Antibody data
- Antibody Data
- Antigen structure
- References [28]
- Comments [0]
- Validations
- Immunocytochemistry [4]
- Other assay [24]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 33-8800 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Ankyrin G Monoclonal Antibody (4G3F8)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Canine, Rabbit
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 4G3F8
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Glucose increases the length and spacing of the lattice structure of the axon initial segment.
Differences in action potential propagation speed and axon initial segment plasticity between neurons from Sprague-Dawley rats and C57BL/6 mice.
Schwann cell nodal membrane disruption triggers bystander axonal degeneration in a Guillain-Barré syndrome mouse model.
Centrosome-mediated microtubule remodeling during axon formation in human iPSC-derived neurons.
Intrinsic Morphologic and Physiologic Development of Human Derived Retinal Ganglion Cells In Vitro.
Quantitative mapping of transcriptome and proteome dynamics during polarization of human iPSC-derived neurons.
Inhibitory Parvalbumin Basket Cell Activity is Selectively Reduced during Hippocampal Sharp Wave Ripples in a Mouse Model of Familial Alzheimer's Disease.
Neurodevelopmental mutation of giant ankyrin-G disrupts a core mechanism for axon initial segment assembly.
The cell-cell junctions of mammalian testes: II. The lamellar smooth muscle monolayer cells of the peritubular wall are laterally connected by vertical adherens junctions-a novel architectonic cell-cell junction system.
KIF2A regulates the development of dentate granule cells and postnatal hippocampal wiring.
NF-κB regulates neuronal ankyrin-G via a negative feedback loop.
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
MAP2 Defines a Pre-axonal Filtering Zone to Regulate KIF1- versus KIF5-Dependent Cargo Transport in Sensory Neurons.
Enrichment of GABAA Receptor α-Subunits on the Axonal Initial Segment Shows Regional Differences.
KCNQ Potassium Channels Modulate Sensitivity of Skin Down-hair (D-hair) Mechanoreceptors.
A system for studying mechanisms of neuromuscular junction development and maintenance.
Effects of Morphology Constraint on Electrophysiological Properties of Cortical Neurons.
Genetically engineered SCN5A mutant pig hearts exhibit conduction defects and arrhythmias.
Contactin-1 regulates myelination and nodal/paranodal domain organization in the central nervous system.
A human Dravet syndrome model from patient induced pluripotent stem cells.
Remodeling of mechanical junctions and of microtubule-associated proteins accompany cardiac connexin43 lateralization.
Regulation of the intermediate filament protein nestin at rodent neuromuscular junctions by innervation and activity.
Voltage-gated sodium channels confer excitability to human odontoblasts: possible role in tooth pain transmission.
The ammonium transporter RhBG: requirement of a tyrosine-based signal and ankyrin-G for basolateral targeting and membrane anchorage in polarized kidney epithelial cells.
Oligodendroglial modulation of fast axonal transport in a mouse model of hereditary spastic paraplegia.
Structural requirements for interaction of sodium channel beta 1 subunits with ankyrin.
ELF a beta-spectrin is a neuronal precursor cell marker in developing mammalian brain; structure and organization of the elf/beta-G spectrin gene.
Essential role of oligodendrocytes in the formation and maintenance of central nervous system nodal regions.
Wang Y, Guan M, Zhang Y, Zhanghao K, Xi P
Microscopy research and technique 2022 Jul;85(7):2679-2691
Microscopy research and technique 2022 Jul;85(7):2679-2691
Differences in action potential propagation speed and axon initial segment plasticity between neurons from Sprague-Dawley rats and C57BL/6 mice.
Chen ZY, Peng L, Zhao M, Li Y, Takahiko M, Tao L, Zou P, Zhang Y
Zoological research 2022 Jul 18;43(4):615-633
Zoological research 2022 Jul 18;43(4):615-633
Schwann cell nodal membrane disruption triggers bystander axonal degeneration in a Guillain-Barré syndrome mouse model.
McGonigal R, Campbell CI, Barrie JA, Yao D, Cunningham ME, Crawford CL, Rinaldi S, Rowan EG, Willison HJ
The Journal of clinical investigation 2022 Jul 15;132(14)
The Journal of clinical investigation 2022 Jul 15;132(14)
Centrosome-mediated microtubule remodeling during axon formation in human iPSC-derived neurons.
Lindhout FW, Portegies S, Kooistra R, Herstel LJ, Stucchi R, Hummel JJA, Scheefhals N, Katrukha EA, Altelaar M, MacGillavry HD, Wierenga CJ, Hoogenraad CC
The EMBO journal 2021 May 17;40(10):e106798
The EMBO journal 2021 May 17;40(10):e106798
Intrinsic Morphologic and Physiologic Development of Human Derived Retinal Ganglion Cells In Vitro.
Risner ML, Pasini S, Chamling X, McGrady NR, Goldberg JL, Zack DJ, Calkins DJ
Translational vision science & technology 2021 Aug 12;10(10):1
Translational vision science & technology 2021 Aug 12;10(10):1
Quantitative mapping of transcriptome and proteome dynamics during polarization of human iPSC-derived neurons.
Lindhout FW, Kooistra R, Portegies S, Herstel LJ, Stucchi R, Snoek BL, Altelaar AM, MacGillavry HD, Wierenga CJ, Hoogenraad CC
eLife 2020 Sep 17;9
eLife 2020 Sep 17;9
Inhibitory Parvalbumin Basket Cell Activity is Selectively Reduced during Hippocampal Sharp Wave Ripples in a Mouse Model of Familial Alzheimer's Disease.
Caccavano A, Bozzelli PL, Forcelli PA, Pak DTS, Wu JY, Conant K, Vicini S
The Journal of neuroscience : the official journal of the Society for Neuroscience 2020 Jun 24;40(26):5116-5136
The Journal of neuroscience : the official journal of the Society for Neuroscience 2020 Jun 24;40(26):5116-5136
Neurodevelopmental mutation of giant ankyrin-G disrupts a core mechanism for axon initial segment assembly.
Yang R, Walder-Christensen KK, Lalani S, Yan H, García-Prieto ID, Álvarez S, Fernández-Jaén A, Speltz L, Jiang YH, Bennett V
Proceedings of the National Academy of Sciences of the United States of America 2019 Sep 24;116(39):19717-19726
Proceedings of the National Academy of Sciences of the United States of America 2019 Sep 24;116(39):19717-19726
The cell-cell junctions of mammalian testes: II. The lamellar smooth muscle monolayer cells of the peritubular wall are laterally connected by vertical adherens junctions-a novel architectonic cell-cell junction system.
Domke LM, Franke WW
Cell and tissue research 2019 Feb;375(2):451-482
Cell and tissue research 2019 Feb;375(2):451-482
KIF2A regulates the development of dentate granule cells and postnatal hippocampal wiring.
Homma N, Zhou R, Naseer MI, Chaudhary AG, Al-Qahtani MH, Hirokawa N
eLife 2018 Jan 9;7
eLife 2018 Jan 9;7
NF-κB regulates neuronal ankyrin-G via a negative feedback loop.
König HG, Schwamborn R, Andresen S, Kinsella S, Watters O, Fenner B, Prehn JH
Scientific reports 2017 Feb 9;7:42006
Scientific reports 2017 Feb 9;7:42006
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
O'Hare Doig RL, Chiha W, Giacci MK, Yates NJ, Bartlett CA, Smith NM, Hodgetts SI, Harvey AR, Fitzgerald M
BMC neuroscience 2017 Aug 14;18(1):62
BMC neuroscience 2017 Aug 14;18(1):62
MAP2 Defines a Pre-axonal Filtering Zone to Regulate KIF1- versus KIF5-Dependent Cargo Transport in Sensory Neurons.
Gumy LF, Katrukha EA, Grigoriev I, Jaarsma D, Kapitein LC, Akhmanova A, Hoogenraad CC
Neuron 2017 Apr 19;94(2):347-362.e7
Neuron 2017 Apr 19;94(2):347-362.e7
Enrichment of GABAA Receptor α-Subunits on the Axonal Initial Segment Shows Regional Differences.
Gao Y, Heldt SA
Frontiers in cellular neuroscience 2016;10:39
Frontiers in cellular neuroscience 2016;10:39
KCNQ Potassium Channels Modulate Sensitivity of Skin Down-hair (D-hair) Mechanoreceptors.
Schütze S, Orozco IJ, Jentsch TJ
The Journal of biological chemistry 2016 Mar 11;291(11):5566-5575
The Journal of biological chemistry 2016 Mar 11;291(11):5566-5575
A system for studying mechanisms of neuromuscular junction development and maintenance.
Vilmont V, Cadot B, Ouanounou G, Gomes ER
Development (Cambridge, England) 2016 Jul 1;143(13):2464-77
Development (Cambridge, England) 2016 Jul 1;143(13):2464-77
Effects of Morphology Constraint on Electrophysiological Properties of Cortical Neurons.
Zhu G, Du L, Jin L, Offenhäusser A
Scientific reports 2016 Apr 7;6:23086
Scientific reports 2016 Apr 7;6:23086
Genetically engineered SCN5A mutant pig hearts exhibit conduction defects and arrhythmias.
Park DS, Cerrone M, Morley G, Vasquez C, Fowler S, Liu N, Bernstein SA, Liu FY, Zhang J, Rogers CS, Priori SG, Chinitz LA, Fishman GI
The Journal of clinical investigation 2015 Jan;125(1):403-12
The Journal of clinical investigation 2015 Jan;125(1):403-12
Contactin-1 regulates myelination and nodal/paranodal domain organization in the central nervous system.
Çolakoğlu G, Bergstrom-Tyrberg U, Berglund EO, Ranscht B
Proceedings of the National Academy of Sciences of the United States of America 2014 Jan 21;111(3):E394-403
Proceedings of the National Academy of Sciences of the United States of America 2014 Jan 21;111(3):E394-403
A human Dravet syndrome model from patient induced pluripotent stem cells.
Higurashi N, Uchida T, Lossin C, Misumi Y, Okada Y, Akamatsu W, Imaizumi Y, Zhang B, Nabeshima K, Mori MX, Katsurabayashi S, Shirasaka Y, Okano H, Hirose S
Molecular brain 2013 May 2;6:19
Molecular brain 2013 May 2;6:19
Remodeling of mechanical junctions and of microtubule-associated proteins accompany cardiac connexin43 lateralization.
Chkourko HS, Guerrero-Serna G, Lin X, Darwish N, Pohlmann JR, Cook KE, Martens JR, Rothenberg E, Musa H, Delmar M
Heart rhythm 2012 Jul;9(7):1133-1140.e6
Heart rhythm 2012 Jul;9(7):1133-1140.e6
Regulation of the intermediate filament protein nestin at rodent neuromuscular junctions by innervation and activity.
Kang H, Tian L, Son YJ, Zuo Y, Procaccino D, Love F, Hayworth C, Trachtenberg J, Mikesh M, Sutton L, Ponomareva O, Mignone J, Enikolopov G, Rimer M, Thompson W
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 May 30;27(22):5948-57
The Journal of neuroscience : the official journal of the Society for Neuroscience 2007 May 30;27(22):5948-57
Voltage-gated sodium channels confer excitability to human odontoblasts: possible role in tooth pain transmission.
Allard B, Magloire H, Couble ML, Maurin JC, Bleicher F
The Journal of biological chemistry 2006 Sep 29;281(39):29002-10
The Journal of biological chemistry 2006 Sep 29;281(39):29002-10
The ammonium transporter RhBG: requirement of a tyrosine-based signal and ankyrin-G for basolateral targeting and membrane anchorage in polarized kidney epithelial cells.
Lopez C, Métral S, Eladari D, Drevensek S, Gane P, Chambrey R, Bennett V, Cartron JP, Le Van Kim C, Colin Y
The Journal of biological chemistry 2005 Mar 4;280(9):8221-8
The Journal of biological chemistry 2005 Mar 4;280(9):8221-8
Oligodendroglial modulation of fast axonal transport in a mouse model of hereditary spastic paraplegia.
Edgar JM, McLaughlin M, Yool D, Zhang SC, Fowler JH, Montague P, Barrie JA, McCulloch MC, Duncan ID, Garbern J, Nave KA, Griffiths IR
The Journal of cell biology 2004 Jul 5;166(1):121-31
The Journal of cell biology 2004 Jul 5;166(1):121-31
Structural requirements for interaction of sodium channel beta 1 subunits with ankyrin.
Malhotra JD, Koopmann MC, Kazen-Gillespie KA, Fettman N, Hortsch M, Isom LL
The Journal of biological chemistry 2002 Jul 19;277(29):26681-8
The Journal of biological chemistry 2002 Jul 19;277(29):26681-8
ELF a beta-spectrin is a neuronal precursor cell marker in developing mammalian brain; structure and organization of the elf/beta-G spectrin gene.
Tang Y, Katuri V, Iqbal S, Narayan T, Wang Z, Lu RS, Mishra L, Mishra B
Oncogene 2002 Aug 8;21(34):5255-67
Oncogene 2002 Aug 8;21(34):5255-67
Essential role of oligodendrocytes in the formation and maintenance of central nervous system nodal regions.
Mathis C, Denisenko-Nehrbass N, Girault JA, Borrelli E
Development (Cambridge, England) 2001 Dec;128(23):4881-90
Development (Cambridge, England) 2001 Dec;128(23):4881-90
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
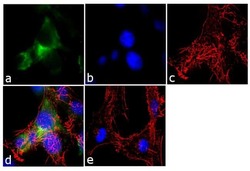
- Experimental details
- Immunofluorescence analysis of Ankyrin G was performed using 70% confluent log phase U-87 MG cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Ankyrin G (4G3F8) Mouse Monoclonal Antibody (Product # 33-8800) at 2µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
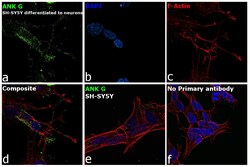
- Experimental details
- Immunofluorescence analysis of Ankyrin G was performed using 70% confluent log phase SH-SY5Y cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Ankyrin G Monoclonal Antibody (4G3F8) (Product # 33-8800) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing Plasma Membrane localization. Panel e represents SH-SY5Y with less expression on ANK G as compared to differentiated neurons. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
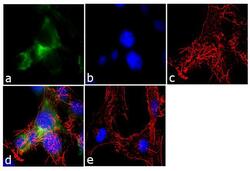
- Experimental details
- Immunofluorescence analysis of Ankyrin G was performed using 70% confluent log phase U-87 MG cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Ankyrin G (4G3F8) Mouse Monoclonal Antibody (Product # 33-8800) at 2µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
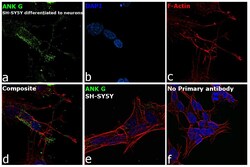
- Experimental details
- Immunofluorescence analysis of Ankyrin G was performed using 70% confluent log phase SH-SY5Y cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with Ankyrin G Monoclonal Antibody (4G3F8) (Product # 33-8800) at 5 µg/mL in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing Plasma Membrane localization. Panel e represents SH-SY5Y with less expression on ANK G as compared to differentiated neurons. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
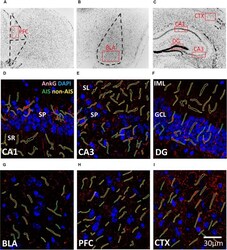
- FIGURE 1 (A-C) Representative illustration of the brain areas where immunofluorescent images were acquired from the (A) prefrontal cortex (PFC), (B) basal lateral amygdala (BLA), and (C) CXT/hippocampal regions (CA1, CA3, and DG). Areas were first located under 10x objective lens based on the contours of DAPI staining, and high magnification images were subsequently acquired using a 63x objective lens within the boundaries of each brain area (red boxes). (D-I) Examples of AIS (green) and Non-AIS (yellow) ROI contours delineated based on the AnkG (red) and DAPI (blue) confocal images of the (D) CA1, (E) CA3, (F) DG, (G) BLA, (H) PFC, and (I) CTX. Abbreviations: DG, dentate gyrus; BLA, basal lateral amygdala; PFC, prefrontal cortex; CXT, somatosensory barrel cortex; SP, stratum pyramidale; SR, stratum radiatum; SL, stratum lucidum; IML, inner molecular layer; GCL, granule cell layer.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
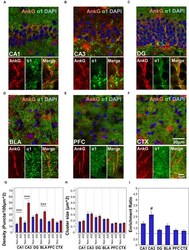
- FIGURE 2 (A-F) Immunofluorescent double-labeled montage images of AnkG (red) and alpha1-subunits (green) taken from single optical sections of the hippocampal sub-regions (CA1, CA3, DG), BLA, PFC, and CTX in C57BL/6J mice. (G,H) Average cluster density and size of alpha1-subunits on the AIS and non-AIS locations were reported as means +- SEM. Significant differences of cluster density were identified in the CA1, CA3, and the BLA, while no statistical significant difference was observed for the cluster size. Asterisks represent significant difference from the AIS and the non-AIS locations using the Bonferroni method for planned multiple comparison, *** p < 0.001. (I) Enrichment ratios of alpha1-subunits across different brain areas were reported. Tukey-Kramer post hoc pair-wise multiple comparison revealed the enrichment ratio of alpha1-subunits in the CA3 was significantly higher than the other brain areas, # p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
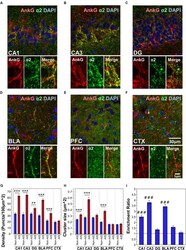
- FIGURE 3 (A-F) Immunofluorescent double-labeled montage images of AnkG (red) and alpha2-subunits (green) taken from single optical sections of the hippocampal sub-regions (CA1, CA3, DG), BLA, PFC, and CTX in C57BL/6J mice. (G,H) Average cluster density and size of alpha2-subunits on the AIS and non-AIS locations were reported as means +- SEM. Significant differences of cluster density were identified in the CA1, CA3, DG, BLA, and PFC. Significant differences of cluster size were identified in the CA1, CA3, and BLA. Asterisks represent significant difference from the AIS and the non-AIS locations using the Bonferroni method for planned multiple comparison, ** p < 0.01, *** p < 0.001. (I) Enrichment ratios of alpha2-subunits across different brain areas were reported. Tukey-Kramer post hoc pair-wise multiple comparison revealed the enrichment ratios of alpha2-subunits in the CA1, CA3, and BLA were significantly higher than the other brain areas, ### p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
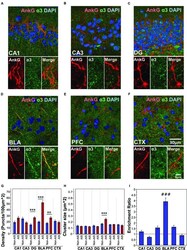
- FIGURE 4 (A-F) Immunofluorescent double-labeled montage images of AnkG (red) and alpha3-subunits (green) taken from single optical sections of the hippocampal sub-regions (CA1, CA3, DG), BLA, PFC, and CTX in C57BL/6J mice. (G,H) Average cluster density and size of alpha3-subunits on the AIS and non-AIS locations were reported as means +- SEM. Significant differences of cluster density were identified in the DG, BLA, and PFC. Significant difference of cluster size was identified in the BLA. Asterisks represent significant difference from the AIS and the non-AIS locations using the Bonferroni method for planned multiple comparison, ** p < 0.01, *** p < 0.001. (I) Enrichment ratios of alpha3-subunits across different brain areas were reported. Tukey-Kramer post hoc pair-wise multiple comparison revealed the enrichment ratio of alpha3-subunits in the BLA was significantly higher than the other brain areas, ### p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
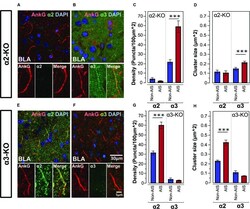
- FIGURE 5 (A,B) Immunofluorescent double-labeled montage images of AnkG (red) and alpha2- and alpha3-subunits (green) taken from single optical sections of the BLA in alpha2-KO mouse. (C,D) Average cluster density and size of the alpha2- and alpha3-subunits on the AIS and non-AIS locations in alpha2-KO were reported as means +- SEM. Asterisks represent significant difference from the AIS and the non-AIS locations using the Bonferroni method for planned multiple comparison, *** p < 0.001. (E,F) Immunofluorescent double-labeled montage images of AnkG (red) and alpha2- and alpha3-subunits (green) taken from single optical sections of the BLA in alpha3-KO mouse. (G,H) Average cluster density and size of the alpha2- and alpha3-subunits on the AIS and non-AIS locations in alpha3-KO were reported as means +- SEM. Asterisks represent significant difference from the AIS and the non-AIS locations using the Bonferroni method for planned multiple comparison, *** p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
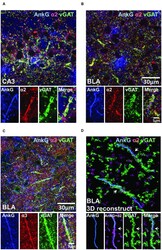
- FIGURE 6 (A-C) Immunofluorescent triple-labeled montage images of AnkG (blue), alpha2/3-subunits (red), and vGAT (green) taken from single optical sections of the CA3 and BLA in C57BL/6J mice. Prominent triple-colocalizations in all images were evident. (D) 3D-reconstructed contours of AnkG, alpha2-subunit, and vGAT from z-stack image series taken in the BLA. The arrows indicated the turning points where the vGAT-positive terminals wrap around the AnkG-positive AIS.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
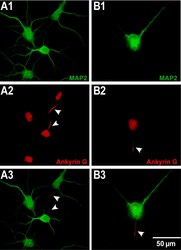
- Figure 3 The morphology of ( A ) un-patterneds and ( B ) patterned neurons. Neurons were stained by MAP2 (green) and Ankyrin G (red) to indicate dendrites and axons, respectively. Un-patterned neurons have 3 ~ 8 dendrites (A1) and one axon for each neuron (A2). A patterned neuron has 2 dendrites (B1) and one axon (B2). (A3) and (B3) are the merged fluorescent images of MAP2 (green) and Ankyrin G (red). All the axons are indicated by arrows. The scale bar represents 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
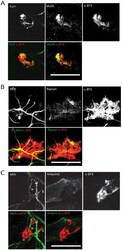
- Fig. 6. Characterization of post-synaptic specialization at Day 14. (A) Representative z -projection of differentiated myofibers stained for alpha-BTX (red) and TuJ1 or MuSK (green). (B) Representative z -projection of differentiated myofibers stained for alpha-BTX (red) and NFH or Rapsyn (green). (C) Representative image of differentiated myofibers stained for alpha-BTX (red) and NFH or ankyrin G (green). Scale bars: 20 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
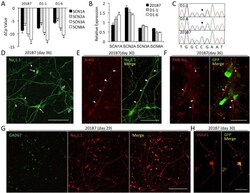
- Figure 2 Na v channel expression in iPSC-derived neurons. ( A ) Real-time PCR addressing neuronal Na v expression at 30 days of differentiation ( N = 3 in each cell line) Crossing point differences to beta-actin (DeltaCp = Cp beta-actin - Cp Nav ) closer to zero indicate higher expression. PCR efficiencies were nearly identical (Additional file 2 ). Asterisks indicate a significant difference to SCN1A ( P < 0.5, one-way ANOVA). Expression strength of the indicated Na v genes was constant across the cell lines ( P = 0.92, two-way ANOVA) ( B ) Normalized expression levels for each Na v gene ( SCN1A + SCN2A + SCN3A + SCN8A )/4 = 1. Compared to the control, SCN1A expression tended to be higher in D1-1 ( P = 0.0929, one-way ANOVA), and it was significantly higher in D1-6 ( * P = 0.0078). The distribution of Na v genes expression ratios in each cell line was significantly different between the control and the patient lines ( P =0.0086 and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
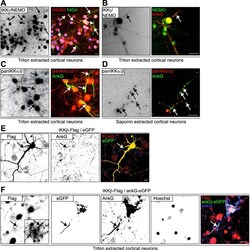
- Figure 2 The IKK-complex co-localizes with ankyrin-G to the axon initial segment. ( A - D ) Cortical neurons were cultured until maturity in vitro and were cytosol-extracted for the visualization of cytoskeletal-associated proteins. They were incubated for 5 minutes in 1% (m/V) Triton-X-100 (A-C) or 0.5% saponin (D) in 10 mM Na 3 PO 4 -buffer (pH 7.4), 1 mM MgCl 2 , 3 mM CaCl 2 , 150 mM NaCl. Extractions were performed at 4 degC, prior to fixation and immunostaining. Anti-IKKgamma/NEMO immunoreactivity is highlighted in red (A) or green (B), the AIS-specific marker 44 antibody 14D4 (green,A) or anti-AnkG (red, B) were used to identify the AIS (green). (C,D) Extractions were similarly performed followed by immunolabeling using pan-IKKalpha/beta - specific antibodies (red, rabbit polyclonal, H-470, SCBT) with the AIS labeled by anti-ankyrinG in green. Color overlays are depicted following background substraction and sharpening, arrows highlight AISs, scale bars, 10 mum. ( E ) IKKbeta-Flag and eGFP vectors were transfected into mouse cortical neurons on DIV13 using Ca-precipitation. 2 days following transfection, neurons were fixed and immunolabeled using a rabbit polyclonal anti-Flag-specific antibody (Sigma, red), as well as a mouse monoclonal ankyrin-G-specific antibody (Zymed). Note the Flag-tag-specific immunoreactivity within the ankyrin-G positive AIS stretch (arrow, yellow). ( F ) IKKbeta-Flag and ankG-eGFP vectors were transfected into mouse cortical neurons as above. 2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 The transcription factor p65/NF-kappaB localizes to the axon initial segment. ( A - D ) Mature cortical neurons were cytosol-extracted using a Triton-X100-based buffer. Extractions were performed at 4 degC. Then, neurons were either immunolabeled using a rabbit monoclonal anti-p65 (green, D14E12, CST), a rabbit monoclonal anti-cJun (green, Cell Signaling Technology) or a mouse monoclonal p65/NF-kappaB-specific antibody (green, pan-p65, F-6, SCBT) together with either mouse monoclonal (413) or rabbit polyclonal ankyrin-G-specific (H-215, both SCBT) antibodies. AIS highlighted by arrows. (D) Experiment was conducted as in C, with single AIS magnified. Note the labeling of pan-p65 immunoreactivities along the axon initial segment. Color overlay is depicted following background substraction. Scale bars, 10 mum. ( E ) p65-paGFP exhibits decreased mobility in the AIS. Following photo-activation of p65-paGFP or paGFP in DIV12-19 rat hippocampal neurons, fluorescence intensity decline was monitored. Representative time course images of photo-activated p65-paGFP or paGFP in the AIS are depicted. DsRed2 fluorescence (not shown) was used to identify the axon initial segment of transfected neurons based on morphological criteria. Times indicate time points after photo-activation. Regions marked with blue boxes were irradiated with 405 nm light to photo-activate the paGFP/p65-paGFP. Note that 1 second after photo-activation, p65-paGFP fluorescence increase is mainly confined to t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
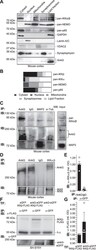
- Figure 1 Ankyrin-G and the IKK-complex accumulate in synaptosomal and lipid fractions in the adult neocortex and co-immunoprecipitate. ( A ) Fresh adult mouse neocortex tissue was isolated and separated using gradient centrifugation. Proteins were extracted from cellular fractions and analyzed by Western blotting. Note the co-localization of pan-IKKalpha/beta, NEMO and p65/NF-kappaB immunoreactivities to the ankyrin-G (clone 463, SCBT) positive lipid fraction. ( B ) Optical densities (OD) of protein immunoreactivity from all fractions were determined and mapped as percentage of total OD of all fractions in bar graphs. ( C ) Ankyrin-G and the IKK-complex co-immunoprecipitate from adult mouse cortical lysates. Mouse cortices were homogenized and minced in Na-Sucrose buffer. Equivalent amounts were used for immunoprecipitation (IP) using anti-ankyrin-G, control anti-IgG, anti-MAP2 or anti-alpha-Tubulin antisera, followed by immuno-blotting (IB) using anti-ankyrin-G (463), anti-pan-IKKalpha/beta (H-470) and anti-MAP2 antibodies. Full-blot views including spliced supernatant control bands in Suppl. Fig. 1A . ( D ) For the reverse experiment, cortical tissue was processed and immunoprecipitated using a rabbit polyclonal antibody to IKKalpha/beta (H-470, SCBT), a rabbit polyclonal anti-IgG antiserum served as control. Rabbit-polyclonal antibodies against ankyrin-G (S.E. Lux) and an alternative commercial antibody raised against ankyrin-G (H-215) were applied to additional samples. F
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
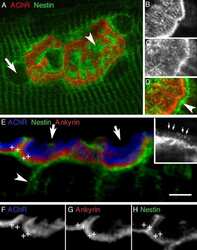
- Figure 2 Centriole loss in NSCs perturbs subsequent axonal Trim46 targeting and action potential maturation A Typical examples of Centrinone-B-treated or control human iPSC-derived NSCs immunostained for Pericentrin and Centrin. Inserts represent centriole(s). Scale bar = 5 um in overview, 2 um in inserts. B Quantifications of the percentage of cells with 0, 1, or 2 centrioles per cell after 0 (control), 2 or 5 days Centrinone-B treatment and prior to neuronal induction. n = 48-51 cells in two independent experiments. C Typical examples of Centrinone-B-treated or control human iPSC-derived neurons (12-15 days) immunostained for AnkG, Trim46, MAP2, and Centrin. Arrowheads mark AIS structures, arrows mark centrosomes. Inserts represent centrosomes, zooms on the right represent AIS structures. Scale bar = 20 um in overview, 2 um in insert, 10 um in zooms. D Quantifications of percentage of human iPSC-derived neurons (12-15 days) treated with Centrinone-B containing a Trim46-positive or Trim46-negative process. Neurons are subdivided in populations containing 2 centrioles, or less than 2 centrioles, based on Centrin immunostaining. n = 31-53 cells in three independent experiments. E Quantifications of percentage of human iPSC-derived neurons (12-15 days) treated with Centrinone-B containing an AnkG-positive or AnkG-negative process. Neurons are subdivided in populations containing 2 centrioles, or less than 2 centrioles, based on Centrin immunostaining. n = 27-32 cells in two ind
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
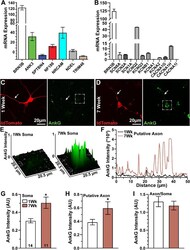
- Figure 3. AnkG expression increases over time. ( A ) Messenger RNA (mRNA) expression (fragments per kilobase million) of BRN3B- and AIS-related genes in hRGCs. ( B ) mRNA expression of BRN3B and select voltage-gated ion channel genes. (C, D) Representative tdTomato-positive ( red ) hRGCs after 1 and 7 weeks in culture immunolabeled against AnkG ( green ). Arrows indicate axon-like neurites. Putative axon was identified by AnkG localization or as the longest neurite. Scale bars = 20 um. ( E ) Surface plot of AnkG intensity distribution in somas indicated by dashed boxes above ( C , D ). ( F ) AnkG fluorescence intensity profiles along axon-like neurites indicated above ( C , D ). Quantification of AnkG immunolabeling within hRGC ( G ) somas and ( H ) putative axons relative to tdTomato intensity. AnkG localization significantly increases within somas ( P = 0.003) and putative axons ( P = 0.039) after 7 weeks in culture. ( I ) However, the ratio of AnkG labeling in putative axons and somas (axonal divided by somatic AnkG intensity) does not change between 1 and 7 weeks ( P = 0.68). Statistics: t -test ( G , H ), Mann-Whitney U test ( I ). Data presented as mean +- standard error of the mean.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
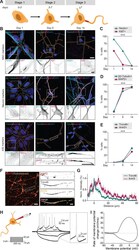
- Figure 1. Successful and protracted transition through early developmental stages in human iPSC-derived neurons. ( A ) Schematic illustration and timing of neurodevelopmental stages 1, 2 and 3 in human iPSC-derived NSCs/neurons. ( B ) Representative images of stage 1 (day 1), 2 (day 5) and 3 (day 14) hiPSC-derived NSCs/neurons. Cells were transduced with FUGW-GFP lentivirus and immunostained for NSC marker Nestin and proliferation marker Ki67, neuron markers beta3-Tubulin and MAP2, or AIS markers AnkG and Trim46. Outlines of cells were defined by the FUGW-GFP signal. Scale bar: 15 um in overview, 5 um in zooms. ( C,D,E ) Quantifications of percentage of human iPSC-derived NSCs positive for Ki67 or Nestin ( C ), beta3-Tubulin or MAP2 ( D ) and AnkG or Trim46 ( E ) at 1, 5 or 14 days in culture (N = 2, n = 100-109 cells). ( F ) Representative image of a polarized human iPSC-derived neuron immunostained for MAP2, Trim46 and AnkG. Zoom represents the AIS structure. Scale bar: 20 um in overview, 5 um in zoom. ( G ) Quantifications of average normalized fluorescent intensity profiles for Trim46 and AnkG at proximal axons of human iPSC-derived neurons (day 15) (n = 9). Distances are normalized to Trim46 peak intensities. ( H ) Left: Schematic illustration of the experimental electrophysiology setup. To determine AP frequency, somatic current injections ranging from -10 pA to 50 pA (steps of 5 pA, 400 ms) were applied. Right: Representative example of evoked AP firing in a human iPSC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
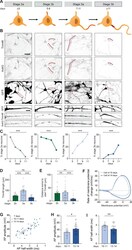
- Figure 3. Identification of intermediate developmental stages during onset of axon formation. ( A ) Schematic illustration and timing of neurodevelopmental stages 2a, 2b, 3a and 3b in human iPSC-derived NSCs/neurons. ( B ) Representative images of stage 2a, 2b, 3a, and 3b hiPSC-derived neurons. Cells were transduced with FUGW-GFP lentivirus and immunostained for AnkG and Trim46. Arrowheads mark Trim46 and AnkG accumulations. Zooms represent a nonpolarized neurite in a stage 2 neuron or a developing axon in a 3 neuron. Scale bar: 40 um overview, 5 um zooms. ( C ) Quantifications of the relative abundance of stage 2a, 2b, 3a or 3b human iPSC-derived neurons (N = 2, n = 50-55 cells). ( D ) Quantifications of the total length of Trim46 structures in neurites of stage 2b, 3a and 3b human iPSC-derived neurons (N = 2, n = 20 cells). ( E ) Quantifications of distance from soma to start of the Trim46 signal in neurites of stage 2b, 3a and 3b human iPSC-derived neurons (N = 2, n = 20 cells). ( F ) Phase plots of a representative AP recorded of a human iPSC-derived neuron at 10 days and 14 days. ( G ) Scatter plot of AP amplitude versus AP half-width grouped by days after plating (N = 4; 7 days: n = 7 cells, 10-11 days: n = 15 cells, 13-14 days: n = 36 cells). ( H ) AP amplitude recorded in human iPSC-derived neurons of 10-11 days (N = 4, n = 15 cells) and 13-14 days (N = 4, n = 36 cells). ( I ) AP half-width recorded in human iPSC-derived neurons of 10-11 days (N = 4, n = 15 cells) and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
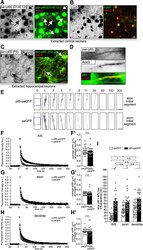
- 1 FIGURE AnkG and Na v 1.2 signals in the AIS showed increased length and bimodal spacing distributions with high GC treatment. To analyze the elongation of the AIS after glucose treatment, superresolution images of hippocampal neurons from the control (10 mM glucose) and high GC groups (20 mM glucose) were obtained by SIM. AnkG and Na v 1.2 showed single-peak distributions and dual-peak distributions in the control and high GC groups, respectively. (a, f) 3D SIM images of the AIS from the control, high GC and recovery groups immunolabeled with AnkG (a) and Na v 1.2 (f) showed ring-like structures (middle, bar: 2 mum); the figure in the upper left, framed by white dotted lines, shows a wide-field image of the same neurons (magenta: AnkG or Na v 1.2, blue: DAPI, bar: 10 mum); the bottom image was magnified and straightened from the yellow dotted box in the middle image (240 nm spacing in orange, bar: 2 mum). (b-d) Histogram of the spacing frequency between adjacent AnkG signals from the control (b), high GC (c) and recovery (d) groups. (e) Quantification of the soma distance (left) and length (right) by AnkG immunostaining in the control, high GC and recovery groups. N >= 51 from each of three independent experiments, one-way ANOVA (distance to soma: F (2,176) = 1.128, p = .326; length: F (2,176) = 18.100, p < .001). (g-i) Histogram of the spacing frequency between adjacent Na v 1.2 signals from the control (g), high GC (h) and recovery groups (i)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
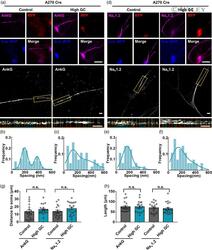
- 4 FIGURE AnkG and Na v 1.2 signals showed blocked alterations and nonperiodic distributions with high GC treatment in A270 Cre neurons. (a, d) (TOP) Representative immunostaining images of A270 Cre neurons; magenta: AnkG (a) and Na v 1.2 (d); red: anti-RFP immunostaining indicating 270 kDa AnkG expression; blue: anti-BFP immunostaining indicating successful Cre transfection. Bar: 10 mum. (Middle) SIM images of AnkG (a) and Na v 1.2 (d) signals from A270 Cre neurons. Bar: 2 mum. (Bottom) the magnified figures of yellow dotted box. Bar: 2 mum. (b, c) Histogram of the spacing frequency between adjacent AnkG signals with control (b) and high GC (c) treatments. (e, f) Histogram of the spacing frequency between adjacent Na v 1.2 signals with control (e) and high GC (f) treatments. (g, h) Quantification of the soma distance (g) and length (h) by immunostaining of two components of AIS with control and high GC treatments from A270 Cre neurons. N >= 17 from each of three independent experiments, one-way ANOVA (distance to soma: F (3,70) = 1.160, p = .331; length: F (3,70) = 0.583, p = .628). GC, glucose concentration; SIM, structured illumination microscopy
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
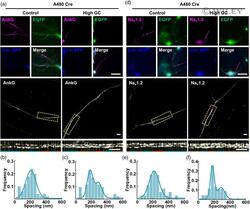
- 5 FIGURE AnkG and Na v 1.2 signals showed increased lengths and bimodal spacing distributions with high GC treatment in A480 Cre neurons. (a, d) (TOP) Representative immunostaining images of A480 Cre neurons; magenta: AnkG (a) and Na v 1.2 (d); green: anti-EGFP immunostaining indicating 480 kDa AnkG expression; blue: anti-BFP immunostaining indicating successful Cre transfection. Bar: 10 mum. (Middle) SIM images of AnkG (a) and Na v 1.2 (d) signals from A480 Cre neurons. Bar: 2 mum. (Bottom) the magnified figures of yellow dotted box. Bar: 2 mum. (b, c) Histogram of the spacing frequency between adjacent AnkG signals with control (b) and high GC (c) treatments. (e, f) Histogram of the spacing frequency between adjacent Na v 1.2 signals with control (e) and high GC (f) treatments. GC, glucose concentration; SIM, structured illumination microscopy
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
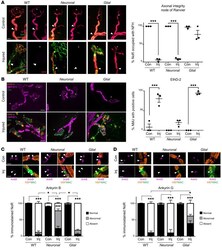
- Experimental details
- Distal motor nerve integrity following selective targeting and acute injury of neural membranes ex vivo. Triangularis sterni nerve-muscle preparations from WT, Neuronal , and Glial mice were treated ex vivo with anti-GM1 Ab and a source of complement (injury, Inj) or anti-GM1 Ab alone (control, Con). ( A ) Loss of axonal integrity due to injury at the motor nerve terminal (MNT, identified by alpha-bungarotoxin, BTx, orange, asterisk) and node of Ranvier (NoR, orange, arrowheads) was monitored by presence of neurofilament H immunostaining (NFH, magenta). Membrane attack complex (MAC) complement pore deposition (green) was present in all injured preparations compared with control. ( B ) Ethidium homodimer-positive (EthD-2-positive, orange) cells overlying MNT (magenta, asterisk) were compared among treatment groups. Representative images show the presence of complement deposition (green) in all injured tissue. ( C and D ) The sites where ankyrin B (AnkB) or AnkG immunostaining should be located are indicated by arrowheads. The presence of normal (black bars, statistical comparisons indicated with asterisks) or abnormal (gray bars) AnkB and AnkG immunostaining was compared to associated controls for each genotype. A lengthened gap between AnkB domains is shown in a representative image from injured Neuronal tissue. Weakened, uneven AnkG staining in injured Glial tissue is shown in the representative image. Scale bars: 20 mum ( A ), 50 mum ( B ), and 5 mum ( C and D ). Results ar
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
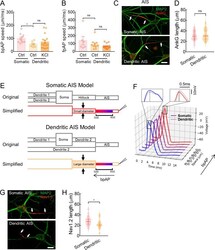
- Experimental details
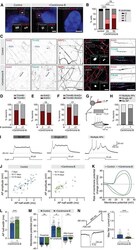
- Figure 5 bpAP speed was lower at dendritic AISs than somatic AISs A: Speed of bpAP at somatic AISs (pink circles, 99.47+-11.35 um/ms, n =10) was much higher than that at dendritic AISs (light orange circles, 55.65+-7.85 um/ms, n =7), * : P 0.05. B: Speed of fpAP at somatic AISs (pink circles, 137.5+-52.43 um/ms, n =6) was similar than that at dendritic AISs (light orange circles, 91.13+-19.42 um/ms, n =10), ns: P >0.05; at dendritic AISs, fpAP speed showed no difference between untreated group and KCl treatment group (dark orange, KCl treatment group, 66.52+-7.78 um/ms, n =15), ns: P >0.05. C: Immunofluorescence images of AIS length indicated by AnkG, distance between two white arrows represents AIS length (Scale bars: 20 um). D: Length indicated by AnkG between somatic AISs (30.54+-1.14 um, n =45) and dendritic AISs (32.21+-1.51 um, n =42) was similar, ns: P >0.05. E: Reformed multi-compartment neuronal models and their simplified equivalent cables. Stimulus was a 1 nA current that lasted for 1 ms. Stimulus was injected into one end of the thin cable. In the somatic model (pink), the three cables were 20 umx50 um (left), 2 umx50 um (middle), and 1.2 umx500 um (right). In the dendritic model (orange), the three cables were 20 umx50 um (left), 3 umx50 um (middle), and 1.2 umx500 um (right). Middle cable in dendritic
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
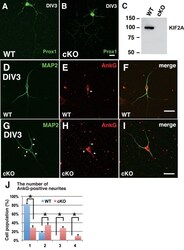
- Experimental details
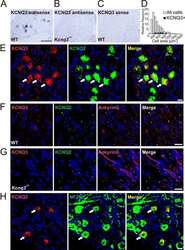
- Figure 6--figure supplement 1. The loss of KIF2A induced MF sprouting in dissociated cultured DGCs. ( A-C ) Characterization of dissociated cultured granule cells and confirmation of their loss of KIF2A. A-B. Representative images of cultured DGCs. Granule cells from the hippocampal dentate gyri of the WT ( A ) and Kif2a -cKO ( B ) mice were cultured for 4 days (DIV4) and then the cells were fixed and stained with an anti-Prox1 antibody. C. KIF2A deletion in cultured DGCs. KIF2A expression was analyzed using western blot analysis in extracts from the WT and Kif2a -cKO cultured cells obtained on DIV4. ( D-I ) Representative images of ankyrin G-positive neurites in WT ( D-F ) and P3- Kif2a -cKO ( G-I ) cultured DGCs. At an early stage of DGC development, all neurites are MAP2-positive around the cell body (D and G, see also Figure 6A ). Among those developing neurites, there were more neurites with ankyrin G at the neck in cKO DGCs (arrowheads in H) than in WT DGCs (arrowheads in E). Statistical analysis of the population of DGCs with the number of ankyrin G-positive neurites/cell is shown in ( J ). The results are shown as the mean +- SDs for approximately 60 cells each; n = 4. (*p
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry