12-4774-41
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [22]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-4774-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (150D/E4), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: eBioscience offers a panel of monoclonal antibodies to different epitopes of human/primate and mouse/rat Foxp3 protein, providing useful tools for investigating the complete expression pattern of Foxp3 at the protein level, and discerning the precise subsets of Foxp3+ cells. Other antibodies to Foxp3 available from eBioscience, which have been used significantly in scientific literature, include the anti-human Foxp3 PCH101 (Product # 72-5776) and ebio7979 (Product # 12-7979), and the anti-mouse/rat Foxp3 FJK-16s (Product # 72-5775). The 150D/E4 has been mapped to the splice region in Exon 2 found in human cells. The splicing of this region has not been shown to occur in mouse. Characterization of this antibody to the splice variant has been limited to epitope mapping. It should be noted that other antibodies such as PCH101 and 236A/E7 will recognize both the spliced and full length forms of the Foxp3 protein and stain more intensely than 150D/E4 and FJK-16s in human cells. The 150D/E4 antibody reacts with mouse/rat/human Foxp3 also known as FORKHEAD BOX P3, SCURFIN, and JM2. Cross reactivity of this antibody to other proteins has not been determined. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of FoxP3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of Foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells. Applications Reported: This 150D/E4 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This 150D/E4 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of mouse splenocytes using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523-00) and protocol. Please see Best Protocols Section (Staining intracellular Antigens for Flow Cytometry) for staining protocol (refer to Protocol B: One-step protocol for intracellular (nuclear) proteins). This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- 150D/E4
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Nanoliposome C6-Ceramide in combination with anti-CTLA4 antibody improves anti-tumor immunity in hepatocellular cancer.
Retinoic Acid Induces Functionally Suppressive Foxp3(+)RORγt(+) T Cells In Vitro.
Regulatory T-cell Transcriptomic Reprogramming Characterizes Adverse Events by Checkpoint Inhibitors in Solid Tumors.
Bendamustine with total body irradiation conditioning yields tolerant T-cells while preserving T-cell-dependent graft-versus-leukemia.
Differential expression of tissue-restricted antigens among mTEC is associated with distinct autoreactive T cell fates.
Regulatory T Cells License Macrophage Pro-Resolving Functions During Atherosclerosis Regression.
Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response.
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
A fully-virulent retargeted oncolytic HSV armed with IL-12 elicits local immunity and vaccine therapy towards distant tumors.
Cutting Edge: Increased Autoimmunity Risk in Glycogen Storage Disease Type 1b Is Associated with a Reduced Engagement of Glycolysis in T Cells and an Impaired Regulatory T Cell Function.
Microbial, metabolomic, and immunologic dynamics in a relapsing genetic mouse model of colitis induced by T-synthase deficiency.
Characteristics of CD4(+)CD25(+)Foxp3(+) regulatory T cells in patients with multiple organ dysfunction syndrome.
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
The effect of citrus peel extracts on cytokines levels and T regulatory cells in acute liver injury.
Elimination of IL-10-inducing T-helper epitopes from an IGFBP-2 vaccine ensures potent antitumor activity.
A CD8 T cell/indoleamine 2,3-dioxygenase axis is required for mesenchymal stem cell suppression of human systemic lupus erythematosus.
CD40 deficiency in mice exacerbates obesity-induced adipose tissue inflammation, hepatic steatosis, and insulin resistance.
Regulatory T cell lineage specification by the forkhead transcription factor foxp3.
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
The origin of FOXP3-expressing CD4+ regulatory T cells: thymus or periphery.
Control of regulatory T cell development by the transcription factor Foxp3.
Qi X, Wu F, Kim SH, Kaifi JT, Kimchi ET, Snyder H, Illendula A, Fox T, Kester M, Staveley-O'Carroll KF, Li G
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2022 Apr;36(4):e22250
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2022 Apr;36(4):e22250
Retinoic Acid Induces Functionally Suppressive Foxp3(+)RORγt(+) T Cells In Vitro.
Martínez-Blanco M, Lozano-Ojalvo D, Pérez-Rodríguez L, Benedé S, Molina E, López-Fandiño R
Frontiers in immunology 2021;12:675733
Frontiers in immunology 2021;12:675733
Regulatory T-cell Transcriptomic Reprogramming Characterizes Adverse Events by Checkpoint Inhibitors in Solid Tumors.
Grigoriou M, Banos A, Hatzioannou A, Kloetgen A, Kouzis P, Aggouraki D, Zakopoulou R, Bamias G, Kassi E, Mavroudis D, Bamias A, Boumpas DT, Tsirigos A, Gogas H, Alissafi T, Verginis P
Cancer immunology research 2021 Jul;9(7):726-734
Cancer immunology research 2021 Jul;9(7):726-734
Bendamustine with total body irradiation conditioning yields tolerant T-cells while preserving T-cell-dependent graft-versus-leukemia.
Stokes J, Hoffman EA, Molina MS, Kummet N, Simpson RJ, Zeng Y, Katsanis E
Oncoimmunology 2020;9(1):1758011
Oncoimmunology 2020;9(1):1758011
Differential expression of tissue-restricted antigens among mTEC is associated with distinct autoreactive T cell fates.
Lebel MÈ, Coutelier M, Galipeau M, Kleinman CL, Moon JJ, Melichar HJ
Nature communications 2020 Jul 24;11(1):3734
Nature communications 2020 Jul 24;11(1):3734
Regulatory T Cells License Macrophage Pro-Resolving Functions During Atherosclerosis Regression.
Sharma M, Schlegel MP, Afonso MS, Brown EJ, Rahman K, Weinstock A, Sansbury BE, Corr EM, van Solingen C, Koelwyn GJ, Shanley LC, Beckett L, Peled D, Lafaille JJ, Spite M, Loke P, Fisher EA, Moore KJ
Circulation research 2020 Jul 17;127(3):335-353
Circulation research 2020 Jul 17;127(3):335-353
Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response.
Court AC, Le-Gatt A, Luz-Crawford P, Parra E, Aliaga-Tobar V, Bátiz LF, Contreras RA, Ortúzar MI, Kurte M, Elizondo-Vega R, Maracaja-Coutinho V, Pino-Lagos K, Figueroa FE, Khoury M
EMBO reports 2020 Feb 5;21(2):e48052
EMBO reports 2020 Feb 5;21(2):e48052
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
Agarwal S, Kraus Z, Dement-Brown J, Alabi O, Starost K, Tolnay M
Cell reports 2020 Feb 4;30(5):1292-1299.e3
Cell reports 2020 Feb 4;30(5):1292-1299.e3
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
Zhang Y, Liu Y, Xu Y
Frontiers in oncology 2019;9:1401
Frontiers in oncology 2019;9:1401
A fully-virulent retargeted oncolytic HSV armed with IL-12 elicits local immunity and vaccine therapy towards distant tumors.
Leoni V, Vannini A, Gatta V, Rambaldi J, Sanapo M, Barboni C, Zaghini A, Nanni P, Lollini PL, Casiraghi C, Campadelli-Fiume G
PLoS pathogens 2018 Aug;14(8):e1007209
PLoS pathogens 2018 Aug;14(8):e1007209
Cutting Edge: Increased Autoimmunity Risk in Glycogen Storage Disease Type 1b Is Associated with a Reduced Engagement of Glycolysis in T Cells and an Impaired Regulatory T Cell Function.
Melis D, Carbone F, Minopoli G, La Rocca C, Perna F, De Rosa V, Galgani M, Andria G, Parenti G, Matarese G
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3803-3808
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3803-3808
Microbial, metabolomic, and immunologic dynamics in a relapsing genetic mouse model of colitis induced by T-synthase deficiency.
Jacobs JP, Lin L, Goudarzi M, Ruegger P, McGovern DP, Fornace AJ Jr, Borneman J, Xia L, Braun J
Gut microbes 2017 Jan 2;8(1):1-16
Gut microbes 2017 Jan 2;8(1):1-16
Characteristics of CD4(+)CD25(+)Foxp3(+) regulatory T cells in patients with multiple organ dysfunction syndrome.
Zang X, Jiang Y, Li X, DU Y, Niu J
Experimental and therapeutic medicine 2016 May;11(5):1908-1912
Experimental and therapeutic medicine 2016 May;11(5):1908-1912
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
De Rosa V, Galgani M, Porcellini A, Colamatteo A, Santopaolo M, Zuchegna C, Romano A, De Simone S, Procaccini C, La Rocca C, Carrieri PB, Maniscalco GT, Salvetti M, Buscarinu MC, Franzese A, Mozzillo E, La Cava A, Matarese G
Nature immunology 2015 Nov;16(11):1174-84
Nature immunology 2015 Nov;16(11):1174-84
The effect of citrus peel extracts on cytokines levels and T regulatory cells in acute liver injury.
Pantsulaia Ia, Iobadze M, Pantsulaia N, Chikovani T
BioMed research international 2014;2014:127879
BioMed research international 2014;2014:127879
Elimination of IL-10-inducing T-helper epitopes from an IGFBP-2 vaccine ensures potent antitumor activity.
Cecil DL, Holt GE, Park KH, Gad E, Rastetter L, Childs J, Higgins D, Disis ML
Cancer research 2014 May 15;74(10):2710-8
Cancer research 2014 May 15;74(10):2710-8
A CD8 T cell/indoleamine 2,3-dioxygenase axis is required for mesenchymal stem cell suppression of human systemic lupus erythematosus.
Wang D, Feng X, Lu L, Konkel JE, Zhang H, Chen Z, Li X, Gao X, Lu L, Shi S, Chen W, Sun L
Arthritis & rheumatology (Hoboken, N.J.) 2014 Aug;66(8):2234-45
Arthritis & rheumatology (Hoboken, N.J.) 2014 Aug;66(8):2234-45
CD40 deficiency in mice exacerbates obesity-induced adipose tissue inflammation, hepatic steatosis, and insulin resistance.
Guo CA, Kogan S, Amano SU, Wang M, Dagdeviren S, Friedline RH, Aouadi M, Kim JK, Czech MP
American journal of physiology. Endocrinology and metabolism 2013 May 1;304(9):E951-63
American journal of physiology. Endocrinology and metabolism 2013 May 1;304(9):E951-63
Regulatory T cell lineage specification by the forkhead transcription factor foxp3.
Fontenot JD, Rasmussen JP, Williams LM, Dooley JL, Farr AG, Rudensky AY
Immunity 2005 Mar;22(3):329-41
Immunity 2005 Mar;22(3):329-41
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Roncador G, Brown PJ, Maestre L, Hue S, Martínez-Torrecuadrada JL, Ling KL, Pratap S, Toms C, Fox BC, Cerundolo V, Powrie F, Banham AH
European journal of immunology 2005 Jun;35(6):1681-91
European journal of immunology 2005 Jun;35(6):1681-91
The origin of FOXP3-expressing CD4+ regulatory T cells: thymus or periphery.
Sakaguchi S
The Journal of clinical investigation 2003 Nov;112(9):1310-2
The Journal of clinical investigation 2003 Nov;112(9):1310-2
Control of regulatory T cell development by the transcription factor Foxp3.
Hori S, Nomura T, Sakaguchi S
Science (New York, N.Y.) 2003 Feb 14;299(5609):1057-61
Science (New York, N.Y.) 2003 Feb 14;299(5609):1057-61
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
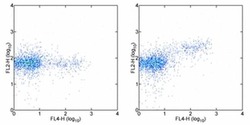
- Experimental details
- Surface staining of C57BL/6 splenocytes with Anti-Mouse CD25 APC (Product # 17-0251-82) followed bY Fixation and Permeabilization witt the Foxp3 Buffer Set (Product # 00-5523-00). Cells were subsequently stained with Mouse IgG1 K Isotype Control PE (Product # 12-4714-81) (left) or Anti-Foxp3 PE (right). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
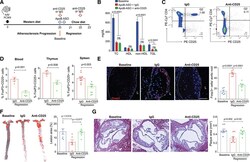
- Experimental details
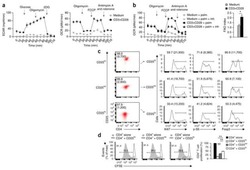
- Figure 2. Regulatory T cell (Treg) depletion abrogates atherosclerosis regression. A , Experimental design: Atherosclerosis was established in C57BL6&solJ mice by injection of adeno-associated virus (AAV)-PCSK (proprotein convertase subtilisin&solkexin type)-9 followed by Western diet feeding for 20 wk (Baseline). Mice were then switched to chow diet and atherosclerosis regression was initiated by apoB (apolipoprotein B)-antisense oligonucleotide (ASO)-mediated lipid lowering in conjunction with anti-CD25 or IgG isotype control antibody treatment for 3 wk. B , Plasma total cholesterol, HDL (high-density lipoprotein) and non-HDL cholesterol, and triglyceride levels at baseline and after regression &plus IgG anti-CD25 treatment (n=11 mice&solgroup). C , Representative flow cytometric analyses of CD4 &plus CD25 &plus cells in the circulation of mice treated with IgG or anti-CD25 during atherosclerosis regression. D , Quantification of FoxP3 &plus CD25 &plus cells in peripheral blood (n=5&solgroup), thymus (n=5&solgroup), and spleen (n=9&solgroup) of mice by flow cytometry. Data are mean+-SEM and are representative of 2 independent experiments. P values were determined by a Student t test. E , Representative immunofluorescence staining for FoxP3 in aortic root plaques from mice at baseline and after regression &plus IgG or anti-CD25 treatment (n=6 mice&solgroup). Quantification shown at right. Scale bars=100 &mgrm. F and G , Representative images and quantification of atheroscler
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
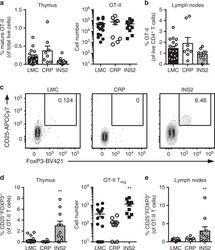
- Experimental details
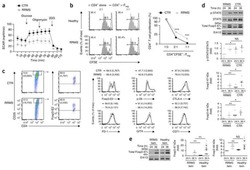
- Fig. 4 mTEC hi -restricted expression of USA preferentially induces T reg differentiation. Littermate control (LMC), CRP, and INS2 Tg mice (CD45.2, Thy1.2) were irradiated and reconstituted with 1% OT-II (CD45.2, Thy1.1) and 99% WT (CD45.1, Thy1.2) bone marrow. The hematopoietic chimeras were analyzed 6-8 weeks post-irradiation. a Proportion of mature TCRbeta hi CD4 + SP OT-II thymocytes among total live cells and absolute number of live CD4 + SP OT-II thymocytes. b Proportion of OT-II cells within the CD4 + T cell population in the peripheral lymph nodes. c Representative flow plots of CD25 and FoxP3 expression on OT-II T cells. d Proportion and absolute cell number of CD25 + Foxp3 + OT-II T cells in the thymus. e Proportion of CD25 + Foxp3 + OT-II T cells in the lymph nodes. (mean +- SEM, n = 17 LMC, 8 CRP, 10 INS2, from three independent experiments). All p -values indicated are calculated by Kruskal-Wallis test followed by Dunn''s post-hoc comparisons to LMC, two-sided. ** p < 0.01 relative to LMC. Source data are provided as a Source Data file.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
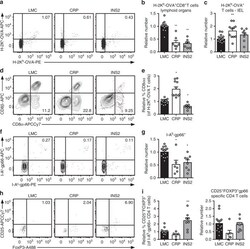
- Experimental details
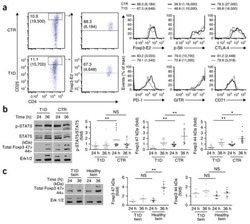
- Fig. 5 TRA expression by distinct mTEC compartments impacts the mode of tolerance induction in the polyclonal T cell repertoire in naive mice. a Representative flow plots of H-2K b -OVA tetramer on pooled peripheral lymph nodes and spleen cells from naive mice after enrichment, gated on live TCRalphabeta + CD8 + T cells. Relative number of H-2K b -OVA-specific TCRalphabeta + CD8 + T cells in the peripheral secondary lymphoid organs (mean +- SEM, n = 12 LMC, 7 CRP, 7 INS2) ( b ) and intraepithelial lymphocytes (IEL) (mean +- SEM, n = 17 LMC, 11 CRP, 8 INS2) ( c ) from CRP and INS2 Tg mice as compared to their respective littermate controls (LMC). d Representative flow plot of CD8alpha and CD8beta and e relative proportion of TCRalphabeta + CD8alphaalpha within the H-2K b -OVA tetramer double positive TCRalphabeta IEL of CRP and INS2 Tg mice as compared to their respective LMC (mean +- SEM, n = 17 LMC, 11 CRP, 8 INS2). f Representative flow plots of I-A b -gp66 tetramer on pooled peripheral lymph nodes and spleen cells from naive mice, gated on live TCRalphabeta + CD4 + T cells. g Relative number of I-A b -gp66 tetramer DP CD4 + T cells in the peripheral lymph nodes and the spleen of naive mice. h Representative flow plots of CD25 and FoxP3, gated on live I-A b -gp66 tetramer + CD4 + T cells. i Relative proportion of CD25 + FoxP3 + T reg of gp66-specific CD4 + T cells and relative number of gp66-specific CD4 + CD25 + FoxP3 + T reg from CRP and INS2 peripheral lymph nodes and sp
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
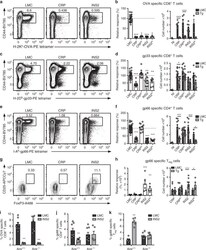
- Experimental details
- Fig. 6 TRA expression by distinct mTEC compartments impacts the mode of tolerance induction in the polyclonal T cell repertoire in LCMV infected mice. Mice were infected with LCMV-OVA and the antigen-specific response was measured in the spleen 8 days post-infection (NI, not infected mice) a, c Representative CD44 and H-2K b -OVA or H-2D b -gp33 tetramer staining on CD8 + T cells from littermate controls (LMC), CRP, and INS2 Tg mice. Relative response and absolute number of OVA- ( b ) and gp33- ( d ) specific CD8 + T cell response in USA expressing mice as compared to LMC. e Representative CD44 and I-A b -gp66 tetramer staining on CD4 + T cells from LMC, CRP, and INS2 Tg mice. f Relative and absolute gp66-specific response in USA expressing mice. g Representative flow plot of CD25 and FoxP3, gated on live I-A b -gp66-specific CD4 + T cells. h Relative proportion of CD25 + FoxP3 + T reg among gp66-specific CD4 + T cells and absolute number of gp66-specific CD4 + CD25 + FoxP3 + T reg (mean +- SEM, b left panel n = 22 LMC, 7 CRP, 5 CRP lo , 9 INS2, 3 INS2 lo ; b right panel n = 3 NI, 8 CRP LMC, 10 CRP Tg, 9 CRP lo LMC, 9 CRP lo Tg, 10 INS2 LMC, 9 INS2 Tg, 3 INS2 lo LMC, 3 INS2 lo Tg; d and f left panel n = 32 LMC, 12 CRP, 12 CRP lo , 14 INS2, 8 INS2 lo , d right panel n = 6 NI, 11 CRP LMC, 12 CRP Tg, 11 CRP lo LMC, 12 CRP lo Tg, 13 INS2 LMC, 11 INS2 Tg, 6 INS2 lo LMC, 6 INS2 lo Tg, f right panel n = 6 NI, 11 CRP LMC, 13 CRP Tg, 12 CRP lo LMC, 13 CRP lo Tg, 14 INS2 LMC, 12 INS2 T
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry