Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Flow cytometry [3]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MHCD0800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD8 Monoclonal Antibody (3B5)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3B5
- Vial size
- 500 µL
- Storage
- 4° C, store in dark
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
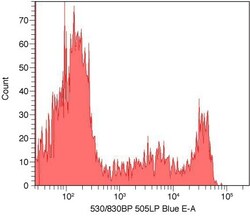
- Experimental details
- Flow cytometry histogram showing human mononuclear cells stained with anti-CD8 complexed with Alexa Fluor® 488 Zenon® labeling reagent. An antibody complex was made by using the Zenon® Alexa Fluor® 488 Mouse IgG2a Labeling Kit (Product # Z-25102) and mouse anti human CD8 (Product # MHCD0800). Human mononuclear cells were stained with the antibody complex and analyzed by flow cytometry using a 488 nm Laser.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
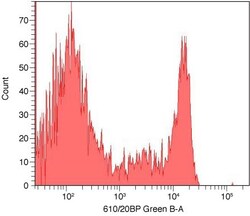
- Experimental details
- An antibody complex was made by using the Zenon® Alexa Fluor® 594 Mouse IgG2a Labeling Kit (Product # Z-25107) and mouse anti human CD8 (Product # MHCD0800). Human mononuclear cells were stained with the antibody complex and analyzed by flow cytometry using a 532 nm Laser.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
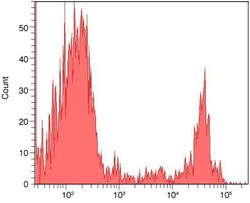
- Experimental details
- Flow cytometry histogram showing human mononuclear cells incubated with anti-CD8 complexed with biotin-XX Zenon® labeling reagent and followed by staining with Alexa Fluor® 488 streptavidin. An antibody complex was made by using the Zenon® Biotin-XX Mouse IgG2a Labeling Kit (Product # Z-25152) and mouse anti human CD8 (Product # MHCD0800). Human mononuclear cells were incubated with the antibody complex, washed, stained with streptavidin, Alexa Fluor® 488 conjugate (Product # S-11223) and analyzed by flow cytometry using a 488 nm Laser.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
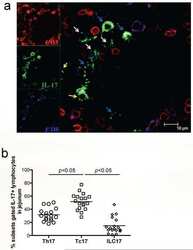
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
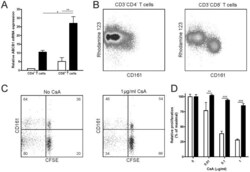
- Figure 2 CD161 hi CD8 + T cells escape from functional inhibition by CsA. (A) Relative ABCB1 expression determined by qPCR on sort-purified CD161 - CD4 + , CD161 neg/low CD8 + (white bars) and CD161 + CD4 + , CD161 hi CD8 + (black bars) T cells. Expression is normalized to that of CD161 - CD4 + T cells. Data represent the mean +- SEM of 2 independent healthy donors. (B) PBMCs were labelled with Rh123, cultured for 30 minutes, and measured by flow cytometry for CD3, CD4, CD8, and CD161. A representative sample is shown gated on CD3 + CD4 + and CD3 + CD8 + T cells. (C-D) CFSE-labelled PBMCs were stimulated in an MLR with irradiated allo-PBMCs in the presence or absence of CsA. CFSE levels were determined after 7 days of culture. (C) Flow cytometry data of one representative experiment is shown gated on CD3 + CD8 + T cells. Numbers represent percentages of proliferation per subset. (D) Relative proliferation is depicted as the percentage proliferating CD161 neg/low T cells (white bars) and CD161 hi CD8 + T cells (black bars) compared to the condition without CsA, as mean+SD ( n = 3). One representative experiment out of 3 independent healthy donors is shown. Statistical analysis was performed using One way ANOVA followed by a Bonferroni post-hoc test. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
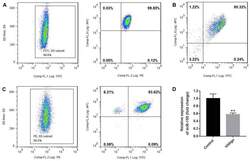
- Figure 1. Purity of CD3 + CD4 + CD45RA + T cells, CD3 + CD8 + T cells and CD4 + CD25 + FoxP3 + Treg cells. CD3 + CD4 + CD45RA + T cells and CD3 + CD8 + T cells were purified by magnetic cell sorting, and their purity was determined by flow cytometry. (A) The purity of CD3 + CD4 + CD45RA + T cells was 99.45% (CD3 + T cells, 99.6%; CD4 + CD45RA + T cells, 99.85%). (B) The purity of CD3 + CD8 + T cells was 95.32%. (C) The purity of CD4 + CD25 + FoxP3 + Treg cells was 93.15% (CD4 + T cells, 99.5%; CD25 + FoxP3 + T cells, 93.62%). (D) miR-155 expression in T cells of the patients with vitiligo and healthy donor was detected by reverse transcription quantitative polymerase chain reaction. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
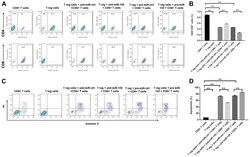
- Figure 4. miR-155 decreased the percentage of CD8 + T cells via promoting apoptosis in the cell culture medium. Anti-miR-ctrl, anti-miR-155, pre-miR-ctrl and pre-miR-155 were transfected into Treg cells. (A) Flow cytometry images and (B) the percentage of CD8 + cells, as determined at 3 days after transfection. Representative fluorescence-activated cell sorting images from a single case are shown. (C) The apoptosis of CD8 + T cells was detected using the Annexin V-FITC/PI Apoptosis Detection kit. (D) Cell apoptosis rate was quantified by flow cytometry. Data are representative of three independent experiments. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
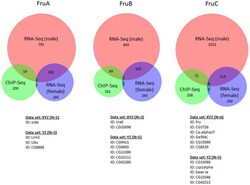
- Figure 5 ChIP-Seq identified genes do not show expression changes in the female fly brain. The ChIP-Seq gene lists were also compared to RNA-Seq performed in the female fly brain 25 following overexpression of the male specific isoform. Although the male and female brain RNA-Seq experiments showed some overlap, very few of these genes were also identified in the ChIP-Seq experiments. In fact, less than 4% of genes identifed in any of the isoform specific ChIP-Seq gene lists demonstrated any expression differences in the female fly brain, compared to more than 20% of genes that were affected in the male brain. Overlap was visualised using BioVenn 43 .
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry