13-7096-85
antibody from Invitrogen Antibodies
Targeting: CCL2
GDCF-2, HC11, MCAF, MCP-1, MCP1, MGC9434, SCYA2, SMC-CF
Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-7096-85 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CCL2 (MCP-1) Monoclonal Antibody (2H5), Biotin, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 2H5 monoclonal antibody reacts with mouse, rat, and human monocyte chemoattractant protein-1 (MCP-1), also known as CCL2 and MCAF.
- Conjugate
- Biotin
- Antibody clone number
- 2H5
- Concentration
- 0.5 mg/mL
Submitted references Apoptotic vesicles restore liver macrophage homeostasis to counteract type 2 diabetes.
Cholangiocyte-Derived Exosomal lncRNA H19 Promotes Macrophage Activation and Hepatic Inflammation under Cholestatic Conditions.
Development and validation of an immunosensor for monocyte chemotactic protein 1 using a silicon photonic microring resonator biosensing platform.
Magnetically-actuated, bead-enhanced silicon photonic immunosensor.
Conditioned medium from umbilical cord mesenchymal stem cells induces migration and angiogenesis.
Oxidized low-density lipoprotein induces long-term proinflammatory cytokine production and foam cell formation via epigenetic reprogramming of monocytes.
The liver X receptor agonist GW3965 improves recovery from mild repetitive traumatic brain injury in mice partly through apolipoprotein E.
CCR2 defines a distinct population of NK cells and mediates their migration during influenza virus infection in mice.
Synergistic drug combinations tend to improve therapeutically relevant selectivity.
Mechanisms underlying renoprotection during renin-angiotensin system blockade.
Mechanisms underlying renoprotection during renin-angiotensin system blockade.
Macrophage inflammatory protein-2 and KC induce chemokine production by mouse astrocytes.
Zheng C, Sui B, Zhang X, Hu J, Chen J, Liu J, Wu D, Ye Q, Xiang L, Qiu X, Liu S, Deng Z, Zhou J, Liu S, Shi S, Jin Y
Journal of extracellular vesicles 2021 May;10(7):e12109
Journal of extracellular vesicles 2021 May;10(7):e12109
Cholangiocyte-Derived Exosomal lncRNA H19 Promotes Macrophage Activation and Hepatic Inflammation under Cholestatic Conditions.
Li X, Liu R, Wang Y, Zhu W, Zhao D, Wang X, Yang H, Gurley EC, Chen W, Hylemon PB, Zhou H
Cells 2020 Jan 11;9(1)
Cells 2020 Jan 11;9(1)
Development and validation of an immunosensor for monocyte chemotactic protein 1 using a silicon photonic microring resonator biosensing platform.
Valera E, Shia WW, Bailey RC
Clinical biochemistry 2016 Jan;49(1-2):121-6
Clinical biochemistry 2016 Jan;49(1-2):121-6
Magnetically-actuated, bead-enhanced silicon photonic immunosensor.
Valera E, McClellan MS, Bailey RC
Analytical methods : advancing methods and applications 2015 Oct 21;7(20):8539-8544
Analytical methods : advancing methods and applications 2015 Oct 21;7(20):8539-8544
Conditioned medium from umbilical cord mesenchymal stem cells induces migration and angiogenesis.
Shen C, Lie P, Miao T, Yu M, Lu Q, Feng T, Li J, Zu T, Liu X, Li H
Molecular medicine reports 2015 Jul;12(1):20-30
Molecular medicine reports 2015 Jul;12(1):20-30
Oxidized low-density lipoprotein induces long-term proinflammatory cytokine production and foam cell formation via epigenetic reprogramming of monocytes.
Bekkering S, Quintin J, Joosten LA, van der Meer JW, Netea MG, Riksen NP
Arteriosclerosis, thrombosis, and vascular biology 2014 Aug;34(8):1731-8
Arteriosclerosis, thrombosis, and vascular biology 2014 Aug;34(8):1731-8
The liver X receptor agonist GW3965 improves recovery from mild repetitive traumatic brain injury in mice partly through apolipoprotein E.
Namjoshi DR, Martin G, Donkin J, Wilkinson A, Stukas S, Fan J, Carr M, Tabarestani S, Wuerth K, Hancock RE, Wellington CL
PloS one 2013;8(1):e53529
PloS one 2013;8(1):e53529
CCR2 defines a distinct population of NK cells and mediates their migration during influenza virus infection in mice.
van Helden MJ, Zaiss DM, Sijts AJ
PloS one 2012;7(12):e52027
PloS one 2012;7(12):e52027
Synergistic drug combinations tend to improve therapeutically relevant selectivity.
Lehár J, Krueger AS, Avery W, Heilbut AM, Johansen LM, Price ER, Rickles RJ, Short GF 3rd, Staunton JE, Jin X, Lee MS, Zimmermann GR, Borisy AA
Nature biotechnology 2009 Jul;27(7):659-66
Nature biotechnology 2009 Jul;27(7):659-66
Mechanisms underlying renoprotection during renin-angiotensin system blockade.
Taal MW, Chertow GM, Rennke HG, Gurnani A, Jiang T, Shahsafaei A, Troy JL, Brenner BM, Mackenzie HS
American journal of physiology. Renal physiology 2001 Feb;280(2):F343-55
American journal of physiology. Renal physiology 2001 Feb;280(2):F343-55
Mechanisms underlying renoprotection during renin-angiotensin system blockade.
Taal MW, Chertow GM, Rennke HG, Gurnani A, Jiang T, Shahsafaei A, Troy JL, Brenner BM, Mackenzie HS
American journal of physiology. Renal physiology 2001 Feb;280(2):F343-55
American journal of physiology. Renal physiology 2001 Feb;280(2):F343-55
Macrophage inflammatory protein-2 and KC induce chemokine production by mouse astrocytes.
Luo Y, Fischer FR, Hancock WW, Dorf ME
Journal of immunology (Baltimore, Md. : 1950) 2000 Oct 1;165(7):4015-23
Journal of immunology (Baltimore, Md. : 1950) 2000 Oct 1;165(7):4015-23
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
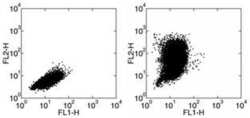
- Experimental details
- Staining of mouse thiogylcolate induced peritoneal exudate cells (PECs) stimulated with LPS in the presence of Brefeldin A for 24 hours. with Anti-mouse F4/80 FITC (Product # 11-4801-82) followed by intracellular staining with Rat IgG2a kappa Isotype Control PE (Product # 12-4321-82) (left) or Anti-Mouse CCL2 PE.
- Conjugate
- Biotin
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
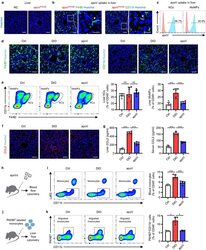
- Experimental details
- FIGURE 3 Efferocytosis of MSC-derived apoVs by liver macrophages alleviates macrophage infiltration in the type 2 diabetes (T2D) liver. (a) Representative confocal microscopy images showing distribution of PKH26-labeled apoVs (red) in the liver, counterstained by Hoechst (blue). After removal of unbound PKH, the stained apoVs were resuspended in PBS and underwent centrifugation, after which the supernatant was used as the negative control (NC) and injected. Scale bars, 50 mum. (b) Representative confocal microscopy images showing uptake of apoVs (red) by macrophages (green) in the liver, counterstained by Hoechst (blue). Scale bars, 50 mum in low magnification images and 25 mum in high magnification images. (c) Flow cytometric analysis showing the uptake of apoVs by macrophages in the liver. KCs, Kupffer cells; MoMFs, monocyte-derived macrophages. (d) Representative immunofluorescent (IF) staining images of F4/80 (green) and CD11b (green) in the liver, counterstained by Hoechst (blue). Ctrl, control mice; DIO, mice with diet-induced obesity; apoV, DIO mice treated by apoVs. Scale bars, 50 mum. (e) Flow cytometric analysis and the corresponding quantification of the percentages of KCs and MoMFs in hepatic CD45 + cells. N = 6 per group. (f) Representative IF staining images of chemokine (C-C motif) ligand 2 (CCL2) (red) in the liver, counterstained by Hoechst (blue). Scale bars, 50 mum. (g) Enzyme-linked immunosorbent assay (ELISA) analysis of CCL2 in liver lysate and serum. N
- Conjugate
- Biotin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
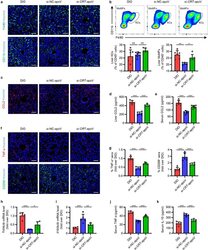
- Experimental details
- FIGURE 8 CRT mediates efferocytosis of MSC-derived apoVs to modulate T2D liver macrophages in vivo. (a) Representative immunofluorescent (IF) staining images of F4/80 (green) and CD11b (green) in the liver, counterstained by Hoechst (blue). DIO, mice with diet-induced obesity; si-NC-apoV, DIO mice treated by apoVs derived from MSCs transfected by siRNA-negative control; si- CRT -apoV, DIO mice treated by apoVs derived from MSCs transfected by siRNA- CRT . Scale bars, 50 mum. (b) Flow cytometric analysis and the corresponding quantification of the percentages of KCs and MoMFs in hepatic CD45 + cells. KCs, Kupffer cells; MoMFs, monocyte-derived macrophages. N = 5-6 per group. (c) Representative IF staining images of chemokine (C-C motif) ligand 2 (CCL2) (red) in the liver, counterstained by Hoechst (blue). Scale bars, 50 mum. (d and e) ELISA analysis of CCL2 in liver lysate (d) and serum (e). N = 6 per group. (f and g) Representative IF staining images of tumor necrosis factor-alpha (TNF-alpha) (red) and CD206 (green) in the liver, counterstained by Hoechst (blue), and the corresponding quantification of fold changes over the DIO group. Scale bars, 50 mum. N = 5-6 per group. (h and i) Quantitative real time polymerase chain reaction (qRT-PCR) analysis of mRNA expression levels of Tnf (h) and interleukin 10 ( Il10 ) (i) in the liver, normalized to beta-actin ( Actb ), and quantification of fold changes over the DIO group. N = 5 per group. (j and k) Enzyme-linked immunosorbent as
- Conjugate
- Biotin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
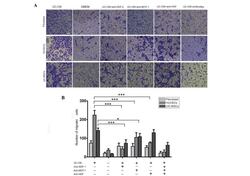
- Experimental details
- Figure 5 Migration of fibroblasts, HUVECs and UC-MSCs in response to UC-CM. (A) A total of 5x10 4 cells were collected and allowed to migrate. Lane 1, UC-CM; lane 2, DMEM; lanes 3-6, in the presence or absence of anti-SDF-1 (20 mu g/ml), anti-MCP-1 (20 mu g/ml) or anti-HGF (20 mu g/ml), respectively. Results are from a representative experiment and are expressed as the mean number of migrated cells in three random fields, scale bar=200 mu m. Cells that crossed the matrigel membrane were stained with crystal violet (magnification, x40). (B) Graphical presentation of the quantified data, presented as the number of migrated cells and expressed as the mean +- standard error of the mean. HUVECs, human umbilical vein endothelial cells; UC-MSCs, umbilical cord mesenchymal stem cells; UC-CM, UC-MSCs conditioned medium; DMEM, Dulbecco's modified Eagle's medium; SDF-1, stromal cell-derived factor 1; MCP-1, monocyte chemotactic protein 1; HGF, hepatocyte growth factor.
- Conjugate
- Biotin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
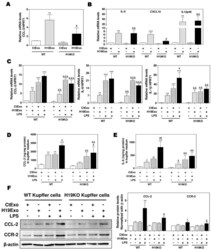
- Experimental details
- Figure 1 Cholangiocyte-derived exosomal H19 promotes Kupffer activation. ( A , B ) Mouse Kupffer cells were treated with control mouse large cholangiocytes (MLE)-derived exosomes (CtExo) and H19-overexpressing MLE-derived exosomes (H19Exo) for 24 h. ( A , B ) The relative mRNA levels of chemokine (C-C motif) ligand 2 (CCL-2), interleukin-6 (IL-6), chemokine (C-X-C motif) ligand 10 (CXCL10), and IL12p40 were measured by real-time RT-PCR and normalized using HPRT1 (Hypoxanthine-guanine phosphoribosyltransferase 1). ( C - F ) Mouse Kupffer cells were treated with CtExo, H19Exo, and lipopolysaccharide (LPS) (10 ng/mL) for 24 h. ( C ) The relative mRNA levels of CCL-2, IL-6, and IL-1beta were measured by real-time RT-PCR and normalized using HPRT1. ( D , E ) The protein levels of CCL-2 and IL-6 in the conditioned medium of Kupffer cells were measured by ELISA assay and normalized using protein concentration. ( F ) Representative immune blot images of CCL-2 and CCR-2 are shown. Relative protein levels were normalized using beta-actin. Results from at least three independent experiments are presented as Mean +- SEM. Statistical significance: * p < 0.05, ** p < 0.01, *** p < 0.001, compared with WT control group; # p < 0.05, ## p < 0.01, compared with WT H19Exo group; & p < 0.05, && p < 0.01, &&& p < 0.001, compared with H19KO control group; $ p < 0.05, compared with H19KO H19Exo group.
- Conjugate
- Biotin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
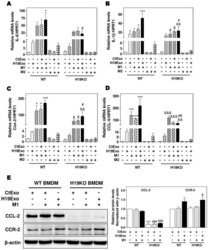
- Experimental details
- Figure 2 Cholangiocyte-derived exosomal H19 promotes BMDM polarization. ( A , B ) Mouse bone marrow-derived macrophage (BMDM) cells were treated with CtExo, H19Exo, M1 (LPS, 10 ng/mL and IFN-gamma, 100 ng/mL) or M2 stimulators (IL-4, 20 ng/mL and IL-13, 20 ng/mL) for 24 h. ( A - D ) The relative mRNA levels of IL-6, IL-1beta, Cox-2, and CCL-5 were measured by real-time RT-PCR and normalized using HPRT1. ( E ) Representative immune blot images of CCL-2 and CCR-2 are shown. Relative protein levels were normalized using beta-actin. Results from at least three independent experiments are presented as Mean +- SEM. Statistical significance: * p < 0.05, *** p < 0.001, compared with WT control group; # p < 0.05, ## p < 0.01, ### p < 0.001, compared with WT H19Exo group; $$$ p < 0.001, compared with WT H19Exo+M1 group; & p < 0.05, && p < 0.01, &&& p < 0.001, compared with H19KO control group.
- Conjugate
- Biotin
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
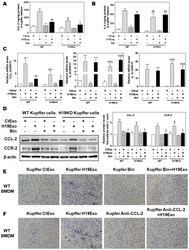
- Experimental details
- Figure 4 Effect of CCL-2 on exosomal H19-induced macrophage activation and migration. ( A-F ) Mouse Kupffer cells were treated with CtExo or H19Exo for 24 h, with or without pretreatment with Bindarit (Bin, 300 muM) and purified CCL-2 antibody (Anti-CCL-2, 20 mug/mL) for 2 h. ( A , B ) The levels of CCL-2 and IL-6 in the conditioned medium of Kupffer cells were measured by ELISA assay and normalized using protein concentration. ( C ) The relative mRNA levels of CCL-2, IL-6, and IL-1beta were measured by real-time RT-PCR and normalized using HPRT1. ( D ) Representative immune blot images of CCL-2 and CCR-2 are shown. Relative protein levels were normalized using beta-actin. ( E , F ) Representative images of migration assay are shown. Results from at least three independent experiments are presented as Mean +- SEM. Statistical significance: * p < 0.05, ** p < 0.01, *** p < 0.001, compared with WT control group; # p < 0.05, ## p < 0.01, compared with WT H19Exo group; & p < 0.05, && p < 0.01, &&& p < 0.001, compared with H19KO control group; $ p < 0.05, compared with H19KO H19Exo group.
- Conjugate
- Biotin
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry