Antibody data
- Antibody Data
- Antigen structure
- References [25]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-7052-81 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-5 Monoclonal Antibody (TRFK5), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The TRFK5 antibody reacts with mouse and human interleukin-5 (IL-5). The TRFK5 antibody is a neutralizing antibody. Mouse IL-5 is a disulfide-linked homodimer, containing two 113 amino acid peptides; these resolve as 32-34 kDa bands in SDS-PAGE. IL-5 is produced by T cells and has been known as eosinophil-differentiating factor (EDF), B cell growth factor II (BCGFII), and T cell-replacing factor (TRF). IL-5 induces eosinophil differentiation and promotes eosinophil survival and activation. In mice, IL-5 has been shown to stimulate B cell proliferation and antibody production.
- Conjugate
- Yellow dye
- Antibody clone number
- TRFK5
- Concentration
- 0.2 mg/mL
Submitted references Lysosomal Acid Lipase Is Required for Donor T Cells to Induce Graft-versus-Host Disease.
CD4(+) T Cells Cross-Reactive with Dengue and Zika Viruses Protect against Zika Virus Infection.
Ovalbumin induces natural killer cells to secrete Th2 cytokines IL‑5 and IL‑13 in a mouse model of asthma.
An Id2(RFP)-Reporter Mouse Redefines Innate Lymphoid Cell Precursor Potentials.
Dephosphorylated Polymerase I and Transcript Release Factor Prevents Allergic Asthma Exacerbations by Limiting IL-33 Release.
Bcl11b is essential for licensing Th2 differentiation during helminth infection and allergic asthma.
Toxoplasma Co-infection Prevents Th2 Differentiation and Leads to a Helminth-Specific Th1 Response.
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Water-soluble chitosan inhibits nerve growth factor and attenuates allergic inflammation in mite allergen-induced allergic rhinitis.
Intestinal helminth infection induces highly functional resident memory CD4(+) T cells in mice.
Transcription Factors Oct-1 and GATA-3 Cooperatively Regulate Th2 Cytokine Gene Expression via the RHS5 within the Th2 Locus Control Region.
Characterization and Quantification of Innate Lymphoid Cell Subsets in Human Lung.
Lack of autophagy induces steroid-resistant airway inflammation.
The IL-10 polarized cytokine pattern in innate and adaptive immunity cells contribute to the development of FVIII inhibitors.
TNF superfamily member TL1A elicits type 2 innate lymphoid cells at mucosal barriers.
Thymic stromal lymphopoietin induces corticosteroid resistance in natural helper cells during airway inflammation.
Group V secretory phospholipase A2 is involved in macrophage activation and is sufficient for macrophage effector functions in allergic pulmonary inflammation.
The acute environment, rather than T cell subset pre-commitment, regulates expression of the human T cell cytokine amphiregulin.
Lack of IL-4 receptor expression on T helper cells reduces T helper 2 cell polyfunctionality and confers resistance in allergic bronchopulmonary mycosis.
Donor B cells in transplants augment clonal expansion and survival of pathogenic CD4+ T cells that mediate autoimmune-like chronic graft-versus-host disease.
Transcription factor RORα is critical for nuocyte development.
Bronchial epithelial cells produce IL-5: implications for local immune responses in the airways.
Pancreatic lipase-related protein 2 (PLRP2) induction by IL-4 in cytotoxic T lymphocytes (CTLs) and reevaluation of the negative effects of its gene ablation on cytotoxicity.
Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples.
The characterization of four monoclonal antibodies specific for mouse IL-5 and development of mouse and human IL-5 enzyme-linked immunosorbent.
Nguyen HD, Ticer T, Bastian D, Kuril S, Li H, Du H, Yan C, Yu XZ
Cell reports 2020 Oct 27;33(4):108316
Cell reports 2020 Oct 27;33(4):108316
CD4(+) T Cells Cross-Reactive with Dengue and Zika Viruses Protect against Zika Virus Infection.
Wen J, Wang YT, Valentine KM, Dos Santos Alves RP, Xu Z, Regla-Nava JA, Ngono AE, Young MP, Ferreira LCS, Shresta S
Cell reports 2020 Apr 28;31(4):107566
Cell reports 2020 Apr 28;31(4):107566
Ovalbumin induces natural killer cells to secrete Th2 cytokines IL‑5 and IL‑13 in a mouse model of asthma.
Chen Z, Wang L
Molecular medicine reports 2019 Apr;19(4):3210-3216
Molecular medicine reports 2019 Apr;19(4):3210-3216
An Id2(RFP)-Reporter Mouse Redefines Innate Lymphoid Cell Precursor Potentials.
Xu W, Cherrier DE, Chea S, Vosshenrich C, Serafini N, Petit M, Liu P, Golub R, Di Santo JP
Immunity 2019 Apr 16;50(4):1054-1068.e3
Immunity 2019 Apr 16;50(4):1054-1068.e3
Dephosphorylated Polymerase I and Transcript Release Factor Prevents Allergic Asthma Exacerbations by Limiting IL-33 Release.
Ni Y, Hao J, Hou X, Du W, Yu Y, Chen T, Wei Z, Li Y, Zhu F, Wang S, Liang R, Li D, Lu Y, Liao K, Li B, Shi G
Frontiers in immunology 2018;9:1422
Frontiers in immunology 2018;9:1422
Bcl11b is essential for licensing Th2 differentiation during helminth infection and allergic asthma.
Lorentsen KJ, Cho JJ, Luo X, Zuniga AN, Urban JF Jr, Zhou L, Gharaibeh R, Jobin C, Kladde MP, Avram D
Nature communications 2018 Apr 26;9(1):1679
Nature communications 2018 Apr 26;9(1):1679
Toxoplasma Co-infection Prevents Th2 Differentiation and Leads to a Helminth-Specific Th1 Response.
Ahmed N, French T, Rausch S, Kühl A, Hemminger K, Dunay IR, Steinfelder S, Hartmann S
Frontiers in cellular and infection microbiology 2017;7:341
Frontiers in cellular and infection microbiology 2017;7:341
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Wang S, Xia P, Chen Y, Qu Y, Xiong Z, Ye B, Du Y, Tian Y, Yin Z, Xu Z, Fan Z
Cell 2017 Sep 21;171(1):201-216.e18
Cell 2017 Sep 21;171(1):201-216.e18
Water-soluble chitosan inhibits nerve growth factor and attenuates allergic inflammation in mite allergen-induced allergic rhinitis.
Chen PC, Hsieh MH, Kuo WS, Kao HF, Hsu CL, Wang JY
The Journal of allergy and clinical immunology 2017 Oct;140(4):1146-1149.e8
The Journal of allergy and clinical immunology 2017 Oct;140(4):1146-1149.e8
Intestinal helminth infection induces highly functional resident memory CD4(+) T cells in mice.
Steinfelder S, Rausch S, Michael D, Kühl AA, Hartmann S
European journal of immunology 2017 Feb;47(2):353-363
European journal of immunology 2017 Feb;47(2):353-363
Transcription Factors Oct-1 and GATA-3 Cooperatively Regulate Th2 Cytokine Gene Expression via the RHS5 within the Th2 Locus Control Region.
Kim K, Kim N, Lee GR
PloS one 2016;11(2):e0148576
PloS one 2016;11(2):e0148576
Characterization and Quantification of Innate Lymphoid Cell Subsets in Human Lung.
De Grove KC, Provoost S, Verhamme FM, Bracke KR, Joos GF, Maes T, Brusselle GG
PloS one 2016;11(1):e0145961
PloS one 2016;11(1):e0145961
Lack of autophagy induces steroid-resistant airway inflammation.
Suzuki Y, Maazi H, Sankaranarayanan I, Lam J, Khoo B, Soroosh P, Barbers RG, James Ou JH, Jung JU, Akbari O
The Journal of allergy and clinical immunology 2016 May;137(5):1382-1389.e9
The Journal of allergy and clinical immunology 2016 May;137(5):1382-1389.e9
The IL-10 polarized cytokine pattern in innate and adaptive immunity cells contribute to the development of FVIII inhibitors.
Silveira AC, Santana MA, Ribeiro IG, Chaves DG, Martins-Filho OA
BMC hematology 2015;15(1):1
BMC hematology 2015;15(1):1
TNF superfamily member TL1A elicits type 2 innate lymphoid cells at mucosal barriers.
Yu X, Pappu R, Ramirez-Carrozzi V, Ota N, Caplazi P, Zhang J, Yan D, Xu M, Lee WP, Grogan JL
Mucosal immunology 2014 May;7(3):730-40
Mucosal immunology 2014 May;7(3):730-40
Thymic stromal lymphopoietin induces corticosteroid resistance in natural helper cells during airway inflammation.
Kabata H, Moro K, Fukunaga K, Suzuki Y, Miyata J, Masaki K, Betsuyaku T, Koyasu S, Asano K
Nature communications 2013;4:2675
Nature communications 2013;4:2675
Group V secretory phospholipase A2 is involved in macrophage activation and is sufficient for macrophage effector functions in allergic pulmonary inflammation.
Ohta S, Imamura M, Xing W, Boyce JA, Balestrieri B
Journal of immunology (Baltimore, Md. : 1950) 2013 Jun 15;190(12):5927-38
Journal of immunology (Baltimore, Md. : 1950) 2013 Jun 15;190(12):5927-38
The acute environment, rather than T cell subset pre-commitment, regulates expression of the human T cell cytokine amphiregulin.
Qi Y, Operario DJ, Georas SN, Mosmann TR
PloS one 2012;7(6):e39072
PloS one 2012;7(6):e39072
Lack of IL-4 receptor expression on T helper cells reduces T helper 2 cell polyfunctionality and confers resistance in allergic bronchopulmonary mycosis.
Müller U, Piehler D, Stenzel W, Köhler G, Frey O, Held J, Grahnert A, Richter T, Eschke M, Kamradt T, Brombacher F, Alber G
Mucosal immunology 2012 May;5(3):299-310
Mucosal immunology 2012 May;5(3):299-310
Donor B cells in transplants augment clonal expansion and survival of pathogenic CD4+ T cells that mediate autoimmune-like chronic graft-versus-host disease.
Young JS, Wu T, Chen Y, Zhao D, Liu H, Yi T, Johnston H, Racine J, Li X, Wang A, Todorov I, Zeng D
Journal of immunology (Baltimore, Md. : 1950) 2012 Jul 1;189(1):222-33
Journal of immunology (Baltimore, Md. : 1950) 2012 Jul 1;189(1):222-33
Transcription factor RORα is critical for nuocyte development.
Wong SH, Walker JA, Jolin HE, Drynan LF, Hams E, Camelo A, Barlow JL, Neill DR, Panova V, Koch U, Radtke F, Hardman CS, Hwang YY, Fallon PG, McKenzie AN
Nature immunology 2012 Jan 22;13(3):229-36
Nature immunology 2012 Jan 22;13(3):229-36
Bronchial epithelial cells produce IL-5: implications for local immune responses in the airways.
Wu CA, Peluso JJ, Zhu L, Lingenheld EG, Walker ST, Puddington L
Cellular immunology 2010;264(1):32-41
Cellular immunology 2010;264(1):32-41
Pancreatic lipase-related protein 2 (PLRP2) induction by IL-4 in cytotoxic T lymphocytes (CTLs) and reevaluation of the negative effects of its gene ablation on cytotoxicity.
Alves BN, Leong J, Tamang DL, Elliott V, Edelnant J, Redelman D, Singer CA, Kuhn AR, Miller R, Lowe ME, Hudig D
Journal of leukocyte biology 2009 Sep;86(3):701-12
Journal of leukocyte biology 2009 Sep;86(3):701-12
Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples.
Abrams JS, Roncarolo MG, Yssel H, Andersson U, Gleich GJ, Silver JE
Immunological reviews 1992 Jun;127:5-24
Immunological reviews 1992 Jun;127:5-24
The characterization of four monoclonal antibodies specific for mouse IL-5 and development of mouse and human IL-5 enzyme-linked immunosorbent.
Schumacher JH, O'Garra A, Shrader B, van Kimmenade A, Bond MW, Mosmann TR, Coffman RL
Journal of immunology (Baltimore, Md. : 1950) 1988 Sep 1;141(5):1576-81
Journal of immunology (Baltimore, Md. : 1950) 1988 Sep 1;141(5):1576-81
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
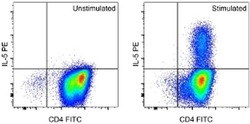
- Experimental details
- Normal human peripheral blood cells were polarized under Th2 conditions and then unstimulated (left) or stimulated for 5 hours with Cell Stimulation Cocktail (Product # 00-4970-03) in the presence of Brefeldin A Solution (Product # 00-4506-51) (right). Cells were intracellularly stained with Anti-Human CD4 FITC (Product # 11-0048-42) and 0.125 µg of Anti-Human/Mouse IL-5 PE using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00) and protocol. Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Frequencies of CD4 + T-cells expressing Gata3 and Th2 cytokines in asthma and helminth infection are reduced in Bcl11b fl/fl dLck-iCre mice by an intrinsic defect. a Flow cytometry analysis of IL-4 (left column), IL-5 (middle column), and IL-13 (right column) against GATA3 in the CD4 + T-cell population of the lung parenchyma of HDM asthma-induced mice. b Frequencies of IL-4 + ( n = 4), IL-5 + ( n = 4), and IL-13 + ( n = 8) (top panel) and GATA3 + ( n = 10) (bottom panel) CD4 + T-cells from the lungs of the indicated groups. Data are derived from two (IL-4, IL-5, and IL-13 data) or five (GATA3 data) independent experiments. c Flow cytometry analysis of IL-4 (left column), IL-5 (middle column), and IL-13 (right column) against GATA3 in the CD4 + T-cell population from the mesenteric (m)LNs of H. polygyrus infected mice. d Frequencies of IL-4 + ( n = 4), IL-5 + ( n = 4), and IL-13 + ( n = 4) (top panel) and GATA3 + ( n = 8) (bottom panel) CD4 + T-cells from mLNs of the indicated groups. Data are derived from two (IL-4, IL-5, and IL-13) or four (GATA3) independent experiments. e Flow cytometry analysis of GATA3 in CD4 + T-cells (left column) and IL-4 (middle and right columns) in the GATA3 hi (middle column) and GATA3 lo (right column) CD4 + T-cells from the mLNs of H. polygyrus bakeri infected mice. f The ratio of GATA3 hi /GATA3 lo CD4 + T-cells (top) and frequencies of IL-4 secreting cells within the GATA3 hi and GATA3 lo CD4 + T-cells (bottom) from the mLNs of H. poly
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry