Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Other assay [41]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-682G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-JNK1/JNK2 (Thr183, Tyr185) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- This antibody is reactive to human and rat JNK1&2. Other species of JNK1&2 have not been tested, and JNK3 (found primarily in neuronal cell lines) has not been detected. The antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated JNK1&2. The final product is generated by affinity chromatography using a JNK1&2-derived peptide that is phosphorylated at threonine 183 and tyrosine 185. Positive controls used: HEK 293 +/- UV irradiation treatment; PC12 cells +/- sorbitol.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis. JNK (pTpY183/185) phosphospecific antibody. 293 and PC12 cells were untreated or treated with either UV light or sorbitol, respectively. Western Blots were performed using either JNK (pTpY183/185) (Product # 44-682G) or p38 (pTpY180/182) (Product # 44-684G) primary antibodies. Anti-rabbit secondary antibody conjugated to Alexa fluor 680 was used for detection. Data was analyzed on the LI-COR Odyssey® Infrared Imaging System. JNK (pTpY183/185): Lane 1 - 293 control; 5 µg; Lane 2 - 293 + UV, 5 µg; Lane 3 - PC12 control, 20 µg; Lane 4 - PC12 + sorbitol, 20 µg. p38 (pTpY180/182): Lane 6 - 293 control, 5 µg; Lane 7 - 293 + UV, 5 µg. (Product # 44-682G)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of JNK1 + JNK2 (pT183 + pT185) was performed by loading 20 µg of HEK-293 (lane1), HEK-293 treated for 5 minutes with 200 mM of Anisomycin (lane2), HEK-293 treated for 20 minutes with UV (lane3), MCF7 (lane4), MCF7 treated for 5 minutes with 200 mM of Anisomycin (lane5), K562 (lane6), K562 treated for 20 minutes with UV (lane7), HeLa (lane8) and HeLa treated for 20 minutes with UV (lane9) cell lysate using Novex®NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5% skim milk for 1 hour at room temperature. JNK1 + JNK2 (pT183 + pT185) was detected at ~ 46 and 55 kDa using JNK1 + JNK2 (pT183 + pT185) Rabbit Polyclonal Antibody (Product # 44-682G) at 1:1000 dilution in 5% skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
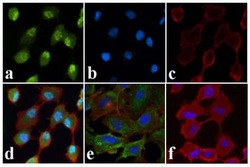
- Experimental details
- Immunofluorescent analysis of JNK1/2 (pT183/pT185) was done on 70% confluent log phase A549 cells treated with Anisomycin (25 µg/mL for 30 min). The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with JNK1/2 (pT183/pT185) Rabbit polyclonal Antibody (Product # 44-682G) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing translocation of JNK1/2 (pT183/pT185) to the nucleus upon Anisomycin treatment. Panel e is untreated cells showing cytoplasmic localization. Panel f shows no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
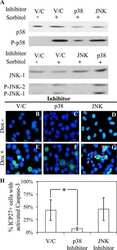
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
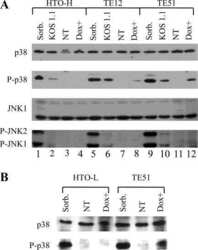
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 2 JNK and ERK activation is required for Chal-24-induced cell death (A) A549 cells were treated with Chal-24 (8 muM) for indicated times. The indicated proteins were detected by Western blot. GAPDH was detected as an input control. (B) A549 were pretreated with the indicated inhibitors for 30 min and then treated with Chal-24 (8 muM) for 30 min. The indicated proteins were detected by Western blot. GAPDH was detected as an input control. (C) A549 cells were pretreated with the indicated inhibitors, SP600125 (10 muM), U0126 (10 muM), SB203580 (5 muM) for 30 min and then treated with Chal-24 (8 muM) for an additional 30 h. Cytotoxicity was detected by LDH release assay. Data shown are the mean +- SD. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 6 Chal-24 induces degradation of c-IAPs depending on autophagy and ERK (A) A549 cells were treated with Chal-24 (8 muM) for indicated times. The indicated proteins were detected by Western blot. beta-actin or GAPDH was detected as an input control. (B) A549 cells were transfected with combination of ERK1 and ERK2 siRNAs or JNK1 and JNK2 siRNAs (10 nM each) for 24 h and then treated with Chal-24 (8 muM) for an additional 30 h. The indicated proteins were detected by Western blot. GAPDH was detected as an input control. (C) A549 cells were pretreated with 3MA (10 mM), CQ (20 muM), or Wortmannin (WTM, 1 muM) for 30 min and then treated with Chal-24 (8 muM) for an additional 30 h. The indicated proteins were detected by Western blot. GAPDH was detected as an input control. (D) A549 were transfected with the indicated siRNAs(10nM) for 24 h and then treated with Chal-24 (8 muM) for an additional 30 h. GAPDH was detected as an input control. (E) A549 cells were pretreated with U0126 (10 muM) for 30 min and then treated with Chal-24 (8 muM) for indicated times. Lambda phosphatase treatment was as described in 5E. The indicated proteins were detected by Western blot. GAPDH was detected as an input control. (F) A549 were treated with Chal-24 (8 muM) for the indicated times. The indicated proteins were detected by Western blot after co-Immunoprecipitation with an antibody for RIP1 (G) A549 were pretreated with CQ (20 muM) for 30 min and then treated with Chal-24 (8 muM) for an addit
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 RIP1 knockdown potentiates cisplatin-induced cytotoxicity involving JNK activation (A, B) A549 and H460 cells (control and RIP1 stable knockdown clones 1 and 2) were treated with cisplatin (A549, 20 muM; H460, 10 muM) for 48 h. Cell death was detected with LDH release assay. Data shown are mean+-SD. **p< 0.01. RIP1 knockdown was confirmed by Western blot, beta-actin was used as an input control. (C, D) Cells were untreated or treated with cisplatin (A549, 20 muM; H460, 10 muM) for the indicated times. JNK and phospho-JNK were examined with Western blot. beta-actin was detected as an input control. (E, F) Cells were pretreated with SP600125 (10 muM) for 30 min and then treated with cisplatin (A549, 20 muM; H460, 10 muM) for an additional 48 h, cell death was detected by LDH assay. Columns shown are mean+-SD. *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Reduced MKP1 expression contributes to cisplatin-induced JNK activation and cytotoxicity in RIP1 knockdown cells (A) MKP1 was detected with Western blot. beta-actin was used as the input control. (B) pcDNA-MKP1 EE or the empty vector pcDNA were co-transfected with HA-JNK1 in A549 RIP1 knockdown cells (1#). The cells were treated with cisplatin (20 muM) for indicated times and JNK1, phospho-JNK1 and Xp-MKP1 EE were detected with Western blot with anti-HA, -phospho-JNK and -Xpress antibody, respectively. beta-actin was used as an input control. (C) The cells were transfected with pcDNA-MKP1 EE or the empty vector pcDNA with EGFP in A549 RIP1 knockdown cells (1#) and treated with cisplatin (20 muM) for 24 h. Photographs were taken under a fluorescence microscope. Cell survival was quantified by counting cells with green fluorescence and normal morphology. Data shown was the mean +- SD, *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2 PRD phosphorylation of MAP2 is necessary and sufficient for microtubule stabilization and MAP2-induced protrusion growth. (A) COS-7 cell were transfected with GFP, GFP-MAP2, GFP-MAP2 T1619D,T1622D,T1625D (henceforth GFP-MAP2-DDD), or GFP-MAP2 T1619A,T1622A,T1625A (henceforth GFP-MAP2-AAA), with or without MKK7-JNK1 as shown. GFP-MAP2-induced protrusion growth while MKK7-JNK1 alone did not. (B) The proportion of cells that generated processes upon 48 h expression of MAP2 variant is shown. GFP-MAP2-AAA expressing cells failed to produce protrusions. (C) The number of processes per cell was counted. Expression of MKK7-JNK1 augmented the number of MAP2-generated processes. GFP-MAP2-DDD alone induced significantly more processes than did GFP-MAP2. (D) Fibroblasts transfected with Venus-tubulin together with GFP, GFP-MAP2, GFP-MAP2 T1619A,T1622A,T1625A (MAP2-AAA), or GFP-MAP2 T1619D,T1622D,T1625D (MAP2-DDD) were permeablized and the distribution of MAP2 or Venus-tubulin to the TX-100 soluble or insoluble fractions were quantified using immunoblotting. (E) Quantified data shows that GFP-MAP2 T1619A,T1622A,T1625A partitions to the soluble phase, while GFP-MAP2 T1619D,T1622D,T1625D remains in the TX-100 insoluble phase. (F) Quantitative data on Venus-tubulin stability. GFP-MAP2 T1619D,T1622D,T1625D stabilizes microtubules whereas GFP-MAP2 T1619A,T1622A,T1625A induces microtubule destabilization. (G) Polarized neurons at 12 days in vitro were immunostained for JNK1 (green) and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Combination of cisplatin and Chal-24 activates JNK-and phosphorylation of Bcl-2 (A) A549 cells were treated with cisplatin (10 muM) and Chal-24 (1 muM) alone or in combination for indicated times. JNK1 and phospho-JNK were examined by Western blot. GAPDH was detected as an input control. (B) the cells were pretreated with SP600125 (10 muM) for 30 min, and then treated with cisplatin (10 muM) and Chal-24 (1 muM) for an additional 48 h, cell death were detected by LDH assay. ** p < 0.01. (C) A549 cells were treated with cisplatin (10 muM) and Chal-24 (1 muM) alone or in combination for indicated times. The indicated proteins were detected by Western blot. GAPDH was used as an input control. (D, E) A549 cells were pretreated with CQ (20 muM) or MG132 (5 muM) for 30 min, respectively, then treated with cisplatin (10 muM), Chal-24 (1 muM) or in combination for an additional 8 h. The indicated proteins were detected by Western blot, GAPDH was detected as an input control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 The poly(I:C)- or LPS-induced association of c-Jun with the Ifnb promoter is prevented by BET inhibitors without inhibiting the phosphorylation of c-Jun or ATF2 ( A and B ) The experiment was performed as in Figure 4 except that an anti-c-Jun antibody was used. ( C and D ) RAW cells were incubated for 1 h with 1.0 muM BI-2536, 1.0 muM JQ1 or 10 muM of the JNK inhibitor JNK-IN-8 and then stimulated for 1 h with 10 mug/ml poly(I:C) ( C ) or for 30 min with 100 ng/ml LPS ( D ). The cells were lysed and 20 mug of cell extract protein was denatured in SDS and subjected to SDS/PAGE followed by immunoblotting with antibodies that recognize the phosphorylated forms of JNK1/2, c-Jun and ATF2 as well as GAPDH. JNK-IN-8 binds covalently to JNK1 and JNK2 leading to a small decrease in their electrophoretic mobilities. IP, immunoprecipitation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Injury-induced signaling in avulsed murine epiphyses involves transforming growth factor beta-activated kinase 1 (TAK-1) and is retained in TRAF-6- and MyD88- mice. A , Cre recombination-mediated cartilage-specific TAK-1 deletion was induced by tamoxifen (TM; 50 mg/kg) as described in Materials and Methods. Murine proximal femoral epiphyses (2 hips per time point) were avulsed and either snap-frozen (time point 0 minutes) or cultured for the indicated times in serum-free medium. Cartilage lysates were analyzed by Western blotting for TAK-1, phospho-JNK (ph-JNK), and total ERK (T-ERK; loading control). B , Murine femoral epiphyses were avulsed, and lysates were analyzed by Western blotting for ubiquitinated proteins and ERK. Ub-HRP = horseradish peroxidase-conjugated ubiquitin. C , TRAF-6- mice were generated as described in Materials and Methods. Top, DNA was extracted from the tips of the tails of 6-day-old animals for TRAF-6 genotyping polymerase chain reaction. Bottom, Murine femoral epiphyses (1 hip per time point) were avulsed, and lysates were analyzed by Western blotting for phosphorylated JNK and total ERK (loading control). D , Murine femoral epiphyses from MyD88- mice were avulsed, and lysates were analyzed by Western blotting for phospho-TAK-1, TAK-1, phospho-JNK, total ERK, phospho-p38, and phospho-ERK. HO = homozygous; HET = heterozygous; WT = wild-type; KO = knockout.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Wnt9b signals through the noncanonical pathway to regulate tubule diameter Western blots of total protein extracted from wild-type and Wnt9b neo/neo kidneys probed with an antibody specific to the dephosphorylated (active) form of beta-catenin show no significant differences in canonical Wnt activity compared to wild type (a). Section in situ hybridization with a probe for the beta-catenin target axin-2 also shows no significant decrease in canonical activity in P1 Wnt9b neo/neo kidneys (c) compared to wild type (b). Note that there is no ectopic axin2 expression in cystic proximal tubules (asterisks in c). (d) Western blots indicate that activated Rho is significantly decreased in Wnt9b neo/neo kidneys at P1 relative to total (+GTP control) Rho levels. Addition of GDP (+GDP) to inactivate Rho was used as a negative control. Phosphorylated Jnk2 is also significantly decreased in the Wnt9b neo/neo kidneys at P1 relative to total levels of Jnk2 (d). Blots shown are representative examples of data gathered from at least 3 different blots from 3 independent protein extractions.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Cartilage injury leads to activation of transforming growth factor beta-activated kinase 1 (TAK-1), and inhibition of TAK-1 abolishes injury-dependent inflammatory gene expression. A, Porcine metacarpophalangeal (MCP) joints were equilibrated at 37degC for 1 hour. Articular cartilage was dissected and either snap-frozen or kept in serum-free medium for the indicated times. Cartilage was then washed with 1x phosphate buffered saline and lysed in radioimmunoprecipitation assay buffer. Lysates were analyzed by Western blotting (WB) for phospho-TAK-1 (ph-T187), phospho-JNK, and ERK. B, A TAK-1 activity assay was carried out as described in Materials and Methods to confirm TAK-1 activation. IP = immunoprecipitation; ph-MBP = phospho-myelin basic protein. C and D, Porcine MCP joints were injected with 5 mu M TAK-1 inhibitor 5z-7-oxozeanol (OXO) or DMSO (vehicle [V]), as described in Materials and Methods, and kept at 37degC for 1 hour. Joints were then opened and cartilage was dissected and snap-frozen (time point 0 minutes) or cultured for the indicated times with or without TAK-1 inhibitor. Cartilage lysates were analyzed for phospho-JNK and total ERK (T-ERK) ( C ) and for IkappaB and total ERK ( D ). E, Cartilage was snap-frozen (time point 0 hours) or kept at 37degC for 4 hours with or without 5 mu M TAK-1 inhibitor (OXO). RNA was extracted from the tissue and used in quantitative reverse transcriptase-polymerase chain reaction studies of a panel of typical inflammator
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Higher molecular weight (Mwt) species of phosphorylated transforming growth factor beta-activated kinase 1 (TAK-1) in injured porcine and murine cartilage. A, Porcine metacarpophalangeal joints were processed as described in Figure 1. Cartilage was then dissected and either snap-frozen (time point 0 minutes) or cultured for 5 or 10 minutes. Cartilage lysates were analyzed by Western blotting for TAK-1 phosphorylated at T187 (ph-T187), T184/T187, and S192, phospho-JNK, and total ERK (T-ERK; loading control). B, Murine proximal femoral epiphyses (2 per time point) were subjected to avulsion injury, and were then either snap-frozen (time point 0 minutes) or cultured for 5 or 10 minutes in serum-free medium. Epiphyses were analyzed by Western blotting for phospho-TAK-1, phospho-JNK, and total ERK (loading control) as described in A . Representative results from 4 independent experiments are shown. NS = nonspecific.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Increased K 63 -linked polyubiquitin in injured porcine articular cartilage. Porcine metacarpophalangeal (MCP) joints were equilibrated at 37degC for 1 hour. A, Cartilage was dissected and either snap-frozen (time point 0 minutes) or cultured for the indicated times. K 63 -linked ubiquitin (UB-K 63 ) proteins were obtained from cartilage lysates using a pull-down assay with receptor associated protein 80 (RAP-80) ubiquitin interacting motifs (UIMs) as described in Materials and Methods, and proteins bound to the beads were analyzed by Western blotting (WB) with an anti-K-63-linked ubiquitin antibody. Lysates were analyzed by Western blotting for phospho-JNK (ph-JNK) and total ERK (T-ERK). Representative results from 3 independent experiments are shown. B, Porcine MCP cartilage sections (5-7 mum) were deparaffinized, rehydrated, and treated for 10 minutes with proteinase K for antigen retrieval. Endogenous peroxidase activity was blocked with 3% H 2 O 2 for 10 minutes, followed by blocking with avidin-biotin for another 10 minutes. Sections were then blocked for 2 hours at room temperature with 10% goat serum in phosphate buffered saline, and incubated overnight at 4degC with anti-K 63 ubiquitin antibody (1:50 dilution). Sections were washed and then incubated for 2 hours in the dark with secondary antibodies conjugated with streptavidin. Antibody binding was visualized using a Vectastain kit and DAB Peroxidase Substrate Kit (Vector). Slides were then counterstained w
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Additional file 1: Figure S1. Representative western blots of phospho-JNK (46 kDa), phospho-ERK (44 kDa), phospho-p38 MAPK (43 kDa), phospho-TAK1 (82 kDa), phospho-AMPK (62 kDa), phospho-GSK3 (46 kDa), and GAPDH (37 kDa) in midguts of mosquitoes at 30 min post-infection with P. falciparum with or without 100 nM ABA. (PDF 872 kb)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Increased miR-940 expression is involved in MKP1 suppression in RIP1 knockdown cells (A) Total RNA isolated from A549 cells (control and RIP1 knockdown) was used for detection of miR-940 with qPCR. (B) The cells were transfected with negative control or miR-940 miScript miRNA inhibitor for 48 h, MKP1 expression was detected with Western blot, and beta-actin was detected as an input control. The intensity of the individual bands was quantified by Quantity One(r) Software and normalized to the corresponding input control (beta-actin) bands. (C) The cells were transfected with the indicated miRNA inhibitor (10 nM) for 24h, and treated with cisplatin (20 muM) for 8 h. JNK1 and phospho-JNK1 were detected with Western blot. beta-actin was detected as an input control. (D) The cells transfected with negative control or miR-940 inhibitor (10nM) for 24h, then the cells were left untreated or treated with cisplatin (20 muM) for an additional 48 h. Cell death was detected with LDH assay. Data shown are the mean+-SD. *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Progesterone up-regulates the JNK pathway via GADD45alpha activation to inhibit progression of colonic carcinoma. (A) Heat map depicting progesterone-induced changes in the expression profile of genes that were assessed on the PCR microarray. Blue and red represent low and high gene expression levels in pinnae, respectively. (B) Selection of genes that were altered the most in SW620 cells. Genes are presented alphabetically. RT-PCR was used to investigate the effect of progesterone on proliferation-related genes such as c-Jun, JNK1, JNK2, GADD45alpha, P15, BAX, BCL2L1 and NCF1 in (C) SW620 and (D) LoVo cells Efficiency of knockdown GADD45alpha in (E) SW620 and (I) LoVo cells, respectively. Expression of GADD45alpha, JNK1, JNK2, c-Jun, BCL-2, and Ki67 in (F) SW620 and (J) LoVo cells was analysed using RT-PCR, and GADD45alpha, phosphorylation of JNK, JNK, c-Jun, BCL-2 and Ki67 in (G) SW620 and (K) LoVo cells were analysed using western blot. Relative expression of phosphorylation of JNK to total protein of JNK in (H) SW620 and (L) LoVo cells. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Activation of the NF-kappaB and JNK pathways and their upstream regulators in injured porcine cartilage. Porcine metacarpophalangeal joints were equilibrated at 37degC for 1 hour. Articular cartilage was dissected and either snap-frozen or kept in serum-free medium for the indicated times. A and B, Cartilage was washed with 1x phosphate buffered saline and lysed in radioimmunoprecipitation assay buffer. Lysates were analyzed by Western blotting for phospho-IKK (ph-IKK), phospho-IkappaB, and IkappaB ( A ) and for phospho-MKK-4, phospho-JNK, phospho-activating transcription factor (phsopho-ATF-2), phospho-p38, and phospho-ERK ( B ). ERK was blotted to check that the lanes were equally loaded. Results are representative of several replicate experiments. C and D, After incubation, cartilage was embedded in OCT, and cryosections were stained with antibodies against the p65 subunit of NF-kappaB ( C ) or phospho-ATF-2 ( D ). The localization of the 2 proteins in different cartilage zones was determined using confocal microscopy. In each cryosection, a total of 500 cells were counted (100 cells in the superficial and deep zones and 300 in the middle zone). Percentages are the proportion of activated cells in the different zones. Representative images are shown. Original magnification x 60.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry