Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- CD3-4-8-A - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD3/CD4/CD8 Antibody Cocktail, FITC, PE, TRI-COLOR™
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Vial size
- 750 µL
- Storage
- 4° C, store in dark
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
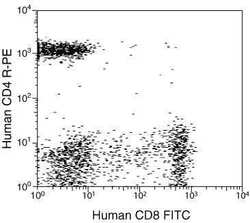
- Experimental details
- Differentiation of human peripheral blood T-cell populations using TRI-COLOR® (PE-Cy™5) conjugated anti-human CD3 mAb (clone: S4.1), FITC conjugated anti-human CD8 mAb (clone: 3B5), and R-PE conjugated anti-human CD4 mAb (clone: S3.5).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Lung neutrophils are the predominant leukocyte population expressing IL-17A during pulmonary infection with C. neoformans strain H99gamma. BALB/c mice received an intranasal inoculum of 1x10 4 CFU of C. neoformans strain H99gamma in 50 ul of sterile PBS (gray bars). Naive Balb/c mice (white bars) are shown as controls. The lungs were excised at day 7 post-inoculation and a single cell suspension generated using enzymatic digestion. The leukocytes were stained with anti-mouse antibodies (CD45, 1A8 (Neut) CD4, CD8, F4/80 (Mac), CD11b/CD11c (DC), CD4/Fox3p (Treg), CD4/DX5 (NKT),gammadelta, B220 (B cell), SiglecF/CD11b (Eosinophil), FcepsilonR1alpha/CD117/CD34 (Mast cell)), fixed, permeabilized, and incubated with anti-mouse antibodies specific for IL-17A and quantified by flow cytometry. Flow cytometry data are cumulative results of five independent experiments using pooled leukocytes from 5 mice per group per experiment. Results shown are the percentage of leukocytes expressing the indicated surface markers and IL-17A. Asterisks (*) indicate where significant differences were observed ( P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Lung gammadelta + T cells are the predominant leukocyte population expressing IL-17A during pulmonary infection with C. neoformans strain H99gamma during neutrophil depletion. BALB/c mice received an intranasal inoculum of 1 x 10 4 CFU of C. neoformans strain H99gamma in 50 mul of sterile PBS. Mice were treated with isotype control antibody (white bars) or with anti-1A8 antibody (gray bars). The lungs were excised at day 7 post-inoculation and a single cell suspension generated using enzymatic digestion. The leukocytes were stained with anti-mouse antibodies (CD45, 1A8 (Neut), CD4, CD8, F4/80 (Mac), CD11b/CD11c (DC), CD4/Fox3p (Treg), CD3/NKp46 (NKT), NKp46/CD45 (NK), gammadelta/CD45 (gammadelta + T cells), CD19 (B cell), SiglecF/CD11b (Eosinophil), fixed, permeabilized, and incubated with anti-IL-17A antibodies and evaluated by flow cytometry. Flow cytometry data are cumulative results of three independent experiments using pooled leukocytes from 4 mice per group per experiment. Results shown are the percentage of leukocytes expressing the indicated surface markers and IL-17A. Asterisks (*) indicate where significant differences ( P < 0.05) were observed between isotype control-treated and anti-1A8-treated mice infected with C. neoformans strain H99gamma.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
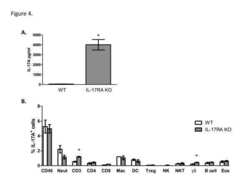
- Experimental details
- Figure 4 Induction of IL-17A production during C. neoformans strain H99gamma infection requires signaling through the IL-17A receptor. BALB/c (white bars) and IL-17RA KO (gray bars) mice were given an intranasal inoculation with C. neoformans strain H99gamma. Lungs were excised at day 7 post-inoculation, and pulmonary cytokine production quantified ( A ) and intracellular IL-17A production was determined in leukocyte populations ( B ). The lungs were excised at day 7 post-inoculation and a single cell suspension generated using enzymatic digestion. The leukocytes were stained with anti-mouse antibodies (CD45, 1A8 (Neut), CD4, CD8, F4/80 (Mac), CD11b/CD11c (DC), CD4/Fox3p (Treg), CD3/NKp46 (NKT), NKp46/CD45 (NK), gammadelta/CD45 (gammadelta + T cells), CD19 (B cell), SiglecF/CD11b (Eosinophil), fixed, permeabilized, and incubated with anti-mouse antibodies specific for IL-17A and quantified by flow cytometry. Asterisks (*) indicate where significant differences ( P < 0.05) were observed between WT and IL-17RA KO mice infected with C. neoformans strain H99gamma. Data are cumulative of three experiments using 3 mice per group.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry