25-4777-41
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [27]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-4777-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (236A/E7), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 236A/E7 antibody reacts with human Foxp3 protein also known as FORKHEAD BOX P3, SCURFIN, and JM2. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of Foxp3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of Foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells. Intracellular staining and flow cytometric analysis of freshly isolated human peripheral blood mononuclear cells (PBMCs) with the 236A/E7 antibody using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523) and protocol reveals staining of the CD4+CD25bright population. The epitope from 236A/E7 is different from that of PCH101 (Product # 72-5776). This antibody has also been shown to recognize rhesus macaque, sooty mangabey and cynomolgus macaque. Applications Reported: This 236A/E7 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This 236A/E7 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of normal human peripheral blood cells using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523) and protocol. Please refer to Best Protocols: Protocol B: One step protocol for (nuclear) intracellular proteins. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Light sensitivity: This tandem dye is sensitive photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 236A/E7
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references A human CD137×PD-L1 bispecific antibody promotes anti-tumor immunity via context-dependent T cell costimulation and checkpoint blockade.
A conserved intratumoral regulatory T cell signature identifies 4-1BB as a pan-cancer target.
Phenotypic Characterization of SLe(x)+ and CLA+ CD4+ T Cells.
Sialyl-Lewis(X) Glycoantigen Is Enriched on Cells with Persistent HIV Transcription during Therapy.
Regulatory T cell features in chronic granulomatous disease.
Unleashing Type-2 Dendritic Cells to Drive Protective Antitumor CD4(+) T Cell Immunity.
Proteomic Analyses of Human Regulatory T Cells Reveal Adaptations in Signaling Pathways that Protect Cellular Identity.
A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments.
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
Effects of conventional therapeutic interventions on the number and function of regulatory T cells.
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Geuijen C, Tacken P, Wang LC, Klooster R, van Loo PF, Zhou J, Mondal A, Liu YB, Kramer A, Condamine T, Volgina A, Hendriks LJA, van der Maaden H, Rovers E, Engels S, Fransen F, den Blanken-Smit R, Zondag-van der Zande V, Basmeleh A, Bartelink W, Kulkarni A, Marissen W, Huang CY, Hall L, Harvey S, Kim S, Martinez M, O'Brien S, Moon E, Albelda S, Kanellopoulou C, Stewart S, Nastri H, Bakker ABH, Scherle P, Logtenberg T, Hollis G, de Kruif J, Huber R, Mayes PA, Throsby M
Nature communications 2021 Jul 21;12(1):4445
Nature communications 2021 Jul 21;12(1):4445
A conserved intratumoral regulatory T cell signature identifies 4-1BB as a pan-cancer target.
Freeman ZT, Nirschl TR, Hovelson DH, Johnston RJ, Engelhardt JJ, Selby MJ, Kochel CM, Lan RY, Zhai J, Ghasemzadeh A, Gupta A, Skaist AM, Wheelan SJ, Jiang H, Pearson AT, Snyder LA, Korman AJ, Tomlins SA, Yegnasubramanian S, Drake CG
The Journal of clinical investigation 2020 Mar 2;130(3):1405-1416
The Journal of clinical investigation 2020 Mar 2;130(3):1405-1416
Phenotypic Characterization of SLe(x)+ and CLA+ CD4+ T Cells.
Kuri-Cervantes L, Pampena MB, Betts MR
STAR protocols 2020 Dec 18;1(3):100154
STAR protocols 2020 Dec 18;1(3):100154
Sialyl-Lewis(X) Glycoantigen Is Enriched on Cells with Persistent HIV Transcription during Therapy.
Colomb F, Giron LB, Kuri-Cervantes L, Adeniji OS, Ma T, Dweep H, Battivelli E, Verdin E, Palmer CS, Tateno H, Kossenkov AV, Roan NR, Betts MR, Abdel-Mohsen M
Cell reports 2020 Aug 4;32(5):107991
Cell reports 2020 Aug 4;32(5):107991
Regulatory T cell features in chronic granulomatous disease.
van de Geer A, Cuadrado E, Slot MC, van Bruggen R, Amsen D, Kuijpers TW
Clinical and experimental immunology 2019 Aug;197(2):222-229
Clinical and experimental immunology 2019 Aug;197(2):222-229
Unleashing Type-2 Dendritic Cells to Drive Protective Antitumor CD4(+) T Cell Immunity.
Binnewies M, Mujal AM, Pollack JL, Combes AJ, Hardison EA, Barry KC, Tsui J, Ruhland MK, Kersten K, Abushawish MA, Spasic M, Giurintano JP, Chan V, Daud AI, Ha P, Ye CJ, Roberts EW, Krummel MF
Cell 2019 Apr 18;177(3):556-571.e16
Cell 2019 Apr 18;177(3):556-571.e16
Proteomic Analyses of Human Regulatory T Cells Reveal Adaptations in Signaling Pathways that Protect Cellular Identity.
Cuadrado E, van den Biggelaar M, de Kivit S, Chen YY, Slot M, Doubal I, Meijer A, van Lier RAW, Borst J, Amsen D
Immunity 2018 May 15;48(5):1046-1059.e6
Immunity 2018 May 15;48(5):1046-1059.e6
A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments.
Barry KC, Hsu J, Broz ML, Cueto FJ, Binnewies M, Combes AJ, Nelson AE, Loo K, Kumar R, Rosenblum MD, Alvarado MD, Wolf DM, Bogunovic D, Bhardwaj N, Daud AI, Ha PK, Ryan WR, Pollack JL, Samad B, Asthana S, Chan V, Krummel MF
Nature medicine 2018 Aug;24(8):1178-1191
Nature medicine 2018 Aug;24(8):1178-1191
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Kishore M, Cheung KCP, Fu H, Bonacina F, Wang G, Coe D, Ward EJ, Colamatteo A, Jangani M, Baragetti A, Matarese G, Smith DM, Haas R, Mauro C, Wraith DC, Okkenhaug K, Catapano AL, De Rosa V, Norata GD, Marelli-Berg FM
Immunity 2017 Nov 21;47(5):875-889.e10
Immunity 2017 Nov 21;47(5):875-889.e10
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Zanin-Zhorov A, Weiss JM, Trzeciak A, Chen W, Zhang J, Nyuydzefe MS, Arencibia C, Polimera S, Schueller O, Fuentes-Duculan J, Bonifacio KM, Kunjravia N, Cueto I, Soung J, Fleischmann RM, Kivitz A, Lebwohl M, Nunez M, Woodson J, Smith SL, West RF, Berger M, Krueger JG, Ryan JL, Waksal SD
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Terranova-Barberio M, Thomas S, Ali N, Pawlowska N, Park J, Krings G, Rosenblum MD, Budillon A, Munster PN
Oncotarget 2017 Dec 26;8(69):114156-114172
Oncotarget 2017 Dec 26;8(69):114156-114172
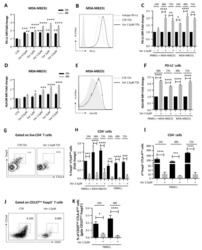
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
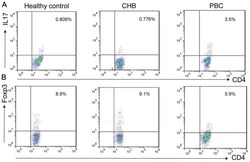
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Liang DY, Hou YQ, Luo LJ, Ao L
International journal of molecular medicine 2016 Jul;38(1):131-8
International journal of molecular medicine 2016 Jul;38(1):131-8
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
MacDonald KG, Hoeppli RE, Huang Q, Gillies J, Luciani DS, Orban PC, Broady R, Levings MK
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Kovacsovics-Bankowski M, Kelley TW, Efimova O, Kim SJ, Wilson A, Swierczek S, Prchal J
Experimental hematology & oncology 2015;5:28
Experimental hematology & oncology 2015;5:28
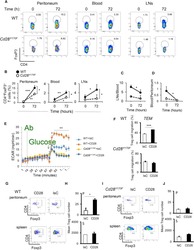
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
Ye SB, Li ZL, Luo DH, Huang BJ, Chen YS, Zhang XS, Cui J, Zeng YX, Li J
Oncotarget 2014 Jul 30;5(14):5439-52
Oncotarget 2014 Jul 30;5(14):5439-52
Effects of conventional therapeutic interventions on the number and function of regulatory T cells.
Roselli M, Cereda V, di Bari MG, Formica V, Spila A, Jochems C, Farsaci B, Donahue R, Gulley JL, Schlom J, Guadagni F
Oncoimmunology 2013 Oct 1;2(10):e27025
Oncoimmunology 2013 Oct 1;2(10):e27025
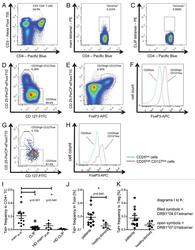
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
Schmidt HH, Ge Y, Hartmann FJ, Conrad H, Klug F, Nittel S, Bernhard H, Domschke C, Schuetz F, Sohn C, Beckhove P
Oncoimmunology 2013 Jun 1;2(6):e24962
Oncoimmunology 2013 Jun 1;2(6):e24962
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Green AM, Mattila JT, Bigbee CL, Bongers KS, Lin PL, Flynn JL
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Ahmadzadeh M, Rosenberg SA
Blood 2006 Mar 15;107(6):2409-14
Blood 2006 Mar 15;107(6):2409-14
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Roncador G, Brown PJ, Maestre L, Hue S, Martínez-Torrecuadrada JL, Ling KL, Pratap S, Toms C, Fox BC, Cerundolo V, Powrie F, Banham AH
European journal of immunology 2005 Jun;35(6):1681-91
European journal of immunology 2005 Jun;35(6):1681-91
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
Alvaro T, Lejeune M, Salvadó MT, Bosch R, García JF, Jaén J, Banham AH, Roncador G, Montalbán C, Piris MA
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
Roncador G, Garcia JF, Garcia JF, Maestre L, Lucas E, Menarguez J, Ohshima K, Nakamura S, Banham AH, Piris MA
Leukemia 2005 Dec;19(12):2247-53
Leukemia 2005 Dec;19(12):2247-53
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Wolf D, Wolf AM, Rumpold H, Fiegl H, Zeimet AG, Muller-Holzner E, Deibl M, Gastl G, Gunsilius E, Marth C
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
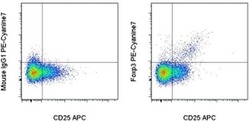
- Experimental details
- Intracellular staining of normal human peripheral blood cells with Anti-Human CD25 APC (Product # 17-0259-42) and Mouse IgG1 K Isotype Control PE-Cyanine7 (Product # 25-4714-80) (left) or Anti-Human Foxp3 PE-Cyanine7 (right) using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523-00) and protocol. Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
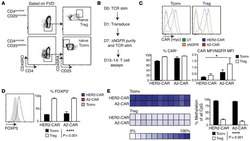
- Figure 3. Tetramer staining of conventional and regulatory T cells from breast cancer patients. ( A-G ) Peripheral blood mononuclear cells (PBMCs) of a breast cancer patient were analyzed, upon gating on living CD3 + CD4 + T cells. The patient sample was stained with tetramers presenting either mam 34-48 ( B ) or the CLIP peptide ( C ). Numbers indicate the percentage of cells in the respective gate, referring to lymphocytes ( A ) or CD3 + CD4 + T cells ( B ) and ( C ). Within CD3 + CD4 + T cells, regulatory T cells (Tregs) were identified as CD25 high CD127 low ( D ) and ( G ) and CD25 high CD127 low FOXP3 + ( E ), ( F ) and ( H ). ( G ) reports the percentage of Tregs within tetramer-positive cells in a representative patient. ( I ) Frequency of mam 34-48 - and CLIP-presenting tetramer-positive cells among CD3 + CD4 + T cells of breast cancer patients and healthy donors (HD) (p values as per Mann-Whitney U tests are indicated). ( J and K ) Frequency of mam 34-48 -specific Tcon ( J ) cells and Tregs ( K ) of breast cancer patients and HDs.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
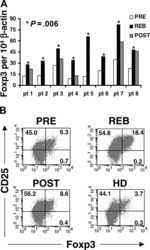
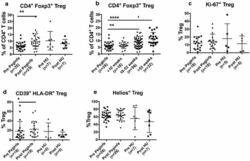
- Fig. 1 PegINFalpha increases Treg and highly suppressive Treg cells in peripheral blood of PV and ET patients. PBMC were collected from patients with PV or ET treated with PegINFalpha or HU for at least 70 days (range 70-616, median 112 for PegINFalpha and range 113-2422, median 175 for HU treated patients). Lymphocytes were analyzed by flow cytometry using surface markers, CD3, CD4, CD25, CD39, HLA-DR and intracellular markers Foxp3, Ki-67 and Helios. a represents CD4 + CD25 + Foxp3 + Treg cells. In b the frequency of Treg was analyzed at different time points after initiation of PegINFalpha treatment. c-e show frequency of Ki-67 + Treg, Helios + Treg, and CD39 + /HLA-DR + ) Treg
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
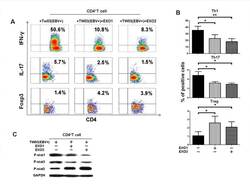
- Figure 3 NPC tumor-released exosomes inhibited Th1 and Th17 cell induction but promoted Tregs by altering STAT protein phosphorylation A. Differentiation of naive CD4 + T cells into Th1 and Th17 cells and Tregs after stimulation with tumor cells with or without EXOs. Purified naive CD4 + T cells from healthy donors were co-cultured with the irradiated NPC cell line TW03 (EBV + ) and then treated with EXOs or left or untreated in the presence of OKT3 in IL-2 medium for 5 days. INFgamma, IL-17, and Foxp3 staining following by FACS analysis was performed after stimulation with PMA/ionomycin. Representative data of five experiments are shown. B. Numerous data showing the mean percentage +- S.E.M. of positive cell subsets in three independent experiments. * means P < 0.05. C. Western blot analysis for the expression of P-STAT1, P-STAT3, and P-STAT5 proteins. Stimulation with EXOs decreased the expression of P-STAT1 and P-STAT3 but increased the expression of P-STAT5 in OKT3 and NPC cells stimulated by CD4 + T cells; the GAPDH gene was included as a control. Representative data of three experiments are shown.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
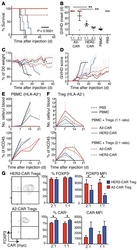
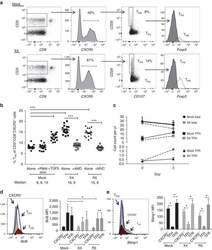
- Figure 3 HIV entry and replication promote T FR expansion. Disaggregated tonsil cells were spinoculated with X4 or R5 HIV and T FR populations were analysed by flow cytometry ( n =15). ( a ) A representative example of tonsil cell flow gating. From viable CD3 + CD8 - cells, T FR are defined as CXCR5 + and CD25 hi CD127 - . T FR cells contain Foxp3 + cells, whereas remaining T FH (CXCR5 + CD25 lo/- ) cells are Foxp3 - . ( b ) Percentages of T FR determined by gating strategies in a are shown. Experimental conditions include mock-spinoculated cells cultured with PMA (50 ng ml -1 ) and ionomycin (1 mug ml -1 ) or exogenous TGF-beta (100 ng ml -1 ) for 24 h and cells pretreated to block CXCR4 (AMD, 200 muM) and CCR5 (MVC, 2 muM). ( c ) Using flow cytometry counting beads, the number of cells per mul were determined for total (CD3 + CD8 - ), T FH (CXCR5 + CD25 lo/- ) and T FR (CXCR5 + CD25 hi CD127 - ) subsets in mock- and X4-spinoculated samples ( n =3). ( d ) Bcl-6 expression is shown in CXCR5- (grey), T FH (blue) and T FR (red) populations after mock-, X4- or R5-spinoculation ( n =5). ( e ) Blimp-1 expression was also determined as in d . The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman nonparametric tests ( b , d , e ) and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
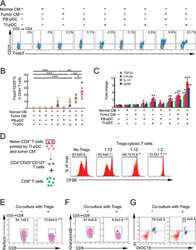
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
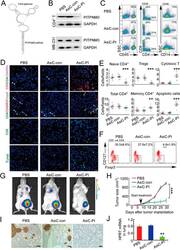
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry