Antibody data
- Antibody Data
- Antigen structure
- References [78]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [4]
- Immunohistochemistry [3]
- Other assay [33]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA3-030A - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- iNOS Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA3-030A detects inducible nitric oxide synthase (iNOS) from human and mouse tissues. This antibody does not detect endothelial NOS (eNOS) or brain NOS (bNOS). PA3-030A has been successfully used in Western blot and immunohistochemistry (paraffin and frozen) procedures. By Western blot, this antibody detects an ~130 kDa protein representing iNOS in induced RAW 264.7 cell extracts. The PA3-030A immunogen is a synthetic peptide corresponding to residues C K(1131) K G S A L E E P K A T R L(1144) of mouse macrophage NOS. PA3-030A can be used with blocking peptide PEP-008. Antibodies to this protein (and modification) were previously sold as part of a Thermo Scientific Cellomics High Content Screening Kit. This replacement antibody is now recommended for researchers who need an antibody for high content cell based assays. It has been thoroughly tested and validated for cellular immunofluorescence (IF) applications. Further optimization including the selection of the most appropriate fluorescent Dylight conjugated secondary antibody may have to be performed for your high content assay.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- Conc. Not Determined
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references 3D printing polylactic acid polymer-bioactive glass loaded with bone cement for bone defect in weight-bearing area.
Adipocyte-derived lactate is a signalling metabolite that potentiates adipose macrophage inflammation via targeting PHD2.
Dual functions of microRNA-17 in maintaining cartilage homeostasis and protection against osteoarthritis.
Anti-Inflammatory CDGSH Iron-Sulfur Domain 2: A Biomarker of Central Nervous System Insult in Cellular, Animal Models and Patients.
Pharmacological Effects of Gami-Yukmijihwang-Tang on the Lipopolysaccharide-Induced Hippocampus Oxidation and Inflammation via Regulation of Sirt6.
Distinct Stromal and Immune Features Collectively Contribute to Long-Term Survival in Pancreatic Cancer.
HA-coated collagen nanofibers for urethral regeneration via in situ polarization of M2 macrophages.
Monascin and Ankaflavin of Monascus purpureus Prevent Alcoholic Liver Disease through Regulating AMPK-Mediated Lipid Metabolism and Enhancing Both Anti-Inflammatory and Anti-Oxidative Systems.
The Gastroprotective Effect of Naringenin against Ethanol-Induced Gastric Ulcers in Mice through Inhibiting Oxidative and Inflammatory Responses.
Requirement of direct contact between chondrocytes and macrophages for the maturation of regenerative cartilage.
Coumestrol mitigates retinal cell inflammation, apoptosis, and oxidative stress in a rat model of diabetic retinopathy via activation of SIRT1.
ASS1 and ASL suppress growth in clear cell renal cell carcinoma via altered nitrogen metabolism.
Effects of mechanical strain on periodontal ligament fibroblasts in presence of Aggregatibacter actinomycetemcomitans lysate.
Chewing Behavior Attenuates the Tumor Progression-Enhancing Effects of Psychological Stress in a Breast Cancer Model Mouse.
CISD2 Attenuates Inflammation and Regulates Microglia Polarization in EOC Microglial Cells-As a Potential Therapeutic Target for Neurodegenerative Dementia.
Graft IL-33 regulates infiltrating macrophages to protect against chronic rejection.
Evaluation of Glycosylated PTGS2 in Colorectal Cancer for NSAIDS-Based Adjuvant Therapy.
Antioxidant effect of acetyl-l-carnitine against cisplatin-induced cardiotoxicity.
Antifungal drug miconazole ameliorated memory deficits in a mouse model of LPS-induced memory loss through targeting iNOS.
Melatonin MT2 receptor agonist IIK-7 produces antinociception by modulation of ROS and suppression of spinal microglial activation in neuropathic pain rats.
Effect of intra-articular hyaluronan injection on inflammation and bone remodeling in the epiphyses and metaphyses of the knee in a murine model of joint injury.
Aberrant oligodendroglial-vascular interactions disrupt the blood-brain barrier, triggering CNS inflammation.
Immunomodulatory nanodiamond aggregate-based platform for the treatment of rheumatoid arthritis.
The immunophenotype of decidual macrophages in acute atherosis.
Different Patterns of Neurodegeneration and Glia Activation in CA1 and CA3 Hippocampal Regions of TgCRND8 Mice.
Lactobacillus casei Strain Shirota Enhances the In Vitro Antiproliferative Effect of Geniposide in Human Oral Squamous Carcinoma HSC-3 Cells.
Tetramethylpyrazine Ameliorates Rotenone-Induced Parkinson's Disease in Rats: Involvement of Its Anti-Inflammatory and Anti-Apoptotic Actions.
Short-Term Intake of a Fructose-, Fat- and Cholesterol-Rich Diet Causes Hepatic Steatosis in Mice: Effect of Antibiotic Treatment.
TRPV4 activation triggers protective responses to bacterial lipopolysaccharides in airway epithelial cells.
TLR-exosomes exhibit distinct kinetics and effector function.
Crocin Inhibits Oxidative Stress and Pro-inflammatory Response of Microglial Cells Associated with Diabetic Retinopathy Through the Activation of PI3K/Akt Signaling Pathway.
Bubble CPAP Support after Discontinuation of Mechanical Ventilation Protects Rat Lungs with Ventilator-Induced Lung Injury.
Nitric oxide synthase in diabetic rat testicular tissue and the effects of pentoxifylline therapy.
Ameliorative effects of pentoxifylline on NOS induced by diabetes in rat kidney.
Dysfunctional MnSOD leads to redox dysregulation and activation of prosurvival AKT signaling in uterine leiomyomas.
Therapeutic effects of pentoxifylline on diabetic heart tissue via NOS.
Distinctly perturbed metabolic networks underlie differential tumor tissue damages induced by immune modulator β-glucan in a two-case ex vivo non-small-cell lung cancer study.
Agrimonolide from Agrimonia pilosa suppresses inflammatory responses through down-regulation of COX-2/iNOS and inactivation of NF-κB in lipopolysaccharide-stimulated macrophages.
Geraniol attenuates 2-acetylaminofluorene induced oxidative stress, inflammation and apoptosis in the liver of wistar rats.
Genistein improves 3-NPA-induced memory impairment in ovariectomized rats: impact of its antioxidant, anti-inflammatory and acetylcholinesterase modulatory properties.
Scutellarin regulates microglia-mediated TNC1 astrocytic reaction and astrogliosis in cerebral ischemia in the adult rats.
The effect of pomegranate fruit extract on testosterone-induced BPH in rats.
Intra-articular resveratrol injection prevents osteoarthritis progression in a mouse model by activating SIRT1 and thereby silencing HIF-2α.
Local pulmonary immune responses in domestic cats naturally infected with Cytauxzoon felis.
Minocycline attenuates pain by inhibiting spinal microglia activation in diabetic rats.
Infusion of bone marrow mononuclear cells reduces lung fibrosis but not inflammation in the late stages of murine silicosis.
Histopathologic, biochemical and genotoxic investigations on chronic sodium nitrite toxicity in mice.
Pretreatment with mineralocorticoid receptor blocker reduces intestinal injury induced by ischemia and reperfusion: involvement of inhibition of inflammatory response, oxidative stress, nuclear factor κB, and inducible nitric oxide synthase.
The cytotoxic, neurotoxic, apoptotic and antiproliferative activities of extracts of some marine algae on the MCF-7 cell line.
GSPE is superior to NAC in the prevention of contrast-induced nephropathy: might this superiority be related to caspase 1 and calpain 1?
Intravenous glutamine administration reduces lung and distal organ injury in malnourished rats with sepsis.
Mechanisms of the protective effects of curcumin against indomethacin-induced gastric ulcer in rats.
Multimechanistic antifibrotic effect of biochanin a in rats: implications of proinflammatory and profibrogenic mediators.
Perspective on rhabdomyolysis-induced acute kidney injury and new treatment options.
Inducible nitric oxide synthase (iNOS) in gingival tissues of chronic periodontitis with and without diabetes: immunohistochemistry and RT-PCR study.
Prognostic value of arginase-II expression and regulatory T-cell infiltration in head and neck squamous cell carcinoma.
How does colistin-induced nephropathy develop and can it be treated?
Inducible nitric oxide synthase inhibition attenuates physical stress-induced lung hyper-responsiveness and oxidative stress in animals with lung inflammation.
Effect of valproic acid treatment on penile structure in prepubertal rats.
Immunohistochemical study of the angiogenetic network of VEGF, HIF1α, VEGFR-2 and endothelial nitric oxide synthase (eNOS) in human breast cancer.
Evaluation of antioxidant activity and inhibitory effect on nitric oxide production of some common vegetables.
Expression of nitric oxide synthase isoforms in the ovine fetal brain: alteration by hormonal and hemodynamic stimuli.
Cyclooxygenase-2 and inducible nitric oxide synthase expression in thyroid neoplasms and their clinicopathological correlation.
Expression of endothelial NO synthase, inducible NO synthase, and estrogen receptors alpha and beta in placental tissue of normal, preeclamptic, and intrauterine growth-restricted pregnancies.
Expression of nitric oxide synthase isoforms is reduced in late-gestation ovine fetal brainstem.
Role of nitric oxide in vascular permeability after combined burns and smoke inhalation injury.
Suppression effect of soy isoflavones on nitric oxide production in RAW 264.7 macrophages.
Mechanisms of vascular instability in a transgenic mouse model of sickle cell disease.
GnRH agonist-suppressed expression of nitric oxide synthases and generation of peroxynitrite in adenomyosis.
GnRH agonist-suppressed expression of nitric oxide synthases and generation of peroxynitrite in adenomyosis.
Granulated metrial gland cells contain nitric oxide synthases during pregnancy in the rat.
Experimental allergic encephalomyelitis in the rat is inhibited by aminoguanidine, an inhibitor of nitric oxide synthase.
Experimental allergic encephalomyelitis in the rat is inhibited by aminoguanidine, an inhibitor of nitric oxide synthase.
Induction of nitric oxide synthase in the human cardiac allograft is associated with contractile dysfunction of the left ventricle.
Inducible and endothelial nitric oxide synthase expression during development of transplant arteriosclerosis in rat aortic grafts.
Metal-induced modulation of nitric oxide production in vitro by murine macrophages: lead, nickel, and cobalt utilize different mechanisms.
Inhibition of macrophages with gadolinium chloride abrogates ozone-induced pulmonary injury and inflammatory mediator production.
Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase.
Ding Y, Liu X, Zhang J, Lv Z, Meng X, Yuan Z, Long T, Wang Y
Frontiers in bioengineering and biotechnology 2022;10:947521
Frontiers in bioengineering and biotechnology 2022;10:947521
Adipocyte-derived lactate is a signalling metabolite that potentiates adipose macrophage inflammation via targeting PHD2.
Feng T, Zhao X, Gu P, Yang W, Wang C, Guo Q, Long Q, Liu Q, Cheng Y, Li J, Cheung CKY, Wu D, Kong X, Xu Y, Ye D, Hua S, Loomes K, Xu A, Hui X
Nature communications 2022 Sep 5;13(1):5208
Nature communications 2022 Sep 5;13(1):5208
Dual functions of microRNA-17 in maintaining cartilage homeostasis and protection against osteoarthritis.
Zhang Y, Li S, Jin P, Shang T, Sun R, Lu L, Guo K, Liu J, Tong Y, Wang J, Liu S, Wang C, Kang Y, Zhu W, Wang Q, Zhang X, Yin F, Sun YE, Cui L
Nature communications 2022 May 4;13(1):2447
Nature communications 2022 May 4;13(1):2447
Anti-Inflammatory CDGSH Iron-Sulfur Domain 2: A Biomarker of Central Nervous System Insult in Cellular, Animal Models and Patients.
Kung WM, Lin CC, Chen WJ, Jiang LL, Sun YY, Hsieh KH, Lin MS
Biomedicines 2022 Mar 27;10(4)
Biomedicines 2022 Mar 27;10(4)
Pharmacological Effects of Gami-Yukmijihwang-Tang on the Lipopolysaccharide-Induced Hippocampus Oxidation and Inflammation via Regulation of Sirt6.
Kang JY, Lee JS, Seol IC, Kim YS, Park MS, Yoo HR
Pharmaceuticals (Basel, Switzerland) 2022 Feb 28;15(3)
Pharmaceuticals (Basel, Switzerland) 2022 Feb 28;15(3)
Distinct Stromal and Immune Features Collectively Contribute to Long-Term Survival in Pancreatic Cancer.
Sadozai H, Acharjee A, Eppenberger-Castori S, Gloor B, Gruber T, Schenk M, Karamitopoulou E
Frontiers in immunology 2021;12:643529
Frontiers in immunology 2021;12:643529
HA-coated collagen nanofibers for urethral regeneration via in situ polarization of M2 macrophages.
Niu Y, Stadler FJ, Yang X, Deng F, Liu G, Xia H
Journal of nanobiotechnology 2021 Sep 22;19(1):283
Journal of nanobiotechnology 2021 Sep 22;19(1):283
Monascin and Ankaflavin of Monascus purpureus Prevent Alcoholic Liver Disease through Regulating AMPK-Mediated Lipid Metabolism and Enhancing Both Anti-Inflammatory and Anti-Oxidative Systems.
Lai JR, Hsu YW, Pan TM, Lee CL
Molecules (Basel, Switzerland) 2021 Oct 18;26(20)
Molecules (Basel, Switzerland) 2021 Oct 18;26(20)
The Gastroprotective Effect of Naringenin against Ethanol-Induced Gastric Ulcers in Mice through Inhibiting Oxidative and Inflammatory Responses.
Li WS, Lin SC, Chu CH, Chang YK, Zhang X, Lin CC, Tung YT
International journal of molecular sciences 2021 Nov 5;22(21)
International journal of molecular sciences 2021 Nov 5;22(21)
Requirement of direct contact between chondrocytes and macrophages for the maturation of regenerative cartilage.
Kanda K, Asawa Y, Inaki R, Fujihara Y, Hoshi K, Hikita A
Scientific reports 2021 Nov 18;11(1):22476
Scientific reports 2021 Nov 18;11(1):22476
Coumestrol mitigates retinal cell inflammation, apoptosis, and oxidative stress in a rat model of diabetic retinopathy via activation of SIRT1.
Xu Y, Zhang Y, Liang H, Liu X
Aging 2021 Feb 1;13(4):5342-5357
Aging 2021 Feb 1;13(4):5342-5357
ASS1 and ASL suppress growth in clear cell renal cell carcinoma via altered nitrogen metabolism.
Khare S, Kim LC, Lobel G, Doulias PT, Ischiropoulos H, Nissim I, Keith B, Simon MC
Cancer & metabolism 2021 Dec 3;9(1):40
Cancer & metabolism 2021 Dec 3;9(1):40
Effects of mechanical strain on periodontal ligament fibroblasts in presence of Aggregatibacter actinomycetemcomitans lysate.
Schröder A, Stumpf J, Paddenberg E, Neubert P, Schatz V, Köstler J, Jantsch J, Deschner J, Proff P, Kirschneck C
BMC oral health 2021 Aug 18;21(1):405
BMC oral health 2021 Aug 18;21(1):405
Chewing Behavior Attenuates the Tumor Progression-Enhancing Effects of Psychological Stress in a Breast Cancer Model Mouse.
Zhou Q, Katano M, Zhang JH, Liu X, Wang KY, Iinuma M, Kubo KY, Azuma K
Brain sciences 2021 Apr 9;11(4)
Brain sciences 2021 Apr 9;11(4)
CISD2 Attenuates Inflammation and Regulates Microglia Polarization in EOC Microglial Cells-As a Potential Therapeutic Target for Neurodegenerative Dementia.
Lin MS
Frontiers in aging neuroscience 2020;12:260
Frontiers in aging neuroscience 2020;12:260
Graft IL-33 regulates infiltrating macrophages to protect against chronic rejection.
Li T, Zhang Z, Bartolacci JG, Dwyer GK, Liu Q, Mathews LR, Velayutham M, Roessing AS, Lee YC, Dai H, Shiva S, Oberbarnscheidt MH, Dziki JL, Mullet SJ, Wendell SG, Wilkinson JD, Webber SA, Wood-Trageser M, Watkins SC, Demetris AJ, Hussey GS, Badylak SF, Turnquist HR
The Journal of clinical investigation 2020 Oct 1;130(10):5397-5412
The Journal of clinical investigation 2020 Oct 1;130(10):5397-5412
Evaluation of Glycosylated PTGS2 in Colorectal Cancer for NSAIDS-Based Adjuvant Therapy.
Venè R, Costa D, Augugliaro R, Carlone S, Scabini S, Casoni Pattacini G, Boggio M, Zupo S, Grillo F, Mastracci L, Pitto F, Minghelli S, Ferrari N, Tosetti F, Romairone E, Mingari MC, Poggi A, Benelli R
Cells 2020 Mar 11;9(3)
Cells 2020 Mar 11;9(3)
Antioxidant effect of acetyl-l-carnitine against cisplatin-induced cardiotoxicity.
Bayrak S, Aktaş S, Altun Z, Çakir Y, Tütüncü M, Kum Özşengezer S, Yilmaz O, Olgun N
The Journal of international medical research 2020 Aug;48(8):300060520951393
The Journal of international medical research 2020 Aug;48(8):300060520951393
Antifungal drug miconazole ameliorated memory deficits in a mouse model of LPS-induced memory loss through targeting iNOS.
Yeo IJ, Yun J, Son DJ, Han SB, Hong JT
Cell death & disease 2020 Aug 14;11(8):623
Cell death & disease 2020 Aug 14;11(8):623
Melatonin MT2 receptor agonist IIK-7 produces antinociception by modulation of ROS and suppression of spinal microglial activation in neuropathic pain rats.
Kuthati Y, Goutham Davuluri VN, Yang CP, Chang HC, Chang CP, Wong CS
Journal of pain research 2019;12:2473-2485
Journal of pain research 2019;12:2473-2485
Effect of intra-articular hyaluronan injection on inflammation and bone remodeling in the epiphyses and metaphyses of the knee in a murine model of joint injury.
Shen Q, Li J, Chan D, Sandy JD, Takeuchi J, Ross RD, Plaas A
American journal of translational research 2019;11(6):3280-3300
American journal of translational research 2019;11(6):3280-3300
Aberrant oligodendroglial-vascular interactions disrupt the blood-brain barrier, triggering CNS inflammation.
Niu J, Tsai HH, Hoi KK, Huang N, Yu G, Kim K, Baranzini SE, Xiao L, Chan JR, Fancy SPJ
Nature neuroscience 2019 May;22(5):709-718
Nature neuroscience 2019 May;22(5):709-718
Immunomodulatory nanodiamond aggregate-based platform for the treatment of rheumatoid arthritis.
Pentecost A, Kim MJ, Jeon S, Ko YJ, Kwon IC, Gogotsi Y, Kim K, Spiller KL
Regenerative biomaterials 2019 Jun;6(3):163-174
Regenerative biomaterials 2019 Jun;6(3):163-174
The immunophenotype of decidual macrophages in acute atherosis.
Gill N, Leng Y, Romero R, Xu Y, Panaitescu B, Miller D, Arif A, Mumuni S, Qureshi F, Hsu CD, Hassan SS, Staff AC, Gomez-Lopez N
American journal of reproductive immunology (New York, N.Y. : 1989) 2019 Apr;81(4):e13098
American journal of reproductive immunology (New York, N.Y. : 1989) 2019 Apr;81(4):e13098
Different Patterns of Neurodegeneration and Glia Activation in CA1 and CA3 Hippocampal Regions of TgCRND8 Mice.
Ugolini F, Lana D, Nardiello P, Nosi D, Pantano D, Casamenti F, Giovannini MG
Frontiers in aging neuroscience 2018;10:372
Frontiers in aging neuroscience 2018;10:372
Lactobacillus casei Strain Shirota Enhances the In Vitro Antiproliferative Effect of Geniposide in Human Oral Squamous Carcinoma HSC-3 Cells.
Qian Y, Song JL, Sun P, Yi R, Liu H, Feng X, Park KY, Zhao X
Molecules (Basel, Switzerland) 2018 May 3;23(5)
Molecules (Basel, Switzerland) 2018 May 3;23(5)
Tetramethylpyrazine Ameliorates Rotenone-Induced Parkinson's Disease in Rats: Involvement of Its Anti-Inflammatory and Anti-Apoptotic Actions.
Michel HE, Tadros MG, Esmat A, Khalifa AE, Abdel-Tawab AM
Molecular neurobiology 2017 Sep;54(7):4866-4878
Molecular neurobiology 2017 Sep;54(7):4866-4878
Short-Term Intake of a Fructose-, Fat- and Cholesterol-Rich Diet Causes Hepatic Steatosis in Mice: Effect of Antibiotic Treatment.
Brandt A, Jin CJ, Nolte K, Sellmann C, Engstler AJ, Bergheim I
Nutrients 2017 Sep 14;9(9)
Nutrients 2017 Sep 14;9(9)
TRPV4 activation triggers protective responses to bacterial lipopolysaccharides in airway epithelial cells.
Alpizar YA, Boonen B, Sanchez A, Jung C, López-Requena A, Naert R, Steelant B, Luyts K, Plata C, De Vooght V, Vanoirbeek JAJ, Meseguer VM, Voets T, Alvarez JL, Hellings PW, Hoet PHM, Nemery B, Valverde MA, Talavera K
Nature communications 2017 Oct 20;8(1):1059
Nature communications 2017 Oct 20;8(1):1059
TLR-exosomes exhibit distinct kinetics and effector function.
Srinivasan S, Su M, Ravishankar S, Moore J, Head P, Dixon JB, Vannberg F
Scientific reports 2017 Mar 14;7:41623
Scientific reports 2017 Mar 14;7:41623
Crocin Inhibits Oxidative Stress and Pro-inflammatory Response of Microglial Cells Associated with Diabetic Retinopathy Through the Activation of PI3K/Akt Signaling Pathway.
Yang X, Huo F, Liu B, Liu J, Chen T, Li J, Zhu Z, Lv B
Journal of molecular neuroscience : MN 2017 Apr;61(4):581-589
Journal of molecular neuroscience : MN 2017 Apr;61(4):581-589
Bubble CPAP Support after Discontinuation of Mechanical Ventilation Protects Rat Lungs with Ventilator-Induced Lung Injury.
Wu CS, Chou HC, Huang LT, Lin YK, Chen CM
Respiration; international review of thoracic diseases 2016;91(2):171-9
Respiration; international review of thoracic diseases 2016;91(2):171-9
Nitric oxide synthase in diabetic rat testicular tissue and the effects of pentoxifylline therapy.
Sönmez MF, Kılıç E, Karabulut D, Çilenk K, Deligönül E, Dündar M
Systems biology in reproductive medicine 2016;62(1):22-30
Systems biology in reproductive medicine 2016;62(1):22-30
Ameliorative effects of pentoxifylline on NOS induced by diabetes in rat kidney.
Sönmez MF, Dündar M
Renal failure 2016;38(4):605-13
Renal failure 2016;38(4):605-13
Dysfunctional MnSOD leads to redox dysregulation and activation of prosurvival AKT signaling in uterine leiomyomas.
Vidimar V, Gius D, Chakravarti D, Bulun SE, Wei JJ, Kim JJ
Science advances 2016 Nov;2(11):e1601132
Science advances 2016 Nov;2(11):e1601132
Therapeutic effects of pentoxifylline on diabetic heart tissue via NOS.
Karabulut D, Ulusoy HB, Kaymak E, Sönmez MF
Anatolian journal of cardiology 2016 May;16(5):310-5
Anatolian journal of cardiology 2016 May;16(5):310-5
Distinctly perturbed metabolic networks underlie differential tumor tissue damages induced by immune modulator β-glucan in a two-case ex vivo non-small-cell lung cancer study.
Fan TW, Warmoes MO, Sun Q, Song H, Turchan-Cholewo J, Martin JT, Mahan A, Higashi RM, Lane AN
Cold Spring Harbor molecular case studies 2016 Jul;2(4):a000893
Cold Spring Harbor molecular case studies 2016 Jul;2(4):a000893
Agrimonolide from Agrimonia pilosa suppresses inflammatory responses through down-regulation of COX-2/iNOS and inactivation of NF-κB in lipopolysaccharide-stimulated macrophages.
Chen L, Teng H, Fang T, Xiao J
Phytomedicine : international journal of phytotherapy and phytopharmacology 2016 Jul 15;23(8):846-55
Phytomedicine : international journal of phytotherapy and phytopharmacology 2016 Jul 15;23(8):846-55
Geraniol attenuates 2-acetylaminofluorene induced oxidative stress, inflammation and apoptosis in the liver of wistar rats.
Hasan SK, Sultana S
Toxicology mechanisms and methods 2015;25(7):559-73
Toxicology mechanisms and methods 2015;25(7):559-73
Genistein improves 3-NPA-induced memory impairment in ovariectomized rats: impact of its antioxidant, anti-inflammatory and acetylcholinesterase modulatory properties.
Menze ET, Esmat A, Tadros MG, Abdel-Naim AB, Khalifa AE
PloS one 2015;10(2):e0117223
PloS one 2015;10(2):e0117223
Scutellarin regulates microglia-mediated TNC1 astrocytic reaction and astrogliosis in cerebral ischemia in the adult rats.
Fang M, Yuan Y, Rangarajan P, Lu J, Wu Y, Wang H, Wu C, Ling EA
BMC neuroscience 2015 Nov 25;16:84
BMC neuroscience 2015 Nov 25;16:84
The effect of pomegranate fruit extract on testosterone-induced BPH in rats.
Ammar AE, Esmat A, Hassona MD, Tadros MG, Abdel-Naim AB, Guns ES
The Prostate 2015 May;75(7):679-92
The Prostate 2015 May;75(7):679-92
Intra-articular resveratrol injection prevents osteoarthritis progression in a mouse model by activating SIRT1 and thereby silencing HIF-2α.
Li W, Cai L, Zhang Y, Cui L, Shen G
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2015 Jul;33(7):1061-70
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2015 Jul;33(7):1061-70
Local pulmonary immune responses in domestic cats naturally infected with Cytauxzoon felis.
Frontera-Acevedo K, Sakamoto K
Veterinary immunology and immunopathology 2015 Jan 15;163(1-2):1-7
Veterinary immunology and immunopathology 2015 Jan 15;163(1-2):1-7
Minocycline attenuates pain by inhibiting spinal microglia activation in diabetic rats.
Sun JS, Yang YJ, Zhang YZ, Huang W, Li ZS, Zhang Y
Molecular medicine reports 2015 Aug;12(2):2677-82
Molecular medicine reports 2015 Aug;12(2):2677-82
Infusion of bone marrow mononuclear cells reduces lung fibrosis but not inflammation in the late stages of murine silicosis.
Lopes-Pacheco M, Ventura TG, de Oliveira HD, Monção-Ribeiro LC, Gutfilen B, de Souza SA, Rocco PR, Borojevic R, Morales MM, Takiya CM
PloS one 2014;9(10):e109982
PloS one 2014;9(10):e109982
Histopathologic, biochemical and genotoxic investigations on chronic sodium nitrite toxicity in mice.
Özen H, Kamber U, Karaman M, Gül S, Atakişi E, Özcan K, Atakişi O
Experimental and toxicologic pathology : official journal of the Gesellschaft fur Toxikologische Pathologie 2014 Oct;66(8):367-75
Experimental and toxicologic pathology : official journal of the Gesellschaft fur Toxikologische Pathologie 2014 Oct;66(8):367-75
Pretreatment with mineralocorticoid receptor blocker reduces intestinal injury induced by ischemia and reperfusion: involvement of inhibition of inflammatory response, oxidative stress, nuclear factor κB, and inducible nitric oxide synthase.
Ozacmak HS, Ozacmak VH, Barut F, Araslı M, Ucan BH
The Journal of surgical research 2014 Oct;191(2):350-61
The Journal of surgical research 2014 Oct;191(2):350-61
The cytotoxic, neurotoxic, apoptotic and antiproliferative activities of extracts of some marine algae on the MCF-7 cell line.
Kurt O, Ozdal-Kurt F, Tuğlu MI, Akçora CM
Biotechnic & histochemistry : official publication of the Biological Stain Commission 2014 Nov;89(8):568-76
Biotechnic & histochemistry : official publication of the Biological Stain Commission 2014 Nov;89(8):568-76
GSPE is superior to NAC in the prevention of contrast-induced nephropathy: might this superiority be related to caspase 1 and calpain 1?
Ulusoy S, Ozkan G, Mungan S, Orem A, Yulug E, Alkanat M, Yucesan FB
Life sciences 2014 May 17;103(2):101-10
Life sciences 2014 May 17;103(2):101-10
Intravenous glutamine administration reduces lung and distal organ injury in malnourished rats with sepsis.
de Oliveira GP, Silva JD, de Araújo CC, Prota LF, Abreu SC, Madeira C, Morales MM, Takiya CM, Diaz BL, Capelozzi VL, Panizzutti R, Pelosi P, Rocco PR
Shock (Augusta, Ga.) 2014 Mar;41(3):222-32
Shock (Augusta, Ga.) 2014 Mar;41(3):222-32
Mechanisms of the protective effects of curcumin against indomethacin-induced gastric ulcer in rats.
Morsy MA, El-Moselhy MA
Pharmacology 2013;91(5-6):267-74
Pharmacology 2013;91(5-6):267-74
Multimechanistic antifibrotic effect of biochanin a in rats: implications of proinflammatory and profibrogenic mediators.
Breikaa RM, Algandaby MM, El-Demerdash E, Abdel-Naim AB
PloS one 2013;8(7):e69276
PloS one 2013;8(7):e69276
Perspective on rhabdomyolysis-induced acute kidney injury and new treatment options.
Ulusoy S, Ozkan G, Alkanat M, Mungan S, Yuluğ E, Orem A
American journal of nephrology 2013;38(5):368-78
American journal of nephrology 2013;38(5):368-78
Inducible nitric oxide synthase (iNOS) in gingival tissues of chronic periodontitis with and without diabetes: immunohistochemistry and RT-PCR study.
Shaker O, Ghallab NA, Hamdy E, Sayed S
Archives of oral biology 2013 Oct;58(10):1397-406
Archives of oral biology 2013 Oct;58(10):1397-406
Prognostic value of arginase-II expression and regulatory T-cell infiltration in head and neck squamous cell carcinoma.
Bron L, Jandus C, Andrejevic-Blant S, Speiser DE, Monnier P, Romero P, Rivals JP
International journal of cancer 2013 Feb 1;132(3):E85-93
International journal of cancer 2013 Feb 1;132(3):E85-93
How does colistin-induced nephropathy develop and can it be treated?
Ozkan G, Ulusoy S, Orem A, Alkanat M, Mungan S, Yulug E, Yucesan FB
Antimicrobial agents and chemotherapy 2013 Aug;57(8):3463-9
Antimicrobial agents and chemotherapy 2013 Aug;57(8):3463-9
Inducible nitric oxide synthase inhibition attenuates physical stress-induced lung hyper-responsiveness and oxidative stress in animals with lung inflammation.
Marques RH, Reis FG, Starling CM, Cabido C, de Almeida-Reis R, Dohlnikoff M, Prado CM, Leick EA, Martins MA, Tibério IF
Neuroimmunomodulation 2012;19(3):158-70
Neuroimmunomodulation 2012;19(3):158-70
Effect of valproic acid treatment on penile structure in prepubertal rats.
Kutlu O, Cansu A, Karagüzel E, Gürgen SG, Koç O, Gür M, Ozgür GK
Epilepsy research 2012 May;99(3):306-11
Epilepsy research 2012 May;99(3):306-11
Immunohistochemical study of the angiogenetic network of VEGF, HIF1α, VEGFR-2 and endothelial nitric oxide synthase (eNOS) in human breast cancer.
Kafousi M, Vrekoussis T, Tsentelierou E, Pavlakis K, Navrozoglou I, Dousias V, Sanidas E, Tsiftsis D, Georgoulias V, Stathopoulos EN
Pathology oncology research : POR 2012 Jan;18(1):33-41
Pathology oncology research : POR 2012 Jan;18(1):33-41
Evaluation of antioxidant activity and inhibitory effect on nitric oxide production of some common vegetables.
Bor JY, Chen HY, Yen GC
Journal of agricultural and food chemistry 2006 Mar 8;54(5):1680-6
Journal of agricultural and food chemistry 2006 Mar 8;54(5):1680-6
Expression of nitric oxide synthase isoforms in the ovine fetal brain: alteration by hormonal and hemodynamic stimuli.
Wood CE, Giroux D
Journal of the Society for Gynecologic Investigation 2006 Jul;13(5):329-37
Journal of the Society for Gynecologic Investigation 2006 Jul;13(5):329-37
Cyclooxygenase-2 and inducible nitric oxide synthase expression in thyroid neoplasms and their clinicopathological correlation.
Kim KH, Kim SH, Kim SH, Back JH, Park MJ, Kim JM
Journal of Korean medical science 2006 Dec;21(6):1064-9
Journal of Korean medical science 2006 Dec;21(6):1064-9
Expression of endothelial NO synthase, inducible NO synthase, and estrogen receptors alpha and beta in placental tissue of normal, preeclamptic, and intrauterine growth-restricted pregnancies.
Schiessl B, Mylonas I, Hantschmann P, Kuhn C, Schulze S, Kunze S, Friese K, Jeschke U
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2005 Dec;53(12):1441-9
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2005 Dec;53(12):1441-9
Expression of nitric oxide synthase isoforms is reduced in late-gestation ovine fetal brainstem.
Wood CE, Chen GF, Keller-Wood M
American journal of physiology. Regulatory, integrative and comparative physiology 2005 Aug;289(2):R613-R619
American journal of physiology. Regulatory, integrative and comparative physiology 2005 Aug;289(2):R613-R619
Role of nitric oxide in vascular permeability after combined burns and smoke inhalation injury.
Soejima K, Traber LD, Schmalstieg FC, Hawkins H, Jodoin JM, Szabo C, Szabo E, Virag L, Salzman A, Traber DL
American journal of respiratory and critical care medicine 2001 Mar;163(3 Pt 1):745-52
American journal of respiratory and critical care medicine 2001 Mar;163(3 Pt 1):745-52
Suppression effect of soy isoflavones on nitric oxide production in RAW 264.7 macrophages.
Sheu F, Lai HH, Yen GC
Journal of agricultural and food chemistry 2001 Apr;49(4):1767-72
Journal of agricultural and food chemistry 2001 Apr;49(4):1767-72
Mechanisms of vascular instability in a transgenic mouse model of sickle cell disease.
Nath KA, Shah V, Haggard JJ, Croatt AJ, Smith LA, Hebbel RP, Katusic ZS
American journal of physiology. Regulatory, integrative and comparative physiology 2000 Dec;279(6):R1949-55
American journal of physiology. Regulatory, integrative and comparative physiology 2000 Dec;279(6):R1949-55
GnRH agonist-suppressed expression of nitric oxide synthases and generation of peroxynitrite in adenomyosis.
Kamada Y, Nakatsuka M, Asagiri K, Noguchi S, Habara T, Takata M, Kudo T
Human reproduction (Oxford, England) 2000 Dec;15(12):2512-9
Human reproduction (Oxford, England) 2000 Dec;15(12):2512-9
GnRH agonist-suppressed expression of nitric oxide synthases and generation of peroxynitrite in adenomyosis.
Kamada Y, Nakatsuka M, Asagiri K, Noguchi S, Habara T, Takata M, Kudo T
Human reproduction (Oxford, England) 2000 Dec;15(12):2512-9
Human reproduction (Oxford, England) 2000 Dec;15(12):2512-9
Granulated metrial gland cells contain nitric oxide synthases during pregnancy in the rat.
Sladek SM, Kanbour-Shakir A, Watkins S, Berghorn KA, Hoffman GE, Roberts JM
Placenta 1998 Jan;19(1):55-65
Placenta 1998 Jan;19(1):55-65
Experimental allergic encephalomyelitis in the rat is inhibited by aminoguanidine, an inhibitor of nitric oxide synthase.
Zhao W, Tilton RG, Corbett JA, McDaniel ML, Misko TP, Williamson JR, Cross AH, Hickey WF
Journal of neuroimmunology 1996 Feb;64(2):123-33
Journal of neuroimmunology 1996 Feb;64(2):123-33
Experimental allergic encephalomyelitis in the rat is inhibited by aminoguanidine, an inhibitor of nitric oxide synthase.
Zhao W, Tilton RG, Corbett JA, McDaniel ML, Misko TP, Williamson JR, Cross AH, Hickey WF
Journal of neuroimmunology 1996 Feb;64(2):123-33
Journal of neuroimmunology 1996 Feb;64(2):123-33
Induction of nitric oxide synthase in the human cardiac allograft is associated with contractile dysfunction of the left ventricle.
Lewis NP, Tsao PS, Rickenbacher PR, Xue C, Johns RA, Haywood GA, von der Leyen H, Trindade PT, Cooke JP, Hunt SA, Billingham ME, Valantine HA, Fowler MB
Circulation 1996 Feb 15;93(4):720-9
Circulation 1996 Feb 15;93(4):720-9
Inducible and endothelial nitric oxide synthase expression during development of transplant arteriosclerosis in rat aortic grafts.
Akyürek LM, Fellström BC, Yan ZQ, Hansson GK, Funa K, Larsson E
The American journal of pathology 1996 Dec;149(6):1981-90
The American journal of pathology 1996 Dec;149(6):1981-90
Metal-induced modulation of nitric oxide production in vitro by murine macrophages: lead, nickel, and cobalt utilize different mechanisms.
Tian L, Lawrence DA
Toxicology and applied pharmacology 1996 Dec;141(2):540-7
Toxicology and applied pharmacology 1996 Dec;141(2):540-7
Inhibition of macrophages with gadolinium chloride abrogates ozone-induced pulmonary injury and inflammatory mediator production.
Pendino KJ, Meidhof TM, Heck DE, Laskin JD, Laskin DL
American journal of respiratory cell and molecular biology 1995 Aug;13(2):125-32
American journal of respiratory cell and molecular biology 1995 Aug;13(2):125-32
Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase.
Bredt DS, Hwang PM, Glatt CE, Lowenstein C, Reed RR, Snyder SH
Nature 1991 Jun 27;351(6329):714-8
Nature 1991 Jun 27;351(6329):714-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
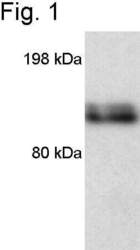
- Experimental details
- Western blot of mouse macrophage iNOS using Product # PA3-030A.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of iNOS using Anti-iNOS Polyclonal Antibody (Product # PA3-030A) shows staining in NIH-3T3 Cells. iNOS staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing iNOS (Product # PA3-030A) at a dilution of 1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of iNOS using Anti-iNOS Polyclonal Antibody (Product # PA3-030A) shows staining in Hela Cells. iNOS staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing iNOS (Product # PA3-030A) at a dilution of 1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of iNOS using Anti-iNOS Polyclonal Antibody (Product # PA3-030A) shows staining in Neuro-2a Cells. iNOS staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing iNOS (Product # PA3-030A) at a dilution of 1:200 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35552, Goat Anti-Rabbit). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of iNOS was performed using 70% confluent log phase A549 cells treated with a combination of TNF-α (5 ng/mL), IFNγ (50 U/mL) and IL-1β (50 U/mL) for 18 h. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with iNOS Rabbit Polyclonal Antibody (Product # PA3-030A) at 1:250 dilution in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic and nuclear localization. Panel e shows the untreated cells with no signal. Panel f represents the no primary control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
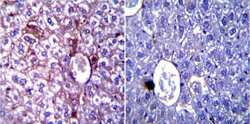
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Mouse liver tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a rabbit polyclonal antibody recognizing iNOS (Product # PA3-030A) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Mouse lung tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:100 with a rabbit polyclonal antibody recognizing iNOS (Product # PA3-030A) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
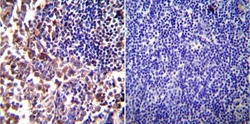
- Experimental details
- Immunohistochemistry was performed on normal biopsies of deparaffinized Mouse lymph node tissue. To expose target proteins, heat induced antigen retrieval was performed using 10mM sodium citrate (pH6.0) buffer, microwaved for 8-15 minutes. Following antigen retrieval tissues were blocked in 3% BSA-PBS for 30 minutes at room temperature. Tissues were then probed at a dilution of 1:200 with a rabbit polyclonal antibody recognizing iNOS (Product # PA3-030A) or without primary antibody (negative control) overnight at 4°C in a humidified chamber. Tissues were washed extensively with PBST and endogenous peroxidase activity was quenched with a peroxidase suppressor. Detection was performed using a biotin-conjugated secondary antibody and SA-HRP, followed by colorimetric detection using DAB. Tissues were counterstained with hematoxylin and prepped for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Protein expression of GFAP, Notch-1, NICD, HES-1, TNF-alpha, IL-1beta and iNOS in TNC1 following different treatments. a Expression levels of GFAP, Notch-1, NICD, and HES-1 were significantly increased after treatment with CM + L when compared with the control in CM; likewise, the expression of TNF-alpha, IL-1beta and iNOS was significantly increased. b In CM + SL treated TNC1, the expression levels of the above markers were further elevated as compared with that of CM + L treated cells. Bar graphs represent expression changes of the respective markers. Significant differences in protein levels are expressed as * # P < 0.05. The values represent the mean +- SD in triplicate. # CM + L vs CM, *CM + SL vs CM + L
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 TRPV4 activation by LPS triggers NO production in human bronchial EC. a Immunoblotting of 16HBE cells lysate probed for NOS isoforms. b Left, average intracellular Ca 2+ responses of human bronchial epithelial cells to 20 mug ml -1 LPS and 10 nM GSK1016790A. The black, blue, and red solid traces correspond to data means of cells responding to LPS and GSK1016790A, GSK1016790A alone or to none of the stimuli, respectively. The thin dashed traces represent the means +- standard errors. Right, stack bar graph showing the percentages of cells responding to LPS and/or GSK1016790A (color-coded as the graph on the left; n = 116). c Immunoblotting of 16HBE cells lysate incubated in LPS (20 mug ml -1 ) or GSK1016790A (10 nM). Where indicated, cells were previously incubated with vehicle (Veh, 0.8% DMSO) or the TRPV4 inhibitor HC067047 (+HC, 10 muM). d Average intracellular Ca 2+ signals (black solid traces) and normalized fluorescence of the NO-sensitive dye DAF-FM (F/F 0 , red solid traces) recorded in 16HBE cells ( n = 52). The thin dashed traces represent the means +- standard errors. LPS, 20 ug ml -1 ; HC067047, 10 uM; GSK1016790A, 10 nM. e Average changes in intracellular Ca 2+ (left panel) and NO production (right panel) induced by LPS. The dark cyan bars correspond to cells pre-incubated with the TRPV4 inhibitor HC067047 (10 uM). n > 74 per bar; ** P < 0.01, two-tailed t -test. f Confocal microscopy images of 16HBE cells immunostained for iNOS. The arrow head points to an
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. UK021 cancerous (CA) lung tissue is less mitotic, has less nitric oxide (NO)-producing potential, and is more necrotic than UK049 CA lung tissues. UK021 and UK049 CA and noncancerous (NC) lung tissues were sampled in the operating room and immediately preserved in formalin for immunohistochemical analysis for PCNA (mitotic index marker), RIP-1 (necrosis marker), and iNOS (inducible nitric oxide synthase, marker for M1-like macrophages [ Ohri et al. 2009 , 2011 ]) as green fluorescence, CD68 (general macrophage marker) as red fluorescence, and DAPI (nuclear marker) as blue fluorescence. ( A , C ) An example frame of the PCNA, RIP-1 (60x magnification), and iNOS + CD68 fluorescence images (60x magnification), as well as their normalized image intensity averaged over three to seven image frames for UK021 tissues. The fluorescence intensity of PCNA/RIP-1 and iNOS was respectively normalized to that of DAPI and CD68. ( B,D ) The corresponding image frames and normalized image intensity for UK049 tissues are shown. The UK021 CA lung tissue displayed a relatively lower mitotic index, higher necrotic state, and lower iNOS to CD68 ratio than the UK049 CA tissue. For both patients, the CA lung tissues were more mitotic and necrotic with a higher iNOS/CD68 ratio than the NC lung tissues proximal and distal to the CA tissues.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7. beta-Glucan-enhanced iNOS expression in UK021 but not UK049 CA lung tissue slices. WGP treatment, tissue preservation, and immunohistochemical analysis are as in Figure 6 . As in Figure 1 , the green fluorescence (iNOS) was normalized to the red fluorescence (CD68) to reflect WGP-induced increase in iNOS expression relative to CD68 expression for UK021 ( A ) but not for UK049 ( B ) CA lung tissue slices.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Immunohistochemical staining demonstrates the intensity of iNOS expression in the cytoplasm of papillary carcinoma cases. ( A ) score 0; ( B ) score 1; ( C ) score 2; ( D ) score 3 (x400).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunolocalization of M1-like macrophages in the decidual vessels with acute atherosis. Multiplex immunofluorescence staining showing nuclear staining (4',6-diamidino-2-phenylindole, DAPI, blue), smooth muscle actin (SMA, green), cytokeratin-7 (CK7, yellow), CD68 (red), CD80 (pink), interleukin (IL)-12A (purple), and inducible nitric oxide synthase (iNOS, cyan) in the decidua basalis with acute atherosis. Representative images showing (A) the immunohistochemistry and fluorescence views and (B) magnification of an M1-like macrophage in the vessel wall of a non-transformed decidual vessel with acute atherosis. Representative images showing (C) the immunohistochemistry and fluorescence views and (D) magnification of an M1-like macrophage near a transformed decidual vessel with acute atherosis. Phenoptics was performed to generate separate and merged immunofluorescence images (B & D) and to convert fluorescence images to the immunohistochemistry view (A & C). Black arrows in the immunohistochemistry view and dotted boxes in the fluorescence view indicate an M1-like macrophage. Images are representative of three experiments per group. Images were taken at 200x magnification, and a close-up of an M1-like macrophage is shown. Scale bars = 50 um (original image) or 5 um (close-up image)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Representative images for immunohistological staining (brown) for iNOS in the hindlimbs of CIA mice that were untreated ( A and B ), or treated with Dex ( C and D ), low ND-ODA ( E and F ), high ND-ODA ( G and H ) or ND-ODA-Dex ( I and J ). N = 2-3 hindlimbs on 1-3 mice. Images magnified at x4 (left column) and x20 (right column). The boxed sections represent the area that has been magnified. All scale bars are 200 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Multiplex IHC analysis of PTGS2 co-localization with macrophage markers CD68, iNOS, Arg1, MRC1/CD206, and CD163: ( a ) two examples of co-localization signals in the M1 series (CD68-iNOS-PTGS2), pseudocolors have been obtained overlaying the black&white mask of each marker on the first slide of the series; ( b ) two examples of co-localization signals in the M2 series (Arg1-MRC1-CD163-PTGS2); ( c ) left, mean Pearson's coefficients of PTGS2 and CD68 co-localization; right: a simplified representation of the mean overlay between PTGS2 and each marker using Manders' overlap coefficients. Slices indicate the extent of PTGS2 signal in the positive areas of each marker (complete analysis is shown in Supplementary Figure S2 ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Miconazole reduces neuroinflammation in LPS-induced mice hippocampus. The immunostaining of GFAP ( a ), Iba-1 ( b ), iNOS ( c ), and COX-2 ( d ) proteins in the hippocampus was performed in 10-um-thick sections of mice brains with specific primary antibodies and biotinylated secondary antibodies. In the upper-right values mean the dyed density in the hippocampus area (DG, CA1, CA3). Quantifications of the areas positively labeled with GFAP, Iba-1, iNOS, or COX-2 were performed using the ImageJ. Quantification results ( n = 3 mice per group) were analyzed using two-way ANOVA, followed by Bonferroni posttests. Data are expressed as the mean +- SEM (two-way ANOVA for repeated measures followed by post-hoc Bonferroni, # p < 0.05 ## p < 0.01, ### p < 0.0001 vs. Con, $ p < 0.05, $$ p < 0.01, $$$ p < 0.0001 vs. MCZ, * p < 0.05, ** p < 0.01, *** p < 0.0001 vs. MCZ + LPS). Representative images of immunohistochemistry staining with respective antibodies are shown here. Full immunohistochemistry results are included in Supplementary Fig. 4 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Knockdown of CISD2 expression augmented tendency for M1 microglial polarization and enhanced proinflammatory response in non-stressed EOC microglial cells. The conditions were set as follows: (i) scrambled RNA-transfected control cells; (ii) siCID2-transfected cells. (A) Confirmation of knockdown efficiency of CISD2 mRNA and protein (B) expression in microglia. (C) Expression of TNF-alpha mRNA, (D) IL-1beta mRNA, (E) iNOS mRNA, and (F) COX2 mRNA under all conditions. (G) iNOS protein expression in siCID2-transfected EOC microglial cells. The upper panel demonstrates the immunoblot results of iNOS at 135 kDa, and beta-actin (45 kDa) serves as an internal control; the lower panel indicates the mean (+-SEM) of iNOS/beta-actin band intensity in ratio with the control group. Vertical bars indicate the mean +- (SEM) of mRNA or protein expression ( n = 3). * p < 0.05; ** p < 0.01; *** p < 0.001 indicate statistically significant difference when compared to scrambled control cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 CMS reduced oxidative stress, inflammation, and apoptosis in DR rats. STZ-treated rats were treated with DMSO, 10 mg/kg CMS, 50 mg/kg CMS, and 100 mg/kg CMS. n = 15 per treatment. ( A ) Expression pattern of iNOS as determined by RT-qPCR in rat retinal tissues, normalized to beta-actin. ( B ) Representative Western blots of iNOS protein and its quantitation in rat retinal tissues, normalized to beta-actin. ( C ) Expression of NO measured by nitrite test in rat retinal tissues. ( D - F ) Expression patterns of IL-6 ( D ), TNF-alpha ( E ), and CRP ( F ) measured by ELISA in the cell supernatant. ( G , H ) Representative images of apoptotic cells (x 400) (scale bar = 25 mum) ( G ) and cell apoptosis in rat retinal tissues ( H ) by TUNEL. ( I ) Representative Western blots of cleaved caspase-3 protein and its quantitation in rat retinal tissues, normalized to beta-actin. ( J , K ) Representative Western blots of Cyt-C protein and its quantitation in the cytoplasm and mitochondria, normalized to beta-actin. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to the sham-operated rats, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to the rats injected with STZ and treated with DMSO. The results were measurement data, which were expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test (n = 15). iNOS, inducible nitric oxide synthase; CMS, coumestrol, STZ, streptozotocin; DMSO, dimethyl sulfoxi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 CMS suppressed HG-induced oxidative stress and inflammation in hRMECs through activating SIRT1. hRMECs were treated with sh-NC, sh-SIRT1, sh-SIRT1 + DMSO and sh-SIRT1 + CMS, respectively. ( A ) Expression pattern of iNOS as determined by RT-qPCR in hRMECs, normalized to beta-actin. ( B ) Representative Western blots of iNOS protein and its quantitation in hRMECs, normalized to beta-actin. ( C ), Expression pattern of NO as determined by nitrite test in hRMECs. ( D - F ), Expression patterns of IL-6 ( D ), TNF-alpha ( E ), and CRP ( F ) as measured by ELISA in the cell supernatant. * p < 0.05, ** p < 0.01, *** p < 0.001, compared to sh-NC-treated cells, and # p < 0.05, ## p < 0.01, ### p < 0.001, compared to cells stimulated with sh-SIRT1 and treated with DMSO. The results were measurement data and expressed as mean +- standard deviation. Comparisons between multiple groups were analyzed by one-way ANOVA with Tukey''s post hoc test. The cell experiments were repeated three times independently. NC, negative control; CMS, coumestrol, HG, high glucose; hRMECs, human retinal microvascular endothelial cells; SIRT1, sirtuin 1; DMSO, dimethyl sulfoxide; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; IL-6, interleukin-6; TNF-alpha, tumor necrosis factor alpha; CRP, C-reactive protein; ELISA, Enzyme linked immunosorbent assay; ANOVA, analysis of variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Treatment with Agac lysate in combination with tensile strain increased NADPH oxidase 4 (NOX-4) and nitric oxide synthase-2 (NOS-2). Gene ( a ) and protein expression of NOX-4 ( b ) and NOS-2 ( c ) after treatment with Agac lysate and tensile strain (cropped blots from Additional file 1 : Figure S4); n = 6; AU arbitrary units. Statistics: ordinary ANOVA with Holm-Sidak's post hoc (NOX-4 mRNA and protein) or Welch-corrected ANOVA with Games-Howell post-hoc tests (NOS-2); * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Impact of Agac lysate in combination with compressive strain on development of reactive oxygen species (ROS) and expression of nitric oxide synthase-2 (NOS-2). Gene ( a ) and protein expression of NADPH oxidase-4 ( b NOX-4), development of ROS ( c ) and NOS-2 ( d ) after treatment with Agac lysate and compressive strain (cropped blots from Additional file 1 : Figure S5); n >= 6; AU arbitrary units. Statistics: ordinary ANOVA with Holm-Sidak's post hoc or Welch-corrected ANOVA with Games-Howell post-hoc tests (NOS-2); * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 The polarization of macrophages to M2 phenotype is related to the elongated cell shape. A Fluorescence micrographs of Raw 264.7 macrophages immune-stained for arginase-1 (green), iNOS (red), and nuclear counterstain (blue) on cell plate (control), collagen and HA-collagen nanofibrous films. Scale bars: 15 mum. B Representative Western blot of arginase-1, iNOS, and tubulin of control, collagen and HA-collagen nanofibrous films and quantification of average across three separate experiments. Quantified TNF-alpha C and IL-10 D secretion from macrophages cultured on different nanofibrous scaffolds or culture plates using ELISA assay. n = 3, ** p < 0.01, *** p < 0.001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Chewing behavior during psychological stress improved oxidative stress in the tumor. ( A ) The expression level of iNOS protein in the tumor. ( B ) The expression level of 4HNE protein in the tumor. ( C ) The expression level of SOD2 protein in the tumor (** p < 0.01 vs. control group, ## p < 0.01 vs. stress group, n = 9/group). All data are expressed as mean +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Hepatic inflammatory factor expressions in the mice fed with the liquid Lieber-DeCarli ethanol diet. ( A ) Immunoblotting photograph of the ( B ) TNF-alpha, ( C ) IL-1beta, ( D ) IL-6, ( E ) NF-kappaB, ( F ) iNOS, ( G ) COX-2, ( H ) ERK1/2, ( I ) p-ERK/ERK1/2 ratio, ( J ) p38, and ( K ) p-p38/p38 ratio. Two groups of the mice were fed a normal diet (NOR group) or a liquid Lieber-DeCarli ethanol diet (EtOH group) without the administration of test materials. The other ethanol-induced liver injury mice were administered silymarin (200 mg/kg bw/day), a low dose of monascin (0.615 mg/kg bw/day; MS-L group), a high dose of monascin (3.075 mg/kg bw/day; MS-H group), ankaflavin (0.3075 mg/kg bw/day; AK-L group), or a high dose of ankaflavin (1.5375 mg/kg bw day; AK-H group). Data are presented as mean +- SD ( n = 8). * p < 0.05, ** p < 0.01, and *** p < 0.001 vs. EtOH group. Abbreviations: TNF-alpha, tumor Necrosis Factor-alpha; IL-1beta, Interleukin 1 beta; IL-6, Interleukin 6; NF-kappaB, nuclear factor kappa-light-chain-enhancer of activated B; iNOS, inducible nitric oxide synthase; COX-2, cyclooxygenase-2; and ERK1/2, extracellular signal-regulated kinase 1/2.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
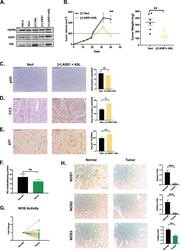
- Experimental details
- Fig. 4 Combined re-expression of ASS1 and ASL suppresses growth in vivo. A Combined doxycycline-induced ectopic expression of ASS1+ ASL in 786-O cells. HSP90 is used as a loading control. B 786-O subcutaneous xenograft tumor growth in nude mice, where cells have doxycycline induced expression of ASS1 and ASL or empty vector ( n = 6). C Immunohistochemistry staining and quantification for phosphorylated histone H3 as a marker for proliferation in xenograft tumors. Scale bars are at 100 mum. D Immunohistochemistry staining and quantification for cleaved caspase 3 as a marker for apoptosis in xenograft tumors. Scale bars are at 100 mum. ** p < 0.01. E Immunohistochemistry staining and quantification for p21 as a marker for proliferation in xenograft tumors. Scale bars are at 100 mum. *** p < 0.001. F Nitric oxide metabolite levels in matched ccRCC tumors and normal kidney as measured using gas-phase chemiluminescence ( n = 20). G Nitric oxide synthase activity measured by radiolabeled citrulline generation, plotted as a fold change between matched normal and ccRCC tumor tissues ( n = 20). H Representative immunohistochemistry images with quantification for NOS1,2,3 protein expression in matched tumor-normal pairs (Biomax OD-CT-UrKid03-002) ( n = 31). Scale bars represent 200 mum. **** p < 0.001, paired t -test
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot and gene expression analysis of LPS-induced hippocampal injury, resulting inflammatory signaling pathways. Mice were sacrificed and brain tissues were removed after the final LPS injection. The hippocampus was isolated from brain tissue and prepared either protein lysates ( n = 6-9 to each group) or mRNA ( n = 4-6 to each group) samples were prepared for further analysis. qPCR analysis was used to determine the mRNA expression levels of pro-inflammatory cytokines including ( A ) Tnfa, ( B ) Il1b, and ( C ) Il6. Western blot analysis of ( D ) NLRP3, iNOS, p-p65, p-65, IkappaBalpha, TLR4, and actinin were performed ( n = 4 to each group). The data were expressed as mean +- SEM. ## p < 0.01 and ### p < 0.001 for Con vs. LPS, * p < 0.05, and ** p < 0.01 for LPS vs. drug-administrated groups.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Spinal cord injury (SCI) downregulates Cisd2 and enhances inflammatory response in the injured spinal cord of mice subjected to spinal cord hemisection. The secretion of various inflammatory mediators in the cerebrospinal fluid (CSF) of rats with SCI. ( A ) Experimental groups and the schedule of treatment with anti-CISD2 antibody in a mouse model of SCI. ( B ) The spinal cord levels of Cisd2 mRNA in the 1, 24, and 48 h post-SCI groups were significantly decreased compared with those in the sham operation group. ( C ) Under the same conditions as those in ( B ), the spinal cord levels of Tnfa mRNA were significantly increased. For B and C, vertical bars indicate mean +- standard error of mean (SEM) ( n = 6 for each group). * p < 0.05, ** p < 0.01, and *** p < 0.001 (pair-wise multiple comparisons between groups were performed using the Newman-Keuls test). The spinal cord levels of the Cisd2 protein were decreased ( D ) and those of Nos2 were increased ( E ) in the 24 h post-SCI group compared with those in the sham operation group. For D and E, the upper panel indicates representative immunoblot of the protein expression of Cisd2 (15 kDa) ( D ) and Nos2 (135 kDa); ( E ) Gapdh (35 kDa) was used as an internal control. For ( D ) and ( E ), vertical bars indicate mean +- SEM of protein levels ( n = 3 for each group). * p < 0.05, sham operation group vs. SCI groups (independent two-sample t -tests). ( F ) Detection of inflammatory mediators in the CSF using the cytokine antibody
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 miR-17 inhibits cartilage destruction by targeting multiple pathological catabolic factors. a Luciferase activity of 293 T cells co-transfected with dual-luciferase reporter constructs containing wild-type or mutated 3'-UTRs, as well as negative control (mimic NC) or miR-17 mimic. The data presented are mean percentage changes over mimic NC +- s.e.m. n = 6 biologically independent samples (NC + wt UTR, miR-17+wt UTR); n = 4 biologically independent samples (NC + mut UTR, miR-17 + mut UTR). b Mouse articular chondrocytes were transfected with miR-17 mimic (50 nM) or mimic NC and treated with or without IL-1beta (5 ng/mL) for 24 h. The protein levels of catabolic and anabolic factors were determined by western blot followed by densitometry analysis. Blots are representative of three independent experiments. c , d qRT-PCR analysis of catabolic genes in knee cartilage ( c ), and representative images of IHC staining and quantification of MMP13 + , ADAMTS5 + and NOS2 + cells in joint sections ( d ) of mice subjected to sham or DMM surgery and intra-articular injections of agomir-NC or agomir-17 (1.5 nmol) for 4 weeks, beginning at 4 weeks after surgery. n = 5 biologically independent samples per group in c . For MMP13 and ADAMTS5 staining, n = 4 mice (sham); n = 5 mice (DMM, DMM + agomir-17). For NOS2 staining, n = 4 mice per group. e , f qRT-PCR analysis of catabolic genes ( e ) and representative images of MMP13, ADAMTS5, and NOS2 immunostaining, and quantification of pos
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Endogenous miR-17 was upregulated by GDF-5 to suppress cartilage destruction in DMM mice. a , b FISH for miR-17 and safranin O/fast green staining of joint sections ( a ), and quantification of miR-17-positive cells and OARSI scores ( b ). Veh (PBS) or GDF-5 (100 ng per injection) was injected weekly for 4 weeks, beginning at 4 weeks after sham or DMM surgery. n = 4 mice per group for cell counting. For OARSI grade, n = 4 mice (sham); n = 6 mice (DMM); n = 5 mice (DMM + GDF-5). c - e qRT-PCR analysis of catabolic genes ( c ); safranin O/fast green staining and IHC staining for MMP13, ADAMTS5 and NOS2 ( d ); OARSI scores; and quantification of cells positive for immunostaining ( e ) from knee joints. A combination of GDF-5 (0 or 100 ng) and 3 nmol of the antagomir (antagomir-17 or antagomir-NC) was injected intra-articularly. The injections were performed weekly for 4 weeks, beginning at 4 weeks after sham or DMM surgery. n = 4 biologically independent samples (sham + antagomir-NC) and n = 5 biologically independent samples for other groups in ( c ). For OARSI scores, n = 4 mice (sham); n = 7 mice (DMM); n = 9 mice (GDF-5 + antagomir-NC, GDF-5 + antagomir-17). For MMP13 staining, n = 5 mice (antagomir-17); n = 4 mice in other groups. For NOS2 and ADAMTS5 staining, n = 4 mice per group. All scale bars, 100 mum. Data were presented as boxplots or mean +- s.e.m. Boxplots: center line, median; box limits, 25th to 75th percentiles; whiskers, min to max. * P < 0.05, ** P < 0.01, and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Cartilage degeneration was developed in miR-17~92 cKO mice. a , b Safranin O/fast green staining and H&E staining of knee joint cartilage ( a ), OARSI grade ( n = 6 mice in control and cKO 8wk; n = 9 mice in cKO; n = 8 mice in cKO + agomir-17), cartilage thickness ( n = 6 mice per group) and cell counts of superficial chondrocytes ( n = 5 mice in control; n = 6 mice for other groups) in femoral condylar cartilage ( b ) from miR-17~92 fl/fl (control), miR-17~92 cKO and agomir-17-injected cKO mice at 4 or 8 weeks after tamoxifen injection, were presented. The arrows in ( a ) indicated cartilage degeneration (left panels) and superficial chondrocytes (right panels). The triangles in ( a ) indicated splitting in cartilage matrix. c , d IHC staining of MMP13, ADAMTS5 and NOS2 ( c ), and qRT-PCR analysis of catabolic factors ( d ) in cartilage of control and miR-17~92 cKO mice at 4 weeks after tamoxifen injection. n = 4 mice per group in ( c ) and n = 3 mice per group in ( d ). Data were presented as boxplots (center line, median; box limits, 25 to 75th percentiles; whiskers, min to max), or the mean +- s.e.m. values. * P < 0.05, and *** P < 0.001. Mann-Whitney U test (two sided) for OARSI grade; one-way ANOVA with Bonferroni's test for cartilage thickness and cell number; two-sided Student's t test for qRT-PCR. All scale bars, 100 mum. Exact P values in ( d ): 0.2236 ( Mmp3 ), 0.7143 ( Mmp13 ),0.0513 ( Adamts5 ), 0.8648 ( Nos2 ). Source data are provided as a Source Data file .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 5 Inducing effect on macrophage polarization of cells RAW264 cultured with leaching solutions from PLA, 1% and 5% BG-PLA. (A) . RT-PCR; (B) Immunofluorescence.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Obesity-induced adipose inflammation is attenuated by lactate depletion in adipocytes. Wildtype (WT) and adipocyte-specific Ldha knockout (AKO) mice were fed with standard chow (STC) or high fat diet (HFD) for 4 months before epididymal white adipose tissue (eWAT) was isolated for analysis. a HE staining of eWAT; scale bar, 100 mum. b Immunofluorescence staining of iNOS and F4/80 in eWAT; scale bar, 100 mum. c - f SVF in eWAT was subjected to flowcytometry analysis for macrophage subtypes. STC-WT: n = 5 ( c - f ); STC-AKO: n = 4 ( c-f ); HFD-WT: n = 12 ( d ) or n = 11( e ) or n = 10 ( f ); HFD-AKO: n = 7 ( c - f ) biologically independent animals. g - j mRNA expression of g Il-1beta (STC-WT: n = 5; STC-AKO: n = 4; HFD-WT: n = 6; HFD-AKO: n = 6 biologically independent animals), h Cd11c (STC-WT: n = 4; STC-AKO: n = 4; HFD-WT: n = 7; HFD-AKO: n = 6 biologically independent animals), i Tnf (STC-WT: n = 4; STC-AKO: n = 4; HFD-WT: n = 9; HFD-AKO: n = 6 biologically independent animals) and j Ccl12 (STC-WT: n = 4; STC-AKO: n = 4; HFD-WT: n = 8; HFD-AKO: n = 7 biologically independent samples) in eWAT. k - l Serum levels of k IL-1beta and l MCP1. n = 4 biologically independent animals. Data represent mean +- SEM; Significance was calculated by two-way ANOVA with post hoc Bonferroni correction ( d-i ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Lactate potentiates IL-1beta expression in mouse and human inflammatory macrophage. a - d Conditioned medium (CM) of epididymal white adipose tissue (eWAT) was collected from wildtype (WT) and adipocyte-specific Ldha knockout (AKO) mice. Bone marrow derived macrophages (BMDM) were cocultured in CM for 24 hr. a Illustration of the co-culture experiment. b , c BMDM mRNA expression of b Il-1beta , n = 4 biologically independent animals, and c Tnf , STC-WT: n = 4; STC-AKO: n = 3; HFD-WT: n = 3; HFD-AKO: n = 4 biologically independent animals. d Il-1beta mRNA in BMDMs cocultured in CM from WT and AKO eWAT. One group of BMDM was cocultured in CM from obese eWAT with AZD3965 (100 nM). n = 4 biologically independent samples. e - i Unelicited or inflammatory BMDMs were treated with 20 mM lactate or 20 mM NaCl as control in vitro. e - f Lactate content in cell lysate was measured after BMDM was cultured with medium containing 20 mM C3- 13 C lactate for 24 hr. e Lactate containing zero, 1, 2 and 3 13 Carbons (m + 0, m + 1, m + 2, m + 3) in cell lysate. n = 3 biologically independent samples. f Relative enrichment of endogenous and m + 1 13 C lactate in cell lysate. g , h mRNA level of g Il-1beta and h Tnf . n = 4 biologically independent samples. i Concentration of IL-1beta in BMDM medium. n = 4 biologically independent samples. j mRNA level of iNOS . n = 4 biologically independent samples. k Western blotting of iNOS. l - n Human CD14 + monocytes were differentiated to macrophages and t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Human adipose lactate level is positively correlated with adipose inflammation and insulin resistance independent of BMI. a - e Correlation between lactate levels in human omental adipose tissues with a BMI, b fasting insulin, c fasting glucose, d HOMA-IR and e HOMA-beta index. n = 65 subjects. f - m 7 omental fat samples were randomly selected from low and high lactate groups (Low_Lac vs. High_Lac). f Inflammatory cytokines mRNA level in adipose. n = 7 individuals. g HE staining of omental fat; scale bar, 200 mum. Arrows point at the crown like structures (CLSs). h CLS numbers in omental fat. n = 7 individuals. i , j Immunofluorescence staining of iNOS and HIF-1alpha and human macrophage marker CD14; scale bar, 40 mum. k , l RNASeq of omental fat ( n = 7 individuals). k GO enrichment of upregulated pathways in High_Lac group. l Heatmap showing differentially expressed genes in GO:0002274 and GO:0002253. m Transcription factor (TF) enrichment in upregulated genes in High_Lac group. Significantly enriched TFs ( p < 0.05) were highlighted in blue. HIF-1alpha was shown in the enlarged region. n Illustration of the study hypothesis. Data represent mean +- SEM; Statistics were performed using spearman correlation analysis ( a - e ), two-way ANOVA with post hoc Bonferroni correction ( f ) or two-tailed student's t test ( h ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Continuous infusion of IIK-7 attenuated PSNT-induced activation of HMGB-1, pERK1/2, iNOS, STAT3 and caspase-3 in vivo. Representative immunoblots are HMGB-1, P44/42 MAPK, iNOS, STAT 3 and casp-3 from the left dorsal quadrant portion of the lumbar spinal cord lysate of sham surgery or PSNT rats continuously infused with either vehicle or IIK-7 for 7 days using an osmotic pump. Abbreviations: IIK-7, N-Butanoyl 2-(9-methoxy-6H-iso-indolo[2,1-a]indol-11-yl)-ethan-amine; P44/42 MAPK, P44/42 mitogen-activated protein kinase; iNOS, Inducible nitric oxide synthase; STAT3, Signal transducer and activator of transcription 3; CASP-3, caspase3; HMGB-1, High mobility group box 1.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot