Antibody data
- Antibody Data
- Antigen structure
- References [24]
- Comments [0]
- Validations
- Immunohistochemistry [1]
- Other assay [11]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 37-0100 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- c-Met Monoclonal Antibody (3D4)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- This antibody is specific for the cytoplasmic domain of the c-Met protein. On western blotting, this antibody recognizes a band at approximately 145 kDa. During development reactivity was confirmed with human HT-29 breast carcinoma lysates and c-Met cytoplasmic domain recombinant protein by both western blot, and with formalin-fixed, paraffin-embedded breast carcinoma tissues by immunohistochemistry. For immunohistochemistry with formalin-fixed, paraffin-embedded tissues, heat-induced epitope retrieval (HIER) with citrate buffer, pH 6.0, is required prior to staining. Recommended antibody incubation period is overnight at 4°C.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3D4
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Targeting of c-MET and AXL by cabozantinib is a potential therapeutic strategy for patients with head and neck cell carcinoma.
Alpha-fetoprotein can promote gastric cancer progression via upregulation of metastasis-associated colon cancer 1.
Dimerization of kringle 1 domain from hepatocyte growth factor/scatter factor provides a potent MET receptor agonist.
Down-regulation of the tumor suppressor miR-34a contributes to head and neck cancer by up-regulating the MET oncogene and modulating tumor immune evasion.
A cysteine selenosulfide redox switch for protein chemical synthesis.
Targeting the MET Receptor Tyrosine Kinase as a Strategy for Radiosensitization in Locoregionally Advanced Head and Neck Squamous Cell Carcinoma.
Identification of genomic and molecular traits that present therapeutic vulnerability to HGF-targeted therapy in glioblastoma.
Genes co-amplified with ERBB2 or MET as novel potential cancer-promoting genes in gastric cancer.
Development of a Novel c-MET-Based CTC Detection Platform.
Defective DNA single-strand break repair is responsible for senescence and neoplastic escape of epithelial cells.
H19 non coding RNA-derived miR-675 enhances tumorigenesis and metastasis of breast cancer cells by downregulating c-Cbl and Cbl-b.
MET is required for the recruitment of anti-tumoural neutrophils.
c-Met is a novel tumor associated antigen for T-cell based immunotherapy against NK/T cell lymphoma.
Novel receptor tyrosine kinase targeted combination therapies for imatinib-resistant gastrointestinal stromal tumors (GIST).
Increase of MET gene copy number confers resistance to a monovalent MET antibody and establishes drug dependence.
Senescent fibroblasts enhance early skin carcinogenic events via a paracrine MMP-PAR-1 axis.
Gene silencing of c-Met leads to brain metastasis inhibitory effects.
Identification of prognostic biomarkers for glioblastomas using protein expression profiling.
Detection of hepatocyte growth factor (HGF) ligand-c-MET receptor activation in formalin-fixed paraffin embedded specimens by a novel proximity assay.
MET amplification identifies a small and aggressive subgroup of esophagogastric adenocarcinoma with evidence of responsiveness to crizotinib.
Prognostic significance of c-Met expression in glioblastomas.
The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma.
Tissue microarray analysis of hepatocyte growth factor/Met pathway components reveals a role for Met, matriptase, and hepatocyte growth factor activator inhibitor 1 in the progression of node-negative breast cancer.
Tissue microarray-based studies of patients with lymph node negative breast carcinoma show that met expression is associated with worse outcome but is not correlated with epidermal growth factor family receptors.
Hagege A, Saada-Bouzid E, Ambrosetti D, Rastoin O, Boyer J, He X, Rousset J, Montemagno C, Doyen J, Pedeutour F, Parola J, Bourget I, Luciano F, Bozec A, Cao Y, Pagès G, Dufies M
Cell reports. Medicine 2022 Sep 20;3(9):100659
Cell reports. Medicine 2022 Sep 20;3(9):100659
Alpha-fetoprotein can promote gastric cancer progression via upregulation of metastasis-associated colon cancer 1.
Mao X, Wang J, Luo F
Oncology letters 2022 Mar;23(3):84
Oncology letters 2022 Mar;23(3):84
Dimerization of kringle 1 domain from hepatocyte growth factor/scatter factor provides a potent MET receptor agonist.
de Nola G, Leclercq B, Mougel A, Taront S, Simonneau C, Forneris F, Adriaenssens E, Drobecq H, Iamele L, Dubuquoy L, Melnyk O, Gherardi E, de Jonge H, Vicogne J
Life science alliance 2022 Jul 29;5(12)
Life science alliance 2022 Jul 29;5(12)
Down-regulation of the tumor suppressor miR-34a contributes to head and neck cancer by up-regulating the MET oncogene and modulating tumor immune evasion.
Wu X, Cheng YL, Matthen M, Yoon A, Schwartz GK, Bala S, Taylor AM, Momen-Heravi F
Journal of experimental & clinical cancer research : CR 2021 Feb 17;40(1):70
Journal of experimental & clinical cancer research : CR 2021 Feb 17;40(1):70
A cysteine selenosulfide redox switch for protein chemical synthesis.
Diemer V, Ollivier N, Leclercq B, Drobecq H, Vicogne J, Agouridas V, Melnyk O
Nature communications 2020 May 22;11(1):2558
Nature communications 2020 May 22;11(1):2558
Targeting the MET Receptor Tyrosine Kinase as a Strategy for Radiosensitization in Locoregionally Advanced Head and Neck Squamous Cell Carcinoma.
Nisa L, Francica P, Giger R, Medo M, Elicin O, Friese-Hamim M, Wilm C, Stroh C, Bojaxhiu B, Quintin A, Caversaccio MD, Dettmer MS, Buchwalder M, Brodie TM, Aebersold DM, Zimmer Y, Carey TE, Medová M
Molecular cancer therapeutics 2020 Feb;19(2):614-626
Molecular cancer therapeutics 2020 Feb;19(2):614-626
Identification of genomic and molecular traits that present therapeutic vulnerability to HGF-targeted therapy in glioblastoma.
Sa JK, Kim SH, Lee JK, Cho HJ, Shin YJ, Shin H, Koo H, Kim D, Lee M, Kang W, Hong SH, Kim JY, Park YW, Song SW, Lee SJ, Joo KM, Nam DH
Neuro-oncology 2019 Feb 14;21(2):222-233
Neuro-oncology 2019 Feb 14;21(2):222-233
Genes co-amplified with ERBB2 or MET as novel potential cancer-promoting genes in gastric cancer.
Kwon MJ, Kim RN, Song K, Jeon S, Jeong HM, Kim JS, Han J, Hong S, Oh E, Choi JS, An J, Pollack JR, Choi YL, Park CK, Shin YK
Oncotarget 2017 Nov 3;8(54):92209-92226
Oncotarget 2017 Nov 3;8(54):92209-92226
Development of a Novel c-MET-Based CTC Detection Platform.
Zhang T, Boominathan R, Foulk B, Rao C, Kemeny G, Strickler JH, Abbruzzese JL, Harrison MR, Hsu DS, Healy P, Li J, Pi C, Prendergast KM, Hobbs C, Gemberling S, George DJ, Hurwitz HI, Connelly M, Garcia-Blanco MA, Armstrong AJ
Molecular cancer research : MCR 2016 Jun;14(6):539-47
Molecular cancer research : MCR 2016 Jun;14(6):539-47
Defective DNA single-strand break repair is responsible for senescence and neoplastic escape of epithelial cells.
Nassour J, Martien S, Martin N, Deruy E, Tomellini E, Malaquin N, Bouali F, Sabatier L, Wernert N, Pinte S, Gilson E, Pourtier A, Pluquet O, Abbadie C
Nature communications 2016 Jan 29;7:10399
Nature communications 2016 Jan 29;7:10399
H19 non coding RNA-derived miR-675 enhances tumorigenesis and metastasis of breast cancer cells by downregulating c-Cbl and Cbl-b.
Vennin C, Spruyt N, Dahmani F, Julien S, Bertucci F, Finetti P, Chassat T, Bourette RP, Le Bourhis X, Adriaenssens E
Oncotarget 2015 Oct 6;6(30):29209-23
Oncotarget 2015 Oct 6;6(30):29209-23
MET is required for the recruitment of anti-tumoural neutrophils.
Finisguerra V, Di Conza G, Di Matteo M, Serneels J, Costa S, Thompson AA, Wauters E, Walmsley S, Prenen H, Granot Z, Casazza A, Mazzone M
Nature 2015 Jun 18;522(7556):349-53
Nature 2015 Jun 18;522(7556):349-53
c-Met is a novel tumor associated antigen for T-cell based immunotherapy against NK/T cell lymphoma.
Kumai T, Matsuda Y, Ohkuri T, Oikawa K, Ishibashi K, Aoki N, Kimura S, Harabuchi Y, Celis E, Kobayashi H
Oncoimmunology 2015 Feb;4(2):e976077
Oncoimmunology 2015 Feb;4(2):e976077
Novel receptor tyrosine kinase targeted combination therapies for imatinib-resistant gastrointestinal stromal tumors (GIST).
Mahadevan D, Theiss N, Morales C, Stejskal AE, Cooke LS, Zhu M, Kurtzman D, Swart R, Ong E, Qi W
Oncotarget 2015 Feb 10;6(4):1954-66
Oncotarget 2015 Feb 10;6(4):1954-66
Increase of MET gene copy number confers resistance to a monovalent MET antibody and establishes drug dependence.
Martin V, Corso S, Comoglio PM, Giordano S
Molecular oncology 2014 Dec;8(8):1561-74
Molecular oncology 2014 Dec;8(8):1561-74
Senescent fibroblasts enhance early skin carcinogenic events via a paracrine MMP-PAR-1 axis.
Malaquin N, Vercamer C, Bouali F, Martien S, Deruy E, Wernert N, Chwastyniak M, Pinet F, Abbadie C, Pourtier A
PloS one 2013;8(5):e63607
PloS one 2013;8(5):e63607
Gene silencing of c-Met leads to brain metastasis inhibitory effects.
Lee SJ, Seol HJ, Lee HW, Kang WY, Kang BG, Jin J, Jo MY, Jin Y, Lee JI, Joo KM, Nam DH
Clinical & experimental metastasis 2013 Oct;30(7):845-54
Clinical & experimental metastasis 2013 Oct;30(7):845-54
Identification of prognostic biomarkers for glioblastomas using protein expression profiling.
Jung Y, Joo KM, Seong DH, Choi YL, Kong DS, Kim Y, Kim MH, Jin J, Suh YL, Seol HJ, Shin CS, Lee JI, Kim JH, Song SY, Nam DH
International journal of oncology 2012 Apr;40(4):1122-32
International journal of oncology 2012 Apr;40(4):1122-32
Detection of hepatocyte growth factor (HGF) ligand-c-MET receptor activation in formalin-fixed paraffin embedded specimens by a novel proximity assay.
Dua R, Zhang J, Parry G, Penuel E
PloS one 2011 Jan 21;6(1):e15932
PloS one 2011 Jan 21;6(1):e15932
MET amplification identifies a small and aggressive subgroup of esophagogastric adenocarcinoma with evidence of responsiveness to crizotinib.
Lennerz JK, Kwak EL, Ackerman A, Michael M, Fox SB, Bergethon K, Lauwers GY, Christensen JG, Wilner KD, Haber DA, Salgia R, Bang YJ, Clark JW, Solomon BJ, Iafrate AJ
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2011 Dec 20;29(36):4803-10
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2011 Dec 20;29(36):4803-10
Prognostic significance of c-Met expression in glioblastomas.
Kong DS, Song SY, Kim DH, Joo KM, Yoo JS, Koh JS, Dong SM, Suh YL, Lee JI, Park K, Kim JH, Nam DH
Cancer 2009 Jan 1;115(1):140-8
Cancer 2009 Jan 1;115(1):140-8
The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma.
Seiwert TY, Jagadeeswaran R, Faoro L, Janamanchi V, Nallasura V, El Dinali M, Yala S, Kanteti R, Cohen EE, Lingen MW, Martin L, Krishnaswamy S, Klein-Szanto A, Christensen JG, Vokes EE, Salgia R
Cancer research 2009 Apr 1;69(7):3021-31
Cancer research 2009 Apr 1;69(7):3021-31
Tissue microarray analysis of hepatocyte growth factor/Met pathway components reveals a role for Met, matriptase, and hepatocyte growth factor activator inhibitor 1 in the progression of node-negative breast cancer.
Kang JY, Dolled-Filhart M, Ocal IT, Singh B, Lin CY, Dickson RB, Rimm DL, Camp RL
Cancer research 2003 Mar 1;63(5):1101-5
Cancer research 2003 Mar 1;63(5):1101-5
Tissue microarray-based studies of patients with lymph node negative breast carcinoma show that met expression is associated with worse outcome but is not correlated with epidermal growth factor family receptors.
Tolgay Ocal I, Dolled-Filhart M, D'Aquila TG, Camp RL, Rimm DL
Cancer 2003 Apr 15;97(8):1841-8
Cancer 2003 Apr 15;97(8):1841-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
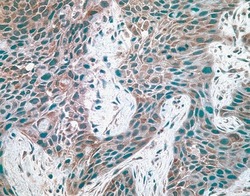
- Experimental details
- Immunohistochemical staining of breast carcinoma tissue using mouse anti-c-Met monoclonal antibody (clone 3D4) (Product # 37-0100).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
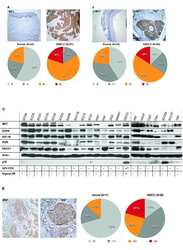
- Figure 2 Classification of 56 patients by prognosis based on 108 protein expression values. (A) Boxplot of protein expression values before (left) and after (right) quantile normalization. The plot shows the quantile normalized distribution of protein expression values for each patient. Horizontal axis represents individual patients. Vertical axis represents H-score. (B) Heatmap and dendrogram as a result of hierarchical clustering of GBM samples. Top dendrogram represents clustering of patients. Left dendrogram represents clustering of proteins. Of two patient branches, samples in the left branch represent cluster 1 and samples in the right branch represent cluster 2, consisting of 19 and 37 patients, respectively. (C) Univariate survival analysis of overall survival by Kaplan-Meier method. Kaplan-Meier survival plot of the two clusters of patients defined by the hierarchical clustering. Cluster 1 is the poor survival group. The log-rank test shows that the difference between two curves is significant (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
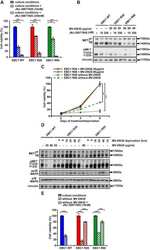
- Fig. 6 Folding and biological activity of biotinylated K1 cyclic analogs. a Primary and tertiary structure of HGF/SF K1 domain (pdb entry 1BHT). b Biotinylated K1 analogs tested for their capacity to bind MET receptor and induce MET-specific phenotypes. The pattern of disulfide bonds determined experimentally corresponds to the native pattern found in K1 domain X-ray crystal structures. c LC-MS monitoring of the folding of cK1-1 peptide into cK1-1f . d Competitive AlphaScreen (r) assay with recombinant NK1 protein. K1B or cK1-1f or cK1-2f were mixed with increasing concentrations of NK1 and with extracellular MET domain fused with human IgG1-Fc (MET-Fc) and incubated with streptavidin AlphaScreen (r) donor beads and Protein A acceptor beads. Data are presented as normalized percentage of maximal expected signal, i.e. without NK1 competition. Error bars represent the standard deviation (SD) of technical replicates ( n = 3). e MET phosphorylation assay. HeLa cells were treated for 10 min with 300 pM mature HGF/SF (HGF), or with 10 nM/100 nM K1/S , cK1-1f/S , and cK1-2f/S . Cell lysates were then analyzed by specific total MET and ERK or phospho-MET, phospho-Akt and phospho-ERK western blot. Total MET and ERK were used as loading controls after membrane stripping and re-probing. This western blot is representative of two independent experiments ( n = 2). f Cell scattering assay. Capan isolated cell islets were incubated for 18 h in culture media with 300 pM mature HGF/SF (HGF),
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure S5. MDCK cell morphogenesis assay and signalling pathway activation using K1K1, K1K1S2, and K1K1S4 mutants. (A) MDCK cell (~500) were seeded into a thick layer of a 1:1 collagen/Growth Factor Reduced Matrigel and treated with semi-log dilutions (100 pM to 300 nM) of hepatocyte growth factor/scatter factor, K1K1, K1K1S2, and K1K1S4 twice a week for 1 mo. The cells were fixed and stained with Evans blue and DAPI. The image of the full plate is presented. (B) Colonies were observed in bright field using inverted microscope observed 4x objective on a Nikon Eclipse TS100 microscope. (C) Phosphorylation analysis of MET signalling pathway by Western blot on HeLa cell lysates after stimulation with semi-log dilution (0.3-30 nM) of K1K1, K1K1S2, or K1KS4 for 10 min. Loading controls are based on total MET, total Akt, and total ERK present in each lane. Source data are available for this figure. Source Data for Figure S5
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
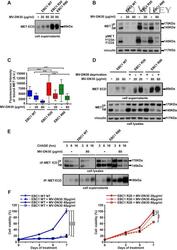
- Experimental details
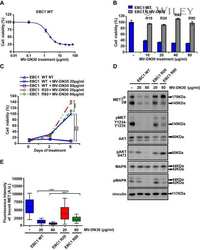
- Resistance is not due to loss of cell responsiveness to MV-DN30. (A) Western blot of MET ECD in cell supernatants of EBC1 WT, R20 and R80 cells in the absence or in the presence of MV-DN30 for 24 h. (B) Western blot revealing MET protein (top) and MET tyrosine phosphorylation (Y1234-Y1235) (middle) levels in EBC1 WT, R20 and R80 cells grown either in the absence (24 h deprivation) or in the presence (24 h) of MV-DN30. Vinculin (bottom) was used as loading control. (C) Box-plot of Fluorescence intensity of plasma-membrane bound MET (A.U. : Arbitrary Units) in EBC1 WT, R20 and R80 cells, either in the absence (24 h deprivation) or in the presence (24 h) of MV-DN30. Plasma-membrane staining as in Fig. 1E. (D) Western blot of MET ECD (top) and total MET protein levels (middle) in EBC1 WT, R20 and R80 cells upon MV-DN30 treatment (24 h). Resistant cells were antibody-deprivated for 24 h before treatment. Vinculin (bottom) was used as loading control. (E) Immunoprecipitation assays of MET (from cell lysates, top) and of MET ECD (from cell supernatants, bottom) upon Pulse-Chase metabolic protein labeling in EBC1 WT and R80 cells, in the absence or in the presence of MV-DN30. (F) Viability of EBC1 WT (left graph) and R20 cells (right graph) in the presence of increasing concentrations of MV-DN30. Results are normalized to WT untreated cells (100%) (left graph) or to R20 cells treated with 20 mug/mL (100%) (right graph) +- s.d. (***P < 0.001, *P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
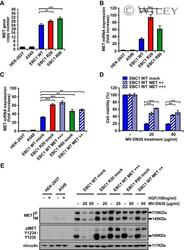
- Experimental details
- MV-DN30 resistant cells display increased MET synthesis. (A) Real-Time qPCR analysis of MET gene copy number in EBC1 WT, R20 and R80 cells. MET copy number +- s.d. (**P < 0.01; ***P < 0.001) was normalized to diploid control cells (HEK-293T and A549 cells). (B) Real-time qPCR of MET mRNA expression level in EBC1 WT, R20 and R80. (C) Real-time qPCR of MET mRNA level in EBC1 WT cells transduced with a control mock vector or with two different doses (MET++ : white oblique lines and MET+++: white squares) of a MET encoding lentiviral vector and in R20 and R80 cells transduced with the control vector alone (mock). (B, C) MET mRNA fold increase +- s.d. (**P < 0.01; ***P < 0.001) was normalized to that of normo-expressing cells (HEK-293T and A549 cells). (D) Viability of EBC1 WT cells transduced with different amounts of MET cDNA or mock vector, kept in the presence or in the absence of MV-DN30, was evaluated 72 h after treatment. Results are normalized with the untreated cells (100%) +- s.d. (**P < 0.01; ***P < 0.001). (E) Western blot showing MET protein and MET Tyrosine phosphorylation levels of transduced WT, R20 and R80 EBC1 cells kept for 24 h in the presence or in the absence of the indicated MV-DN30 concentrations. A549 and HEK-293T cells were stimulated with HGF (100 ng/mL) as a comparison for physiological levels of MET phosphorylation.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunohistochemistry
Immunohistochemistry