Antibody data
- Antibody Data
- Antigen structure
- References [15]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- A-11130 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-Transferrin Receptor Monoclonal Antibody (236-15375)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- The anti-human transferrin receptor antibody should be useful for the localization of transferrin receptors by immunofluorescence techniques. This antibody recognizes the human transferrin receptor and does not block binding of transferrin to the receptor. Although fluorescent transferrin conjugates have been widely used to study transferrin receptor-mediated endocytosis, antibodies to the transferrin receptor have proven to be useful for localizing transferrin receptors, studying endocytic recycling pathways and performing enzyme-linked immunosorbent assays (ELISAs). Upon receipt, store desiccated at -20°C. Stored properly, the antibody should remain stable for at least one year. To prepare a stock solution, reconstitute the antibody in PBS with 1% BSA and 2 mM sodium azide. Reconstituted solutions should remain stable for 6 months when stored at 4°C. For longer storage, divide into aliquots and freeze at -20°C.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 236-15375
- Vial size
- 50 µg
- Storage
- -20°C
Submitted references Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.
A perfused human blood-brain barrier on-a-chip for high-throughput assessment of barrier function and antibody transport.
High-Content Imaging Reveals Expansion of the Endosomal Compartment during Coxiella burnetii Parasitophorous Vacuole Maturation.
Quantitative Analysis of Endocytic Recycling of Membrane Proteins by Monoclonal Antibody-Based Recycling Assays.
Loss of F-box only protein 2 (Fbxo2) disrupts levels and localization of select NMDA receptor subunits, and promotes aberrant synaptic connectivity.
Oligomerization of Clostridium perfringens epsilon toxin is dependent upon caveolins 1 and 2.
MARCH ubiquitin ligases alter the itinerary of clathrin-independent cargo from recycling to degradation.
The activation of P2X7 receptor impairs lysosomal functions and stimulates the release of autophagolysosomes in microglial cells.
Recycling of the human prostacyclin receptor is regulated through a direct interaction with Rab11a GTPase.
The Rab5 activator ALS2/alsin acts as a novel Rac1 effector through Rac1-activated endocytosis.
Abl tyrosine kinase regulates endocytosis of the epidermal growth factor receptor.
Compartmental signal modulation: Endosomal phosphatidylinositol 3-phosphate controls endosome morphology and selective cargo sorting.
Tumour necrosis factor alpha causes hypoferraemia and reduced intestinal iron absorption in mice.
Copper and zinc cause delivery of the prion protein from the plasma membrane to a subset of early endosomes and the Golgi.
The coxsackie B virus and adenovirus receptor resides in a distinct membrane microdomain.
Salman MM, Marsh G, Kusters I, Delincé M, Di Caprio G, Upadhyayula S, de Nola G, Hunt R, Ohashi KG, Gray T, Shimizu F, Sano Y, Kanda T, Obermeier B, Kirchhausen T
Frontiers in bioengineering and biotechnology 2020;8:573775
Frontiers in bioengineering and biotechnology 2020;8:573775
A perfused human blood-brain barrier on-a-chip for high-throughput assessment of barrier function and antibody transport.
Wevers NR, Kasi DG, Gray T, Wilschut KJ, Smith B, van Vught R, Shimizu F, Sano Y, Kanda T, Marsh G, Trietsch SJ, Vulto P, Lanz HL, Obermeier B
Fluids and barriers of the CNS 2018 Aug 31;15(1):23
Fluids and barriers of the CNS 2018 Aug 31;15(1):23
High-Content Imaging Reveals Expansion of the Endosomal Compartment during Coxiella burnetii Parasitophorous Vacuole Maturation.
Larson CL, Heinzen RA
Frontiers in cellular and infection microbiology 2017;7:48
Frontiers in cellular and infection microbiology 2017;7:48
Quantitative Analysis of Endocytic Recycling of Membrane Proteins by Monoclonal Antibody-Based Recycling Assays.
Blagojević Zagorac G, Mahmutefendić H, Maćešić S, Karleuša L, Lučin P
Journal of cellular physiology 2017 Mar;232(3):463-476
Journal of cellular physiology 2017 Mar;232(3):463-476
Loss of F-box only protein 2 (Fbxo2) disrupts levels and localization of select NMDA receptor subunits, and promotes aberrant synaptic connectivity.
Atkin G, Moore S, Lu Y, Nelson RF, Tipper N, Rajpal G, Hunt J, Tennant W, Hell JW, Murphy GG, Paulson H
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Apr 15;35(15):6165-78
The Journal of neuroscience : the official journal of the Society for Neuroscience 2015 Apr 15;35(15):6165-78
Oligomerization of Clostridium perfringens epsilon toxin is dependent upon caveolins 1 and 2.
Fennessey CM, Sheng J, Rubin DH, McClain MS
PloS one 2012;7(10):e46866
PloS one 2012;7(10):e46866
MARCH ubiquitin ligases alter the itinerary of clathrin-independent cargo from recycling to degradation.
Eyster CA, Cole NB, Petersen S, Viswanathan K, Früh K, Donaldson JG
Molecular biology of the cell 2011 Sep;22(17):3218-30
Molecular biology of the cell 2011 Sep;22(17):3218-30
The activation of P2X7 receptor impairs lysosomal functions and stimulates the release of autophagolysosomes in microglial cells.
Takenouchi T, Nakai M, Iwamaru Y, Sugama S, Tsukimoto M, Fujita M, Wei J, Sekigawa A, Sato M, Kojima S, Kitani H, Hashimoto M
Journal of immunology (Baltimore, Md. : 1950) 2009 Feb 15;182(4):2051-62
Journal of immunology (Baltimore, Md. : 1950) 2009 Feb 15;182(4):2051-62
Recycling of the human prostacyclin receptor is regulated through a direct interaction with Rab11a GTPase.
Wikström K, Reid HM, Hill M, English KA, O'Keeffe MB, Kimbembe CC, Kinsella BT
Cellular signalling 2008 Dec;20(12):2332-46
Cellular signalling 2008 Dec;20(12):2332-46
The Rab5 activator ALS2/alsin acts as a novel Rac1 effector through Rac1-activated endocytosis.
Kunita R, Otomo A, Mizumura H, Suzuki-Utsunomiya K, Hadano S, Ikeda JE
The Journal of biological chemistry 2007 Jun 1;282(22):16599-611
The Journal of biological chemistry 2007 Jun 1;282(22):16599-611
Abl tyrosine kinase regulates endocytosis of the epidermal growth factor receptor.
Tanos B, Pendergast AM
The Journal of biological chemistry 2006 Oct 27;281(43):32714-23
The Journal of biological chemistry 2006 Oct 27;281(43):32714-23
Compartmental signal modulation: Endosomal phosphatidylinositol 3-phosphate controls endosome morphology and selective cargo sorting.
Fili N, Calleja V, Woscholski R, Parker PJ, Larijani B
Proceedings of the National Academy of Sciences of the United States of America 2006 Oct 17;103(42):15473-8
Proceedings of the National Academy of Sciences of the United States of America 2006 Oct 17;103(42):15473-8
Tumour necrosis factor alpha causes hypoferraemia and reduced intestinal iron absorption in mice.
Laftah AH, Sharma N, Brookes MJ, McKie AT, Simpson RJ, Iqbal TH, Tselepis C
The Biochemical journal 2006 Jul 1;397(1):61-7
The Biochemical journal 2006 Jul 1;397(1):61-7
Copper and zinc cause delivery of the prion protein from the plasma membrane to a subset of early endosomes and the Golgi.
Brown LR, Harris DA
Journal of neurochemistry 2003 Oct;87(2):353-63
Journal of neurochemistry 2003 Oct;87(2):353-63
The coxsackie B virus and adenovirus receptor resides in a distinct membrane microdomain.
Ashbourne Excoffon KJ, Moninger T, Zabner J
Journal of virology 2003 Feb;77(4):2559-67
Journal of virology 2003 Feb;77(4):2559-67
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
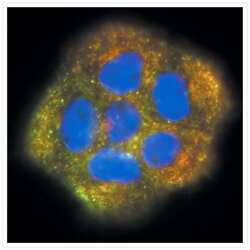
- Experimental details
- Visualization of transferrin and transferrin receptors in A431 cells. A431 cells incubated with green-fluorescent Alexa Fluor® 488 transferrin (Product # T-13342), then fixed and permeabilized. Transferrin receptors were identified with anti-transferrin receptor, mouse IgG1 monoclonal antibody (Product # A-11130) and visualized with red-fluorescent Alexa Fluor® 555 goat anti-mouse IgG antibody (Product # A-21422). Yellow fluorescence indicates regions of co-localization. Nuclei were stained with DAPI (Product # D1306, D3571, D21490).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Transferrin Receptor was performed using 70% confluent log phase MCF-7 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Transferrin Receptor (236-15375) Mouse Monoclonal Antibody (Product # A-11130) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing membranous localization. Panel e shows the control without primary antibody. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Specific co-immunoprecipitation of epsilon-toxin and caveolins. A) ACHN cells were either untreated (control) or treated with GFP-labeled toxin. Cell lysates were incubated with beads crosslinked to CAV2, E-cadherin or transferrin receptor antibodies. Proteins eluted off the beads were analyzed by immunoblotting with anti-GFP antibody. B) ACHN cells were either untreated (control) or treated with GFP-labeled toxin. Cell lysates were incubated with beads crosslinked to CAV1 antibodies. Proteins eluted off the beads were analyzed by immunoblotting with anti-GFP antibody. Representative immunoblots are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Cells infected with C. burnetii exhibit more internalized Tf . (A) HeLa cells infected with C. burnetii or left uninfected were incubated with Tf488 for the indicated times. CellProfiler was used to select only cells that emitted strong C. burnetii signal (Infected--large PV) from the total population of cells in infected cell cultures (Infected). The amount of intracellular Tf was measured by quantitative fluorescence microscopy. Plots depict percent Tf488 intensity calculated relative to uninfected cell cultures (Uninfected) at 0 min. A black asterisk indicates the mean Tf intensity of infected cell cultures is significantly greater than for uninfected cell cultures. A green asterisk indicates the mean Tf intensities of the subset of cells containing large PV is significantly greater than for infected or uninfected cell cultures. Statistical significance ( P < 0.01) was determined by two-way ANOVA using Tukey's test for multiple comparisons with the mean number of cells counted per sample equal to 1038 +- 411. (B) Similar slopes were detected in the linear first 5 min of the Tf endocytosis assay, indicating increased internalized Tf by infected cells is not due to an increase in the rate of uptake (3 days post-infection). The plot depicts mean Tf488 signal intensity (integrated intensity per cell) with the mean number of cells counted at each time point equal to 1553 +- 178. (C) Increased internalized Tf by infected cells (total population) is not a
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2 Cell seeding procedures and formation of a human brainmicrovessel using TY10 cells. (A) Cell seeding, 1st method. Representation of the steps used to increase the cell concentration before injection into the chip. Cells are allowed to settle by gravity (left and central panels) in the cell seeder and then injected as a bolus into the hollow lumen. Prior to injection, the chamber is capped with a plugged pipette tip (central panel). (B) Cell seeding, 2nd method. Representation of the steps used to inject cells into the chip in a more controlled and uniform manner compared to the 1st method. Cells are allowed to settle at the bottom of a syringe, and are then delivered into the hollow lumen at constant flow controlled by a syringe pump. (C) Immunostaining of TY10 monolayers: endothelial cell marker PECAM-1/CD31, transporters Glut-1 and transferrin receptor (TfR), and tight junction proteins ZO-1, Occludin and Claudin-5. Staining for laminin provides evidence that TY10 cells deposit extracellular matrix while in culture. Scale bar, 100 mum. (D) Representative image of a chemically fixed sample of TY10 cells after they were grown in the brain microvessel-on-a-chip with medium flowing from left to right at 1 mµL/min for 7 days. Volumetric image was obtained using a spinning disk confocal microscope. Maximum z-projection is shown for a sample stained with DAPI (nuclei, blue) and phalloidin (actin, green). Scale bar, 100 mum. (D') Representative image of a chemically
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
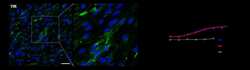
- Experimental details
- Additional file 4. Characterization of the human transferrin receptor in TY10 endothelial cells. ( a ) Immunofluorescent staining of the hTfR in TY10 endothelial cells. Scale bar is 50 um. ( b ) Flow cytometry analysis of cell surface binding of anti-TfR MEM-189 to TY10 endothelial cells in the presence and absence of transferrin (25 ug/mL), EC 50 = 0.44 +- 0.09 nM (-Tf); 0.5 +- 0.1 nM (+Tf).
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry