MAB1660
antibody from R&D Systems
Targeting: CFTR
ABC35, ABCC7, CF, CFTR/MRP, dJ760C5.1, MRP7, TNR-CFTR
Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Immunohistochemistry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MAB1660 - Provider product page

- Provider
- R&D Systems
- Product name
- Human CFTR R Domain Antibody
- Antibody type
- Monoclonal
- Description
- Protein A or G purified from hybridoma culture supernatant. Detects human CFTR. Specifically recognizes in vitro synthesized CFTR, recombinant CFTR protein, and non-recombinant CFTR protein (1, 2).
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Unconjugated
- Antigen sequence
P13569- Isotype
- IgG
- Antibody clone number
- 13-1
- Vial size
- 50 ug
- Concentration
- LYOPH
- Storage
- Use a manual defrost freezer and avoid repeated freeze-thaw cycles. 12 months from date of receipt, -20 to -70 °C, as supplied. 1 month, 2 to 8 °C under sterile conditions after opening. 6 months, -20 to -70 °C under sterile conditions after opening.
Submitted references Synergy of cAMP and calcium signaling pathways in CFTR regulation.
Mutating the Conserved Q-loop Glutamine 1291 Selectively Disrupts Adenylate Kinase-dependent Channel Gating of the ATP-binding Cassette (ABC) Adenylate Kinase Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) and Reduces Channel Function in Primary Human Airway Epithelia.
A novel chimeric adenoassociated virus 2/human bocavirus 1 parvovirus vector efficiently transduces human airway epithelia.
Defective CFTR expression and function are detectable in blood monocytes: development of a new blood test for cystic fibrosis.
Identification of a bone marrow-derived epithelial-like population capable of repopulating injured mouse airway epithelium.
Human-specific cystic fibrosis transmembrane conductance regulator antibodies detect in vivo gene transfer to ovine airways.
Chloride channels in the small intestinal cell line IEC-18.
Protection of Cftr knockout mice from acute lung infection by a helper-dependent adenoviral vector expressing Cftr in airway epithelia.
Mitochondria-rich cells as experimental model in studies of epithelial chloride channels.
Mitochondria-rich cells as experimental model in studies of epithelial chloride channels.
Reciprocal protein kinase A regulatory interactions between cystic fibrosis transmembrane conductance regulator and Na+/H+ exchanger isoform 3 in a renal polarized epithelial cell model.
Localization of cystic fibrosis transmembrane conductance regulator in chloride secretory epithelia.
Bozoky Z, Ahmadi S, Milman T, Kim TH, Du K, Di Paola M, Pasyk S, Pekhletski R, Keller JP, Bear CE, Forman-Kay JD
Proceedings of the National Academy of Sciences of the United States of America 2017 Mar 14;114(11):E2086-E2095
Proceedings of the National Academy of Sciences of the United States of America 2017 Mar 14;114(11):E2086-E2095
Mutating the Conserved Q-loop Glutamine 1291 Selectively Disrupts Adenylate Kinase-dependent Channel Gating of the ATP-binding Cassette (ABC) Adenylate Kinase Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) and Reduces Channel Function in Primary Human Airway Epithelia.
Dong Q, Ernst SE, Ostedgaard LS, Shah VS, Ver Heul AR, Welsh MJ, Randak CO
The Journal of biological chemistry 2015 May 29;290(22):14140-53
The Journal of biological chemistry 2015 May 29;290(22):14140-53
A novel chimeric adenoassociated virus 2/human bocavirus 1 parvovirus vector efficiently transduces human airway epithelia.
Yan Z, Keiser NW, Song Y, Deng X, Cheng F, Qiu J, Engelhardt JF
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Dec;21(12):2181-94
Molecular therapy : the journal of the American Society of Gene Therapy 2013 Dec;21(12):2181-94
Defective CFTR expression and function are detectable in blood monocytes: development of a new blood test for cystic fibrosis.
Sorio C, Buffelli M, Angiari C, Ettorre M, Johansson J, Vezzalini M, Viviani L, Ricciardi M, Verzè G, Assael BM, Melotti P
PloS one 2011;6(7):e22212
PloS one 2011;6(7):e22212
Identification of a bone marrow-derived epithelial-like population capable of repopulating injured mouse airway epithelium.
Wong AP, Keating A, Lu WY, Duchesneau P, Wang X, Sacher A, Hu J, Waddell TK
The Journal of clinical investigation 2009 Feb;119(2):336-48
The Journal of clinical investigation 2009 Feb;119(2):336-48
Human-specific cystic fibrosis transmembrane conductance regulator antibodies detect in vivo gene transfer to ovine airways.
Davidson H, McLachlan G, Wilson A, Boyd AC, Doherty A, MacGregor G, Davies L, Painter HA, Coles R, Hyde SC, Gill DR, Amaral MD, Collie DD, Porteous DJ, Penque D
American journal of respiratory cell and molecular biology 2006 Jul;35(1):72-83
American journal of respiratory cell and molecular biology 2006 Jul;35(1):72-83
Chloride channels in the small intestinal cell line IEC-18.
Basavappa S, Vulapalli SR, Zhang H, Yule D, Coon S, Sundaram U
Journal of cellular physiology 2005 Jan;202(1):21-31
Journal of cellular physiology 2005 Jan;202(1):21-31
Protection of Cftr knockout mice from acute lung infection by a helper-dependent adenoviral vector expressing Cftr in airway epithelia.
Koehler DR, Sajjan U, Chow YH, Martin B, Kent G, Tanswell AK, McKerlie C, Forstner JF, Hu J
Proceedings of the National Academy of Sciences of the United States of America 2003 Dec 23;100(26):15364-9
Proceedings of the National Academy of Sciences of the United States of America 2003 Dec 23;100(26):15364-9
Mitochondria-rich cells as experimental model in studies of epithelial chloride channels.
Willumsen NJ, Amstrup J, Møbjerg N, Jespersen A, Kristensen P, Larsen EH
Biochimica et biophysica acta 2002 Nov 13;1566(1-2):28-43
Biochimica et biophysica acta 2002 Nov 13;1566(1-2):28-43
Mitochondria-rich cells as experimental model in studies of epithelial chloride channels.
Willumsen NJ, Amstrup J, Møbjerg N, Jespersen A, Kristensen P, Larsen EH
Biochimica et biophysica acta 2002 Nov 13;1566(1-2):28-43
Biochimica et biophysica acta 2002 Nov 13;1566(1-2):28-43
Reciprocal protein kinase A regulatory interactions between cystic fibrosis transmembrane conductance regulator and Na+/H+ exchanger isoform 3 in a renal polarized epithelial cell model.
Bagorda A, Guerra L, Di Sole F, Hemle-Kolb C, Cardone RA, Fanelli T, Reshkin SJ, Gisler SM, Murer H, Casavola V
The Journal of biological chemistry 2002 Jun 14;277(24):21480-8
The Journal of biological chemistry 2002 Jun 14;277(24):21480-8
Localization of cystic fibrosis transmembrane conductance regulator in chloride secretory epithelia.
Denning GM, Ostedgaard LS, Cheng SH, Smith AE, Welsh MJ
The Journal of clinical investigation 1992 Jan;89(1):339-49
The Journal of clinical investigation 1992 Jan;89(1):339-49
No comments: Submit comment
Supportive validation
- Submitted by
- R&D Systems (provider)
- Main image
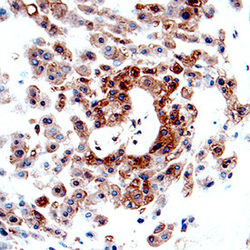
- Experimental details
- CFTR in Human Placenta. CFTR was detected in immersion fixed paraffin-embedded sections of human placenta using Mouse Anti-Human CFTR R Domain Monoclonal Antibody (Catalog # MAB1660) at 15 µg/mL overnight at 4 °C. Before incubation with the primary antibody tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific labeling was localized to the plasma membrane and cytoplasm of decidual cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry