Antibody data
- Antibody Data
- Antigen structure
- References [28]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [8]
- Immunoprecipitation [1]
- Immunohistochemistry [3]
- Flow cytometry [2]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA3-012 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- HSP90 beta Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA3-012 detects heat shock protein 90 beta (HSP90) from human, monkey, mouse, and rat tissues and cells. This antibody does not detect HSP86. PA3-012 has been successfully used in Western blot, immunohistochemistry, immunofluorescence, flow cytometry, and immunoprecipitation procedures. By Western blot, this antibody detects an ~84 kDa protein representing HSP84 from Hepa1 cell lysate. Immunofluorescence staining of HSP84 in Hepa1 cells with PA3-012 yields a pattern consistent with cytoplasmic staining. Immunoprecipitation experiments with this antibody suggest that HSP84 exists primarily as homodimers in Hepa 1 cells. Furthermore, the antibody is capable of precipitating HSP84 that is complexed with other proteins such as the aryl hydrocarbon (Ah) receptor. The PA3-012 immunogen is a synthetic peptide corresponding to residues P(2) E E V H H G E E E V E(13) of mouse HSP84. The N-terminal regions of HSP84 and HSP86 show the largest difference in amino acid sequence.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references HSP90 promotes radioresistance of cervical cancer cells via reducing FBXO6-mediated CD147 polyubiquitination.
Human coronavirus dependency on host heat shock protein 90 reveals an antiviral target.
Heat Shock Protein 90 as a Prognostic Marker and Therapeutic Target for Adrenocortical Carcinoma.
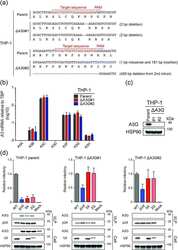
HIV-1 restriction by endogenous APOBEC3G in the myeloid cell line THP-1.
APOBEC3B lysine residues are dispensable for DNA cytosine deamination, HIV-1 restriction, and nuclear localization.
The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling.
Vacuolar H+ ATPase expression and activity is required for Rab27B-dependent invasive growth and metastasis of breast cancer.
Heat shock protein 90α (HSP90α), a substrate and chaperone of DNA-PK necessary for the apoptotic response.
Mutations that increase both Hsp90 ATPase activity in vitro and Hsp90 drug resistance in vivo.
Effect of the secretory small GTPase Rab27B on breast cancer growth, invasion, and metastasis.
Tubulin acetylation favors Hsp90 recruitment to microtubules and stimulates the signaling function of the Hsp90 clients Akt/PKB and p53.
Proteomic identification of differentially expressed genes in mouse neural stem cells and neurons differentiated from embryonic stem cells in vitro.
Changes of endoplasmic reticulum chaperone complexes, redox state, and impaired protein disulfide reductase activity in misfolding alpha1-antitrypsin transgenic mice.
Progressive decrease in chaperone protein levels in a mouse model of Huntington's disease and induction of stress proteins as a therapeutic approach.
Functional and cellular responses in a novel rodent model of anterior ischemic optic neuropathy.
Comparative analysis of the ATP-binding sites of Hsp90 by nucleotide affinity cleavage: a distinct nucleotide specificity of the C-terminal ATP-binding site.
Tyrosine phosphorylation of HSP-90 during mammalian sperm capacitation.
Purification of polyglutamine aggregates and identification of elongation factor-1alpha and heat shock protein 84 as aggregate-interacting proteins.
Purification of polyglutamine aggregates and identification of elongation factor-1alpha and heat shock protein 84 as aggregate-interacting proteins.
A Nucleotide-dependent molecular switch controls ATP binding at the C-terminal domain of Hsp90. N-terminal nucleotide binding unmasks a C-terminal binding pocket.
Overexpression of murine small heat shock protein HSP25 interferes with chondrocyte differentiation and decreases cell adhesion.
Hsp and chaperone distribution during endochondral bone development in mouse embryo.
Hsp and chaperone distribution during endochondral bone development in mouse embryo.
Function and regulation of heat shock factor 2 during mouse embryogenesis.
Function and regulation of heat shock factor 2 during mouse embryogenesis.
Heat shock protein 84 forms a complex with mutant p53 protein predominantly within a cytoplasmic compartment of the cell.
Subunit composition of the heteromeric cytosolic aryl hydrocarbon receptor complex.
Localization and characterization of the 86- and 84-kDa heat shock proteins in Hepa 1c1c7 cells.
Song Q, Wen J, Li W, Xue J, Zhang Y, Liu H, Han J, Ning T, Lu Z
Cancer science 2022 Apr;113(4):1463-1474
Cancer science 2022 Apr;113(4):1463-1474
Human coronavirus dependency on host heat shock protein 90 reveals an antiviral target.
Li C, Chu H, Liu X, Chiu MC, Zhao X, Wang D, Wei Y, Hou Y, Shuai H, Cai J, Chan JF, Zhou J, Yuen KY
Emerging microbes & infections 2020 Dec;9(1):2663-2672
Emerging microbes & infections 2020 Dec;9(1):2663-2672
Heat Shock Protein 90 as a Prognostic Marker and Therapeutic Target for Adrenocortical Carcinoma.
Siebert C, Ciato D, Murakami M, Frei-Stuber L, Perez-Rivas LG, Monteserin-Garcia JL, Nölting S, Maurer J, Feuchtinger A, Walch AK, Haak HR, Bertherat J, Mannelli M, Fassnacht M, Korpershoek E, Reincke M, Stalla GK, Hantel C, Beuschlein F
Frontiers in endocrinology 2019;10:487
Frontiers in endocrinology 2019;10:487
HIV-1 restriction by endogenous APOBEC3G in the myeloid cell line THP-1.
Ikeda T, Molan AM, Jarvis MC, Carpenter MA, Salamango DJ, Brown WL, Harris RS
The Journal of general virology 2019 Jul;100(7):1140-1152
The Journal of general virology 2019 Jul;100(7):1140-1152
APOBEC3B lysine residues are dispensable for DNA cytosine deamination, HIV-1 restriction, and nuclear localization.
Molan AM, Hanson HM, Chweya CM, Anderson BD, Starrett GJ, Richards CM, Harris RS
Virology 2017 Nov;511:74-81
Virology 2017 Nov;511:74-81
The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling.
Van Deun J, Mestdagh P, Sormunen R, Cocquyt V, Vermaelen K, Vandesompele J, Bracke M, De Wever O, Hendrix A
Journal of extracellular vesicles 2014;3
Journal of extracellular vesicles 2014;3
Vacuolar H+ ATPase expression and activity is required for Rab27B-dependent invasive growth and metastasis of breast cancer.
Hendrix A, Sormunen R, Westbroek W, Lambein K, Denys H, Sys G, Braems G, Van den Broecke R, Cocquyt V, Gespach C, Bracke M, De Wever O
International journal of cancer 2013 Aug 15;133(4):843-54
International journal of cancer 2013 Aug 15;133(4):843-54
Heat shock protein 90α (HSP90α), a substrate and chaperone of DNA-PK necessary for the apoptotic response.
Solier S, Kohn KW, Scroggins B, Xu W, Trepel J, Neckers L, Pommier Y
Proceedings of the National Academy of Sciences of the United States of America 2012 Aug 7;109(32):12866-72
Proceedings of the National Academy of Sciences of the United States of America 2012 Aug 7;109(32):12866-72
Mutations that increase both Hsp90 ATPase activity in vitro and Hsp90 drug resistance in vivo.
Zurawska A, Urbanski J, Matuliene J, Baraniak J, Klejman MP, Filipek S, Matulis D, Bieganowski P
Biochimica et biophysica acta 2010 May;1803(5):575-83
Biochimica et biophysica acta 2010 May;1803(5):575-83
Effect of the secretory small GTPase Rab27B on breast cancer growth, invasion, and metastasis.
Hendrix A, Maynard D, Pauwels P, Braems G, Denys H, Van den Broecke R, Lambert J, Van Belle S, Cocquyt V, Gespach C, Bracke M, Seabra MC, Gahl WA, De Wever O, Westbroek W
Journal of the National Cancer Institute 2010 Jun 16;102(12):866-80
Journal of the National Cancer Institute 2010 Jun 16;102(12):866-80
Tubulin acetylation favors Hsp90 recruitment to microtubules and stimulates the signaling function of the Hsp90 clients Akt/PKB and p53.
Giustiniani J, Daire V, Cantaloube I, Durand G, Poüs C, Perdiz D, Baillet A
Cellular signalling 2009 Apr;21(4):529-39
Cellular signalling 2009 Apr;21(4):529-39
Proteomic identification of differentially expressed genes in mouse neural stem cells and neurons differentiated from embryonic stem cells in vitro.
Akama K, Tatsuno R, Otsu M, Horikoshi T, Nakayama T, Nakamura M, Toda T, Inoue N
Biochimica et biophysica acta 2008 May;1784(5):773-82
Biochimica et biophysica acta 2008 May;1784(5):773-82
Changes of endoplasmic reticulum chaperone complexes, redox state, and impaired protein disulfide reductase activity in misfolding alpha1-antitrypsin transgenic mice.
Papp E, Száraz P, Korcsmáros T, Csermely P
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2006 May;20(7):1018-20
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2006 May;20(7):1018-20
Progressive decrease in chaperone protein levels in a mouse model of Huntington's disease and induction of stress proteins as a therapeutic approach.
Hay DG, Sathasivam K, Tobaben S, Stahl B, Marber M, Mestril R, Mahal A, Smith DL, Woodman B, Bates GP
Human molecular genetics 2004 Jul 1;13(13):1389-405
Human molecular genetics 2004 Jul 1;13(13):1389-405
Functional and cellular responses in a novel rodent model of anterior ischemic optic neuropathy.
Bernstein SL, Guo Y, Kelman SE, Flower RW, Johnson MA
Investigative ophthalmology & visual science 2003 Oct;44(10):4153-62
Investigative ophthalmology & visual science 2003 Oct;44(10):4153-62
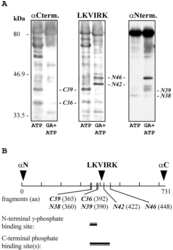
Comparative analysis of the ATP-binding sites of Hsp90 by nucleotide affinity cleavage: a distinct nucleotide specificity of the C-terminal ATP-binding site.
Soti C, Vermes A, Haystead TA, Csermely P
European journal of biochemistry 2003 Jun;270(11):2421-8
European journal of biochemistry 2003 Jun;270(11):2421-8
Tyrosine phosphorylation of HSP-90 during mammalian sperm capacitation.
Ecroyd H, Jones RC, Aitken RJ
Biology of reproduction 2003 Dec;69(6):1801-7
Biology of reproduction 2003 Dec;69(6):1801-7
Purification of polyglutamine aggregates and identification of elongation factor-1alpha and heat shock protein 84 as aggregate-interacting proteins.
Mitsui K, Nakayama H, Akagi T, Nekooki M, Ohtawa K, Takio K, Hashikawa T, Nukina N
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 1;22(21):9267-77
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 1;22(21):9267-77
Purification of polyglutamine aggregates and identification of elongation factor-1alpha and heat shock protein 84 as aggregate-interacting proteins.
Mitsui K, Nakayama H, Akagi T, Nekooki M, Ohtawa K, Takio K, Hashikawa T, Nukina N
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 1;22(21):9267-77
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Nov 1;22(21):9267-77
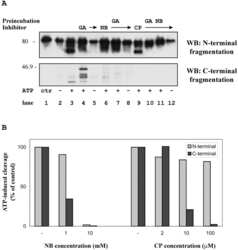
A Nucleotide-dependent molecular switch controls ATP binding at the C-terminal domain of Hsp90. N-terminal nucleotide binding unmasks a C-terminal binding pocket.
Söti C, Rácz A, Csermely P
The Journal of biological chemistry 2002 Mar 1;277(9):7066-75
The Journal of biological chemistry 2002 Mar 1;277(9):7066-75
Overexpression of murine small heat shock protein HSP25 interferes with chondrocyte differentiation and decreases cell adhesion.
Favet N, Duverger O, Loones MT, Poliard A, Kellermann O, Morange M
Cell death and differentiation 2001 Jun;8(6):603-13
Cell death and differentiation 2001 Jun;8(6):603-13
Hsp and chaperone distribution during endochondral bone development in mouse embryo.
Loones MT, Morange M
Cell stress & chaperones 1998 Dec;3(4):237-44
Cell stress & chaperones 1998 Dec;3(4):237-44
Hsp and chaperone distribution during endochondral bone development in mouse embryo.
Loones MT, Morange M
Cell stress & chaperones 1998 Dec;3(4):237-44
Cell stress & chaperones 1998 Dec;3(4):237-44
Function and regulation of heat shock factor 2 during mouse embryogenesis.
Rallu M, Loones M, Lallemand Y, Morimoto R, Morange M, Mezger V
Proceedings of the National Academy of Sciences of the United States of America 1997 Mar 18;94(6):2392-7
Proceedings of the National Academy of Sciences of the United States of America 1997 Mar 18;94(6):2392-7
Function and regulation of heat shock factor 2 during mouse embryogenesis.
Rallu M, Loones M, Lallemand Y, Morimoto R, Morange M, Mezger V
Proceedings of the National Academy of Sciences of the United States of America 1997 Mar 18;94(6):2392-7
Proceedings of the National Academy of Sciences of the United States of America 1997 Mar 18;94(6):2392-7
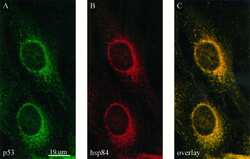
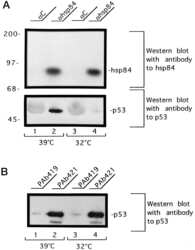
Heat shock protein 84 forms a complex with mutant p53 protein predominantly within a cytoplasmic compartment of the cell.
Sepehrnia B, Paz IB, Dasgupta G, Momand J
The Journal of biological chemistry 1996 Jun 21;271(25):15084-90
The Journal of biological chemistry 1996 Jun 21;271(25):15084-90
Subunit composition of the heteromeric cytosolic aryl hydrocarbon receptor complex.
Chen HS, Perdew GH
The Journal of biological chemistry 1994 Nov 4;269(44):27554-8
The Journal of biological chemistry 1994 Nov 4;269(44):27554-8
Localization and characterization of the 86- and 84-kDa heat shock proteins in Hepa 1c1c7 cells.
Perdew GH, Hord N, Hollenback CE, Welsh MJ
Experimental cell research 1993 Dec;209(2):350-6
Experimental cell research 1993 Dec;209(2):350-6
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
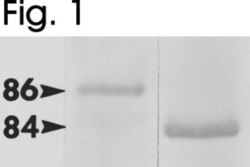
- Experimental details
- Western blot of mouse HSP84 and HSP86 using Product # PA3-013 and Product # PA3-012, respectively.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
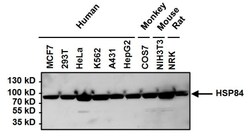
- Experimental details
- Western blot analysis of Heat Shock Protein 84 (HSP84) was performed by loading 50 µg of the indicated whole cell lysates per well onto a 4-20% Tris-HCl polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked with 5% BSA/TBST for at least 1 hour. The membrane was then probed with a HSP84 polyclonal antibody (Product # PA3-012) at a dilution of 1:1000 overnight at 4°C on a rocking platform, washed in TBS-0.1%Tween 20, and probed with a goat anti-rabbit IgG-HRP secondary antibody (Product # 31460) at a dilution of 1:20,000 for at least one hour. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
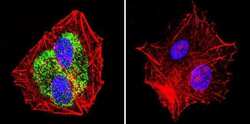
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in HepG2 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
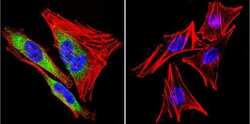
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in A2058 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
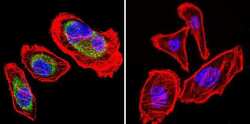
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in U251 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
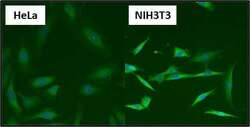
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 (HSP84) (green) in HeLa and NIH3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature. Cells were blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a HSP84 polyclonal antibody (Product # PA3-012), at a dilution of 1:50 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat-anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:400 for 30 minutes at room temperature. Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
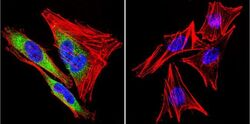
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in A2058 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
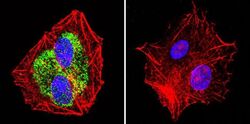
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in HepG2 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
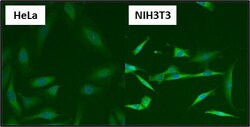
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 using Anti-Heat Shock Protein 84 Polyclonal Antibody (Product # PA3-012) shows staining in U251 Cells. Heat Shock Protein 84 staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Heat Shock Protein 84 (Product # PA3-012) at a dilution of 1:100 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
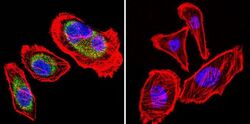
- Experimental details
- Immunofluorescent analysis of Heat Shock Protein 84 (HSP84) (green) in HeLa and NIH3T3 cells. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature. Cells were blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a HSP84 polyclonal antibody (Product # PA3-012), at a dilution of 1:50 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat-anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:400 for 30 minutes at room temperature. Nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
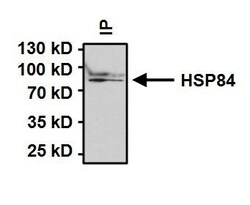
- Experimental details
- Immunoprecipitation of Heat Shock Protein 84 (HSP84) was performed on HeLa cells. Antigen:antibody complexes were formed by incubating 500 µg whole cell lysate with 2 µg of HSP84 polyclonal antibody (Product # PA3-012) overnight on a rocking platform at 4°C. Immune complexes were captured on 50 µL Protein A/G Plus Agarose (Product # 20423), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). Samples were resolved on a 4-20% Tris-HCl polyacrylamide gel, transferred to a PVDF membrane, and blocked with 5% BSA/TBST for at least 1 hour. The membrane was probed with a HSP84 polyclonal antibody (Product # PA3-012) at a dilution of 1:1000 overnight rotating at 4°C, washed in TBST, and probed with Clean-Blot IP Detection Reagent-HRP (Product # 21232) at a dilution of 1:1000 for at least one hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34075).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
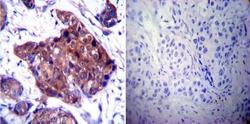
- Experimental details
- Immunohistochemistry was performed on cancer biopsies of deparaffinized Human breast carcinoma tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:100 with a rabbit polyclonal antibody recognizing Heat Shock Protein 84 (Product # PA3-012) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
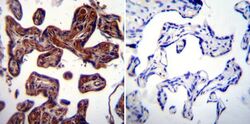
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human placenta tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:20 with a rabbit polyclonal antibody recognizing Heat Shock Protein 84 (Product # PA3-012) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
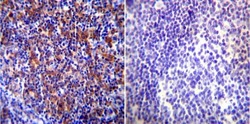
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Human tonsil tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:100 with a rabbit polyclonal antibody recognizing Heat Shock Protein 84 (Product # PA3-012) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
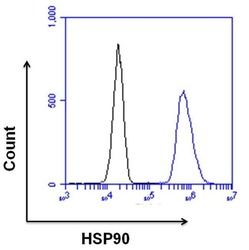
- Experimental details
- Flow cytometry analysis of HSP90 was done on HeLa cells. Cells were fixed, permeabilized and stained with a HSP90 rabbit polyclonal antibody (Product # PA3-012, blue histogram) or a rabbit IgG isotype control (Product # MA5-16384, black histogram) at a dilution of 10 µg/mL. After incubation for 1 hour on ice, the cells were labeled with a Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 647 conjugate (Product # A27040) at a dilution of 1:50 for 1 hour on ice. A representative 10,000 cells were acquired and analyzed for each sample.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
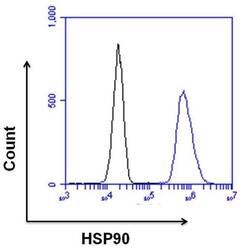
- Experimental details
- Flow cytometry analysis of HSP90 was done on HeLa cells. Cells were fixed, permeabilized and stained with a HSP90 rabbit polyclonal antibody (Product # PA3-012, blue histogram) or a rabbit IgG isotype control (Product # MA5-16384, black histogram) at a dilution of 10 µg/mL. After incubation for 1 hour on ice, the cells were labeled with a Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Secondary Antibody, Alexa Fluor® 647 conjugate (Product # A27040) at a dilution of 1:50 for 1 hour on ice. A representative 10,000 cells were acquired and analyzed for each sample.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
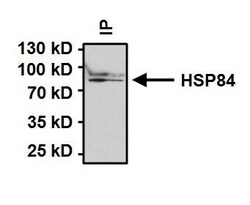
- Immunoprecipitation of Heat Shock Protein 84 (HSP84) was performed on HeLa cells. Antigen:antibody complexes were formed by incubating 500 µg whole cell lysate with 2 µg of HSP84 polyclonal antibody (Product # PA3-012) overnight on a rocking platform at 4°C. Immune complexes were captured on 50 µL Protein A/G Plus Agarose (Product # 20423), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). Samples were resolved on a 4-20% Tris-HCl polyacrylamide gel, transferred to a PVDF membrane, and blocked with 5% BSA/TBST for at least 1 hour. The membrane was probed with a HSP84 polyclonal antibody (Product # PA3-012) at a dilution of 1:1000 overnight rotating at 4°C, washed in TBST, and probed with Clean-Blot IP Detection Reagent-HRP (Product # 21232) at a dilution of 1:1000 for at least one hour. Chemiluminescent detection was performed using SuperSignal West Dura (Product # 34075).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
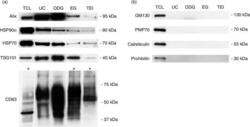
- Fig. 2 Characterization of exosome preparations by Western blot. Western blot analysis of (a) common exosome markers (Alix, HSP90alpha, HSP70, TSG101 and CD63) and (b) cell organelle and apoptosis markers (GM130, PMP70, calreticulin and prohibitin) in 10 ug of exosomes isolated by 4 different methods. MCF-7 Rab27B total cell lysate (TCL) was loaded as positive control. Asterisks indicate loading of 50 ug of protein.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
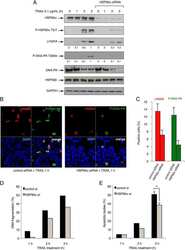
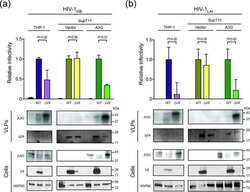
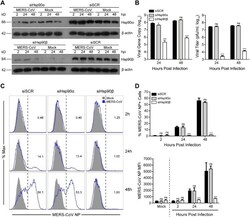
- Figure 3. Genetic depletion of Hsp90beta suppressed MERS-CoV replication and abolished virus spread. At 24 h after transfection of Hsp90alpha, Hsp90beta or scrambled siRNA in triplicate, A549 cells were infected with MERS-CoV at a MOI of 0.1. (A) The expression levels of Hsp90alpha and Hsp90beta in both infected and mock-infected cells were detected by Western blot. (B) At the indicated hours post infection, cell-free culture media were applied to viral load detection and viral titration. (C, D) The infected or mock-infected cells were fixed at the indicated hours post infection and labelled for flow cytometry. (C) Histograms show the infection rates of one representative experiment. (D) Results present mean and SD of infection rate and MFI of one representative experiment independently repeated three times. Student's t test was used for data analysis. *** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
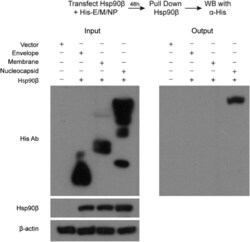
- Experimental details
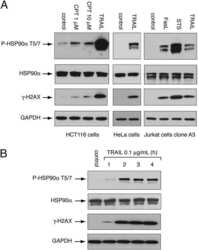
- Figure 4. Co-IP assay identified MERS-CoV NP as an Hsp90beta client protein. 293T cells were transfected with Hsp90beta expression plasmid and His-tag plasmid expressing the indicated MERS-CoV structural proteins or blank vector. Cell lysates of the transfectants were applied to verify the expression of His-tagged viral proteins and Hsp90beta. The cell lysates (input) were used for immunoprecipitation with an alpha-Hsp90beta antibody; the co-precipitated partner (output) was detected by Western blot using an alpha-His antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
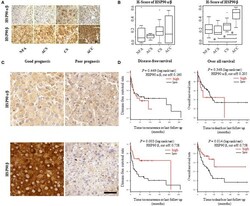
- Experimental details
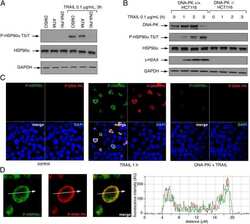
- Figure 1 HSP90alpha/beta and HSP90beta staining in adrenocortical adenomas (NFA, non-functional adenoma, n = 8; ACS, adenoma with autonomous cortisol secretion, n = 4; CS, adenoma with overt Cushing syndrome, n = 14) and adrenocortical carcinoma (ACC, n = 6) with representative sections (A) and semi-quantitative H-scores (B) . Representative images of HSP90alpha/beta and HSP90beta immunohistochemistry from an ACC patient with good prognosis without recurrence and one with poor prognosis with disease related death, respectively (C) . Kaplan-Meier plots for disease-free and overall survival (D) in relation to HSP90alpha/beta and HSP90beta cytoplasmic intensity above (high: red line) or below (low: black line) the cut off. Log rank test was used to statistically compare the curves and P -values are provided (* P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
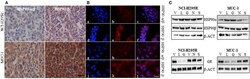
- Experimental details
- Figure 2 Representative pictures of the abundance of HSP90alpha/beta and HSP90beta in NCI-H295R and MUC-1 xenografts (A) . Immunofluorescence staining illustrating the localization of HSP90alpha/beta (a, g: DAPI; b,h: HSP90alpha/beta; c,i: merged) and HSP90beta (d,j: DAPI; e,k: HSP90beta; f,l: merged) in NCI-H295R (a-f) and MUC-1 (g-l) cells (B) . Expression of HSP90alpha, HSP90beta and glucocorticoid receptor (GR) was demonstrated by Western Blot analysis. NCI-H295R and MUC-1 cells were treated with HSP90 inhibitors for 48 h vehicle (V): 1:1000 DMSO; luminespib (L): 0.2 muM; ganetespib (G): 0.2 muM; novobiocin (N): 100 muM; silibinin (S): 40 muM (C) . Scale bars represent 25 mum.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot