Antibody data
- Antibody Data
- Antigen structure
- References [10]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [1]
- Immunohistochemistry [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 34-1000 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NEFM Monoclonal Antibody (RMO 14.9)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Mouse, Rat, Chicken/Avian, Rabbit
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- RMO 14.9
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Long-term cognitive impairment without diffuse axonal injury following repetitive mild traumatic brain injury in rats.
Fasudil may induce the differentiation of bone marrow mesenchymal stem cells into neuron‑like cells via the Wnt/β‑catenin pathway.
Neuronal Injury and Glial Changes Are Hallmarks of Open Field Blast Exposure in Swine Frontal Lobe.
Temporal Sequence of Autolysis in the Cerebellar Cortex of the Mouse.
Neuroaxonal dystrophy in Australian Merino lambs.
Temporal assessment of traumatic axonal injury in the rat corpus callosum and optic chiasm.
A comparison of differentiation protocols for RGC-5 cells.
Selective induction of ultrastructural (neurofilament) compaction in axons by means of a new head-injury apparatus.
Corticotropin-releasing factor 2 receptor localization in skeletal muscle.
Neural differentiation of mouse embryonic stem cells in vitro and after transplantation into eyes of mutant mice with rapid retinal degeneration.
Tadepalli SA, Bali ZK, Bruszt N, Nagy LV, Amrein K, Fazekas B, Büki A, Czeiter E, Hernádi I
Behavioural brain research 2020 Jan 27;378:112268
Behavioural brain research 2020 Jan 27;378:112268
Fasudil may induce the differentiation of bone marrow mesenchymal stem cells into neuron‑like cells via the Wnt/β‑catenin pathway.
Hu Y, Li X, Huang G, Wang J, Lu W
Molecular medicine reports 2019 Apr;19(4):3095-3104
Molecular medicine reports 2019 Apr;19(4):3095-3104
Neuronal Injury and Glial Changes Are Hallmarks of Open Field Blast Exposure in Swine Frontal Lobe.
Kallakuri S, Desai A, Feng K, Tummala S, Saif T, Chen C, Zhang L, Cavanaugh JM, King AI
PloS one 2017;12(1):e0169239
PloS one 2017;12(1):e0169239
Temporal Sequence of Autolysis in the Cerebellar Cortex of the Mouse.
Finnie JW, Blumbergs PC, Manavis J
Journal of comparative pathology 2016 May;154(4):323-8
Journal of comparative pathology 2016 May;154(4):323-8
Neuroaxonal dystrophy in Australian Merino lambs.
Kessell AE, Finnie JW, Blumbergs PC, Manavis J, Jerrett IV
Journal of comparative pathology 2012 Jul;147(1):62-72
Journal of comparative pathology 2012 Jul;147(1):62-72
Temporal assessment of traumatic axonal injury in the rat corpus callosum and optic chiasm.
Zakaria N, Kallakuri S, Bandaru S, Cavanaugh JM
Brain research 2012 Jul 27;1467:81-90
Brain research 2012 Jul 27;1467:81-90
A comparison of differentiation protocols for RGC-5 cells.
Wood JP, Chidlow G, Tran T, Crowston JG, Casson RJ
Investigative ophthalmology & visual science 2010 Jul;51(7):3774-83
Investigative ophthalmology & visual science 2010 Jul;51(7):3774-83
Selective induction of ultrastructural (neurofilament) compaction in axons by means of a new head-injury apparatus.
Pál J, Tóth Z, Farkas O, Kellényi L, Dóczi T, Gallyas F
Journal of neuroscience methods 2006 Jun 15;153(2):283-9
Journal of neuroscience methods 2006 Jun 15;153(2):283-9
Corticotropin-releasing factor 2 receptor localization in skeletal muscle.
Samuelsson S, Lange JS, Hinkle RT, Tarnopolsky M, Isfort RJ
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jul;52(7):967-77
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jul;52(7):967-77
Neural differentiation of mouse embryonic stem cells in vitro and after transplantation into eyes of mutant mice with rapid retinal degeneration.
Meyer JS, Katz ML, Maruniak JA, Kirk MD
Brain research 2004 Jul 16;1014(1-2):131-44
Brain research 2004 Jul 16;1014(1-2):131-44
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of NEFM was performed by loading 20 µg of HEK-293 wild type (Lane 1), HEK-293 Cas9 control (Lane 2), HEK-293 NEFM knockout (Lane 3) membrane enriched cell extracts. The blot was probed with Anti-NEFM Monoclonal Antibody (Product # 34-1000) (1:1000 dilution) and Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177) (1:4000 dilution). Loss of signal upon CRISPR mediated knockout (KO) confirms that antibody is specific to NEFM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-NEFM Monoclonal Antibody (RMO 14.9) (Product # 34-1000) and a 160 kDa band corresponding to NEFM was observed in HEK-293 cell line, Mouse Cerebellum, Rat Brain but not in HeLa, Hep G2 cell line, Mouse Heart and Mouse Spleen which are reported to be negative. Membrane extracts (30 µg lysate) of HEK-293 (Lane 1), HeLa (Lane 2), Hep G2 (Lane 3), tissue extracts of Mouse Cerebellum (Lane 4), Mouse Brain (Lane 5), Rat Brain (Lane 6), Mouse Heart (Lane 7) and Mouse Spleen (Lane 8) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (2ug/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
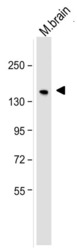
- Experimental details
- Western blot analysis of neurofilament, medium chain was performed by loading 25 µg of mouse brain tissue lysate per well onto a polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked. NF-M was detected at ~160 kD using a neurofilament, medium chain antibody (Product # 13-0400) at a dilution of 2 µg/mL in blocking buffer overnight at 4C, followed by a HRP-labeled secondary antibody for 1 hour at room temperature and detection with a chemiluminescent substrate.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of NF-M was performed using 70% confluent log phase HEK-293 and HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. HEK-293 cells were labeled with NF-M Mouse Monoclonal Antibody (Product # 34-1000) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415). Panel d represents the merged image of HEK-293 showing cytoskeletal (intermediate filaments) localization. Panel e represents the merged image of HeLa cells showing no expression for NF-M protein. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
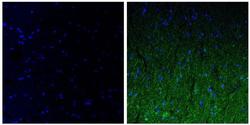
- Experimental details
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament light chain monoclonal antibody (Product # 34-1000) in 0.3% BSA at a dilution of 1:50 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of the neurofilament medium chain in paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). Tissue sections were deparaffinized with xylene, and rehydrated with ethanol. To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were washed with water and PBS, and then blocked in 0.3% BSA for 30 min at room temperature. Tissues were then probed with a neurofilament light chain monoclonal antibody (Product # 34-1000) in 0.3% BSA at a dilution of 1:50 for 1 hour at 37°C. Tissues were then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, DyLight 488 conjugate for 1 hour at 37°C (green). Nuclei (blue) were stained with DAPI. Images were taken at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of the neurofilament medium chain showing staining in the filaments of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0) and microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Neurofilament medium chain monoclonal antibody (Product # 34-1000) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA