25-4776-42
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [40]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [23]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-4776-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (PCH101), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: eBioscience offers a panel of monoclonal antibodies to different epitopes of human Foxp3, providing useful tools for investigating the complete expression pattern of Foxp3 at the protein level, and discerning the precise subsets of Foxp3^+ cells. The PCH101 antibody reacts with the amino terminus of human foxp3 protein also known as FORKHEAD BOX P3, SCURFIN, and JM2; cross reactivity of this antibody to other proteins has not been determined. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of Foxp3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells. Intracellular staining of human peripheral blood mononuclear cells (PBMCs) with PCH101 antibody using the anti-human Foxp3 Staining Set and protocol reveals approximately 0.5-4% of lymphocytes staining, with the majority of staining occurring in the CD25^bright population. This is subject to donor variability. PCH101 crossreacts with rhesus, chimpanzee and cynomolgus. We recommend the use of CD4 (OKT4, Product # 11-0048-42 , or RPA-T4, Product # 11-0049-42 , depending on the species) and CD25 (BC96, Product # 17-0259-42). Applications Reported: This PCH101 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This PCH101 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of normal human peripheral blood cells using the Foxp3/Transcription Factor Buffer Set (Product # 00-5523). Please see Best Protocols Section (Staining intracellular Antigens for Flow Cytometry) for staining protocol (refer to Protocol B: One-step protocol for intracellular (nuclear) proteins). This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Light sensitivity: This tandem dye is sensitive photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- PCH101
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Combined TCR Repertoire Profiles and Blood Cell Phenotypes Predict Melanoma Patient Response to Personalized Neoantigen Therapy plus Anti-PD-1.
Comparative Analysis of the Regulatory T Cells Dynamics in Peripheral Blood in Human and Porcine Polytrauma.
Injection of Adipose-Derived Stromal Cells in the Knee of Patients with Severe Osteoarthritis has a Systemic Effect and Promotes an Anti-Inflammatory Phenotype of Circulating Immune Cells.
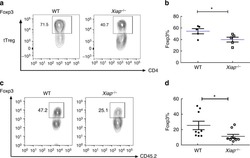
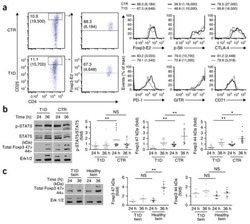
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
Intratumoral FoxP3(+)Helios(+) Regulatory T Cells Upregulating Immunosuppressive Molecules Are Expanded in Human Colorectal Cancer.
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Phenotypic characterization of regulatory T cells from antiretroviral-naive HIV-1-infected people.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Combining FoxP3 and Helios with GARP/LAP markers can identify expanded Treg subsets in cancer patients.
Equilibrium of Treg/Th17 cells of peripheral blood in syphilitic patients with sero-resistance.
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Human Head and Neck Squamous Cell Carcinoma-Associated Semaphorin 4D Induces Expansion of Myeloid-Derived Suppressor Cells.
Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche.
Development of Type 2, But Not Type 1, Leprosy Reactions is Associated with a Severe Reduction of Circulating and In situ Regulatory T-Cells.
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
The immunosuppressive enzyme IL4I1 promotes FoxP3(+) regulatory T lymphocyte differentiation.
PD-1+Tim-3+ CD8+ T Lymphocytes Display Varied Degrees of Functional Exhaustion in Patients with Regionally Metastatic Differentiated Thyroid Cancer.
Synovial Regulatory T Cells Occupy a Discrete TCR Niche in Human Arthritis and Require Local Signals To Stabilize FOXP3 Protein Expression.
Helios, and not FoxP3, is the marker of activated Tregs expressing GARP/LAP.
A CD8 T cell/indoleamine 2,3-dioxygenase axis is required for mesenchymal stem cell suppression of human systemic lupus erythematosus.
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
Human T cells upregulate CD69 after coculture with xenogeneic genetically-modified pig mesenchymal stromal cells.
Impaired function of regulatory T cells in cord blood of children of allergic mothers.
OMIP-006: phenotypic subset analysis of human T regulatory cells via polychromatic flow cytometry.
Engagement of TLR2 reverses the suppressor function of conjunctiva CD4+CD25+ regulatory T cells and promotes herpes simplex virus epitope-specific CD4+CD25- effector T cell responses.
CD40 signalling induces IL-10-producing, tolerogenic dendritic cells.
Safety and T cell modulating effects of high dose vitamin D3 supplementation in multiple sclerosis.
Dynamic development of homing receptor expression and memory cell differentiation of infant CD4+CD25high regulatory T cells.
Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells.
IL-15 and dermal fibroblasts induce proliferation of natural regulatory T cells isolated from human skin.
FOXP3 regulates TLR10 expression in human T regulatory cells.
Mucosal but not peripheral FOXP3+ regulatory T cells are highly increased in untreated HIV infection and normalize after suppressive HAART.
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Foxp3+CD4+CD25+ T cells control virus-specific memory T cells in chimpanzees that recovered from hepatitis C.
Depletion of alloreactive T cells via CD69: implications on antiviral, antileukemic and immunoregulatory T lymphocytes.
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Human CD4+ T cells express TLR5 and its ligand flagellin enhances the suppressive capacity and expression of FOXP3 in CD4+CD25+ T regulatory cells.
Poran A, Scherer J, Bushway ME, Besada R, Balogh KN, Wanamaker A, Williams RG, Prabhakara J, Ott PA, Hu-Lieskovan S, Khondker ZS, Gaynor RB, Rooney MS, Srinivasan L
Cell reports. Medicine 2020 Nov 17;1(8):100141
Cell reports. Medicine 2020 Nov 17;1(8):100141
Comparative Analysis of the Regulatory T Cells Dynamics in Peripheral Blood in Human and Porcine Polytrauma.
Serve R, Sturm R, Schimunek L, Störmann P, Heftrig D, Teuben MPJ, Oppermann E, Horst K, Pfeifer R, Simon TP, Kalbas Y, Pape HC, Hildebrand F, Marzi I, Relja B
Frontiers in immunology 2018;9:435
Frontiers in immunology 2018;9:435
Injection of Adipose-Derived Stromal Cells in the Knee of Patients with Severe Osteoarthritis has a Systemic Effect and Promotes an Anti-Inflammatory Phenotype of Circulating Immune Cells.
Pers YM, Quentin J, Feirreira R, Espinoza F, Abdellaoui N, Erkilic N, Cren M, Dufourcq-Lopez E, Pullig O, Nöth U, Jorgensen C, Louis-Plence P
Theranostics 2018;8(20):5519-5528
Theranostics 2018;8(20):5519-5528
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Hsieh WC, Hsu TS, Chang YJ, Lai MZ
Nature communications 2018 Jan 31;9(1):463
Nature communications 2018 Jan 31;9(1):463
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
Arce Vargas F, Furness AJS, Litchfield K, Joshi K, Rosenthal R, Ghorani E, Solomon I, Lesko MH, Ruef N, Roddie C, Henry JY, Spain L, Ben Aissa A, Georgiou A, Wong YNS, Smith M, Strauss D, Hayes A, Nicol D, O'Brien T, Mårtensson L, Ljungars A, Teige I, Frendéus B, TRACERx Melanoma, TRACERx Renal, TRACERx Lung consortia, Pule M, Marafioti T, Gore M, Larkin J, Turajlic S, Swanton C, Peggs KS, Quezada SA
Cancer cell 2018 Apr 9;33(4):649-663.e4
Cancer cell 2018 Apr 9;33(4):649-663.e4
Intratumoral FoxP3(+)Helios(+) Regulatory T Cells Upregulating Immunosuppressive Molecules Are Expanded in Human Colorectal Cancer.
Syed Khaja AS, Toor SM, El Salhat H, Ali BR, Elkord E
Frontiers in immunology 2017;8:619
Frontiers in immunology 2017;8:619
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Syed Khaja AS, Toor SM, El Salhat H, Faour I, Ul Haq N, Ali BR, Elkord E
Oncotarget 2017 May 16;8(20):33159-33171
Oncotarget 2017 May 16;8(20):33159-33171
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Terranova-Barberio M, Thomas S, Ali N, Pawlowska N, Park J, Krings G, Rosenblum MD, Budillon A, Munster PN
Oncotarget 2017 Dec 26;8(69):114156-114172
Oncotarget 2017 Dec 26;8(69):114156-114172
Phenotypic characterization of regulatory T cells from antiretroviral-naive HIV-1-infected people.
Ambada GN, Ntsama CE, Nji NN, Ngu LN, Sake CN, Lissom A, Tchouangeu FT, Tchadji J, Sosso M, Etoa FX, Nchinda GW
Immunology 2017 Aug;151(4):405-416
Immunology 2017 Aug;151(4):405-416
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
Combining FoxP3 and Helios with GARP/LAP markers can identify expanded Treg subsets in cancer patients.
Abd Al Samid M, Chaudhary B, Khaled YS, Ammori BJ, Elkord E
Oncotarget 2016 Mar 22;7(12):14083-94
Oncotarget 2016 Mar 22;7(12):14083-94
Equilibrium of Treg/Th17 cells of peripheral blood in syphilitic patients with sero-resistance.
Zhao J, Ma J, Zhang X, Li Q, Yang X
Experimental and therapeutic medicine 2016 Jun;11(6):2300-2304
Experimental and therapeutic medicine 2016 Jun;11(6):2300-2304
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Sakthivel P, Grunewald J, Eklund A, Bruder D, Wahlström J
Clinical and experimental immunology 2016 Feb;183(2):294-306
Clinical and experimental immunology 2016 Feb;183(2):294-306
Human Head and Neck Squamous Cell Carcinoma-Associated Semaphorin 4D Induces Expansion of Myeloid-Derived Suppressor Cells.
Younis RH, Han KL, Webb TJ
Journal of immunology (Baltimore, Md. : 1950) 2016 Feb 1;196(3):1419-29
Journal of immunology (Baltimore, Md. : 1950) 2016 Feb 1;196(3):1419-29
Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche.
Clever D, Roychoudhuri R, Constantinides MG, Askenase MH, Sukumar M, Klebanoff CA, Eil RL, Hickman HD, Yu Z, Pan JH, Palmer DC, Phan AT, Goulding J, Gattinoni L, Goldrath AW, Belkaid Y, Restifo NP
Cell 2016 Aug 25;166(5):1117-1131.e14
Cell 2016 Aug 25;166(5):1117-1131.e14
Development of Type 2, But Not Type 1, Leprosy Reactions is Associated with a Severe Reduction of Circulating and In situ Regulatory T-Cells.
Vieira AP, Trindade MÂ, Pagliari C, Avancini J, Sakai-Valente NY, Duarte AJ, Benard G
The American journal of tropical medicine and hygiene 2016 Apr;94(4):721-7
The American journal of tropical medicine and hygiene 2016 Apr;94(4):721-7
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
De Rosa V, Galgani M, Porcellini A, Colamatteo A, Santopaolo M, Zuchegna C, Romano A, De Simone S, Procaccini C, La Rocca C, Carrieri PB, Maniscalco GT, Salvetti M, Buscarinu MC, Franzese A, Mozzillo E, La Cava A, Matarese G
Nature immunology 2015 Nov;16(11):1174-84
Nature immunology 2015 Nov;16(11):1174-84
The immunosuppressive enzyme IL4I1 promotes FoxP3(+) regulatory T lymphocyte differentiation.
Cousin C, Aubatin A, Le Gouvello S, Apetoh L, Castellano F, Molinier-Frenkel V
European journal of immunology 2015 Jun;45(6):1772-82
European journal of immunology 2015 Jun;45(6):1772-82
PD-1+Tim-3+ CD8+ T Lymphocytes Display Varied Degrees of Functional Exhaustion in Patients with Regionally Metastatic Differentiated Thyroid Cancer.
Severson JJ, Serracino HS, Mateescu V, Raeburn CD, McIntyre RC Jr, Sams SB, Haugen BR, French JD
Cancer immunology research 2015 Jun;3(6):620-30
Cancer immunology research 2015 Jun;3(6):620-30
Synovial Regulatory T Cells Occupy a Discrete TCR Niche in Human Arthritis and Require Local Signals To Stabilize FOXP3 Protein Expression.
Bending D, Giannakopoulou E, Lom H, Wedderburn LR
Journal of immunology (Baltimore, Md. : 1950) 2015 Dec 15;195(12):5616-24
Journal of immunology (Baltimore, Md. : 1950) 2015 Dec 15;195(12):5616-24
Helios, and not FoxP3, is the marker of activated Tregs expressing GARP/LAP.
Elkord E, Abd Al Samid M, Chaudhary B
Oncotarget 2015 Aug 21;6(24):20026-36
Oncotarget 2015 Aug 21;6(24):20026-36
A CD8 T cell/indoleamine 2,3-dioxygenase axis is required for mesenchymal stem cell suppression of human systemic lupus erythematosus.
Wang D, Feng X, Lu L, Konkel JE, Zhang H, Chen Z, Li X, Gao X, Lu L, Shi S, Chen W, Sun L
Arthritis & rheumatology (Hoboken, N.J.) 2014 Aug;66(8):2234-45
Arthritis & rheumatology (Hoboken, N.J.) 2014 Aug;66(8):2234-45
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
Liu X, Gao N, Li M, Xu D, Hou Y, Wang Q, Zhang G, Sun Q, Zhang H, Zeng X
PloS one 2013;8(6):e64531
PloS one 2013;8(6):e64531
Human T cells upregulate CD69 after coculture with xenogeneic genetically-modified pig mesenchymal stromal cells.
Li J, Andreyev O, Chen M, Marco M, Iwase H, Long C, Ayares D, Shen Z, Cooper DK, Ezzelarab MB
Cellular immunology 2013 Sep-Oct;285(1-2):23-30
Cellular immunology 2013 Sep-Oct;285(1-2):23-30
Impaired function of regulatory T cells in cord blood of children of allergic mothers.
Hrdý J, Kocourková I, Prokešová L
Clinical and experimental immunology 2012 Oct;170(1):10-7
Clinical and experimental immunology 2012 Oct;170(1):10-7
OMIP-006: phenotypic subset analysis of human T regulatory cells via polychromatic flow cytometry.
Murdoch DM, Staats JS, Weinhold KJ
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 Apr;81(4):281-3
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 Apr;81(4):281-3
Engagement of TLR2 reverses the suppressor function of conjunctiva CD4+CD25+ regulatory T cells and promotes herpes simplex virus epitope-specific CD4+CD25- effector T cell responses.
Dasgupta G, Chentoufi AA, You S, Falatoonzadeh P, Urbano LA, Akhtarmalik A, Nguyen K, Ablabutyan L, Nesburn AB, BenMohamed L
Investigative ophthalmology & visual science 2011 May 17;52(6):3321-33
Investigative ophthalmology & visual science 2011 May 17;52(6):3321-33
CD40 signalling induces IL-10-producing, tolerogenic dendritic cells.
Tuettenberg A, Fondel S, Steinbrink K, Enk AH, Jonuleit H
Experimental dermatology 2010 Jan;19(1):44-53
Experimental dermatology 2010 Jan;19(1):44-53
Safety and T cell modulating effects of high dose vitamin D3 supplementation in multiple sclerosis.
Smolders J, Peelen E, Thewissen M, Cohen Tervaert JW, Menheere P, Hupperts R, Damoiseaux J
PloS one 2010 Dec 13;5(12):e15235
PloS one 2010 Dec 13;5(12):e15235
Dynamic development of homing receptor expression and memory cell differentiation of infant CD4+CD25high regulatory T cells.
Grindebacke H, Stenstad H, Quiding-Järbrink M, Waldenström J, Adlerberth I, Wold AE, Rudin A
Journal of immunology (Baltimore, Md. : 1950) 2009 Oct 1;183(7):4360-70
Journal of immunology (Baltimore, Md. : 1950) 2009 Oct 1;183(7):4360-70
Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells.
Nagata S, Ise T, Pastan I
Journal of immunology (Baltimore, Md. : 1950) 2009 Jun 15;182(12):7518-26
Journal of immunology (Baltimore, Md. : 1950) 2009 Jun 15;182(12):7518-26
IL-15 and dermal fibroblasts induce proliferation of natural regulatory T cells isolated from human skin.
Clark RA, Kupper TS
Blood 2007 Jan 1;109(1):194-202
Blood 2007 Jan 1;109(1):194-202
FOXP3 regulates TLR10 expression in human T regulatory cells.
Bell MP, Svingen PA, Rahman MK, Xiong Y, Faubion WA Jr
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
Mucosal but not peripheral FOXP3+ regulatory T cells are highly increased in untreated HIV infection and normalize after suppressive HAART.
Epple HJ, Loddenkemper C, Kunkel D, Tröger H, Maul J, Moos V, Berg E, Ullrich R, Schulzke JD, Stein H, Duchmann R, Zeitz M, Schneider T
Blood 2006 Nov 1;108(9):3072-8
Blood 2006 Nov 1;108(9):3072-8
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Ahmadzadeh M, Rosenberg SA
Blood 2006 Mar 15;107(6):2409-14
Blood 2006 Mar 15;107(6):2409-14
Foxp3+CD4+CD25+ T cells control virus-specific memory T cells in chimpanzees that recovered from hepatitis C.
Manigold T, Shin EC, Mizukoshi E, Mihalik K, Murthy KK, Rice CM, Piccirillo CA, Rehermann B
Blood 2006 Jun 1;107(11):4424-32
Blood 2006 Jun 1;107(11):4424-32
Depletion of alloreactive T cells via CD69: implications on antiviral, antileukemic and immunoregulatory T lymphocytes.
Hartwig UF, Nonn M, Khan S, Meyer RG, Huber C, Herr W
Bone marrow transplantation 2006 Feb;37(3):297-305
Bone marrow transplantation 2006 Feb;37(3):297-305
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Lim HW, Hillsamer P, Banham AH, Kim CH
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Lim HW, Hillsamer P, Banham AH, Kim CH
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Human CD4+ T cells express TLR5 and its ligand flagellin enhances the suppressive capacity and expression of FOXP3 in CD4+CD25+ T regulatory cells.
Crellin NK, Garcia RV, Hadisfar O, Allan SE, Steiner TS, Levings MK
Journal of immunology (Baltimore, Md. : 1950) 2005 Dec 15;175(12):8051-9
Journal of immunology (Baltimore, Md. : 1950) 2005 Dec 15;175(12):8051-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
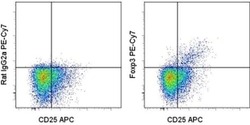
- Experimental details
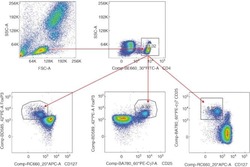
- Intracellular staining of normal human peripheral blood cells with Anti-Human/Non-Human Primate CD25 PerCP-eFluor® 710 (Product # 46-0257-42) and Rat IgG2a K Isotype Control PE-Cyanine7 (Product # 25-4321-82) (left) or Anti-Human Foxp3 PE-Cyanine7 (right) using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523-00). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
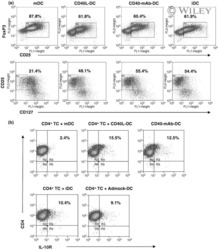
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
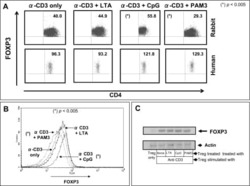
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
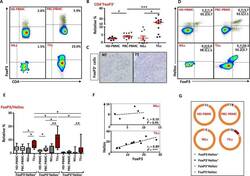
- Experimental details
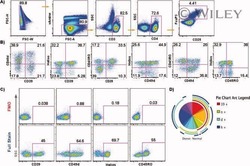
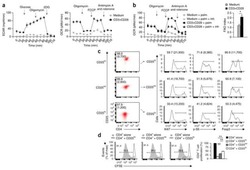
- Figure 6 FoxP3 and Helios expression in CD4 + T cells PBMC from HD and PBC patients, NILs and TILs were stained for CD3 and CD4 antibodies followed by intracellular staining for FoxP3 and Helios. Live cells were first gated using Fixable Viability Dye 660. ( A ). Representative flow cytometric plots of FoxP3 staining from one cancer patient are shown. ( B ). Scatter plot showing the differences in frequencies of FoxP3 + Tregs between different samples. ( C ). IHC staining of FoxP3 + expression in one NT and TT samples. ( D ). Flow cytometric plots of FoxP3 and Helios co-expression in CD4 + T cells from different samples and whisker plots ( E ) showing differences in various FoxP3 and Helios-expressing Treg subsets. ( F ). Non-parametric Spearman's test showing correlations between FoxP3 and Helios expressions in NILs and TILs. ( G ). Pie charts show the relative percentages of different FoxP3 and Helios Treg subsets.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
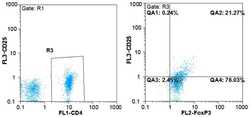
- Experimental details
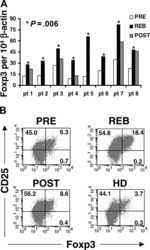
- Figure 1. Flow cytometric mapping of the CD4 + CD25 + Foxp3 + cells in peripheral blood withdrawn from the syphilitic patients with sero-resistance. Foxp3, forkhead box P3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
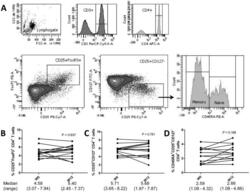
- Experimental details
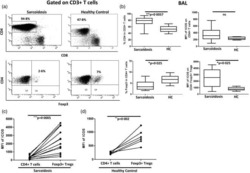
- Figure 2 Phenotypic analysis of circulating regulatory T cells (Treg). A) Isolated PBMC were analyzed directly ex-vivo by flow cytometry. In the lymphogate, CD3 + CD4 + cells were assessed for the proportions of CD25 + FoxP3 + and CD25 + CD127 - Treg cells. Expression of CD45RA was analyzed to phenotype naive (CD45RA + ) and memory (CD45RA - ) Treg. B-D) The proportions of circulating CD25 + FoxP3 + (B) and CD25 + CD127 - (C)Treg, and of naive Treg (D) before and after vitamin D 3 supplementation (week 0 and 12). Significance was assessed with the Wilcoxon signed ranks comparison test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
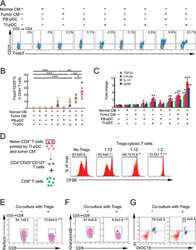
- Experimental details
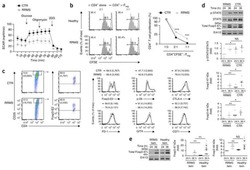
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
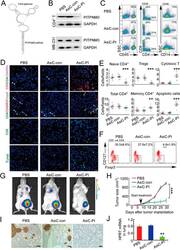
- Experimental details
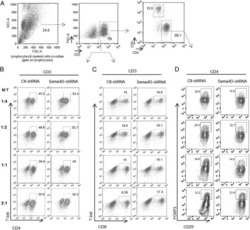
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry