Antibody data
- Antibody Data
- Antigen structure
- References [54]
- Comments [0]
- Validations
- Immunocytochemistry [4]
- Immunohistochemistry [5]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-0700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NEFM Monoclonal Antibody (RMO-270)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- The cloning partner for this antibody is Sp/2. This antibody reacts with the 160 kD polypeptide subunit of human neurofilament. It specifically recognizes a phosphate-independent epitope in the tail (carboxy) domain of NF-M of most vertebrates and invertebrates. This antibody is suitable for immunohistochemical staining of Bouin's and alcohol-fixed paraffin-embedded or frozen tissue sections. To stain, incubate 30-60 minutes at room temperature or overnight at 4°C.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- RMO-270
- Vial size
- 200 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Sox8 remodels the cranial ectoderm to generate the ear.
Endothelial PlexinD1 signaling instructs spinal cord vascularization and motor neuron development.
Atoh7-independent specification of retinal ganglion cell identity.
Endoglycan plays a role in axon guidance by modulating cell adhesion.
CNTF and Nrf2 Are Coordinately Involved in Regulating Self-Renewal and Differentiation of Neural Stem Cell during Embryonic Development.
Microglial modulation through colony-stimulating factor-1 receptor inhibition attenuates demyelination.
Ribosome Incorporation into Somatic Cells Promotes Lineage Transdifferentiation towards Multipotency.
Normal development of spinal axons in early embryo stages and posterior locomotor function is independent of GAL-1.
Innervation of the syrinx of the zebra finch (Taeniopygia guttata).
Motor neurons control blood vessel patterning in the developing spinal cord.
Experience-dependent plasticity of excitatory and inhibitory intertectal inputs in Xenopus tadpoles.
Identification of molecular signatures specific for distinct cranial sensory ganglia in the developing chick.
Regulation of downstream neuronal genes by proneural transcription factors during initial neurogenesis in the vertebrate brain.
Quantitative assessment of angiogenesis, perfused blood vessels and endothelial tip cells in the postnatal mouse brain.
Galanin modulates the neural niche to favour perineural invasion in head and neck cancer.
Distinct adhesion-independent functions of β-catenin control stage-specific sensory neurogenesis and proliferation.
Comprehensive evaluation of peripheral nerve regeneration in the acute healing phase using tissue clearing and optical microscopy in a rodent model.
Excess caffeine exposure impairs eye development during chick embryogenesis.
Evolutionarily conserved morphogenetic movements at the vertebrate head-trunk interface coordinate the transport and assembly of hypopharyngeal structures.
Arylsulfatase B improves locomotor function after mouse spinal cord injury.
Cranial neural crest cells form corridors prefiguring sensory neuroblast migration.
Motoneuronal Sema3C is essential for setting stereotyped motor tract positioning in limb-derived chemotropic semaphorins.
A method to isolate and culture expand human dental pulp stem cells.
Sprouty genes are essential for the normal development of epibranchial ganglia in the mouse embryo.
Ectopic Mitf in the embryonic chick retina by co-transfection of β-catenin and Otx2.
Nectin-like molecules/SynCAMs are required for post-crossing commissural axon guidance.
Implanted adult human dental pulp stem cells induce endogenous axon guidance.
Intermediate filaments exchange subunits along their length and elongate by end-to-end annealing.
Adult human dental pulp stem cells differentiate toward functionally active neurons under appropriate environmental cues.
Semaphorin and neuropilin co-expression in motoneurons sets axon sensitivity to environmental semaphorin sources during motor axon pathfinding.
Delamination of cells from neurogenic placodes does not involve an epithelial-to-mesenchymal transition.
Pre-/post-otic rhombomeric interactions control the emergence of a fetal-like respiratory rhythm in the mouse embryo.
Receptor tyrosine phosphatases guide vertebrate motor axons during development.
Redefining the head-trunk interface for the neural crest.
Distinct roles of different neural cell adhesion molecule (NCAM) isoforms in synaptic maturation revealed by analysis of NCAM 180 kDa isoform-deficient mice.
Wnt2b controls retinal cell differentiation at the ciliary marginal zone.
Fgf signalling is required for formation of cartilage in the head.
Metalloproteases and guidance of retinal axons in the developing visual system.
Exogenous growth factors induce the production of ganglion cells at the retinal margin.
Cues from neuroepithelium and surface ectoderm maintain neural crest-free regions within cranial mesenchyme of the developing chick.
Differential effects of unnatural sialic acids on the polysialylation of the neural cell adhesion molecule and neuronal behavior.
Myelin-associated glycoprotein modulates expression and phosphorylation of neuronal cytoskeletal elements and their associated kinases.
Neurofilament-M interacts with the D1 dopamine receptor to regulate cell surface expression and desensitization.
A critical role for sonic hedgehog signaling in the early expansion of the developing brain.
Distinct regulatory cascades for head and trunk myogenesis.
Early development of the mesencephalic trigeminal nucleus.
Boundary formation and compartition in the avian diencephalon.
Multiple actions of neurturin correlate with spatiotemporal patterns of Ret expression in developing chick cranial ganglion neurons.
Specification of distinct motor neuron identities by the singular activities of individual Hox genes.
Homeotic transformation of rhombomere identity after localized Hoxb1 misexpression.
Mitogen-activated protein kinases (Erk1,2) phosphorylate Lys-Ser-Pro (KSP) repeats in neurofilament proteins NF-H and NF-M.
Tlx-1 and Tlx-3 homeobox gene expression in cranial sensory ganglia and hindbrain of the chick embryo: markers of patterned connectivity.
Migration of hypoglossal myoblast precursors.
Astrocytes and regenerating axons at the proximal stump of the severed frog optic nerve.
Buzzi AL, Chen J, Thiery A, Delile J, Streit A
Proceedings of the National Academy of Sciences of the United States of America 2022 Jul 12;119(28):e2118938119
Proceedings of the National Academy of Sciences of the United States of America 2022 Jul 12;119(28):e2118938119
Endothelial PlexinD1 signaling instructs spinal cord vascularization and motor neuron development.
Vieira JR, Shah B, Dupraz S, Paredes I, Himmels P, Schermann G, Adler H, Motta A, Gärtner L, Navarro-Aragall A, Ioannou E, Dyukova E, Bonnavion R, Fischer A, Bonanomi D, Bradke F, Ruhrberg C, Ruiz de Almodóvar C
Neuron 2022 Dec 21;110(24):4074-4089.e6
Neuron 2022 Dec 21;110(24):4074-4089.e6
Atoh7-independent specification of retinal ganglion cell identity.
Brodie-Kommit J, Clark BS, Shi Q, Shiau F, Kim DW, Langel J, Sheely C, Ruzycki PA, Fries M, Javed A, Cayouette M, Schmidt T, Badea T, Glaser T, Zhao H, Singer J, Blackshaw S, Hattar S
Science advances 2021 Mar;7(11)
Science advances 2021 Mar;7(11)
Endoglycan plays a role in axon guidance by modulating cell adhesion.
Baeriswyl T, Dumoulin A, Schaettin M, Tsapara G, Niederkofler V, Helbling D, Avilés E, Frei JA, Wilson NH, Gesemann M, Kunz B, Stoeckli ET
eLife 2021 Mar 2;10
eLife 2021 Mar 2;10
CNTF and Nrf2 Are Coordinately Involved in Regulating Self-Renewal and Differentiation of Neural Stem Cell during Embryonic Development.
Si ZP, Wang G, Han SS, Jin Y, Hu YX, He MY, Brand-Saberi B, Yang X, Liu GS
iScience 2019 Sep 27;19:303-315
iScience 2019 Sep 27;19:303-315
Microglial modulation through colony-stimulating factor-1 receptor inhibition attenuates demyelination.
Wies Mancini VSB, Pasquini JM, Correale JD, Pasquini LA
Glia 2019 Feb;67(2):291-308
Glia 2019 Feb;67(2):291-308
Ribosome Incorporation into Somatic Cells Promotes Lineage Transdifferentiation towards Multipotency.
Ito N, Katoh K, Kushige H, Saito Y, Umemoto T, Matsuzaki Y, Kiyonari H, Kobayashi D, Soga M, Era T, Araki N, Furuta Y, Suda T, Kida Y, Ohta K
Scientific reports 2018 Jan 26;8(1):1634
Scientific reports 2018 Jan 26;8(1):1634
Normal development of spinal axons in early embryo stages and posterior locomotor function is independent of GAL-1.
Pasquini JM, Barrantes FJ, Quintá HR
The Journal of comparative neurology 2017 Sep 1;525(13):2861-2875
The Journal of comparative neurology 2017 Sep 1;525(13):2861-2875
Innervation of the syrinx of the zebra finch (Taeniopygia guttata).
Faunes M, Botelho JF, Wild JM
The Journal of comparative neurology 2017 Sep 1;525(13):2847-2860
The Journal of comparative neurology 2017 Sep 1;525(13):2847-2860
Motor neurons control blood vessel patterning in the developing spinal cord.
Himmels P, Paredes I, Adler H, Karakatsani A, Luck R, Marti HH, Ermakova O, Rempel E, Stoeckli ET, Ruiz de Almodóvar C
Nature communications 2017 Mar 6;8:14583
Nature communications 2017 Mar 6;8:14583
Experience-dependent plasticity of excitatory and inhibitory intertectal inputs in Xenopus tadpoles.
Gambrill AC, Faulkner R, Cline HT
Journal of neurophysiology 2016 Nov;116(5):2281-2297
Journal of neurophysiology 2016 Nov;116(5):2281-2297
Identification of molecular signatures specific for distinct cranial sensory ganglia in the developing chick.
Patthey C, Clifford H, Haerty W, Ponting CP, Shimeld SM, Begbie J
Neural development 2016 Jan 27;11:3
Neural development 2016 Jan 27;11:3
Regulation of downstream neuronal genes by proneural transcription factors during initial neurogenesis in the vertebrate brain.
Ware M, Hamdi-Rozé H, Le Friec J, David V, Dupé V
Neural development 2016 Dec 7;11(1):22
Neural development 2016 Dec 7;11(1):22
Quantitative assessment of angiogenesis, perfused blood vessels and endothelial tip cells in the postnatal mouse brain.
Wälchli T, Mateos JM, Weinman O, Babic D, Regli L, Hoerstrup SP, Gerhardt H, Schwab ME, Vogel J
Nature protocols 2015 Jan;10(1):53-74
Nature protocols 2015 Jan;10(1):53-74
Galanin modulates the neural niche to favour perineural invasion in head and neck cancer.
Scanlon CS, Banerjee R, Inglehart RC, Liu M, Russo N, Hariharan A, van Tubergen EA, Corson SL, Asangani IA, Mistretta CM, Chinnaiyan AM, D'Silva NJ
Nature communications 2015 Apr 28;6:6885
Nature communications 2015 Apr 28;6:6885
Distinct adhesion-independent functions of β-catenin control stage-specific sensory neurogenesis and proliferation.
Gay MH, Valenta T, Herr P, Paratore-Hari L, Basler K, Sommer L
BMC biology 2015 Apr 11;13:24
BMC biology 2015 Apr 11;13:24
Comprehensive evaluation of peripheral nerve regeneration in the acute healing phase using tissue clearing and optical microscopy in a rodent model.
Jung Y, Ng JH, Keating CP, Senthil-Kumar P, Zhao J, Randolph MA, Winograd JM, Evans CL
PloS one 2014;9(4):e94054
PloS one 2014;9(4):e94054
Excess caffeine exposure impairs eye development during chick embryogenesis.
Ma ZL, Wang G, Cheng X, Chuai M, Kurihara H, Lee KK, Yang X
Journal of cellular and molecular medicine 2014 Jun;18(6):1134-43
Journal of cellular and molecular medicine 2014 Jun;18(6):1134-43
Evolutionarily conserved morphogenetic movements at the vertebrate head-trunk interface coordinate the transport and assembly of hypopharyngeal structures.
Lours-Calet C, Alvares LE, El-Hanfy AS, Gandesha S, Walters EH, Sobreira DR, Wotton KR, Jorge EC, Lawson JA, Kelsey Lewis A, Tada M, Sharpe C, Kardon G, Dietrich S
Developmental biology 2014 Jun 15;390(2):231-46
Developmental biology 2014 Jun 15;390(2):231-46
Arylsulfatase B improves locomotor function after mouse spinal cord injury.
Yoo M, Khaled M, Gibbs KM, Kim J, Kowalewski B, Dierks T, Schachner M
PloS one 2013;8(3):e57415
PloS one 2013;8(3):e57415
Cranial neural crest cells form corridors prefiguring sensory neuroblast migration.
Freter S, Fleenor SJ, Freter R, Liu KJ, Begbie J
Development (Cambridge, England) 2013 Sep;140(17):3595-600
Development (Cambridge, England) 2013 Sep;140(17):3595-600
Motoneuronal Sema3C is essential for setting stereotyped motor tract positioning in limb-derived chemotropic semaphorins.
Sanyas I, Bozon M, Moret F, Castellani V
Development (Cambridge, England) 2012 Oct;139(19):3633-43
Development (Cambridge, England) 2012 Oct;139(19):3633-43
A method to isolate and culture expand human dental pulp stem cells.
Gronthos S, Arthur A, Bartold PM, Shi S
Methods in molecular biology (Clifton, N.J.) 2011;698:107-21
Methods in molecular biology (Clifton, N.J.) 2011;698:107-21
Sprouty genes are essential for the normal development of epibranchial ganglia in the mouse embryo.
Simrick S, Lickert H, Basson MA
Developmental biology 2011 Oct 1;358(1):147-55
Developmental biology 2011 Oct 1;358(1):147-55
Ectopic Mitf in the embryonic chick retina by co-transfection of β-catenin and Otx2.
Westenskow PD, McKean JB, Kubo F, Nakagawa S, Fuhrmann S
Investigative ophthalmology & visual science 2010 Oct;51(10):5328-35
Investigative ophthalmology & visual science 2010 Oct;51(10):5328-35
Nectin-like molecules/SynCAMs are required for post-crossing commissural axon guidance.
Niederkofler V, Baeriswyl T, Ott R, Stoeckli ET
Development (Cambridge, England) 2010 Feb;137(3):427-35
Development (Cambridge, England) 2010 Feb;137(3):427-35
Implanted adult human dental pulp stem cells induce endogenous axon guidance.
Arthur A, Shi S, Zannettino AC, Fujii N, Gronthos S, Koblar SA
Stem cells (Dayton, Ohio) 2009 Sep;27(9):2229-37
Stem cells (Dayton, Ohio) 2009 Sep;27(9):2229-37
Intermediate filaments exchange subunits along their length and elongate by end-to-end annealing.
Colakoğlu G, Brown A
The Journal of cell biology 2009 Jun 1;185(5):769-77
The Journal of cell biology 2009 Jun 1;185(5):769-77
Adult human dental pulp stem cells differentiate toward functionally active neurons under appropriate environmental cues.
Arthur A, Rychkov G, Shi S, Koblar SA, Gronthos S
Stem cells (Dayton, Ohio) 2008 Jul;26(7):1787-95
Stem cells (Dayton, Ohio) 2008 Jul;26(7):1787-95
Semaphorin and neuropilin co-expression in motoneurons sets axon sensitivity to environmental semaphorin sources during motor axon pathfinding.
Moret F, Renaudot C, Bozon M, Castellani V
Development (Cambridge, England) 2007 Dec;134(24):4491-501
Development (Cambridge, England) 2007 Dec;134(24):4491-501
Delamination of cells from neurogenic placodes does not involve an epithelial-to-mesenchymal transition.
Graham A, Blentic A, Duque S, Begbie J
Development (Cambridge, England) 2007 Dec;134(23):4141-5
Development (Cambridge, England) 2007 Dec;134(23):4141-5
Pre-/post-otic rhombomeric interactions control the emergence of a fetal-like respiratory rhythm in the mouse embryo.
Borday C, Coutinho A, Germon I, Champagnat J, Fortin G
Journal of neurobiology 2006 Oct;66(12):1285-301
Journal of neurobiology 2006 Oct;66(12):1285-301
Receptor tyrosine phosphatases guide vertebrate motor axons during development.
Stepanek L, Stoker AW, Stoeckli E, Bixby JL
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Apr 13;25(15):3813-23
The Journal of neuroscience : the official journal of the Society for Neuroscience 2005 Apr 13;25(15):3813-23
Redefining the head-trunk interface for the neural crest.
Ferguson CA, Graham A
Developmental biology 2004 May 1;269(1):70-80
Developmental biology 2004 May 1;269(1):70-80
Distinct roles of different neural cell adhesion molecule (NCAM) isoforms in synaptic maturation revealed by analysis of NCAM 180 kDa isoform-deficient mice.
Polo-Parada L, Bose CM, Plattner F, Landmesser LT
The Journal of neuroscience : the official journal of the Society for Neuroscience 2004 Feb 25;24(8):1852-64
The Journal of neuroscience : the official journal of the Society for Neuroscience 2004 Feb 25;24(8):1852-64
Wnt2b controls retinal cell differentiation at the ciliary marginal zone.
Kubo F, Takeichi M, Nakagawa S
Development (Cambridge, England) 2003 Feb;130(3):587-98
Development (Cambridge, England) 2003 Feb;130(3):587-98
Fgf signalling is required for formation of cartilage in the head.
Walshe J, Mason I
Developmental biology 2003 Dec 15;264(2):522-36
Developmental biology 2003 Dec 15;264(2):522-36
Metalloproteases and guidance of retinal axons in the developing visual system.
Webber CA, Hocking JC, Yong VW, Stange CL, McFarlane S
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Sep 15;22(18):8091-100
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Sep 15;22(18):8091-100
Exogenous growth factors induce the production of ganglion cells at the retinal margin.
Fischer AJ, Dierks BD, Reh TA
Development (Cambridge, England) 2002 May;129(9):2283-91
Development (Cambridge, England) 2002 May;129(9):2283-91
Cues from neuroepithelium and surface ectoderm maintain neural crest-free regions within cranial mesenchyme of the developing chick.
Golding JP, Dixon M, Gassmann M
Development (Cambridge, England) 2002 Mar;129(5):1095-105
Development (Cambridge, England) 2002 Mar;129(5):1095-105
Differential effects of unnatural sialic acids on the polysialylation of the neural cell adhesion molecule and neuronal behavior.
Charter NW, Mahal LK, Koshland DE Jr, Bertozzi CR
The Journal of biological chemistry 2002 Mar 15;277(11):9255-61
The Journal of biological chemistry 2002 Mar 15;277(11):9255-61
Myelin-associated glycoprotein modulates expression and phosphorylation of neuronal cytoskeletal elements and their associated kinases.
Dashiell SM, Tanner SL, Pant HC, Quarles RH
Journal of neurochemistry 2002 Jun;81(6):1263-72
Journal of neurochemistry 2002 Jun;81(6):1263-72
Neurofilament-M interacts with the D1 dopamine receptor to regulate cell surface expression and desensitization.
Kim OJ, Ariano MA, Lazzarini RA, Levine MS, Sibley DR
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Jul 15;22(14):5920-30
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Jul 15;22(14):5920-30
A critical role for sonic hedgehog signaling in the early expansion of the developing brain.
Britto J, Tannahill D, Keynes R
Nature neuroscience 2002 Feb;5(2):103-10
Nature neuroscience 2002 Feb;5(2):103-10
Distinct regulatory cascades for head and trunk myogenesis.
Mootoosamy RC, Dietrich S
Development (Cambridge, England) 2002 Feb;129(3):573-83
Development (Cambridge, England) 2002 Feb;129(3):573-83
Early development of the mesencephalic trigeminal nucleus.
Hunter E, Begbie J, Mason I, Graham A
Developmental dynamics : an official publication of the American Association of Anatomists 2001 Nov;222(3):484-93
Developmental dynamics : an official publication of the American Association of Anatomists 2001 Nov;222(3):484-93
Boundary formation and compartition in the avian diencephalon.
Larsen CW, Zeltser LM, Lumsden A
The Journal of neuroscience : the official journal of the Society for Neuroscience 2001 Jul 1;21(13):4699-711
The Journal of neuroscience : the official journal of the Society for Neuroscience 2001 Jul 1;21(13):4699-711
Multiple actions of neurturin correlate with spatiotemporal patterns of Ret expression in developing chick cranial ganglion neurons.
Hashino E, Johnson EM Jr, Milbrandt J, Shero M, Salvi RJ, Cohan CS
The Journal of neuroscience : the official journal of the Society for Neuroscience 1999 Oct 1;19(19):8476-86
The Journal of neuroscience : the official journal of the Society for Neuroscience 1999 Oct 1;19(19):8476-86
Specification of distinct motor neuron identities by the singular activities of individual Hox genes.
Jungbluth S, Bell E, Lumsden A
Development (Cambridge, England) 1999 Jun;126(12):2751-8
Development (Cambridge, England) 1999 Jun;126(12):2751-8
Homeotic transformation of rhombomere identity after localized Hoxb1 misexpression.
Bell E, Wingate RJ, Lumsden A
Science (New York, N.Y.) 1999 Jun 25;284(5423):2168-71
Science (New York, N.Y.) 1999 Jun 25;284(5423):2168-71
Mitogen-activated protein kinases (Erk1,2) phosphorylate Lys-Ser-Pro (KSP) repeats in neurofilament proteins NF-H and NF-M.
Veeranna, Amin ND, Ahn NG, Jaffe H, Winters CA, Grant P, Pant HC
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 Jun 1;18(11):4008-21
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 Jun 1;18(11):4008-21
Tlx-1 and Tlx-3 homeobox gene expression in cranial sensory ganglia and hindbrain of the chick embryo: markers of patterned connectivity.
Logan C, Wingate RJ, McKay IJ, Lumsden A
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 Jul 15;18(14):5389-402
The Journal of neuroscience : the official journal of the Society for Neuroscience 1998 Jul 15;18(14):5389-402
Migration of hypoglossal myoblast precursors.
Mackenzie S, Walsh FS, Graham A
Developmental dynamics : an official publication of the American Association of Anatomists 1998 Dec;213(4):349-58
Developmental dynamics : an official publication of the American Association of Anatomists 1998 Dec;213(4):349-58
Astrocytes and regenerating axons at the proximal stump of the severed frog optic nerve.
Blanco RE, Orkand PM
Cell and tissue research 1996 Dec;286(3):337-45
Cell and tissue research 1996 Dec;286(3):337-45
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
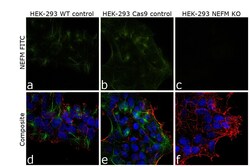
- Experimental details
- Knockout of NEFM was achieved by CRISPR-Cas9 genome editing. Immunofluorescence analysis was performed on wild type HEK-293 cells (panel a,d), HEK-293 Cas9 cells (panels b,e) and HEK-293 NEFM KO cells (panel c,f). Cells were fixed, permeabilized, and labelled with NEFM Monoclonal Antibody (RMO-270) (Product # 13-0700) (5 µg/mL), followed by Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723) (1:2000). Nuclei (blue) were stained using ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962), and Rhodamine Phalloidin (Product # R415) (1:300) was used for cytoskeletal F-actin (red) staining. Loss of signal (panel c,f) upon CRISPR mediated knockout (KO) confirms that antibody is specific to NEFM (green). The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
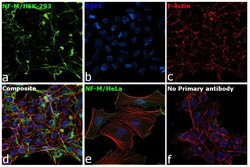
- Experimental details
- Immunofluorescence analysis of NF-M was performed using 70% confluent log phase HEK-293 and HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. HEK-293 cells were labeled with NF-M Mouse Monoclonal Antibody (Product # 13-0700) at 5 µg/mL in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415). Panel d represents the merged image of HEK-293 showing cytoskeletal (intermediate filaments) localization. Panel e represents the merged image of HeLa cells showing no expression for NF-M protein. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
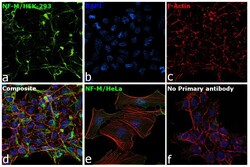
- Experimental details
- Immunofluorescence analysis of NF-M was performed using 70% confluent log phase HEK-293 and HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. HEK-293 cells were labeled with NF-M Mouse Monoclonal Antibody (Product # 13-0700) at 5 µg/mL in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415). Panel d represents the merged image of HEK-293 showing cytoskeletal (intermediate filaments) localization. Panel e represents the merged image of HeLa cells showing no expression for NF-M protein. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
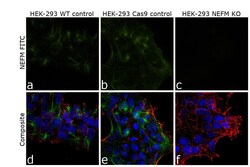
- Experimental details
- Knockout of NEFM was achieved by CRISPR-Cas9 genome editing. Immunofluorescence analysis was performed on wild type HEK-293 cells (panel a,d), HEK-293 Cas9 cells (panels b,e) and HEK-293 NEFM KO cells (panel c,f). Cells were fixed, permeabilized, and labelled with NEFM Monoclonal Antibody (RMO-270) (Product # 13-0700) (5 µg/mL), followed by Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723) (1:2000). Nuclei (blue) were stained using ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962), and Rhodamine Phalloidin (Product # R415) (1:300) was used for cytoskeletal F-actin (red) staining. Loss of signal (panel c,f) upon CRISPR mediated knockout (KO) confirms that antibody is specific to NEFM (green). The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
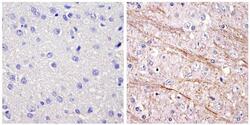
- Immunohistochemistry analysis of the neurofilament medium chain showing staining in the filaments of paraffin-embedded rat brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament medium chain monoclonal antibody (Product # 13-0700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
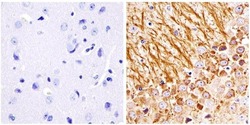
- Immunohistochemistry analysis of the neurofilament medium chain showing staining in the filaments of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament medium chain monoclonal antibody (Product # 13-0700) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
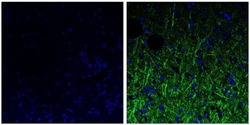
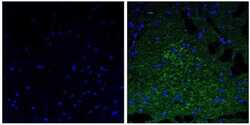
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament medium chain monoclonal antibody (Product # 13-0700) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
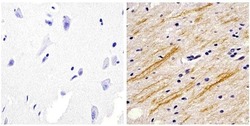
- Immunohistochemistry analysis of the neurofilament medium chain showing staining in the filaments of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament medium chain monoclonal antibody (Product # 13-0700) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament medium chain monoclonal antibody (Product # 13-0700) in 0.3% BSA at a dilution of 1:20 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
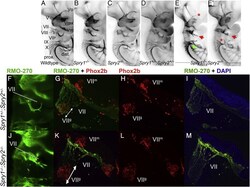
- Fig. 2 Cranial nerve abnormalities in Sprouty mutants. Anti-neurofilament immunohistochemistry revealing developing cranial nerves of wildtype (A), Spry1 -/- (B), Spry2 -/- (C), Spry1 +/- ;Spry2 +/- (D) and Spry1 -/- ;Spry2 -/- (E,E') E10.5 embryos is shown. All embryos were between 34 and 36 somite stage. Standard labelling of the cranial nerves was used, trigeminal ganglion (V) with opthalmic (Vo), maxillary (Vmx) and mandibulary (Vm) branches, facial nerve (VII); vestibulocochlear nerve (VIII); glossopharyngeal nerve (IX) and the vagus nerve (X). Arrows highlight abnormal morphology and asterisks indicate missing portions. The majority of developing cranial nerves present in Spry1 -/- (B), Spry2 -/- (C) and Spry1 +/- ;Spry2 +/- (D) embryos were comparable to wildtype and the latter genotype was used as a control in this study. Spry1 -/- ;Spry2 -/- embryos have trigeminal defects (e.g. absent ophthalmic branch of the trigeminal nerve in E), facial nerve defects and glossopharyngeal and vagus cranial nerves display incomplete or irregular bridging between proximal and distal ganglia (E,E'). (F-M) Neurofilament (RMO-270) and Phox2b immunohistochemistry counter stained with DAPI on sections from E10.5 Spry1 +/- ;Spry2 +/- control (F-I) and Spry1 -/- ;Spry2 -/- (J-M) mutant embryos. Neurofilament staining is indicated with green fluorescence, Phox2b labelling of motor and sensory neuron nuclei in red and DAPI stained nuclei in blue. Labelling of markers and genotypes
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
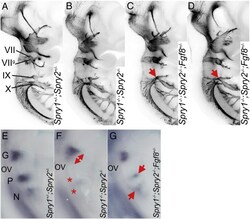
- Fig. 6 Spry1-/-;Spry2-/- epibranchial cranial nerve phenotype is partially rescued by Fgf8 heterozygosity. (A-D) Anti-neurofilament immunohistochemistry showing the developing cranial nerves in Spry1 +/- ;Spry2 +/- , Spry1 -/- ;Spry2 -/- and Spry1 -/- ;Spry2 -/- ; Fgf8 +/- embryos. Note the increased glossopharyngeal nerve fibres in Spry1 -/- ;Spry2 -/- ; Fgf8 +/- embryos (red arrows in C and D) compared to the Spry1 -/- ;Spry2 -/- embryo (B). (E-G) Epibranchial placodes in E9.5 embryos as revealed by a Ngn2 antisense RNA probe. Note the enlarged geniculate (red arrow) and absence of petrosal and nodose placodes (red stars) in the Spry1 -/- ;Spry2 -/- embryo (F), and the rescue of these phenotypes in the Spry1 -/- ;Spry2 -/- ; Fgf8 +/- embryo, especially the presence of the petrosal and nodose placodes (red arrows in G).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
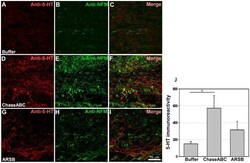
- Figure 7 5-HT immunoreactive axons are more abundant 9 weeks after enzyme treatment than in control mice. One ul ChaseABC (10 U/ml), ARSB (10 U/ml) or buffer was injected at the injury site and 0.5 mm rostral and caudal to this site in mice with severe compression injury. After 9 weeks, the mice were perfused, and sagittal spinal cord sections were analyzed by immunofluorescence. Double immunostaining for serotonin (5-HT) and neurofilament-M (NF-M) shows higher immunoreactivities caudal injury site in the ChaseABC ( D,E,F ) and ARSB ( G,H,I ) treated mice versus buffer treated control mice ( A,B,C ). ( A,D,G ) Immunostainings for 5-HT and ( B,E,H ) NF-M, and ( C,F,I ) merged for 5-HT with NF-M. 5-HT immunoreactive axons are seen beyond the injury site in the ChaseABC and ARSB injected mice. Immunoreactive areas were quantified above threshold using Image J software ( J ). Mean fluorescence intensity of the area between the injury site and 1 mm caudal to it is significantly higher in ChaseABC treated versus buffer treated control mice. Asterisk indicates significant differences between the groups *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
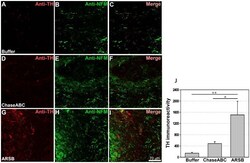
- Figure 8 Tyrosine hydroxylase (TH) immunoreactive axons are more abundant 9 weeks after enzyme treatment than in control mice. One ul ChaseABC (10 U/ml), ARSB (10 U/ml) or buffer was injected at the injury site and 0.5 mm rostral and caudal of this site in mice with severe compression injury. After 9 weeks, the mice were perfused, and sagittal spinal cord sections were analyzed by immunofluorescence. Double immunostaining for TH and neurofilament-M (NF-M) shows higher immunoreactivities caudal to the injury site in the ChaseABC ( D,E,F ) and ARSB ( G,H,I ) treated mice versus the buffer treated control mice ( A,B,C ). ( A,D,G ) Immunostainings for TH and ( B,E,H ) NF-M, and ( C,F,I ) merged for TH with NF-M. Immunoreactive areas were quantified above threshold using Image J software ( J ). Mean fluorescence intensity of the area between the injury site and 1 mm caudal to it shows significantly higher immunofluorescence intensity in ARSB versus buffer treated control and ChaseABC treated mice. Asterisks indicate significant differences between the groups *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
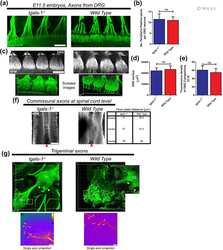
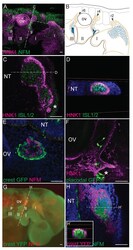
- Fig. 1. Neural crest cells form corridors associated with sensory neuroblasts in chick and mouse. ( A , B ) Lateral view of HH15 + chick embryo showing association of NCCs with neuroblasts in (A) a 3D reconstruction of HNK1 (magenta) and NFM (green) staining, and (B) a schematic representation (NCCs, orange; neuroblasts, blue; general NCC territory is shown by an orange line; the direction of migration is indicated by arrows). ( C , D ) HH17 transverse section (C) and digital coronal re-section (D) reveal HNK1 + NCCs (magenta) peripheral to ISL + neuroblasts (green) (single optical confocal section). ( E ) NCC labelling by GFP electroporation confirms NCCs (green) encircle NFM + neuroblasts (magenta); HH18 coronal section. ( F ) Following placode electroporation, bipolar GFP + cells (green) are seen migrating within the HNK1 + NCC corridor (magenta) (sagittal, HH17, single optical confocal section). ( G ) Lateral view of 29 ss Wnt1cre:R26RYFP mouse embryo shows the association of NCCs (green) with NFM + neuroblasts (red). ( H , H ') Transverse section (H) and digital coronal re-section (H') show YFP + NCCs (magenta) encircling NFM + neuroblasts (green) as in chick (confocal stack projection). Asterisks indicate placodes; I, II and III, pharyngeal arches 1, 2 and 3; NT, neural tube; OV, otic vesicle; p, petrosal; g, geniculate; t, trigeminal ganglia; r, rhombomere. Dotted lines in A indicate the levels of sections in C,E; dotted lines in C indicate the level of the s
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
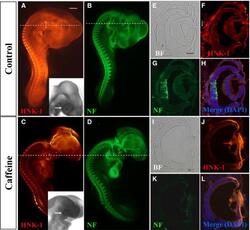
- Figure 2 Caffeine exposure impaired neurofilament (NF) and HNK-1 expression in early chick eyes. HH10 chick embryos were treated with 15 mumol/egg caffeine for 1.5 days and then whole-mount double immunostained with NF and HNK-1 antibodies. (A-D) Control and caffeine-treated embryos stained with HNK-1 (A and C) and NF (B and D) antibodies. Higher magnified bright-field images indicated the eye malformation induced by caffeine (black arrows). (E-H) Transverse sections of A and B (dotted white lines) showing the expression pattern of HNK-1 and NF in the control. There are some HNK-1 + neural crest cells surrounding the eyes in control embryo (white arrow in F), but absent in caffeine-treated ones. (I-L) Transverse sections of C and D (dotted white lines) showing HNK-1 and NF expression in caffeine-treated eyes is distinctly different from control eyes. BF, bright-field. Scale bars = 1 mm in A-D; 100 mum in E-L.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
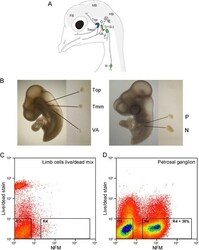
- Fig. 1 Isolation of embryonic chick placode-derived cranial sensory neurons by dissection and FACS. A Schematic of cranial sensory ganglia in embryonic day 12 (HH38) chick (adapted from [ 6 ]). The ganglia are labelled: trigeminal ganglion as two separate lobes (Top: ophthalmic; Tmm: maxillomandibular); vestibulo-acoustic ganglion (VA); epibranchial series as three separate ganglia (G: geniculate; P: petrosal; N: nodose). Also labelled are the neural crest-derived superior- jugular ganglionic complex (S/J); the inner ear (IE); and forebrain (FB); midbrain (MB) and hindbrain (HB) of the CNS. Colours indicate the sensory modality of ganglion: blue: general somatosensory; magenta: special somatosensory; green: viscerosensory. B Representative dissections of cranial sensory ganglia: Top, Tmm and VA at HH18, and P and N at HH23. C , D Representative FACS plots of cells stained for live/dead stain and NFM. C Control cell population: limb bud cells devoid of neurons, containing 50 % dead cells. D Petrosal ganglion cell population: 36 % of cells are NFM positive and dead cell marker negative
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
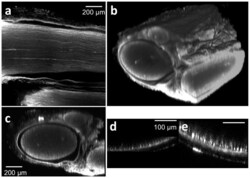
- Figure 1 Tissue clearing following antibody staining allows for visualization of axons throughout entire rat sciatic nerves. Tissue were stained with anti-neurofilament antibodies using a whole mount immunofluorescence method. (a) A lateral (xy) confocal image at the depth of approximately 400 mum from the surface of a stained and cleared wild type rat sciatic nerve. (b) Three dimensional view of a whole stained and cleared sciatic nerve. Non-specific GFP epineurial staining can also be seen secondary to presence of fibroblasts, collagen, and vasa nervorum in the epineurium, which does not affect analysis. (c) Reconstructed transverse cross sectional image. Three fascicles surrounded by the epineural sheath can be recognized. For comparison, a transverse cross sectional image of non-tissue cleared, stained sciatic nerve is shown in (d); note that non-specific staining occurs in the epineurial sheath as well, but only the right portion of the sheath is observed. (e) Two-photon transverse cross sectional image of a sciatic nerve from a transgenic rat (thy-1 GFP).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
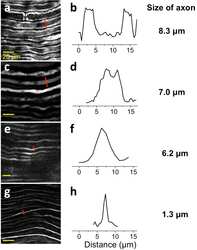
- Figure 2 Deep tissue and high resolution images of tissue cleared nerves by confocal microscopy following anti-neurofilament antibody labeling. (a) Lateral (xy) confocal image. (b) Cross sectional image. The elongated shape of the axons in the z direction is caused by the lower relative axial resolution of confocal microscopy.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
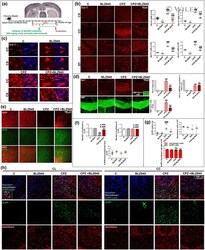
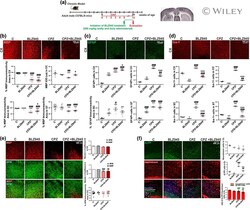
- Impact of preventive BLZ945 treatment on microglia and neurodegeneration in chronic CPZ-induced demyelination. (a) Experimental procedure. (b) Immunohistochemical studies of Iba-1 in the cortex (CX), corpus callosum (CC), external capsule (EC), and striatum (ST). Quantification of Iba-1+ cells. (c) High magnification confocal images of Iba-1+ cells in CX and CC to evaluate morphological changes. (d) Immunohistochemical studies of Iba-1 and MBP in CC. Quantification of Iba-1+ cells, MBP+Iba-1+ cells and MBP+Iba-1+/Iba-1+ ratio in CC. (e) Immunohistochemical studies of MBP and NFM in the CX. (f) Quantitative colocalization assessment by Mander's coefficient . (g) Quantification of Neurotrace+ cells, AbetaPP+ cells and AbetaPP+ /Neurotrace+ ratio. (h) Immunohistochemical studies of AbetaPP+ cells in cingulum (CL) and CC. Values represent the mean +- SD of three different mice: * p < .05, *** p < .001 as compared with C; ### p < .001 as compared with BLZ945; ++ p < .01, +++ p < .001 as compared with CPZ using one-way ANOVA followed by Bonferroni post hoc tests [Color figure can be viewed at wileyonlinelibrary.com ]
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Effect of therapeutic BLZ945 treatment on chronic CPZ-induced demyelination. (a) Experimental procedure . (b) Immunohistochemical studies of MBP in the cortex (CX) . Quantification of MBP immunoreactivity and IOD in the CX, corpus callosum (CC), external capsule (EC), and striatum (ST) . (c) Immunohistochemical studies of GFAP in the CX. Quantification of GFAP+ cells in the CX, CC, EC and ST . (d) Immunohistochemical studies of Iba-1 in the CX. Quantification of Iba-1+ cells in the CX, CC, EC and ST. (e) Immunohistochemical studies of MBP and NFM in the CX. Quantitative colocalization assessment by Mander's coefficient. (f) Immunohistochemical studies of AbetaPP+ cells. Quantification of Neurotrace+ cells, AbetaPP+ cells and AbetaPP+/Neurotrace+ ratio. Values represent the mean +- SD of three different mice: * p < .05, ** p < .01, *** p < .001 as compared with C; # p < .05, ## p < .01, ### p < .001 as compared with BLZ945; + p < .05, +++ p < .001 as compared with CPZ using one-way ANOVA followed by Bonferroni post hoc tests [Color figure can be viewed at wileyonlinelibrary.com ]
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
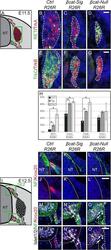
- Figure 4 Late subtype specification is altered and boundary cap cell identity is lost in mutant animals. (A) Illustration of a transverse section at E11.5 displaying cells, which contribute to the DRG in red. Green box represents caption area for subfigures B-G. (B-G) Triple immunohistochemistry at E11.5 for beta-gal expressed by the R26R reporter and for markers for subtype specification of sensory neurons: tyrosine receptor kinases RET and TrkA, TrkB and TrkC. Dashed lines surround the border of R26R -positive cells contributing to the DRG. (H) Quantification of late sensory subtypes per DRG displays an altered distribution of sensory neuron subtypes in mutant animals. (I) Illustration of a transverse section at E12.5 displaying boundary cap cells in red. Green box represents caption area for subfigures J-O, respectively. (J-L) Double immunohistochemistry for Krox20 as a marker for differentiated boundary cap cells and neurofilament (NF) at E12.5. (M-O) Triple immunohistochemistry for Krox20, NF and Islet1/2 as a marker for motor neurons at E12.5. (J',M') In control animals, sensory axons invading the dorsal entry zone of the neural tube and motor axons penetrating the prospective motor exit point of the neural tube are surrounded by boundary cap cells. (K',L',N',O') Dorsal as well as ventral Krox20 expression is lost in both mutant animals. Arrows show migrating motor neurons, which are exiting or have already exited the ventral neural tube du
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
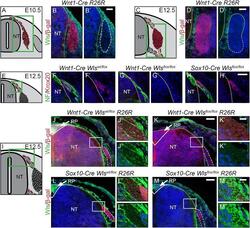
- Figure 7 Development of boundary cap cells depends on neural tube-derived Wnt. (A, C) Illustration of a transverse section at E10.5 (A) and E12.5 (C) displaying cells contributing to the DRG in red. Green box represents caption area for subfigures B and D. respectively. (B, D) Double immunohistochemistry for Wls and beta-gal expressed in Wnt1-Cre R26R mice shows that mesenchymal tissue and not cells populating the DRG express Wls. (B', D') Corresponding single fluorescence channels of the same sections. Dashed lines frame lineage-traced cells. (E, I) Illustration of a transverse section at E12.5 displaying BCCs in red. Green box represents caption area for subfigures F-H and J-M, respectively. (F-H) Double immunohistochemistry for Krox20 and neurofilament (NF). (F'-H') Corresponding single fluorescence channels of the same sections. (G') Dorsal BCCs are lost upon conditional knock-out of Wls using Wnt1-Cre . (H') However, BCCs are rescued in conditional knockout of Wls using Sox10-Cre . (J-M) Double immunohistochemistry for Wls and beta-gal expressed by the Cre reporter line R26R . J'-M' and J""-M"" are higher magnifications of the boxed areas in J-M, respectively. Dashed lines frame sensory axons . (J-J"", L-L"") Wls is expressed at the site where sensory axons enter the dorsal neural tube at the dorsal entry zone and strongly expressed in the roofplate (arrow, J, L) of control animals. (K-K"") Expression of Wls is lost in dorsal neura
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
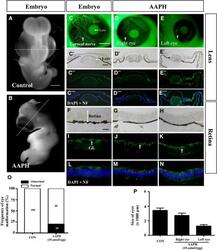
- Figure 6 AAPH exposure replicates the effect of caffeine on the developing eye. Early chick embryos exposed to 10 mumol/egg AAPH for 6 days and immunostained with NF antibody. (A and B) Control (A) and AAPH-treated (B) chick embryos. (C-E) NF immunostaining demonstrates the presence of neurons in control (C) and AAPH-treated (right eye, D) and (left eye, E) lens. The staining shows AAPH-treatment repressed neuron development in the eyes. (C'-E''') Transverse section of the lens at the levels indicated by white line in A and B. Bright-field (C', D', E'), NF immunostaining (C'', D'', E'') and NF immunostaining + DAPI (C''', D''', E'''). (O and P) Bar charts showing AAPH significantly increased the incidence of eye malformation (O) and reduced the size (diameter) of the eyes (P). NF, neurofilament. Scale bars = 2 mm in A and B; 1 mm in C-E; 200 mum in F-N.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry