Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 37-8100 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Ephrin B1 Monoclonal Antibody (2D3E9)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Chicken/Avian
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2D3E9
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references EPH receptor B2 stimulates human monocyte adhesion and migration independently of its EphrinB ligands.
A novel synaptic junction preparation for the identification and characterization of cleft proteins.
Anchoring and synaptic stability of PSD-95 is driven by ephrin-B3.
Vreeken D, Bruikman CS, Cox SML, Zhang H, Lalai R, Koudijs A, van Zonneveld AJ, Hovingh GK, van Gils JM
Journal of leukocyte biology 2020 Sep;108(3):999-1011
Journal of leukocyte biology 2020 Sep;108(3):999-1011
A novel synaptic junction preparation for the identification and characterization of cleft proteins.
Burch A, Tao-Cheng JH, Dosemeci A
PloS one 2017;12(3):e0174895
PloS one 2017;12(3):e0174895
Anchoring and synaptic stability of PSD-95 is driven by ephrin-B3.
Hruska M, Henderson NT, Xia NL, Le Marchand SJ, Dalva MB
Nature neuroscience 2015 Nov;18(11):1594-605
Nature neuroscience 2015 Nov;18(11):1594-605
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
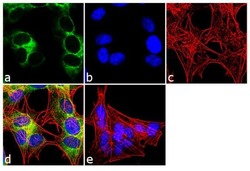
- Experimental details
- Immunofluorescent analysis of Ephrin-B Pan was performed using 70% confluent log phase SH-SY5Y cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Ephrin-B Pan (2D3E9) Mouse Monoclonal Antibody (Product # 37-8100) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
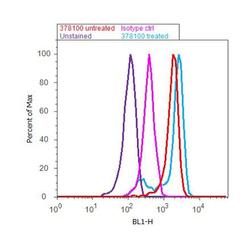
- Experimental details
- Flow Cytometry analysis of Ephrin-B was done on SKOV-3 cells (untreated, red histogram) and SKOV-3 cells treated with 40ng/ml TNF alpha for 24 hours (blue histogram). Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Ephrin-B Mouse Monoclonal Antibody (37-8100) or with mouse isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10, 000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
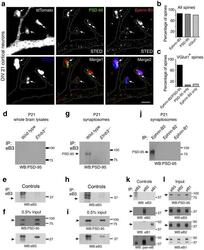
- Experimental details
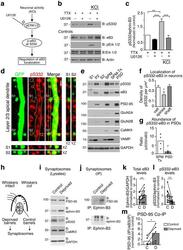
- Figure 1 PSD-95 is in a complex with ephrin-B3 at synapses a) ST imulated E mission D epletion (STED) imaging of PSD-95 and ephrin-B3 at synapses. DIV21 tdTomato transfected rat cortical neurons were labeled with the indicated antibodies. PSD-95 and ephrin-B3 were imaged at super-resolution using STED (~80 nm). VGlut1 and tdTomato were imaged at conventional resolution (~250 nm) using confocal. Images are high contrast examples of PSD-95, ephrin-B3 and vGlut1 immunostaining. Arrows indicate PSD-95, ephrin-B3 and vGlut1 co-clusters in dendritic spines. Scale bar: 2 um. b) PSD-95, ephrin-B3 and vGlut1 are found in most (>=80%) spines (248 spines on 15 neurons, two separate experiments). c) PSD-95-ephrin-B3 co-clusters are found predominantly in vGlut + spines (162 PSD-95-ephrin-B3 containing spines out of 202 vGlut1 + spines). Single PSD-95 (14 out of 202 vGlut + spines) or ephrin-B3 (20 out of 202 vGlut + spines) clusters are rarely found at synapses. d-f) Co-immunoprecipitation (co-IP) of ephrin-B3 and PSD-95 from P21 wild type and Efnb3 -/- mouse whole brain lysates. The efficacy of ephrin-B3 IP (e) and input (f) are shown for each condition. (g-i) PSD-95-ephrin-B3 co-IP from synaptosomes of wild type and Efnb3 -/- animals (g). Control immunoblots indicating the amount of ephrin-B3 IP (h) and input (i) . (j) PSD-95 selectively co-IPs with ephrin-B3 in synaptosomes isolated from P21 wild type brains despite the efficient IP (k) and presence (l) of each of the three ephrin-B f
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
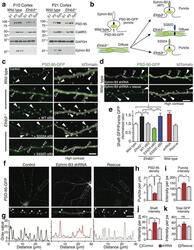
- Experimental details
- Figure 4 Ephrin-B3 regulates subcellular PSD-95 localization (a) Western blots of non-synaptic (S1), crude synaptosomal (P2) and pure synaptosomal (Syn.) fractions from cortices of P10 and P21 wild type and Efnb3 -/- mice probed with the indicated antibodies. Less synaptic enrichment of PSD-95 is observed in synaptosomes from Efnb3 -/- mice. (b) Model and predicted outcomes for the organotypic slice culture experiment. (c-d) Representative two-photon images of cortical pyramidal neurons from wild type and Efnb3 -/- organotypic slices transfected with PSD-95-GFP and the indicated ephrin-B3 rescue constructs. Right panels in c and d are the high contrast representations of tdTomato and PSD-95-GFP. Filled arrowheads indicate PSD-95-GFP localized to a spine; open arrowheads show PSD-95-GFP localized in the dendritic shaft. PSD-95-GFP is diffusely localized in dendritic shafts of Efnb3 -/- neurons (c) and ephrin-B3 shRNA-transfected wild type neurons (d) . Scale bar in c, d : 5 um. (e) The ratio of average GFP pixel intensities in dendritic shafts to the GFP intensities per pixel in puncta was used to measure the fraction of diffuse PSD-95-GFP in c-d (ANOVA, F (4, 93) = 7.155, p-values: wild type// Efnb3 -/- : 0.0105, wild type// Efnb3 -/- + S332D: 0.0233, Efnb3 -/- // Efnb3 -/- + wt-eB3: 0.0285, Efnb3 -/- // Efnb3 -/- + S332A: 0.0015, Efnb3 -/- + wt-eB3// Efnb3 -/- + S332D: 0.0487, Efnb3 -/- + S332A// Efnb3 -/- + S332D: 0.0027; wild type (n = 22), Efnb3 -/- (n = 31), Efnb3 -/- +
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
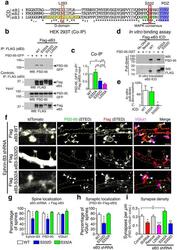
- Figure 2 Ephrin-B3 links MAPK signaling to PSD-95-ephrin-B3 interaction and localization ( a) Sequence alignment of intracellular domains (ICD) of ephrin-B (eB1-3) family. ERK binding D-Domain in the juxtamembrane region of ephrin-B3 (yellow box) and a putative MAPK phosphorylation motif (green box). Red boxes indicate the positions of point mutations in the D-domain (L293A) and in the putative MAPK phosphorylation motif (S332A/S332D). ( b) Immunoprecipitation, using antibodies to Flag, of HEK 293T lysates transfected with PSD-95-GFP and indicated flag-tagged ephrin-B3 (flag-eB3) constructs. (c) Quantification of co-IP experiments shown in b (ANOVA, F (3, 23) = 9.842. WT-S332D: p = 0.0475, WT-L293A: p = 0.0034, S332A-S332D: p = 0.0087, S332A-L293A: p = 0.0006; n = 6 blots from separate transfections). (d) In vitro generated flag-tagged wild type and mutant ephrin-B3 ICDs used to pull down PSD-95-GST. Direct interaction between the two proteins is disrupted by S332D ephrin-B3 ICD. (e) Quantification of pull-down experiments from d (ANOVA, F (2, 9) = 2.823, Fisher's LSD post-hoc, p = 0.0176 (WT-S332D), p = 0.0257 (S332A-S332D); n = 4 independent in vitro binding experiments). (f) Representative high contrast STED images of DIV21 neurons transfected with tdTomato, ephrin-B3 (eB3) shRNA and indicated flag-eB3 rescue constructs. Arrowheads indicate co-localized, flag-ephrin-B3/PSD-95/vGlut1 clusters. Scale bars : , 1 um. (g) Quantification of spine localization of flag-ephrin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3 ERK phosphorylation of S332 regulates subcellular localization of ephrin-B3 (a) Schematic representation of the experiments. (b) Representative immunoblots of lysates from DIV14 rat cortical neurons treated with TTX (1uM, 4 hr.) and U0126 (10uM, 4 hr.) to block activity and ERK activation, respectively. Depolarization with KCl (55mM, 1 hr.) was used to elevate ERK activity. ( c ) Ratio of pS332 to ephrin-B3 signals was used to quantify the effect of neuronal activity on S332 phosphorylation (ANOVA, F (3, 12) = 19.27, Tukey's post-hoc, TTX-KCl: p = 0.0012, TTX+U0126-KCl: p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
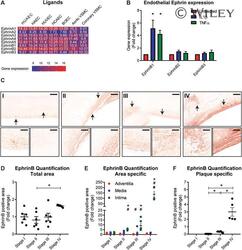
- 2 FIGURE Increased expression of EphrinB in progressive human atherosclerotic lesions . ( A ) Expression heatmap of Ephrin family ligands in endothelial cells and vascular smooth muscle cells. Blue indicates lower and red higher expression. ( B ) EphrinB1/B2/B3 expression in HUVECs stimulated with 20 ng/mL IL-1beta or 10 ng/mL TNF-alpha for 24 h. Results are relative to untreated cells, set as 1. Mean +- sem of n = 3. * P < 0.05. ( C ) Overview pictures of immunohistochemical staining for EphrinB in human aortic sections in different stages of atherosclerosis. Lower images show higher power magnification of the arrow-indicated fields. Scale bars represent 350 and 50 um, respectively. ( D-F ) Quantification of EphrinB signal on total area ( D ), adventitia, media, and intima regions ( E ) and total plaque area ( F ). Results are relative to stage I, set as 1. Mean +- sem of n = 6. ( D-F ) * P < 0.05 compared to indicated stages. ( E ) * P < 0.05 representing interstage variability within a region compared to stage I and # P < 0.05 representing intrastage variability between regions. Stage I: normal, adaptive intimal thickening, intima xantoma; II: pathologic intimal thickening, early fibroatheroma; III: late fibroatheroma, thin cap fibroatheroma, ruptured plaque; IV: healing rupture, fibrous calcifies plaque
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry