):5305-12
Journal of cell science 2013 Nov 15;126(Pt 22):5305-12
Releasable SNAP-tag probes for studying endocytosis and recycling.
Cole NB, Donaldson JG
ACS chemical biology 2012 Mar 16;7(3):464-9
ACS chemical biology 2012 Mar 16;7(3):464-9
EHD2 regulates caveolar dynamics via ATP-driven targeting and oligomerization.
Morén B, Shah C, Howes MT, Schieber NL, McMahon HT, Parton RG, Daumke O, Lundmark R
Molecular biology of the cell 2012 Apr;23(7):1316-29
Molecular biology of the cell 2012 Apr;23(7):1316-29
Disabled-2 protein facilitates assembly polypeptide-2-independent recruitment of cystic fibrosis transmembrane conductance regulator to endocytic vesicles in polarized human airway epithelial cells.
Cihil KM, Ellinger P, Fellows A, Stolz DB, Madden DR, Swiatecka-Urban A
The Journal of biological chemistry 2012 Apr 27;287(18):15087-99
The Journal of biological chemistry 2012 Apr 27;287(18):15087-99
Mistargeting of SH3TC2 away from the recycling endosome causes Charcot-Marie-Tooth disease type 4C.
Roberts RC, Peden AA, Buss F, Bright NA, Latouche M, Reilly MM, Kendrick-Jones J, Luzio JP
Human molecular genetics 2010 Mar 15;19(6):1009-18
Human molecular genetics 2010 Mar 15;19(6):1009-18
Overexpression of myosin VI in prostate cancer cells enhances PSA and VEGF secretion, but has no effect on endocytosis.
Puri C, Chibalina MV, Arden SD, Kruppa AJ, Kendrick-Jones J, Buss F
Oncogene 2010 Jan 14;29(2):188-200
Oncogene 2010 Jan 14;29(2):188-200
Regulation of endosomal membrane traffic by a Gadkin/AP-1/kinesin KIF5 complex.
Schmidt MR, Maritzen T, Kukhtina V, Higman VA, Doglio L, Barak NN, Strauss H, Oschkinat H, Dotti CG, Haucke V
Proceedings of the National Academy of Sciences of the United States of America 2009 Sep 8;106(36):15344-9
Proceedings of the National Academy of Sciences of the United States of America 2009 Sep 8;106(36):15344-9
Functional equivalence of the clathrin heavy chains CHC17 and CHC22 in endocytosis and mitosis.
Hood FE, Royle SJ
Journal of cell science 2009 Jul 1;122(Pt 13):2185-90
Journal of cell science 2009 Jul 1;122(Pt 13):2185-90
Immunocell-array for molecular dissection of multiple signaling pathways in mammalian cells.
Zanardi A, Giorgetti L, Botrugno OA, Minucci S, Milani P, Pelicci PG, Carbone R
Molecular & cellular proteomics : MCP 2007 May;6(5):939-47
Molecular & cellular proteomics : MCP 2007 May;6(5):939-47
Internalization of large double-membrane intercellular vesicles by a clathrin-dependent endocytic process.
Piehl M, Lehmann C, Gumpert A, Denizot JP, Segretain D, Falk MM
Molecular biology of the cell 2007 Feb;18(2):337-47
Molecular biology of the cell 2007 Feb;18(2):337-47
Defective CFTR apical endocytosis and enterocyte brush border in myosin VI-deficient mice.
Ameen N, Apodaca G
Traffic (Copenhagen, Denmark) 2007 Aug;8(8):998-1006
Traffic (Copenhagen, Denmark) 2007 Aug;8(8):998-1006
Recruitment of clathrin onto endosomes by the Tom1-Tollip complex.
Katoh Y, Imakagura H, Futatsumori M, Nakayama K
Biochemical and biophysical research communications 2006 Mar 3;341(1):143-9
Biochemical and biophysical research communications 2006 Mar 3;341(1):143-9
Rapid turnover of stereocilia membrane proteins: evidence from the trafficking and mobility of plasma membrane Ca(2+)-ATPase 2.
Grati M, Schneider ME, Lipkow K, Strehler EE, Wenthold RJ, Kachar B
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Jun 7;26(23):6386-95
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Jun 7;26(23):6386-95
Death-receptor activation halts clathrin-dependent endocytosis.
Austin CD, Lawrence DA, Peden AA, Varfolomeev EE, Totpal K, De Mazière AM, Klumperman J, Arnott D, Pham V, Scheller RH, Ashkenazi A
Proceedings of the National Academy of Sciences of the United States of America 2006 Jul 5;103(27):10283-10288
Proceedings of the National Academy of Sciences of the United States of America 2006 Jul 5;103(27):10283-10288
Expression of the adaptor protein m-Numb in mouse male germ cells.
Corallini S, Fera S, Grisanti L, Falciatori I, Muciaccia B, Stefanini M, Vicini E
Reproduction (Cambridge, England) 2006 Dec;132(6):887-97
Reproduction (Cambridge, England) 2006 Dec;132(6):887-97
SPG3A protein atlastin-1 is enriched in growth cones and promotes axon elongation during neuronal development.
Zhu PP, Soderblom C, Tao-Cheng JH, Stadler J, Blackstone C
Human molecular genetics 2006 Apr 15;15(8):1343-53
Human molecular genetics 2006 Apr 15;15(8):1343-53
Role of the endocytic machinery in the sorting of lysosome-associated membrane proteins.
Janvier K, Bonifacino JS
Molecular biology of the cell 2005 Sep;16(9):4231-42
Molecular biology of the cell 2005 Sep;16(9):4231-42
Exchange of clathrin, AP2 and epsin on clathrin-coated pits in permeabilized tissue culture cells.
Yim YI, Scarselletta S, Zang F, Wu X, Lee DW, Kang YS, Eisenberg E, Greene LE
Journal of cell science 2005 Jun 1;118(Pt 11):2405-13
Journal of cell science 2005 Jun 1;118(Pt 11):2405-13
The DnaJ-domain protein RME-8 functions in endosomal trafficking.
Girard M, Poupon V, Blondeau F, McPherson PS
The Journal of biological chemistry 2005 Dec 2;280(48):40135-43
The Journal of biological chemistry 2005 Dec 2;280(48):40135-43
Highly cooperative control of endocytosis by clathrin.
Moskowitz HS, Yokoyama CT, Ryan TA
Molecular biology of the cell 2005 Apr;16(4):1769-76
Molecular biology of the cell 2005 Apr;16(4):1769-76
In vitro formation of recycling vesicles from endosomes requires adaptor protein-1/clathrin and is regulated by rab4 and the connector rabaptin-5.
Pagano A, Crottet P, Prescianotto-Baschong C, Spiess M
Molecular biology of the cell 2004 Nov;15(11):4990-5000
Molecular biology of the cell 2004 Nov;15(11):4990-5000
MUC1 membrane trafficking is modulated by multiple interactions.
Kinlough CL, Poland PA, Bruns JB, Harkleroad KL, Hughey RP
The Journal of biological chemistry 2004 Dec 17;279(51):53071-7
The Journal of biological chemistry 2004 Dec 17;279(51):53071-7
SHIP2 is recruited to the cell membrane upon macrophage colony-stimulating factor (M-CSF) stimulation and regulates M-CSF-induced signaling.
Wang Y, Keogh RJ, Hunter MG, Mitchell CA, Frey RS, Javaid K, Malik AB, Schurmans S, Tridandapani S, Marsh CB
Journal of immunology (Baltimore, Md. : 1950) 2004 Dec 1;173(11):6820-30
Journal of immunology (Baltimore, Md. : 1950) 2004 Dec 1;173(11):6820-30
RNAi-mediated Hip1R silencing results in stable association between the endocytic machinery and the actin assembly machinery.
Engqvist-Goldstein AE, Zhang CX, Carreno S, Barroso C, Heuser JE, Drubin DG
Molecular biology of the cell 2004 Apr;15(4):1666-79
Molecular biology of the cell 2004 Apr;15(4):1666-79
The adaptor protein ARH escorts megalin to and through endosomes.
Nagai M, Meerloo T, Takeda T, Farquhar MG
Molecular biology of the cell 2003 Dec;14(12):4984-96
Molecular biology of the cell 2003 Dec;14(12):4984-96
Imaging actin and dynamin recruitment during invagination of single clathrin-coated pits.
Merrifield CJ, Feldman ME, Wan L, Almers W
Nature cell biology 2002 Sep;4(9):691-8
Nature cell biology 2002 Sep;4(9):691-8
The beta-appendages of the four adaptor-protein (AP) complexes: structure and binding properties, and identification of sorting nexin 9 as an accessory protein to AP-2.
Lundmark R, Carlsson SR
The Biochemical journal 2002 Mar 15;362(Pt 3):597-607
The Biochemical journal 2002 Mar 15;362(Pt 3):597-607
The actin-binding protein Hip1R associates with clathrin during early stages of endocytosis and promotes clathrin assembly in vitro.
Engqvist-Goldstein AE, Warren RA, Kessels MM, Keen JH, Heuser J, Drubin DG
The Journal of cell biology 2001 Sep 17;154(6):1209-23
The Journal of cell biology 2001 Sep 17;154(6):1209-23
The tripartite motif family identifies cell compartments.
Reymond A, Meroni G, Fantozzi A, Merla G, Cairo S, Luzi L, Riganelli D, Zanaria E, Messali S, Cainarca S, Guffanti A, Minucci S, Pelicci PG, Ballabio A
The EMBO journal 2001 May 1;20(9):2140-51
The EMBO journal 2001 May 1;20(9):2140-51
Nucleocytoplasmic shuttling of endocytic proteins.
Vecchi M, Polo S, Poupon V, van de Loo JW, Benmerah A, Di Fiore PP
The Journal of cell biology 2001 Jun 25;153(7):1511-7
The Journal of cell biology 2001 Jun 25;153(7):1511-7
Tandem arrangement of the clathrin and AP-2 binding domains in amphiphysin 1 and disruption of clathrin coat function by amphiphysin fragments comprising these sites.
Slepnev VI, Ochoa GC, Butler MH, De Camilli P
The Journal of biological chemistry 2000 Jun 9;275(23):17583-9
The Journal of biological chemistry 2000 Jun 9;275(23):17583-9
ADP-Ribosylation factor 1 (ARF1) regulates recruitment of the AP-3 adaptor complex to membranes.
Ooi CE, Dell'Angelica EC, Bonifacino JS
The Journal of cell biology 1998 Jul 27;142(2):391-402
The Journal of cell biology 1998 Jul 27;142(2):391-402
Adaptor self-aggregation, adaptor-receptor recognition and binding of alpha-adaptin subunits to the plasma membrane contribute to recruitment of adaptor (AP2) components of clathrin-coated pits.
Chang MP, Mallet WG, Mostov KE, Brodsky FM
The EMBO journal 1993 May;12(5):2169-80
The EMBO journal 1993 May;12(5):2169-80
Clathrin assembly protein AP-2 induces aggregation of membrane vesicles: a possible role for AP-2 in endosome formation.
Beck KA, Chang M, Brodsky FM, Keen JH
The Journal of cell biology 1992 Nov;119(4):787-96
The Journal of cell biology 1992 Nov;119(4):787-96
100-kD proteins of Golgi- and trans-Golgi network-associated coated vesicles have related but distinct membrane binding properties.
Wong DH, Brodsky FM
The Journal of cell biology 1992 Jun;117(6):1171-9
The Journal of cell biology 1992 Jun;117(6):1171-9
100-kD proteins of Golgi- and trans-Golgi network-associated coated vesicles have related but distinct membrane binding properties.
Wong DH, Brodsky FM
The Journal of cell biology 1992 Jun;117(6):1171-9
The Journal of cell biology 1992 Jun;117(6):1171-9
100-kDa polypeptides in peripheral clathrin-coated vesicles are required for receptor-mediated endocytosis.
Chin DJ, Straubinger RM, Acton S, Näthke I, Brodsky FM
Proceedings of the National Academy of Sciences of the United States of America 1989 Dec;86(23):9289-93
Proceedings of the National Academy of Sciences of the United States of America 1989 Dec;86(23):9289-93
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
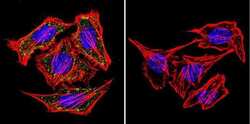
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in Hela Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
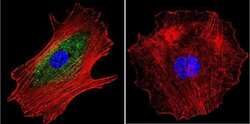
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in MCF-7 Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
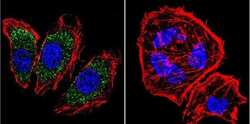
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in U251 Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
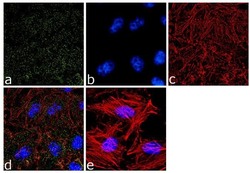
- Immunofluorescence analysis of alpha Adaptin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with alpha Adaptin (AP6) Mouse Monoclonal Antibody (Product # MA1-064) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing punctuated cytoplasmic and nuclear localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
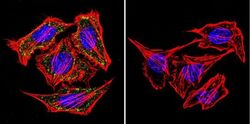
- Experimental details
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in Hela Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
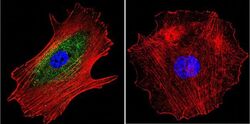
- Experimental details
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in MCF-7 Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
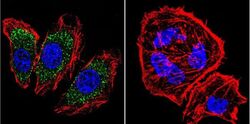
- Experimental details
- Immunofluorescent analysis of alpha Adaptin using alpha Adaptin Monoclonal Antibody (AP6) (Product # MA1-064) shows staining in U251 Cells. alpha Adaptin (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with an antibody recognizing alpha Adaptin (Product # MA1-064) at a dilution of 1:20 over night at 4 °C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552 for GAR, Product # 35503 for GAM). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
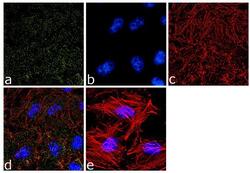
- Experimental details
- Immunofluorescence analysis of alpha Adaptin was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with alpha Adaptin (AP6) Mouse Monoclonal Antibody (Product # MA1-064) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing punctuated cytoplasmic and nuclear localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
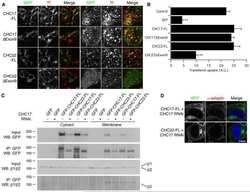
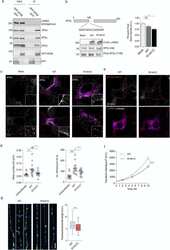
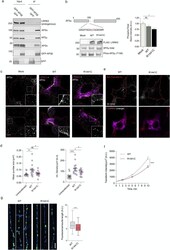
- Fig. 4 Cellular effects of LRRK2 on AP2. (a) We expressed and immunoprecipitated GFP or GFP-tagged AP2beta subunit followed by immunoblot for endogenous members of the AP2 complex and for endogenous LRRK2. Markers on the left of all blots are in kilodaltons (b) R1441C LRRK2 lowers AP2mu phosphorylation. Mock transfected, or HEK293 cells overexpressing WT or R1441C LRRK2 were treated with pcalyculinA and probed for LRRK2, total and T156 phosphorylated AP2mu as indicated. Markers on the left of all blots are in kilodaltons. A significant decrease of phospho-AP2mu/total AP2 was observed in cells expressing R1441C LRRK2 ( n = 9; AdjP, 0.0127, *P < .05; One-way ANOVA with Dunnett's multiple comparisons test). (c) Representative images of HEK293 cells stained for endogenous AP2alpha following either mock, LRRK2 WT or R1441C transfection. Scale bar: 5 muM. (d) Integrated density/area and mean vesicle size of AP2alpha was determined in areas positive for LRRK2 expression (ns, not significant; *, P < .05; **, P < .01; One-way ANOVA with Dunnett's multiple comparisons test) (e) Representative images of HEK293 cells expressing either WT or R1441C LRRK2 and assayed for transferrin uptake at T10. (f) Time course quantification of internalised transferrin at 2,5 and 10 min. Expression of the R1441C mutant resulted in a significant decrease in internalised transferrin relative to WT (****, P < .0001; Unpaired two-tailed t -test with Welch's correction for unequal variance). (g) Representati
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
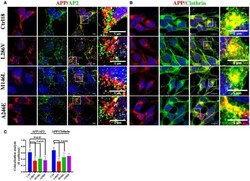
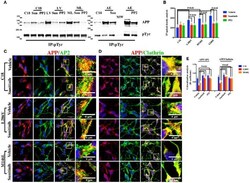
- Figure 2 The extent of Clathrin/APP and AP2/APP colocalization is reduced in NSCs from patients with AD . Confocal microscopy analysis of double staining using rabbit anti-APP and mouse anti-AP2 (A) and mouse anti-Clathrin (B) in control (C18) and AD neurons with PS1 mutations (L286V; M146L; A246E). Colocalization analysis is reported in (C) . The (R) coefficient (Pearson's coefficient) was used for the quantitative and comparative analyses. The extent of colocalization was calculated in at least five separate fields per slide in 10 different slides for each NSCs. The data are expressed as mean +- SEM. Scale bars are 5 mum for APP/AP2 and 6 mum for APP/Clathrin colocalization. Scale bars in high-resolution pictures are 5 mum. Statistically significant differences were calculated by one-way ANOVA and Tukey's post hoc test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
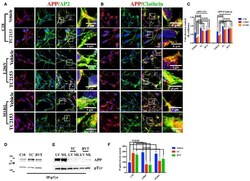
- Figure 3 Tyr kinase inhibitors restore APP colocalization with Clathrin and AP2 in NSCs from patients with AD . (A) IP analysis of control and PS1 neurons that were exposed, or not exposed, to Sunitinib and PP2. Control C18 and AD samples (LV, ML, AE) were immunoprecipitated with anti-pTyr agarose conjugated antibody (4G10) and analyzed with rabbit anti-APP (clone Y188). The image is representative of four independent experiments. Quantification is reported in (B) . Data were normalized with pTyr pulled down levels (input) and expressed as % of C18. Statistically significant differences were calculated by one-way ANOVA for repeated measures followed by Tukey's post hoc test for multiple comparisons. Confocal microscopy analysis of double staining using rabbit anti-APP and mouse anti-AP2 (C) and mouse anti-Clathrin (D) in controls and in neurons carrying L286V or M146L mutation on PS1 gene following exposure to the Tyr kinase inhibitor Sunitinib. The panels are representative of four different experiments performed in triplicate. (E) reports quantitative analysis of APP colocalization to AP2 and Clathrin after 12 h of exposure to Sunitinib. (E) also reports colocalization analysis after PP2 exposure. The (R) coefficient (Pearson's coefficient) was used for the quantitative and comparative analyses. The extent of colocalization was calculated in five separate fields per slide in five different slides for each experimental point. The data are expressed as mean +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
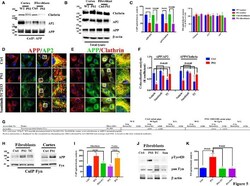
- Figure 4 Tyr phosphatase inhibitors reduce APP colocalization with Clathrin and AP2 and increase phosphorylation of APP Tyr in control neurons . Confocal microscopy analysis of double staining with rabbit anti-APP and mouse anti-AP2 (A) or mouse anti-Clathrin (B) in C18, L286V, and M146L neurons before and after Tyr phosphatase inhibitor (TC2153) exposure. The panels are representative of five different experiments performed in duplicate. (C) Colocalization analysis of APP to AP2 and Clathrin following incubation with TC2153 and BVT948 (BVT) inhibitors in C18 neurons and in AD neurons. The (R) coefficient (Pearson's coefficient) was used for the quantitative and comparative analyses. The extent of colocalization was calculated in five separate fields per slide in four different slides for each experimental point. The data are expressed as mean +- SEM. Scale bars 6 mum and 4 mum. Statistically significant differences were calculated by one-way ANOVA and Tukey's post hoc test. (D,E) . IP analysis of C18 (D) and AD neurons (E) before and after exposure to TC2153 and BVT948 (TC, BVT). Samples were immunoprecipitated with anti-pTyr agarose conjugated antibody (4G10) and analyzed with rabbit anti-APP (clone Y188). Densitometric analysis is reported in (F) . Data were normalized with IgG levels and expressed as % of C18. Statistically significant differences were calculated using Student's t -test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
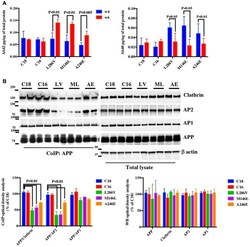
- Figure 5 APP/AP2 and APP/Clathrin binding analysis in cortical tissues and fibroblasts of Gottingen minipigs with a PS1 M146I mutation in the PS1 gene . (A) CoIP analysis of cortical tissues and fibroblasts from control (Ctrl) and PS1 M146I minipigs. Samples were immunoprecipitated with rabbit anti-APP and analyzed with mouse anti-Clathrin (Clath) and mouse anti-AP2. (B) WB analysis of total lysate from controls (WT and Ctrl) and PS1 M146I minipigs. Densitometric analysis is reported in (C) . Data were normalized to the corresponding beta-actin values and expressed as % of Ctrl. Data from CoIP samples were normalized to the corresponding APP input amount and expressed as % of Ctrl. Statistically significant differences were calculated using Student's t -test. (D,E) Confocal microscopy analysis of double staining with rabbit anti-APP and mouse anti-AP2 (D) and mouse anti-Clathrin (E) in fibroblasts from Ctrl and PS1M146I minipigs in the presence of TC2153 or Sunitinib. The panels are representative of five different experiments performed in duplicate in Ctrl and PS1 fibroblasts from two independent control (Ctrl 1-2) and three PS1 (PS1a-c) minipigs. Colocalization analysis is reported in (F) . The (R) coefficient (Pearson's coefficient) was used for the quantitative and comparative analyses. The extent of colocalization was calculated in six separate fields per slide in four different slides for each cell line. Scale bars 1 muM and 5 muM in high magnification. Statis
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 APP binding to Clathrin and AP2 is compromised in AD neurons. (A) ELISA analysis for secreted Abeta42 (Ab42) and Abeta40 (Ab40) levels from controls (C18 and C16) and AD neurons carrying mutations in the PS1 gene (L286V, M146L, and A246E) after 4 and 6 weeks in culture. Abeta levels were assessed from media after 24 h of plating. Data are expressed as pg/mug (pg/ug) of total protein. Each data point is the mean +- SEM of triplicate determinations of four independent experiments. (B) Co-Immunoprecipitation (CoIP) analysis from controls (C18, C16) and AD neurons (LV, ML, AE). Samples were immunoprecipitated with rabbit anti-APP (clone Y188) and analyzed with mouse anti-Clathrin (clone X22), mouse anti-AP2 (clone AP6), and rabbit anti-AP1 (left panel). The right panel shows total levels of APP, Clathrin, AP2, and AP1 expression in the same samples. Densitometric analysis is reported below. Data from total lysate samples were normalized to the corresponding beta-actin values and expressed as % of C18. Data from IP samples were normalized to the corresponding APP input band and expressed as % of C18. The data are representative of five independent experiments. Statistically significant differences were calculated by one-way ANOVA and Tukey's post hoc test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
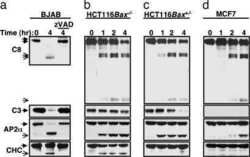
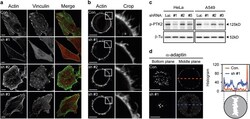
- Figure 2 alpha TAT1 downregulation decreased F-actin and altered alpha -adaptin distribution. ( a ) Confocal images of A549 cells visualized with Alexa Fluor 488 phalloidin (green) and immunostained with anti-vinculin antibody (red). ( b ) Confocal images of metaphase HeLa cells visualized with Alexa Fluor 488 phalloidin. Single optical section of the middle plane is shown. ( c ) pTyr397-PTK2 level determined by western blotting. The same protein extract was used in Figures 1a and 2c , therefore the same blotting of beta -Tu is presented in both figures. ( d ) Confocal images of metaphase HeLa cells immunostained with anti- alpha -adaptin antibody. Single optical section of the bottom or middle planes are shown. The signal intensity across the dashed lines is plotted in the right panel and the cartoon below indicates the relative position of the metaphase cell. Scale bar, 10 mu m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 4 Cellular effects of LRRK2 on AP2. (a) We expressed and immunoprecipitated GFP or GFP-tagged AP2beta subunit followed by immunoblot for endogenous members of the AP2 complex and for endogenous LRRK2. Markers on the left of all blots are in kilodaltons (b) R1441C LRRK2 lowers AP2mu phosphorylation. Mock transfected, or HEK293 cells overexpressing WT or R1441C LRRK2 were treated with pcalyculinA and probed for LRRK2, total and T156 phosphorylated AP2mu as indicated. Markers on the left of all blots are in kilodaltons. A significant decrease of phospho-AP2mu/total AP2 was observed in cells expressing R1441C LRRK2 ( n = 9; AdjP, 0.0127, *P < .05; One-way ANOVA with Dunnett's multiple comparisons test). (c) Representative images of HEK293 cells stained for endogenous AP2alpha following either mock, LRRK2 WT or R1441C transfection. Scale bar: 5 muM. (d) Integrated density/area and mean vesicle size of AP2alpha was determined in areas positive for LRRK2 expression (ns, not significant; *, P < .05; **, P < .01; One-way ANOVA with Dunnett's multiple comparisons test) (e) Representative images of HEK293 cells expressing either WT or R1441C LRRK2 and assayed for transferrin uptake at T10. (f) Time course quantification of internalised transferrin at 2,5 and 10 min. Expression of the R1441C mutant resulted in a significant decrease in internalised transferrin relative to WT (****, P < .0001; Unpaired two-tailed t -test with Welch's correction for unequal variance). (g) Representati
 Explore
Explore Validate
Validate Learn
Learn