Antibody data
- Antibody Data
- Antigen structure
- References [60]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [28]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 45-0037-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD3 Monoclonal Antibody (OKT3), PerCP-Cyanine5.5, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The OKT3 monoclonal antibody reacts with an epitope on the epsilon-subunit within the human CD3 complex. The OKT3 antibody has been reported to have potent immunosuppressive properties in vivo and has been proven effective in the treatment of renal, heart and liver allograft rejection. The CD3 subunits, gamma, delta, and epsilon chains, are required for proper assembly, trafficking and surface expression of the TCR complex. CD3 is expressed by thymocytes in a developmentally regulated manner and by all mature T cells. Crosslinking of TCR initiates an intracellular biochemical pathway resulting in cellular activation and proliferation.
- Antibody clone number
- OKT3
- Concentration
- 5 µL/Test
Submitted references Broad and potent neutralizing human antibodies to tick-borne flaviviruses protect mice from disease.
Ageing promotes early T follicular helper cell differentiation by modulating expression of RBPJ.
Imbalance between T helper 1 and regulatory T cells plays a detrimental role in experimental Parkinson's disease in mice.
Standardized 11-color flow cytometry panel for the functional phenotyping of human T regulatory cells.
HDAC Inhibitor, CG-745, Enhances the Anti-Cancer Effect of Anti-PD-1 Immune Checkpoint Inhibitor by Modulation of the Immune Microenvironment.
Small-molecule MMP2/MMP9 inhibitor SB-3CT modulates tumor immune surveillance by regulating PD-L1.
Relationship between T cell receptor clonotype and PD-1 expression of tumor-infiltrating lymphocytes in colorectal cancer.
IL-10 Restores MHC Class I Expression and Interferes With Immunity in Papillary Thyroid Cancer With Hashimoto Thyroiditis.
NKG2C(pos) NK Cells Regulate the Expansion of Cytomegalovirus-Specific CD8 T Cells.
Selective Killing of Activated T Cells by 5-Aminolevulinic Acid Mediated Photodynamic Effect: Potential Improvement of Extracorporeal Photopheresis.
Accumulation of TNFR2-expressing regulatory T cells in malignant pleural effusion of lung cancer patients is associated with poor prognosis.
Pyridoxal-5'-Phosphate Promotes Immunomodulatory Function of Adipose-Derived Mesenchymal Stem Cells through Indoleamine 2,3-Dioxygenase-1 and TLR4/NF-κB Pathway.
Integrating Ligand-Receptor Interactions and In Vitro Evolution for Streamlined Discovery of Artificial Nucleic Acid Ligands.
Progranulin prevents regulatory NK cell cytotoxicity against antiviral T cells.
MicroRNA‑155 inhibits the proliferation of CD8+ T cells via upregulating regulatory T cells in vitiligo.
Bispecific Antibody Approach for Improved Melanoma-Selective PD-L1 Immune Checkpoint Blockade.
Cdc42 Couples T Cell Receptor Endocytosis to GRAF1-Mediated Tubular Invaginations of the Plasma Membrane.
Quantitative Interactomics in Primary T Cells Provides a Rationale for Concomitant PD-1 and BTLA Coinhibitor Blockade in Cancer Immunotherapy.
Identification and characterization of the cellular subclones that contribute to the pathogenesis of mantle cell lymphoma.
Unleashing Type-2 Dendritic Cells to Drive Protective Antitumor CD4(+) T Cell Immunity.
Site-specific PEGylation of an anti-CEA/CD3 bispecific antibody improves its antitumor efficacy.
Enhanced Anti-lymphoma Activity of CAR19-iNKT Cells Underpinned by Dual CD19 and CD1d Targeting.
PD1 is expressed on exhausted T cells as well as virus specific memory CD8+ T cells in the bone marrow of myeloma patients.
Distinctive features of tumor-infiltrating γδ T lymphocytes in human colorectal cancer.
Restimulation-induced T-cell death through NTB-A/SAP signaling pathway is impaired in tuberculosis patients with depressed immune responses.
Squamous Cell Tumors Recruit γδ T Cells Producing either IL17 or IFNγ Depending on the Tumor Stage.
NKG2D Ligand-Targeted Bispecific T-Cell Engagers Lead to Robust Antitumor Activity against Diverse Human Tumors.
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
Imbalance between subsets of CD8(+) peripheral blood T cells in patients with chronic obstructive pulmonary disease.
Umbilical Cord-Derived Mesenchymal Stem Cells Suppress Autophagy of T Cells in Patients with Systemic Lupus Erythematosus via Transfer of Mitochondria.
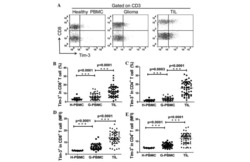
Expression of the galectin-9-Tim-3 pathway in glioma tissues is associated with the clinical manifestations of glioma.
The trajectory of the blood DNA methylome ageing rate is largely set before adulthood: evidence from two longitudinal studies.
CRISPR-Cas9 mediated efficient PD-1 disruption on human primary T cells from cancer patients.
Activation of Innate and Adaptive Immunity by a Recombinant Human Cytomegalovirus Strain Expressing an NKG2D Ligand.
MiRNA-22 inhibits oncogene galectin-1 in hepatocellular carcinoma.
Microfluidic differential immunocapture biochip for specific leukocyte counting.
Durable complete responses off all treatment in patients with metastatic malignant melanoma after sequential immunotherapy followed by a finite course of BRAF inhibitor therapy.
PD-1, PD-L1 and PD-L2 Gene Expression on T-Cells and Natural Killer Cells Declines in Conjunction with a Reduction in PD-1 Protein during the Intensive Phase of Tuberculosis Treatment.
HIV Malaria Co-Infection Is Associated with Atypical Memory B Cell Expansion and a Reduced Antibody Response to a Broad Array of Plasmodium falciparum Antigens in Rwandan Adults.
Length of paternal lifespan is manifested in the DNA methylome of their nonagenarian progeny.
Homeostatic regulation of T cell trafficking by a B cell-derived peptide is impaired in autoimmune and chronic inflammatory disease.
A novel antibody-drug conjugate targeting SAIL for the treatment of hematologic malignancies.
Ageing-associated changes in the human DNA methylome: genomic locations and effects on gene expression.
Towards programming immune tolerance through geometric manipulation of phosphatidylserine.
Transcriptomic and epigenetic analyses reveal a gender difference in aging-associated inflammation: the Vitality 90+ study.
Comparative immune phenotypic analysis of cutaneous Squamous Cell Carcinoma and Intraepidermal Carcinoma in immune-competent individuals: proportional representation of CD8+ T-cells but not FoxP3+ Regulatory T-cells is associated with disease stage.
TL1A induces TCR independent IL-6 and TNF-α production and growth of PLZF⁺ leukocytes.
PD-1(+) CD8(+) T cells are exhausted in tumours and functional in draining lymph nodes of colorectal cancer patients.
Invariant natural killer T cells act as an extravascular cytotoxic barrier for joint-invading Lyme Borrelia.
The combination of type I IFN, TNF-α, and cell surface receptor engagement with dendritic cells enables NK cells to overcome immune evasion by dengue virus.
Bioengineering T cells to target carbohydrate to treat opportunistic fungal infection.
Immune response to JC virus T antigen in patients with and without colorectal neoplasia.
Modulation of NKG2D expression in human CD8(+) T cells corresponding with tuberculosis drug cure.
Transcriptional analysis reveals gender-specific changes in the aging of the human immune system.
Changes of cytokines during a spaceflight analog--a 45-day head-down bed rest.
Humanized mice, a new model to study the influence of drug treatment on neonatal sepsis.
Characterization of the role of distinct plasma cell-free DNA species in age-associated inflammation and frailty.
Penicillium marneffei-stimulated dendritic cells enhance HIV-1 trans-infection and promote viral infection by activating primary CD4+ T cells.
Differentiation of human peripheral blood Vδ1+ T cells expressing the natural cytotoxicity receptor NKp30 for recognition of lymphoid leukemia cells.
A microfabricated electrical differential counter for the selective enumeration of CD4+ T lymphocytes.
Agudelo M, Palus M, Keeffe JR, Bianchini F, Svoboda P, Salát J, Peace A, Gazumyan A, Cipolla M, Kapoor T, Guidetti F, Yao KH, Elsterová J, Teislerová D, Chrdle A, Hönig V, Oliveira T, West AP, Lee YE, Rice CM, MacDonald MR, Bjorkman PJ, Růžek D, Robbiani DF, Nussenzweig MC
The Journal of experimental medicine 2021 May 3;218(5)
The Journal of experimental medicine 2021 May 3;218(5)
Ageing promotes early T follicular helper cell differentiation by modulating expression of RBPJ.
Webb LMC, Fra-Bido S, Innocentin S, Matheson LS, Attaf N, Bignon A, Novarino J, Fazilleau N, Linterman MA
Aging cell 2021 Jan;20(1):e13295
Aging cell 2021 Jan;20(1):e13295
Imbalance between T helper 1 and regulatory T cells plays a detrimental role in experimental Parkinson's disease in mice.
Li W, Luo Y, Xu H, Ma Q, Yao Q
The Journal of international medical research 2021 Apr;49(4):300060521998471
The Journal of international medical research 2021 Apr;49(4):300060521998471
Standardized 11-color flow cytometry panel for the functional phenotyping of human T regulatory cells.
Manuszak C, Brainard M, Thrash E, Hodi FS, Severgnini M
Journal of biological methods 2020;7(2):e131
Journal of biological methods 2020;7(2):e131
HDAC Inhibitor, CG-745, Enhances the Anti-Cancer Effect of Anti-PD-1 Immune Checkpoint Inhibitor by Modulation of the Immune Microenvironment.
Kim YD, Park SM, Ha HC, Lee AR, Won H, Cha H, Cho S, Cho JM
Journal of Cancer 2020;11(14):4059-4072
Journal of Cancer 2020;11(14):4059-4072
Small-molecule MMP2/MMP9 inhibitor SB-3CT modulates tumor immune surveillance by regulating PD-L1.
Ye Y, Kuang X, Xie Z, Liang L, Zhang Z, Zhang Y, Ma F, Gao Q, Chang R, Lee HH, Zhao S, Su J, Li H, Peng J, Chen H, Yin M, Peng C, Yang N, Wang J, Liu J, Liu H, Han L, Chen X
Genome medicine 2020 Sep 28;12(1):83
Genome medicine 2020 Sep 28;12(1):83
Relationship between T cell receptor clonotype and PD-1 expression of tumor-infiltrating lymphocytes in colorectal cancer.
Sukegawa K, Shitaoka K, Hamana H, Kobayashi E, Miyahara Y, Fujii K, Tsuda K, Saeki S, Nagata T, Ozawa T, Saito S, Fujii T, Muraguchi A, Shiku H, Kishi H
European journal of immunology 2020 Oct;50(10):1580-1590
European journal of immunology 2020 Oct;50(10):1580-1590
IL-10 Restores MHC Class I Expression and Interferes With Immunity in Papillary Thyroid Cancer With Hashimoto Thyroiditis.
Lu ZW, Hu JQ, Liu WL, Wen D, Wei WJ, Wang YL, Wang Y, Liao T, Ji QH
Endocrinology 2020 Oct 1;161(10)
Endocrinology 2020 Oct 1;161(10)
NKG2C(pos) NK Cells Regulate the Expansion of Cytomegalovirus-Specific CD8 T Cells.
Grutza R, Moskorz W, Senff T, Bäcker E, Lindemann M, Zimmermann A, Uhrberg M, Lang PA, Timm J, Cosmovici C
Journal of immunology (Baltimore, Md. : 1950) 2020 Jun 1;204(11):2910-2917
Journal of immunology (Baltimore, Md. : 1950) 2020 Jun 1;204(11):2910-2917
Selective Killing of Activated T Cells by 5-Aminolevulinic Acid Mediated Photodynamic Effect: Potential Improvement of Extracorporeal Photopheresis.
Darvekar S, Juzenas P, Oksvold M, Kleinauskas A, Holien T, Christensen E, Stokke T, Sioud M, Peng Q
Cancers 2020 Feb 6;12(2)
Cancers 2020 Feb 6;12(2)
Accumulation of TNFR2-expressing regulatory T cells in malignant pleural effusion of lung cancer patients is associated with poor prognosis.
Ye LL, Peng WB, Niu YR, Xiang X, Wei XS, Wang ZH, Wang X, Zhang SY, Chen X, Zhou Q
Annals of translational medicine 2020 Dec;8(24):1647
Annals of translational medicine 2020 Dec;8(24):1647
Pyridoxal-5'-Phosphate Promotes Immunomodulatory Function of Adipose-Derived Mesenchymal Stem Cells through Indoleamine 2,3-Dioxygenase-1 and TLR4/NF-κB Pathway.
Li C, Huang J, Zhu H, Shi Q, Li D, Ju X
Stem cells international 2019;2019:3121246
Stem cells international 2019;2019:3121246
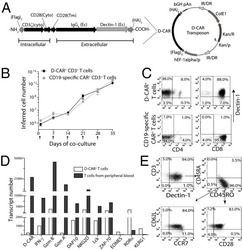
Integrating Ligand-Receptor Interactions and In Vitro Evolution for Streamlined Discovery of Artificial Nucleic Acid Ligands.
Zumrut HE, Batool S, Argyropoulos KV, Williams N, Azad R, Mallikaratchy PR
Molecular therapy. Nucleic acids 2019 Sep 6;17:150-163
Molecular therapy. Nucleic acids 2019 Sep 6;17:150-163
Progranulin prevents regulatory NK cell cytotoxicity against antiviral T cells.
Huang A, Shinde PV, Huang J, Senff T, Xu HC, Margotta C, Häussinger D, Willnow TE, Zhang J, Pandyra AA, Timm J, Weggen S, Lang KS, Lang PA
JCI insight 2019 Sep 5;4(17)
JCI insight 2019 Sep 5;4(17)
MicroRNA‑155 inhibits the proliferation of CD8+ T cells via upregulating regulatory T cells in vitiligo.
Lv M, Li Z, Liu J, Lin F, Zhang Q, Li Z, Wang Y, Wang K, Xu Y
Molecular medicine reports 2019 Oct;20(4):3617-3624
Molecular medicine reports 2019 Oct;20(4):3617-3624
Bispecific Antibody Approach for Improved Melanoma-Selective PD-L1 Immune Checkpoint Blockade.
Koopmans I, Hendriks MAJM, van Ginkel RJ, Samplonius DF, Bremer E, Helfrich W
The Journal of investigative dermatology 2019 Nov;139(11):2343-2351.e3
The Journal of investigative dermatology 2019 Nov;139(11):2343-2351.e3
Cdc42 Couples T Cell Receptor Endocytosis to GRAF1-Mediated Tubular Invaginations of the Plasma Membrane.
Rossatti P, Ziegler L, Schregle R, Betzler VM, Ecker M, Rossy J
Cells 2019 Nov 4;8(11)
Cells 2019 Nov 4;8(11)
Quantitative Interactomics in Primary T Cells Provides a Rationale for Concomitant PD-1 and BTLA Coinhibitor Blockade in Cancer Immunotherapy.
Celis-Gutierrez J, Blattmann P, Zhai Y, Jarmuzynski N, Ruminski K, Grégoire C, Ounoughene Y, Fiore F, Aebersold R, Roncagalli R, Gstaiger M, Malissen B
Cell reports 2019 Jun 11;27(11):3315-3330.e7
Cell reports 2019 Jun 11;27(11):3315-3330.e7
Identification and characterization of the cellular subclones that contribute to the pathogenesis of mantle cell lymphoma.
Tang J, Zhang L, Zhou T, Sun Z, Kong L, Jing L, Xing H, Wu H, Liu Y, Zhou S, Li J, Chen M, Xu F, Tang J, Ma T, Hu M, Liu D, Guo J, Zhu X, Chen Y, Ye T, Wang J, Li X, Xing HR
Genes & diseases 2019 Dec;6(4):407-418
Genes & diseases 2019 Dec;6(4):407-418
Unleashing Type-2 Dendritic Cells to Drive Protective Antitumor CD4(+) T Cell Immunity.
Binnewies M, Mujal AM, Pollack JL, Combes AJ, Hardison EA, Barry KC, Tsui J, Ruhland MK, Kersten K, Abushawish MA, Spasic M, Giurintano JP, Chan V, Daud AI, Ha P, Ye CJ, Roberts EW, Krummel MF
Cell 2019 Apr 18;177(3):556-571.e16
Cell 2019 Apr 18;177(3):556-571.e16
Site-specific PEGylation of an anti-CEA/CD3 bispecific antibody improves its antitumor efficacy.
Pan H, Liu J, Deng W, Xing J, Li Q, Wang Z
International journal of nanomedicine 2018;13:3189-3201
International journal of nanomedicine 2018;13:3189-3201
Enhanced Anti-lymphoma Activity of CAR19-iNKT Cells Underpinned by Dual CD19 and CD1d Targeting.
Rotolo A, Caputo VS, Holubova M, Baxan N, Dubois O, Chaudhry MS, Xiao X, Goudevenou K, Pitcher DS, Petevi K, Kachramanoglou C, Iles S, Naresh K, Maher J, Karadimitris A
Cancer cell 2018 Oct 8;34(4):596-610.e11
Cancer cell 2018 Oct 8;34(4):596-610.e11
PD1 is expressed on exhausted T cells as well as virus specific memory CD8+ T cells in the bone marrow of myeloma patients.
Sponaas AM, Yang R, Rustad EH, Standal T, Thoresen AS, Dao Vo C, Waage A, Slørdahl TS, Børset M, Sundan A
Oncotarget 2018 Aug 10;9(62):32024-32035
Oncotarget 2018 Aug 10;9(62):32024-32035
Distinctive features of tumor-infiltrating γδ T lymphocytes in human colorectal cancer.
Meraviglia S, Lo Presti E, Tosolini M, La Mendola C, Orlando V, Todaro M, Catalano V, Stassi G, Cicero G, Vieni S, Fourniè JJ, Dieli F
Oncoimmunology 2017;6(10):e1347742
Oncoimmunology 2017;6(10):e1347742
Restimulation-induced T-cell death through NTB-A/SAP signaling pathway is impaired in tuberculosis patients with depressed immune responses.
Hernández Del Pino RE, Pellegrini JM, Rovetta AI, Peña D, Álvarez GI, Rolandelli A, Musella RM, Palmero DJ, Malbran A, Pasquinelli V, García VE
Immunology and cell biology 2017 Sep;95(8):716-728
Immunology and cell biology 2017 Sep;95(8):716-728
Squamous Cell Tumors Recruit γδ T Cells Producing either IL17 or IFNγ Depending on the Tumor Stage.
Lo Presti E, Toia F, Oieni S, Buccheri S, Turdo A, Mangiapane LR, Campisi G, Caputo V, Todaro M, Stassi G, Cordova A, Moschella F, Rinaldi G, Meraviglia S, Dieli F
Cancer immunology research 2017 May;5(5):397-407
Cancer immunology research 2017 May;5(5):397-407
NKG2D Ligand-Targeted Bispecific T-Cell Engagers Lead to Robust Antitumor Activity against Diverse Human Tumors.
Godbersen C, Coupet TA, Huehls AM, Zhang T, Battles MB, Fisher JL, Ernstoff MS, Sentman CL
Molecular cancer therapeutics 2017 Jul;16(7):1335-1346
Molecular cancer therapeutics 2017 Jul;16(7):1335-1346
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
Lebedeva A, Vorobyeva D, Vagida M, Ivanova O, Felker E, Fitzgerald W, Danilova N, Gontarenko V, Shpektor A, Vasilieva E, Margolis L
Atherosclerosis 2017 Dec;267:90-98
Atherosclerosis 2017 Dec;267:90-98
Imbalance between subsets of CD8(+) peripheral blood T cells in patients with chronic obstructive pulmonary disease.
Chen L, Chen G, Zhang MQ, Xiong XZ, Liu HJ, Xin JB, Zhang JC, Wu JH, Meng ZJ, Sun SW
PeerJ 2016;4:e2301
PeerJ 2016;4:e2301
Umbilical Cord-Derived Mesenchymal Stem Cells Suppress Autophagy of T Cells in Patients with Systemic Lupus Erythematosus via Transfer of Mitochondria.
Chen J, Wang Q, Feng X, Zhang Z, Geng L, Xu T, Wang D, Sun L
Stem cells international 2016;2016:4062789
Stem cells international 2016;2016:4062789
Expression of the galectin-9-Tim-3 pathway in glioma tissues is associated with the clinical manifestations of glioma.
Liu Z, Han H, He X, Li S, Wu C, Yu C, Wang S
Oncology letters 2016 Mar;11(3):1829-1834
Oncology letters 2016 Mar;11(3):1829-1834
The trajectory of the blood DNA methylome ageing rate is largely set before adulthood: evidence from two longitudinal studies.
Kananen L, Marttila S, Nevalainen T, Kummola L, Junttila I, Mononen N, Kähönen M, Raitakari OT, Hervonen A, Jylhä M, Lehtimäki T, Hurme M, Jylhävä J
Age (Dordrecht, Netherlands) 2016 Jun;38(3):65
Age (Dordrecht, Netherlands) 2016 Jun;38(3):65
CRISPR-Cas9 mediated efficient PD-1 disruption on human primary T cells from cancer patients.
Su S, Hu B, Shao J, Shen B, Du J, Du Y, Zhou J, Yu L, Zhang L, Chen F, Sha H, Cheng L, Meng F, Zou Z, Huang X, Liu B
Scientific reports 2016 Jan 28;6:20070
Scientific reports 2016 Jan 28;6:20070
Activation of Innate and Adaptive Immunity by a Recombinant Human Cytomegalovirus Strain Expressing an NKG2D Ligand.
Tomić A, Varanasi PR, Golemac M, Malić S, Riese P, Borst EM, Mischak-Weissinger E, Guzmán CA, Krmpotić A, Jonjić S, Messerle M
PLoS pathogens 2016 Dec;12(12):e1006015
PLoS pathogens 2016 Dec;12(12):e1006015
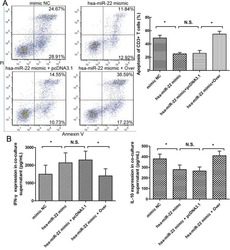
MiRNA-22 inhibits oncogene galectin-1 in hepatocellular carcinoma.
You Y, Tan JX, Dai HS, Chen HW, Xu XJ, Yang AG, Zhang YJ, Bai LH, Bie P
Oncotarget 2016 Aug 30;7(35):57099-57116
Oncotarget 2016 Aug 30;7(35):57099-57116
Microfluidic differential immunocapture biochip for specific leukocyte counting.
Hassan U, Watkins NN, Reddy B Jr, Damhorst G, Bashir R
Nature protocols 2016 Apr;11(4):714-26
Nature protocols 2016 Apr;11(4):714-26
Durable complete responses off all treatment in patients with metastatic malignant melanoma after sequential immunotherapy followed by a finite course of BRAF inhibitor therapy.
Wyluda EJ, Cheng J, Schell TD, Haley JS, Mallon C, Neves RI, Robertson G, Sivik J, Mackley H, Talamo G, Drabick JJ
Cancer biology & therapy 2015;16(5):662-70
Cancer biology & therapy 2015;16(5):662-70
PD-1, PD-L1 and PD-L2 Gene Expression on T-Cells and Natural Killer Cells Declines in Conjunction with a Reduction in PD-1 Protein during the Intensive Phase of Tuberculosis Treatment.
Hassan SS, Akram M, King EC, Dockrell HM, Cliff JM
PloS one 2015;10(9):e0137646
PloS one 2015;10(9):e0137646
HIV Malaria Co-Infection Is Associated with Atypical Memory B Cell Expansion and a Reduced Antibody Response to a Broad Array of Plasmodium falciparum Antigens in Rwandan Adults.
Subramaniam KS, Skinner J, Ivan E, Mutimura E, Kim RS, Feintuch CM, Portugal S, Anastos K, Crompton PD, Daily JP
PloS one 2015;10(4):e0124412
PloS one 2015;10(4):e0124412
Length of paternal lifespan is manifested in the DNA methylome of their nonagenarian progeny.
Marttila S, Kananen L, Jylhävä J, Nevalainen T, Hervonen A, Jylhä M, Hurme M
Oncotarget 2015 Oct 13;6(31):30557-67
Oncotarget 2015 Oct 13;6(31):30557-67
Homeostatic regulation of T cell trafficking by a B cell-derived peptide is impaired in autoimmune and chronic inflammatory disease.
Chimen M, McGettrick HM, Apta B, Kuravi SJ, Yates CM, Kennedy A, Odedra A, Alassiri M, Harrison M, Martin A, Barone F, Nayar S, Hitchcock JR, Cunningham AF, Raza K, Filer A, Copland DA, Dick AD, Robinson J, Kalia N, Walker LSK, Buckley CD, Nash GB, Narendran P, Rainger GE
Nature medicine 2015 May;21(5):467-475
Nature medicine 2015 May;21(5):467-475
A novel antibody-drug conjugate targeting SAIL for the treatment of hematologic malignancies.
Kim SY, Theunissen JW, Balibalos J, Liao-Chan S, Babcock MC, Wong T, Cairns B, Gonzalez D, van der Horst EH, Perez M, Levashova Z, Chinn L, D'Alessio JA, Flory M, Bermudez A, Jackson DY, Ha E, Monteon J, Bruhns MF, Chen G, Migone TS
Blood cancer journal 2015 May 29;5(5):e316
Blood cancer journal 2015 May 29;5(5):e316
Ageing-associated changes in the human DNA methylome: genomic locations and effects on gene expression.
Marttila S, Kananen L, Häyrynen S, Jylhävä J, Nevalainen T, Hervonen A, Jylhä M, Nykter M, Hurme M
BMC genomics 2015 Mar 14;16(1):179
BMC genomics 2015 Mar 14;16(1):179
Towards programming immune tolerance through geometric manipulation of phosphatidylserine.
Roberts RA, Eitas TK, Byrne JD, Johnson BM, Short PJ, McKinnon KP, Reisdorf S, Luft JC, DeSimone JM, Ting JP
Biomaterials 2015 Dec;72:1-10
Biomaterials 2015 Dec;72:1-10
Transcriptomic and epigenetic analyses reveal a gender difference in aging-associated inflammation: the Vitality 90+ study.
Nevalainen T, Kananen L, Marttila S, Jylhä M, Hervonen A, Hurme M, Jylhävä J
Age (Dordrecht, Netherlands) 2015 Aug;37(4):9814
Age (Dordrecht, Netherlands) 2015 Aug;37(4):9814
Comparative immune phenotypic analysis of cutaneous Squamous Cell Carcinoma and Intraepidermal Carcinoma in immune-competent individuals: proportional representation of CD8+ T-cells but not FoxP3+ Regulatory T-cells is associated with disease stage.
Freeman A, Bridge JA, Maruthayanar P, Overgaard NH, Jung JW, Simpson F, Prow TW, Soyer HP, Frazer IH, Freeman M, Wells JW
PloS one 2014;9(10):e110928
PloS one 2014;9(10):e110928
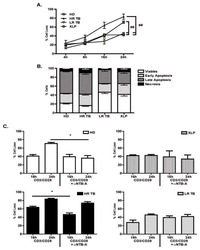
TL1A induces TCR independent IL-6 and TNF-α production and growth of PLZF⁺ leukocytes.
Reichwald K, Jørgensen TZ, Tougaard P, Skov S
PloS one 2014;9(1):e85793
PloS one 2014;9(1):e85793
PD-1(+) CD8(+) T cells are exhausted in tumours and functional in draining lymph nodes of colorectal cancer patients.
Wu X, Zhang H, Xing Q, Cui J, Li J, Li Y, Tan Y, Wang S
British journal of cancer 2014 Sep 23;111(7):1391-9
British journal of cancer 2014 Sep 23;111(7):1391-9
Invariant natural killer T cells act as an extravascular cytotoxic barrier for joint-invading Lyme Borrelia.
Lee WY, Sanz MJ, Wong CH, Hardy PO, Salman-Dilgimen A, Moriarty TJ, Chaconas G, Marques A, Krawetz R, Mody CH, Kubes P
Proceedings of the National Academy of Sciences of the United States of America 2014 Sep 23;111(38):13936-41
Proceedings of the National Academy of Sciences of the United States of America 2014 Sep 23;111(38):13936-41
The combination of type I IFN, TNF-α, and cell surface receptor engagement with dendritic cells enables NK cells to overcome immune evasion by dengue virus.
Lim DS, Yawata N, Selva KJ, Li N, Tsai CY, Yeong LH, Liong KH, Ooi EE, Chong MK, Ng ML, Leo YS, Yawata M, Wong SB
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 15;193(10):5065-75
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 15;193(10):5065-75
Bioengineering T cells to target carbohydrate to treat opportunistic fungal infection.
Kumaresan PR, Manuri PR, Albert ND, Maiti S, Singh H, Mi T, Roszik J, Rabinovich B, Olivares S, Krishnamurthy J, Zhang L, Najjar AM, Huls MH, Lee DA, Champlin RE, Kontoyiannis DP, Cooper LJ
Proceedings of the National Academy of Sciences of the United States of America 2014 Jul 22;111(29):10660-5
Proceedings of the National Academy of Sciences of the United States of America 2014 Jul 22;111(29):10660-5
Immune response to JC virus T antigen in patients with and without colorectal neoplasia.
Butcher LD, Garcia M, Arnold M, Ueno H, Goel A, Boland CR
Gut microbes 2014 Jul 1;5(4):468-75
Gut microbes 2014 Jul 1;5(4):468-75
Modulation of NKG2D expression in human CD8(+) T cells corresponding with tuberculosis drug cure.
Hassan SS, Cho JE, Akram M, Fielding KL, Dockrell HM, Cliff JM
PloS one 2013;8(7):e70063
PloS one 2013;8(7):e70063
Transcriptional analysis reveals gender-specific changes in the aging of the human immune system.
Marttila S, Jylhävä J, Nevalainen T, Nykter M, Jylhä M, Hervonen A, Tserel L, Peterson P, Hurme M
PloS one 2013;8(6):e66229
PloS one 2013;8(6):e66229
Changes of cytokines during a spaceflight analog--a 45-day head-down bed rest.
Xu X, Tan C, Li P, Zhang S, Pang X, Liu H, Li L, Sun X, Zhang Y, Wu H, Chen X, Ge Q
PloS one 2013;8(10):e77401
PloS one 2013;8(10):e77401
Humanized mice, a new model to study the influence of drug treatment on neonatal sepsis.
Ernst W, Zimara N, Hanses F, Männel DN, Seelbach-Göbel B, Wege AK
Infection and immunity 2013 May;81(5):1520-31
Infection and immunity 2013 May;81(5):1520-31
Characterization of the role of distinct plasma cell-free DNA species in age-associated inflammation and frailty.
Jylhävä J, Nevalainen T, Marttila S, Jylhä M, Hervonen A, Hurme M
Aging cell 2013 Jun;12(3):388-97
Aging cell 2013 Jun;12(3):388-97
Penicillium marneffei-stimulated dendritic cells enhance HIV-1 trans-infection and promote viral infection by activating primary CD4+ T cells.
Qin Y, Li Y, Liu W, Tian R, Guo Q, Li S, Li H, Zhang D, Zheng Y, Wu L, Lan K, Wang J
PloS one 2011;6(11):e27609
PloS one 2011;6(11):e27609
Differentiation of human peripheral blood Vδ1+ T cells expressing the natural cytotoxicity receptor NKp30 for recognition of lymphoid leukemia cells.
Correia DV, Fogli M, Hudspeth K, da Silva MG, Mavilio D, Silva-Santos B
Blood 2011 Jul 28;118(4):992-1001
Blood 2011 Jul 28;118(4):992-1001
A microfabricated electrical differential counter for the selective enumeration of CD4+ T lymphocytes.
Watkins NN, Sridhar S, Cheng X, Chen GD, Toner M, Rodriguez W, Bashir R
Lab on a chip 2011 Apr 21;11(8):1437-47
Lab on a chip 2011 Apr 21;11(8):1437-47
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
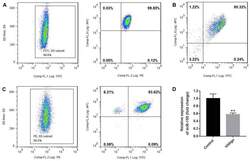
- Staining of normal human peripheral blood cells with Anti-Human CD19 FITC (Product # 11-0199-42) and Mouse IgG2a K Isotype Control PerCP-Cyanine5-5 (Product # 45-4724-82) (left) or Anti-Human CD3 PerCP-Cyanine5-5 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
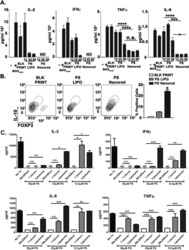
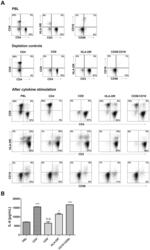
- Figure 9 IL-6 produced by PBLs depleted for CD4 + , CD8 + , HLA-DR + or CD56 + /CD16 + cells. (A) Freshly purified PBLs stained for CD4, CD8, CD3, HLA-DR, CD16, CD56. Depletion controls for PBLs depleted for CD4 + , CD8 + , HLA-DR + or CD16 + /CD56 + cells. PBLs and depletions were stimulated for 7 days with combinations of IL-12 (4 ng/mL), IL-15 (10 ng/mL), IL-18 (40 ng/mL) and TL1A (100 ng/mL) and stained for CD4, CD8, CD3, HLA-DR, CD16 and CD56 expression. (B) IL-6 production after 7 days by PBLs and depletions stimulated with IL-12, IL-15, IL-18 and TL1A as described above. Error bars represent the SEM of two measurements. Statistically significant differences by t-test: *** = p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
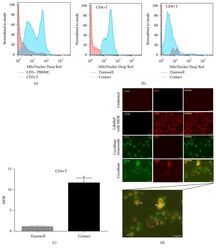
- Figure 7 UC-MSCs transferred mitochondria to activate T cells. PBMCs from SLE patients were labeled with carboxyfluorescein succinimidyl amino ester (CFSE) and treated with anti-CD3/CD28 antibodies for two days. Then they were cocultured with UC-MSCs for 12 h, which had been prelabeled with respiratory mitochondrion specific probe Mitotracker Deep Red (MDR). PBMCs were cultured with UC-MSCs through transwell as control. Then PBMCs were stained with anti-CD3, anti-CD4, or anti-CD8 dye and detected for MDR fluorescence with flow cytometry. Fluorescence microscopy was carried out similarly with anti-CD3/28 stimulation for 12 h and then cocultured for 6 h. All experiments were performed in triplicate. (a) T cells (CD3 positive) rather than non-T cells (CD3 negative) got MDR staining. (b-d) T cells cultured with UC-MSCs directly rather than through transwell got transferred mitochondria ( n = 3). Arrows indicated transferred mitochondria within lymphocytes. * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
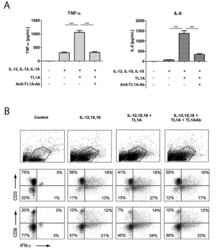
- Figure 2 TL1A induces IL-6 and TNF-alpha. Freshly purified PBMCs were incubated with IL-12 (2 ng/mL), IL-15 (10 ng/mL), IL-18 (10 ng/mL), TL1A (100 ng/mL) and TL1AAb (1 ug/mL, blocking antibody). Extra IL-15 (2 ng/mL) was added on day 3. ( A ) After 6 days, supernatants were collected and different cytokines were measured by bead-based ELISA. Error bars represent the SEM of eight measurements. Statistically significant differences are indicated by ***(t-test, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
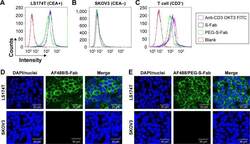
- Figure 4 PEG-S-Fab can bind CEA on tumor cells and CD3 + on T cells. Notes: Flow cytometry analysis of PEG-S-Fab and S-Fab on CEA-positive LS174T cells ( A ), CEA-negative SKOV3 cells ( B ), and CD3 + T cells ( C ). The positive control anti-CD3 antibody OKT3 was used for T-cell flow cytometry. Confocal microscopy of immunofluorescence staining as described in the ""Materials and methods"" section. S-Fab ( D ) and PEG-S-Fab ( E ) on LS174T cells (upper panel) and SKOV3 cells (lower panel), respectively. The scale bar represents 30 mum. AF488, stained by the goat antihuman IgG (H + L)-AlexaFluor 488 antibody; LS174T, human colorectal cell line LS174T; SKOV3, human ovarian cancer cell line SKOV3. Abbreviations: CEA, carcinoembryonic antigen; DAPI, 4',6-diamidino-2-phenylindole; IgG, immunoglobulin G; PEG, polyethylene glycol; PEG-S-Fab, PEGylated S-Fab; S-Fab, single-domain antibody-linked Fab; FITC, fluoresceine isothiocyanate.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
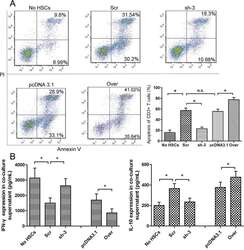
- Figure 4 The expression of galectin-1 in HSCs promotes HSC-induced T cell apoptosis and Th1/Th2 cytokine balance skewing A. Flow cytometry (annexin V-FITC apoptosis detection) analyses to detect T cell apoptosis in CD3+ T cells, cultured alone or co-cultured with HSCs subjected to different pre-treatments (cell transfection for galectin-1 knockdown and overexpression: sh-3 group versus Scr group; Over group versus pcDNA3.1 group) for 48 hours at a ratio of 10:1 (T:HSC), B. ELISA showing the levels of cytokines (IFN-gamma and IL-10) in the supernatant. Data are shown as the means (+- SD) of triplicates (n = 7). *P < 0.05. NC, negative control group; Scr, non-targeting scrambled sequence group; sh, small hairpin RNA sequence transfection group; pcDNA3.1, negative control group; Over, galectin-1 overexpression group; No HSCs, T cells cultured alone.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Cell surface expression of SAIL in CLL, AML and MM patient samples and normal BMMC and PBMC controls. ( a ) Three CLL specimens analyzed by flow cytometry. CLL cells were identified as CD19/CD5 double-positive cells. The histograms present SAIL (filled) and isotype control (open) staining in the live-cell and the CLL population. ( b ) Flow cytometry analysis of three AML specimens. SAIL expression is assessed in live-cells, CD33-positive and CD34-positive cells. ( c ) Flow cytometry analysis of three MM specimens. CD38 high cells with CD56 expression were gated for MM cells. SAIL expression is assessed in the live-cell and the MM population. ( d and e ) Flow cytometry analysis of SAIL expression in BMMC ( d ) and PBMC ( e ) via co-staining with CD19, CD3, CD14, CD56, CD33, CD34 and a cocktail of lineage (LN) markers. Numbers in histograms are median-fluorescence-intensity fold-change values relative to the isotype control. Three and two representative examples are shown for the tumor and normal samples, respectively.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
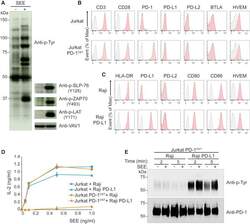
- Figure 3 Outcome of PD-1-PD-L1 Engagement at T Cell-APC Interface (A) Jurkat-PD-1 OST cells were stimulated with Raji cells that have been preincubated in the absence (-) or presence (+) of 200 ng/mL SEE and lysed 2 min after the initiation of cell-cell contact. Immunoblot analysis of equal amounts of lysates from the specified conditions probed with antibody to phosphorylated proteins (Anti-p-Tyr) or with phospho-tyrosine-specific antibodies directed against SLP76 pY128, ZAP70 pY493, LAT pY171 or VAV1 (loading control). Left margin, molecular size in kilodaltons (kDa). Data are representative of three independent experiments. (B) Expression of CD3, CD28, PD-1, PD-L1, PD-L2, BTLA, and HVEM at the surface of Jurkat cells and Jurkat-PD-1 OST cells, analyzed using flow cytometry. (C) Expression of HLA-DR, PD-L1, PD-L2, CD80, CD86, and HVEM at the surface of Raji cells and Raji-PD-L1 cells, analyzed using flow cytometry. In (B) and (C), gray shaded curves correspond to isotype-matched control antibody (negative control), and data are representative of two independent experiments. (D) IL-2 production by Jurkat and Jurkat-PD-1 OST cells stimulated for 24 h with either Raji or Raji-PD-L1 cells in the absence (0) or presence of the specified amounts of SEE. Data are representative of three independent experiments, and mean and SEM are shown. (E) Jurkat-PD-1 OST cells stimulated at 37degC with Raji or Raji-PD-L1 cells preincubated in the absence (-) or presence (+) of 200 ng/mL SEE an
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
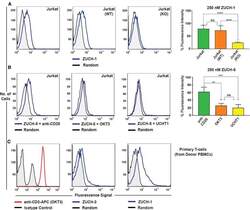
- Figure 6 Characterization of Aptamer Specificity against TCR-CD3epsilon (A) Flow-cytometric analyses of binding of the highest affinity aptamer, ZUCH-1, against Jurkat.E6 cells used in SELEX (left), against wild-type Jurkat cells used for generating CRISPR knockout cell lines (middle), against CRISPR double-knockout Jurkat cells (right), and the overall conclusion from six independent specificity analyses (far right). Aptamer ZUCH-1 does not bind to knockout cells, thereby demonstrating epitope specificity (ordinary one-way ANOVA, using Tukey''s multiple comparisons test performed on GraphPad Prism to obtain statistical significance: ****p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 ALA-PDT of mixed populations of resting and activated cells. Healthy donor PBMCs were activated in vitro with anti-CD3/CD28 antibodies for three days. The activated T cells were then labeled with anti-human CD25-FITC antibody. The resting and CD25-FITC labeled activated T cells were mixed in certain ratios as indicated. The mixed cells were incubated with 3 mM ALA for 1 h at 37 degC and then irradiated with the LED blue light at 0.9 J/cm 2 or 1.8 J/cm 2 . The cell survivals were measured 2 h after light irradiation with flow cytometry as described in Figure 3 . The control samples without light are also included. ( A ) Mixture of 1% CD25-FITC labelled activated T cells with 99% resting PBMCs. ( B ) Mixture of 5% CD25-FITC labelled activated T cells with 95% resting PBMCs. * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
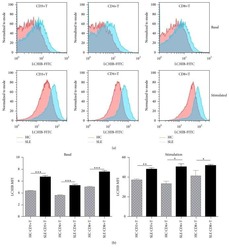
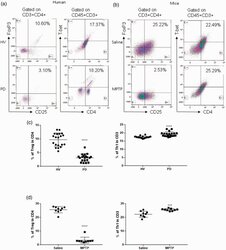
- Figure 1. Patients with Parkinson's disease (PD) and mice with experimental PD exhibit decreased regulatory T (Treg) and increased T helper 1 (Th1) cell numbers in the blood. Treg cells were defined as CD3+CD4+CD25+FoxP3+ cells, whereas Th1 cells were identified as CD3+CD4+T-bet+ cells. (a) Representative plots of Treg and Th1 cells in patients with PD and healthy volunteers (HVs). (b) Representative plots of Treg and Th1 cells in control mice (Saline) and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated mice. (c) Patients with PD exhibited lower Treg cell levels and greater Th1 cell levels in the blood than HVs. n = 20. ****, p < 0.0001 by a two-tailed Student's t tests. (d) MPTP-induced experimental PD mice had fewer Treg and more Th1 cells in the circulation than saline-treated mice. n = 9/group in each experiment performed in triplicate. ***, p < 0.001; ****, p < 0.0001, according to Student's t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
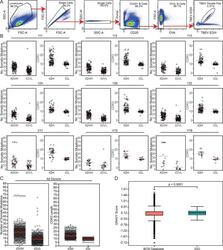
- Figure S2. Sorting strategy and antibody sequence analysis. (A) Sorting strategy. Forward and side scatter (FSC and SSC, respectively) were used to gate on single lymphocytes. Dump channel included CD3, CD8, CD14, CD16, and a viability dye. CD20 + B cells that failed to bind OVA (OVA - ) but did bind to the TBEV EDIII bait coupled with both PE and AF647 fluorophores were purified. (B) For each donor, the number of V gene somatic nucleotide mutations is shown on the left and the amino acid length of the CDR3 is shown on the right. (C) As in B, but for all donors combined. For B and C, horizontal red lines indicate the mean. (D) Distribution of hydrophobicity GRAVY scores at the IGH CDR3 of antibodies from all donors combined and compared with human repertoire (). P < 0.0001 was determined using the Wilcoxon nonparametric test. Related to Fig. 2 , Table S2 , and Table S3 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1. Purity of CD3 + CD4 + CD45RA + T cells, CD3 + CD8 + T cells and CD4 + CD25 + FoxP3 + Treg cells. CD3 + CD4 + CD45RA + T cells and CD3 + CD8 + T cells were purified by magnetic cell sorting, and their purity was determined by flow cytometry. (A) The purity of CD3 + CD4 + CD45RA + T cells was 99.45% (CD3 + T cells, 99.6%; CD4 + CD45RA + T cells, 99.85%). (B) The purity of CD3 + CD8 + T cells was 95.32%. (C) The purity of CD4 + CD25 + FoxP3 + Treg cells was 93.15% (CD4 + T cells, 99.5%; CD25 + FoxP3 + T cells, 93.62%). (D) miR-155 expression in T cells of the patients with vitiligo and healthy donor was detected by reverse transcription quantitative polymerase chain reaction. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
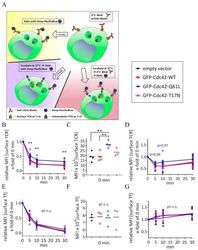
- Figure 4 Expression of Cdc42-Q61L selectively impairs internalization of the TCR-CD3 complex. ( A ) Schematic of the flow-cytometry based internalization assay; cells are labelled at 4 degC with a functional biotinylated anti-CD3epsilon and either (yellow) directly stained with Pacific Blue-streptavidin to measure surface expression of TCR-CD3 complex in resting cells, (blue) activated by incubation at 37 degC and stained with Pacific Blue-streptavidin to detect remaining TCR-CD3 at the cell surface after activation-induced internalization, or (red) activated by incubation at 37 degC, re-labelled with biotinylated anti-CD3epsilon and stained with Pacific Blue-streptavidin to detect total surface TCR-CD3 in activated cells. ( B ) Remaining TCR-CD3 at the cell surface detected by an antibody against CD3epsilon (clone OKT3) after activation-induced internalization in cells expressing an empty vector, GFP-WT-Cdc42, GFP-Cdc42-Q61L, or GFP-Cdc42-T17N. ( C ) Surface expression of TCR-CD3 complex in cells expressing the same constructs as in ( B ). ( D ) Total surface TCR-CD3 in activated cells transfected as in ( B ). ( E ) Internalization of biotinylated Tf detected with Pacific Blue-streptavidin after incubation at 37 degC as described for anti-CD3epsilon in ( A ), in cells activated by soluble anti-CD3epsilon and expressing the same constructs as in ( B ). ( F ) Surface expression of Tf in cells expressing the same constructs as in ( B ). ( G ) Total Tf at the cell surface detect
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Flow cytometric analysis of the proportion and proliferation ability of CD3 + CD8 + T lymphocytes. (a) Proportion of CD3 + CD8 + T lymphocytes. (b) Blank group. (c) Control group, lymphocytes cocultured with A-MSCs stimulated at 0 ng/mL PLP. (d) Treated group, lymphocytes cocultured with A-MSCs stimulated at 50 ng/mL PLP.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 13 Flow cytometric analysis of the proportion of CD3 + CD8 + T lymphocytes after A-MSCs coculture with TLR3 and TLR4 inhibition. There was no significant difference in the proportion of CD3 + CD8 + T lymphocytes between concentrations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 1 Figure Characterization of TILs and PBMCs from colorectal and breast cancer patients. (A) Expression of PD-1 in CD8 + TILs and PBMCs from colorectal cancers and breast cancers. (Left) Representative flow cytometry data. TILs and PBMCs were stained with CD3, CD8, and PD-1 antibodies, and PD-1 expression in CD3 + CD8 + T cells was analyzed by flow cytometry. (Right) Percentages of PD-1 + and PD-1 - cells in CD8 + TILs and CD8 + PBMCs in colorectal cancer (CC) patients and breast cancer (BC) patients. The percentage of PD-1 + and PD-1 - cells in TILs were analyzed by flow cytometry with 34 CC patient's and 10 BC patient's samples. The percentages of PD-1 + and PD-1 - cells in PBMCs were analyzed with samples from 10 CC patients and 6 BC patients. Because of limited cell number, each patient's sample was analyzed once per experiment and mean +- SD is calculated. (B) The TCRs repertoire of PD-1 + CD8 + TILs from CC and BC patients. (Left) Representative TCR repertoires of PD-1 + and PD-1 - TILs from a CC patient and a BC patient. PD-1 + or PD-1 - CD8 + TILs were single cell sorted, and then TCR cDNA was amplified with single cell RT-PCR and sequenced directly. Each pie slice in gray indicates a T-cell population in which T cells expressed the same clonotypic TCRbeta. A white pie slice indicates a T cell population in which each T cell expressed a unique TCR. The total number of analyzed cells is shown in the center of each pie chart. (Right) Clonally expanded populations in CD8
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Immunophenotype analysis of JeKo-1-parental and JeKo-1-spheroid cells. (A) The expression of CD22, CD34, CD10, CD5, CD38 and TDT in CD19 - population from JeKo-1-spheroid cells. (B) The expression of CD19 and IgM in JeKo-1-spheroid total cells. (C) Comparison of marker expression in JeKo-1-spheroid with the normal B cell differentiation stages. Red font indicates the same immunophenotype. Figure 2
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
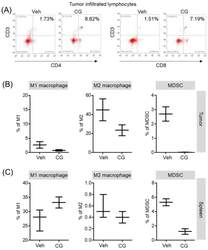
- Figure 6 The proportion of MDSCs and M1/M2 macrophage were changed by CG-745 treatment in Hepa1-6 syngeneic mouse model: (A-C) Hepa1-6 inoculated C57BL/6 mice were treated with vehicle or 15 mg/kg/day of CG (CG-745) by intraperitoneally for 5 days/week for 3 weeks. Tumor (A, B) and spleen (C) were harvested from each individual mouse. Cells were stained with fluorescence-conjugated antibodies specific to CD3, CD4, CD8, CD25 and Foxp3, and subjected to flow cytometry analysis. The graph shows means with error bars. Error bars represent standard deviations for 3 samples in each group. Results are representative of three separate experiments (A). p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
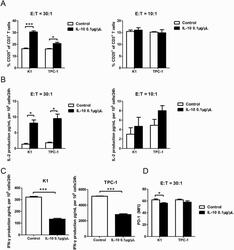
- Figure 4. IL-10 promotes T-cell activation in cocultures of CD3 + CD8 + CD25 + T cells and PTC cell lines. ( A ) Flow cytometry results of the CD25 + fraction of CD3 + T cells in coculture systems containing activated CD3 + CD8 + CD25 + T lymphocytes (effector cells, E) and pretreated PTC cells (K-1 and TPC-1 treated with 0.1 mug/muL of IL-10 for 24 hours; target cells, T) at various E:T ratios (30:1 and 10:1) (* P < 0.05, *** P < 0.001). ( B ) IL-2 production in coculture systems (details described above) measured by ELISA (* P < 0.05). ( C ) IFN-gamma production in coculture systems of CD3 + CD8 + CD25 + T lymphocytes and IL-10-stimulated PTC cells (K-1 and TPC-1) at a 30:1 ratio (*** P < 0.001). ( D ) Mean fluorescence intensity (MFI) of PD-1 expression in coculture systems of activated CD3 + CD8 + CD25 + T lymphocytes and IL-10 pretreated PTC cells at a 30:1 ratio (* P < 0.05). All tests were conducted in triplicate. ELISA, enzyme-linked immunosorbent assay; H, hours; IFN, interferon; IL, interleukin; PD-1, programmed death-1; PTC, papillary thyroid cancer.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
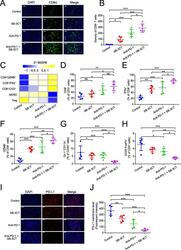
- Fig. 3 Immune features in tumors for B16F10 xenograft mouse model with SB-3CT treatment. a Fluorescence expression and b quantification of CD8 + T cells. c Heatmap of Z -score normalized percentage of immune cell populations ( d - h ) in TILs for B16F10 tumor-bearing mice treated with anti-PD-1 and SB-3CT in combination or alone. d - h In the implanted B16F10 tumors from mice treated with or without SB-3CT and PD-1 blockade, fluorescence-activated cell sorting (FACS) was used to measure d CD8 + in CD3 + T cells, e CD8 + IFNgamma + in CD8 + T cells, f CD8 + GZMB + in CD8 + T cells, g Gr-1 + CD11b + MDSCs in CD45 + cells, and h CD25 + FOXP3 + Treg in CD4 + cells. i Fluorescence expression and j quantification of PD-L1 in B16F10 tumor-bearing wild-type C57/BL6 mice treated with isotype, SB-3CT, anti-PD-1, or combination strategy. Sample size is 5 in each cohort. Scale bars, 50 mum. Results are mean +- s.d. ns, p > 0.05,* p < 0.05, ** p < 0.01, and *** p < 0.001, as determined by one-way ANOVA and Dunnett's multiple comparison test
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry