Antibody data
- Antibody Data
- Antigen structure
- References [1]
- Comments [0]
- Validations
- Immunocytochemistry [4]
- Immunoprecipitation [2]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-167 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- HuR Monoclonal Antibody (3A2)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3A2
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references RNA-binding protein HuR regulates translation of vitamin D receptor modulating rapid epithelial restitution after wounding.
Zhang Y, Cai JZ, Xiao L, Chung HK, Ma XX, Chen LL, Rao JN, Wang JY
American journal of physiology. Cell physiology 2020 Jul 1;319(1):C208-C217
American journal of physiology. Cell physiology 2020 Jul 1;319(1):C208-C217
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
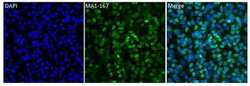
- Experimental details
- Immunofluorescent analysis of HuR (3A2) (green) in HeLa cells. Cells were fixed in 4% paraformaldhyde, permeabilized with 0.1% Triton X-100, and blocked with 1% Blocker BSA (Product # 37525), each for 15 minutes at room temperature. Cells were stained with HuR (3A2) monoclonal antibody (Product # MA1-167) at a dilution of 1:50 in blocking buffer at 37C for at least one hour. Cells were then washed with PBS and then incubated with a DyLight 488-conjugated goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:1000 for 1 hour at room temperature. Nuclei (blue) were stained with with DAPI (Product # 46190). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
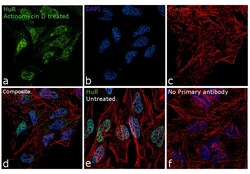
- Experimental details
- Immunofluorescence analysis of HuR was performed using 70% confluent log phase HeLa cells treated with Actinomycin D (1uM for 19hrs). The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with HuR Monoclonal Antibody (3A2) (Product # MA1-167) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing translocation of HuR in to the cytoplasm. Panel e represents the control cells showing exclusive nuclear staining. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
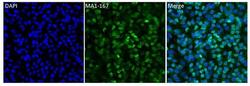
- Experimental details
- Immunofluorescent analysis of HuR (3A2) (green) in HeLa cells. Cells were fixed in 4% paraformaldhyde, permeabilized with 0.1% Triton X-100, and blocked with 1% Blocker BSA (Product # 37525), each for 15 minutes at room temperature. Cells were stained with HuR (3A2) monoclonal antibody (Product # MA1-167) at a dilution of 1:50 in blocking buffer at 37C for at least one hour. Cells were then washed with PBS and then incubated with a DyLight 488-conjugated goat anti-mouse IgG secondary antibody (Product # 35502) at a dilution of 1:1000 for 1 hour at room temperature. Nuclei (blue) were stained with with DAPI (Product # 46190). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
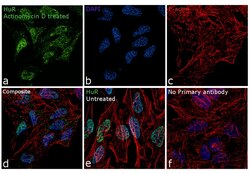
- Experimental details
- Immunofluorescence analysis of HuR was performed using 70% confluent log phase HeLa cells treated with Actinomycin D (1uM for 19hrs). The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with HuR Monoclonal Antibody (3A2) (Product # MA1-167) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing translocation of HuR in to the cytoplasm. Panel e represents the control cells showing exclusive nuclear staining. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
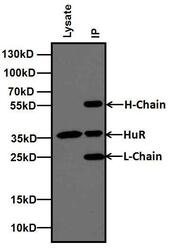
- Experimental details
- Immunoprecipitation of HuR was performed using MCF-7 cells. Antigen-antibody complexes were formed by incubating 500 µg of MCF-7 whole cell lysates with 2 µg of HuR (3A2) monoclonal antibody (Product # MA1-167) overnight on a rocking platform at 4C. The immune complexes were captured on 100 µL Protein A/G Agarose (Product # 20421), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). The IP eluate (right lane) and 20 µg of MCF-7 lysate (left lane) were resolved on a 4-20% Tris-Glycine polyacrylamide gel, transferred to a nitrocellulose membrane, and blocked with 5% Milk in TBST for at least 1 hour. The membrane was probed with a HuR (3A2) monoclonal antibody monoclonal antibody (Product # MA1-167) at a dilution of 1:250 overnight rotating at 4C, washed in TBST, and probed with goat anti-mouse IgG-HRP secondary antibody (Product # 31430) at a dilution of 1:10,000 for at least 30 minutes at room temperature. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
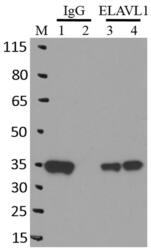
- Experimental details
- Immunoprecipitation of ELAVL1 was performed on K562 cells. Antigen-antibody complexes were formed by incubating approximately 500 ug whole cell lysate with 5 ug of monoclonal ELAVL1 antibody (Product # MA1-167) rotating 60 min at RT. The immune complexes were captured on 625 ug of Dynabeads M-280 sheep anti-mouse IgG (Product # 11202D), washed extensively, and eluted in NuPAGE LDS sample buffer (Product # NP0007). Samples were resolved onto a 4-12% Bis-Tris protein gel (Product # NP0335BOX). Lanes 1 and 3 are input and lanes 2 and 4 are IP. Proteins were transferred to nitrocellulose membrane (Product # IB23001) and blocked in 5% milk. ELAVL1 was detected at approximately 36 kDa using a monoclonal ELAVL1 antibody (Product # MA1-167) at a dilution of 1:2000, followed by incubation with anti-mouse secondary antibody. Chemiluminescent detection was performed using ECL Western Blotting Substrate (Product # 32106). Data courtesy of the Yeo lab as part of the ENCODE project (www.encodeproject.org).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
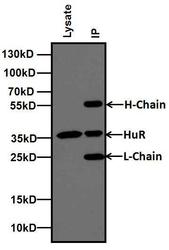
- Experimental details
- Immunoprecipitation of HuR was performed using MCF-7 cells. Antigen-antibody complexes were formed by incubating 500 µg of MCF-7 whole cell lysates with 2 µg of HuR (3A2) monoclonal antibody (Product # MA1-167) overnight on a rocking platform at 4C. The immune complexes were captured on 100 µL Protein A/G Agarose (Product # 20421), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). The IP eluate (right lane) and 20 µg of MCF-7 lysate (left lane) were resolved on a 4-20% Tris-Glycine polyacrylamide gel, transferred to a nitrocellulose membrane, and blocked with 5% Milk in TBST for at least 1 hour. The membrane was probed with a HuR (3A2) monoclonal antibody monoclonal antibody (Product # MA1-167) at a dilution of 1:250 overnight rotating at 4C, washed in TBST, and probed with goat anti-mouse IgG-HRP secondary antibody (Product # 31430) at a dilution of 1:10,000 for at least 30 minutes at room temperature. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
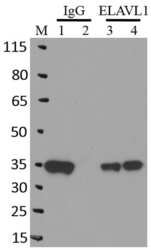
- Experimental details
- Immunoprecipitation of ELAVL1 was performed on K562 cells. Antigen-antibody complexes were formed by incubating approximately 500 ug whole cell lysate with 5 ug of monoclonal ELAVL1 antibody (Product # MA1-167) rotating 60 min at RT. The immune complexes were captured on 625 ug of Dynabeads M-280 sheep anti-mouse IgG (Product # 11202D), washed extensively, and eluted in NuPAGE LDS sample buffer (Product # NP0007). Samples were resolved onto a 4-12% Bis-Tris protein gel (Product # NP0335BOX). Lanes 1 and 3 are input and lanes 2 and 4 are IP. Proteins were transferred to nitrocellulose membrane (Product # IB23001) and blocked in 5% milk. ELAVL1 was detected at approximately 36 kDa using a monoclonal ELAVL1 antibody (Product # MA1-167) at a dilution of 1:2000, followed by incubation with anti-mouse secondary antibody. Chemiluminescent detection was performed using ECL Western Blotting Substrate (Product # 32106). Data courtesy of the Yeo lab as part of the ENCODE project (www.encodeproject.org).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
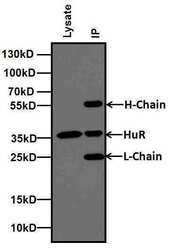
- Experimental details
- Immunoprecipitation of HuR was performed using MCF-7 cells. Antigen-antibody complexes were formed by incubating 500 µg of MCF-7 whole cell lysates with 2 µg of HuR (3A2) monoclonal antibody (Product # MA1-167) overnight on a rocking platform at 4C. The immune complexes were captured on 100 µL Protein A/G Agarose (Product # 20421), washed extensively, and eluted with 5X Lane Marker Reducing Sample Buffer (Product # 39000). The IP eluate (right lane) and 20 µg of MCF-7 lysate (left lane) were resolved on a 4-20% Tris-Glycine polyacrylamide gel, transferred to a nitrocellulose membrane, and blocked with 5% Milk in TBST for at least 1 hour. The membrane was probed with a HuR (3A2) monoclonal antibody monoclonal antibody (Product # MA1-167) at a dilution of 1:250 overnight rotating at 4C, washed in TBST, and probed with goat anti-mouse IgG-HRP secondary antibody (Product # 31430) at a dilution of 1:10,000 for at least 30 minutes at room temperature. Chemiluminescent detection was performed using SuperSignal West Pico (Product # 34080).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
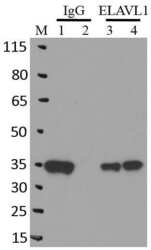
- Experimental details
- RNA immunoprecipitation (RIP) western of ELAVL1 was performed on K562 cells. Antigen-antibody complexes were formed by incubating approximately 500 æg whole cell lysate with 5 æg of monoclonal ELAVL1 antibody (Product # MA1-167) rotating 60 min at RT. The immune complexes were captured on 625 æg of Dynabeads M-280 sheep anti-mouse IgG (Product # 11202D), washed extensively, and eluted in NuPAGE LDS sample buffer (Product # NP0007). Samples were resolved onto a 4-12% Bis-Tris protein gel (Product # NP0335BOX). Lanes 1 and 3 are input and lanes 2 and 4 are IP. Proteins were transferred to nitrocellulose membrane (Product # IB23001) and blocked in 5% milk. ELAVL1 was detected at approximately 36 kDa using a monoclonal ELAVL1 antibody (Product # MA1-167) at a dilution of 1:2000, followed by incubation with anti-mouse secondary antibody. Chemiluminescent detection was performed using ECL Western Blotting Substrate (Product # 32106). Data courtesy of the Yeo lab as part of the ENCODE project (www.encodeproject.org).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
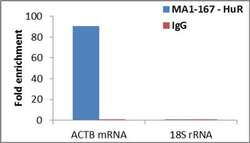
- Experimental details
- Detection of binding of endogenous HuR protein to specific RNA using Anti-HuR Antibody: RNA Immunoprecipitation (RIP) was performed using Anti-HuR Mouse Monoclonal Antibody (Product # MA1-167, 4 µg) on clarified whole cell lysate from 2 million SH-SY5Y cells. Normal Rabbit IgG was used as a negative IP control. Immunoprecipitated RNA was purified by RiboPure™ RNA Purification Kit (Product # AM1924) and analyzed by RT-PCR using the Power SYBR® Green RNA-to-CT™ 1-Step Kit (Product # 4389986) with primer pairs over ACTB mRNA (positive) and 18S rRNA (negative). Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
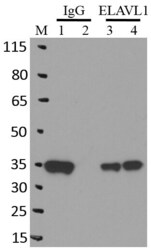
- Experimental details
- RNA immunoprecipitation (RIP) western of ELAVL1 was performed on K562 cells. Antigen-antibody complexes were formed by incubating approximately 500 µg whole cell lysate with 5 µg of monoclonal ELAVL1 antibody (Product # MA1-167) rotating 60 min at RT. The immune complexes were captured on 625 µg of Dynabeads M-280 sheep anti-mouse IgG (Product # 11202D), washed extensively, and eluted in NuPAGE LDS sample buffer (Product # NP0007). Samples were resolved onto a 4-12% Bis-Tris protein gel (Product # NP0335BOX). Lanes 1 and 3 are input and lanes 2 and 4 are IP. Proteins were transferred to nitrocellulose membrane (Product # IB23001) and blocked in 5% milk. ELAVL1 was detected at approximately 36 kDa using a monoclonal ELAVL1 antibody (Product # MA1-167) at a dilution of 1:2000, followed by incubation with anti-mouse secondary antibody. Chemiluminescent detection was performed using ECL Western Blotting Substrate (Product # 32106). Data courtesy of the Yeo lab as part of the ENCODE project (www.encodeproject.org).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry