Antibody data
- Antibody Data
- Antigen structure
- References [9]
- Comments [0]
- Validations
- Flow cytometry [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MHCD0828TR - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-CD8 Monoclonal Antibody (3B5), Pacific Blue
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- For fluorochrome conjugated antibodies only, light exposure should be avoided. Use dim light during handling, incubation with cells, and prior to analysis. It is recommended that cells be analyzed within 18 hours of staining. If the reagent is being diluted, it is recommended that only the quantity to be used within one week be diluted. Each lot is tested by flow cytometry using human peripheral blood leukocytes (PBL). Based on this testing it is recommended that 5 µL of antibody be used per 1 x 10^6 cells in a 100 µL staining volume.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3B5
- Vial size
- 25 tests
- Concentration
- 0.25 mg/mL
- Storage
- 4° C
Submitted references Poly(I:C) primes primary human glioblastoma cells for an immune response invigorated by PD-L1 blockade.
The tumor-associated antigen RHAMM (HMMR/CD168) is expressed by monocyte-derived dendritic cells and presented to T cells.
Concomitant evaluation of PMA+ionomycin-induced kinase phosphorylation and cytokine production in T cell subsets by flow cytometry.
Interleukin-7 facilitates HIV-1 transmission to cervico-vaginal tissue ex vivo.
Establishment of a neonatal rhesus macaque model to study Mycobacterium tuberculosis infection.
A novel method for autophagy detection in primary cells: impaired levels of macroautophagy in immunosenescent T cells.
A CD8α(-) subpopulation of macaque circulatory natural killer cells can mediate both antibody-dependent and antibody-independent cytotoxic activities.
HIV-1 infection abrogates CD8+ T cell mitogen-activated protein kinase signaling responses.
Modulation of cell signaling networks after CTLA4 blockade in patients with metastatic melanoma.
De Waele J, Marcq E, Van Audenaerde JR, Van Loenhout J, Deben C, Zwaenepoel K, Van de Kelft E, Van der Planken D, Menovsky T, Van den Bergh JM, Willemen Y, Pauwels P, Berneman ZN, Lardon F, Peeters M, Wouters A, Smits EL
Oncoimmunology 2018;7(3):e1407899
Oncoimmunology 2018;7(3):e1407899
The tumor-associated antigen RHAMM (HMMR/CD168) is expressed by monocyte-derived dendritic cells and presented to T cells.
Willemen Y, Van den Bergh JM, Bonte SM, Anguille S, Heirman C, Stein BM, Goossens H, Kerre T, Thielemans K, Peeters M, Van Tendeloo VF, Smits EL, Berneman ZN
Oncotarget 2016 Nov 8;7(45):73960-73970
Oncotarget 2016 Nov 8;7(45):73960-73970
Concomitant evaluation of PMA+ionomycin-induced kinase phosphorylation and cytokine production in T cell subsets by flow cytometry.
Crawford TQ, Jalbert E, Ndhlovu LC, Barbour JD
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2014 Mar;85(3):268-76
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2014 Mar;85(3):268-76
Interleukin-7 facilitates HIV-1 transmission to cervico-vaginal tissue ex vivo.
Introini A, Vanpouille C, Lisco A, Grivel JC, Margolis L
PLoS pathogens 2013 Feb;9(2):e1003148
PLoS pathogens 2013 Feb;9(2):e1003148
Establishment of a neonatal rhesus macaque model to study Mycobacterium tuberculosis infection.
Cepeda M, Salas M, Folwarczny J, Leandro AC, Hodara VL, de la Garza MA, Dick EJ Jr, Owston M, Armitige LY, Gauduin MC
Tuberculosis (Edinburgh, Scotland) 2013 Dec;93 Suppl:S51-9
Tuberculosis (Edinburgh, Scotland) 2013 Dec;93 Suppl:S51-9
A novel method for autophagy detection in primary cells: impaired levels of macroautophagy in immunosenescent T cells.
Phadwal K, Alegre-Abarrategui J, Watson AS, Pike L, Anbalagan S, Hammond EM, Wade-Martins R, McMichael A, Klenerman P, Simon AK
Autophagy 2012 Apr;8(4):677-89
Autophagy 2012 Apr;8(4):677-89
A CD8α(-) subpopulation of macaque circulatory natural killer cells can mediate both antibody-dependent and antibody-independent cytotoxic activities.
Vargas-Inchaustegui DA, Demberg T, Robert-Guroff M
Immunology 2011 Nov;134(3):326-40
Immunology 2011 Nov;134(3):326-40
HIV-1 infection abrogates CD8+ T cell mitogen-activated protein kinase signaling responses.
Crawford TQ, Ndhlovu LC, Tan A, Carvidi A, Hecht FM, Sinclair E, Barbour JD
Journal of virology 2011 Dec;85(23):12343-50
Journal of virology 2011 Dec;85(23):12343-50
Modulation of cell signaling networks after CTLA4 blockade in patients with metastatic melanoma.
Comin-Anduix B, Sazegar H, Chodon T, Matsunaga D, Jalil J, von Euw E, Escuin-Ordinas H, Balderas R, Chmielowski B, Gomez-Navarro J, Koya RC, Ribas A
PloS one 2010 Sep 15;5(9):e12711
PloS one 2010 Sep 15;5(9):e12711
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
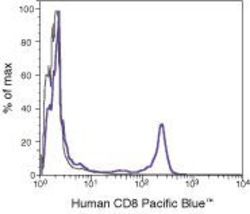
- Experimental details
- Flow Cytometry analysis using a CD8 monoclonal antibody (Product # MHCD0828).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
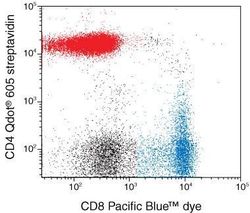
- Experimental details
- Figure 1. Multicolor analysis of CD4-positive and CD8-positive populations using Qdot® 605 streptavidin and a Pacific Blue™ dye-labeled conjugate. Human mononuclear cells were blocked with nonspecific goat IgG and then stained with a biotinylated mouse anti-human CD4 antibody. Cells were incubated with Qdot® 605 streptavidin, then with mouse anti-human CD8 antibody conjugated to Pacific Blue™ dye. Cells were analyzed on a flow cytometer equipped with a 405 nm violet diode laser and 450/50 and 605/20 nm bandpass filters. Compensation was performed using single-color controls. Cells were analyzed using a lymphocyte gate as determined by FSC/SSC. These reagents give clear separation of the CD4-positive and CD8-positive populations using a single excitation source, the violet diode laser.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of CD8 using a monoclonal antibody (Product # MHCD0828).
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry