Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Immunocytochemistry [3]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-26688 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- MTCO1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody is predicted to react with bovine, chicken, mouse, porcine, rat, rabbit and Xenopus and zebrafish based on sequence homology.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 400 μL
- Concentration
- 0.5 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Mitochondrial RNA modifications shape metabolic plasticity in metastasis.
Augmented Liver Uptake of the Membrane Voltage Sensor Tetraphenylphosphonium Distinguishes Early Fibrosis in a Mouse Model.
Dual-Specificity Phosphatase 1 (DUSP1) Has a Central Role in Redox Homeostasis and Inflammation in the Mouse Cochlea.
Evidence of Oxidative Phosphorylation in Zebrafish Photoreceptor Outer Segments at Different Larval Stages.
Deletion of the Mitochondrial Complex-IV Cofactor Heme A:Farnesyltransferase Causes Focal Segmental Glomerulosclerosis and Interferon Response.
RB1 deficiency in triple-negative breast cancer induces mitochondrial protein translation.
Heme Oxygenase-1/Carbon Monoxide System and Embryonic Stem Cell Differentiation and Maturation into Cardiomyocytes.
Depletion of TDP-43 decreases fibril and plaque β-amyloid and exacerbates neurodegeneration in an Alzheimer's mouse model.
Delaunay S, Pascual G, Feng B, Klann K, Behm M, Hotz-Wagenblatt A, Richter K, Zaoui K, Herpel E, Münch C, Dietmann S, Hess J, Benitah SA, Frye M
Nature 2022 Jul;607(7919):593-603
Nature 2022 Jul;607(7919):593-603
Augmented Liver Uptake of the Membrane Voltage Sensor Tetraphenylphosphonium Distinguishes Early Fibrosis in a Mouse Model.
Pandita H, Mezey E, Ganapathy-Kanniappan S
Frontiers in physiology 2021;12:676722
Frontiers in physiology 2021;12:676722
Dual-Specificity Phosphatase 1 (DUSP1) Has a Central Role in Redox Homeostasis and Inflammation in the Mouse Cochlea.
Bermúdez-Muñoz JM, Celaya AM, García-Mato Á, Muñoz-Espín D, Rodríguez-de la Rosa L, Serrano M, Varela-Nieto I
Antioxidants (Basel, Switzerland) 2021 Aug 25;10(9)
Antioxidants (Basel, Switzerland) 2021 Aug 25;10(9)
Evidence of Oxidative Phosphorylation in Zebrafish Photoreceptor Outer Segments at Different Larval Stages.
Calzia D, Garbarino G, Caicci F, Pestarino M, Manni L, Traverso CE, Panfoli I, Candiani S
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2018 Jul;66(7):497-509
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2018 Jul;66(7):497-509
Deletion of the Mitochondrial Complex-IV Cofactor Heme A:Farnesyltransferase Causes Focal Segmental Glomerulosclerosis and Interferon Response.
Baek JH, Gomez IG, Wada Y, Roach A, Mahad D, Duffield JS
The American journal of pathology 2018 Dec;188(12):2745-2762
The American journal of pathology 2018 Dec;188(12):2745-2762
RB1 deficiency in triple-negative breast cancer induces mitochondrial protein translation.
Jones RA, Robinson TJ, Liu JC, Shrestha M, Voisin V, Ju Y, Chung PE, Pellecchia G, Fell VL, Bae S, Muthuswamy L, Datti A, Egan SE, Jiang Z, Leone G, Bader GD, Schimmer A, Zacksenhaus E
The Journal of clinical investigation 2016 Oct 3;126(10):3739-3757
The Journal of clinical investigation 2016 Oct 3;126(10):3739-3757
Heme Oxygenase-1/Carbon Monoxide System and Embryonic Stem Cell Differentiation and Maturation into Cardiomyocytes.
Suliman HB, Zobi F, Piantadosi CA
Antioxidants & redox signaling 2016 Mar 1;24(7):345-60
Antioxidants & redox signaling 2016 Mar 1;24(7):345-60
Depletion of TDP-43 decreases fibril and plaque β-amyloid and exacerbates neurodegeneration in an Alzheimer's mouse model.
LaClair KD, Donde A, Ling JP, Jeong YH, Chhabra R, Martin LJ, Wong PC
Acta neuropathologica 2016 Dec;132(6):859-873
Acta neuropathologica 2016 Dec;132(6):859-873
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
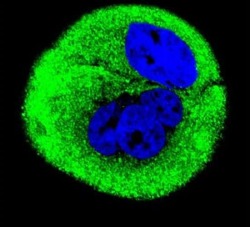
- Experimental details
- Immunofluorescent analysis of MCF-7 cells using a Cytochrome C Oxidase Subunit 1 polyclonal antibody (Product # PA5-26688) at a dilution of 1:10-50, followed by a fluor-conjugated goat anti-rabbit secondary antibody (green). Nuclei were stained with DAPI (blue).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
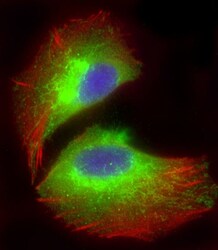
- Experimental details
- Immunocytochemistry analysis of MTCO1 in U-2 OS (human osteosarcoma cell line) cells. Samples were incubated with MTCO1 polyclonal antibody (Product # PA5-26688) using a dilution of 1:25 followed by Dylight® 488-conjugated goat anti-rabbit IgG at a dilution of 1:200 (green). Cells were 4% paraformaldehyde-fixed and 0.1% Triton X-100 permeabilized. Immunofluorescence image showing endoplasmic reticulum staining on U-2 OS cell line. Cytoplasmic actin is detected with Dylight® 554 Phalloidin at 1:100 dilution (red). The nuclear counter stain is DAPI (blue).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
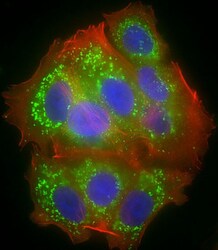
- Experimental details
- Immunocytochemistry analysis of MTCO1 in MCF-7 cells. Samples were incubated with MTCO1 polyclonal antibody (Product # PA5-26688) using a dilution of 1:25 followed by Dylight® 488-conjugated goat anti-rabbit IgG at a dilution of 1:200 (green). Cells were 4% paraformaldehyde-fixed and 0.1% Triton X-100 permeabilized. Immunofluorescence image showing cytoplasm and mitochondrion staining on MCF-7 cell line. Cytoplasmic actin is detected with Dylight® 554 Phalloidin at 1:100 dilution (red). The nuclear counter stain is DAPI (blue).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
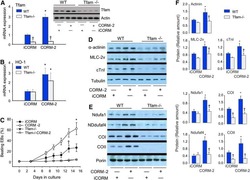
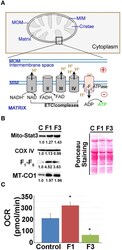
- Figure 3 Mitochondrial (mito-) oxidative phosphorylation (OxPhos) upregulated in early fibrosis. (A) Schematic diagram showing the overview of the regulation of mito-membrane potential. In OxPhos, ETC complexes release protons (H + ) into the space between the inner (MIM) and outer membranes (MOM) of mitochondria. H + accumulation establishes an electrochemical gradient leading to a negative potential (-) in the matrix. Thus, a higher rate of OxPhos (e.g., ATP synthesis) results in increased negative potential in the mito-matrix. (B) Immunoblot of mito-proteins shows an increase in mito-translocation of STAT3, F 1 -F 0 ATP synthase, and MT-CO1. COX IV remained unaltered. Immunoblot was re-probed for different targets to maintain the loading control. Numerical values at the bottom of the immunoblots represent the densitometry quantification of respective signals. Ponceau staining of the membrane shown for overall protein profile. (C) Metabolic flux analysis showing the oxygen consumption rate (OCR) of mitochondria isolated from the control, and F1 and F3 livers. Mitochondria showing a net increase in the OCR in the F1 liver. Unlike the typical cell-based assay, in this study, we used the mitochondria isolated from the respective livers as referred in the ""Materials and methods"" section. Hence, the extracellular acidification rate (ECAR) which is pertinent in the cellular assay is inapplicable. Data represent mean +- SE ( n = 3), t -test (* p < 0.05).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry