53-4777-42
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [49]
- Comments [0]
- Validations
- Other assay [49]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 53-4777-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (236A/E7), Alexa Fluor™ 488, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 236A/E7 antibody reacts with human Foxp3 protein also known as FORKHEAD BOX P3, SCURFIN, and JM2. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of Foxp3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of Foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells. Intracellular staining and flow cytometric analysis of freshly isolated human peripheral blood mononuclear cells (PBMCs) with the 236A/E7 antibody using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523) and protocol reveals staining of the CD4+CD25bright population. The epitope from 236A/E7 is different from that of PCH101 (Product # 72-5776-40). This antibody has also been shown to recognize rhesus macaque, sooty mangabey and cynomolgus macaque. Applications Reported: This 236A/E7 antibody has been reported for use in intracellular staining followed by flow cytometric analysis and IHC. Applications Tested: This 236A/E7 antibody has been pre-titrated and tested using the Foxp3/Transctiption Factor Staining Buffer Set (Product # 00-5523-00) and protocol. Please see Best Protocols Section (Staining intracellular Antigens for Flow Cytometry) for staining protocol (refer to Protocol B: One-step protocol for intracellular (nuclear) proteins). This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 519 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- 236A/E7
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Analysis of Expression of Different Histone Deacetylases in Autoimmune Thyroid Disease.
Common clonal origin of conventional T cells and induced regulatory T cells in breast cancer patients.
Multiple Immunostainings with Different Epitope Retrievals-The FOLGAS Protocol.
Increased CD3(+), CD8(+), or FoxP3(+) T Lymphocyte Infiltrations Are Associated with the Pathogenesis of Colorectal Cancer but Not with the Overall Survival of Patients.
Digital Pathology Analysis Quantifies Spatial Heterogeneity of CD3, CD4, CD8, CD20, and FoxP3 Immune Markers in Triple-Negative Breast Cancer.
Distinct Tumor Microenvironments Are a Defining Feature of Strain-Specific CRISPR/Cas9-Induced MPNSTs.
Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response.
hESC-derived immune suppressive dendritic cells induce immune tolerance of parental hESC-derived allografts.
Single-cell multi-omics analysis presents the landscape of peripheral blood T-cell subsets in human chronic prostatitis/chronic pelvic pain syndrome.
Inhibition of RANK signaling in breast cancer induces an anti-tumor immune response orchestrated by CD8+ T cells.
Biomarker recommendation for PD-1/PD-L1 immunotherapy development in pediatric cancer based on digital image analysis of PD-L1 and immune cells.
Molecular profiling of primary uveal melanomas with tumor-infiltrating lymphocytes.
Highly multiplexed immunofluorescence images and single-cell data of immune markers in tonsil and lung cancer.
Hijacking antibody-induced CTLA-4 lysosomal degradation for safer and more effective cancer immunotherapy.
Spontaneous Regression of Ovarian Carcinoma After Septic Peritonitis; A Unique Case Report.
Increased circulating regulatory T cells in medicated people with schizophrenia.
Multicomponent analysis of the tumour microenvironment reveals low CD8 T cell number, low stromal caveolin-1 and high tenascin-C and their combination as significant prognostic markers in non-small cell lung cancer.
STAT3 Inhibition Combined with CpG Immunostimulation Activates Antitumor Immunity to Eradicate Genetically Distinct Castration-Resistant Prostate Cancers.
Long-lived pancreatic ductal adenocarcinoma slice cultures enable precise study of the immune microenvironment.
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
A novel method for assaying human regulatory T cell direct suppression of B cell effector function.
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Inhibition of the JAK/STAT Signaling Pathway in Regulatory T Cells Reveals a Very Dynamic Regulation of Foxp3 Expression.
The ratio of CD8 to Treg tumor-infiltrating lymphocytes is associated with response to cisplatin-based neoadjuvant chemotherapy in patients with muscle invasive urothelial carcinoma of the bladder.
The immune microenvironment of breast ductal carcinoma in situ.
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Differential tumor infiltration by T-cells characterizes intrinsic molecular subtypes in breast cancer.
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Altered expression of autoimmune regulator in infant down syndrome thymus, a possible contributor to an autoimmune phenotype.
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
IDO and regulatory T cell support are critical for cytotoxic T lymphocyte-associated Ag-4 Ig-mediated long-term solid organ allograft survival.
Expansion of CD4+CD25+FOXP3+ regulatory T cells in infants of mothers with type 1 diabetes.
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Selective expression of latency-associated peptide (LAP) and IL-1 receptor type I/II (CD121a/CD121b) on activated human FOXP3+ regulatory T cells allows for their purification from expansion cultures.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Sacristán-Gómez P, Serrano-Somavilla A, González-Amaro R, Martínez-Hernández R, Marazuela M
The Journal of clinical endocrinology and metabolism 2021 Oct 21;106(11):3213-3227
The Journal of clinical endocrinology and metabolism 2021 Oct 21;106(11):3213-3227
Common clonal origin of conventional T cells and induced regulatory T cells in breast cancer patients.
Xydia M, Rahbari R, Ruggiero E, Macaulay I, Tarabichi M, Lohmayer R, Wilkening S, Michels T, Brown D, Vanuytven S, Mastitskaya S, Laidlaw S, Grabe N, Pritsch M, Fronza R, Hexel K, Schmitt S, Müller-Steinhardt M, Halama N, Domschke C, Schmidt M, von Kalle C, Schütz F, Voet T, Beckhove P
Nature communications 2021 Feb 18;12(1):1119
Nature communications 2021 Feb 18;12(1):1119
Multiple Immunostainings with Different Epitope Retrievals-The FOLGAS Protocol.
von Schoenfeld A, Bronsert P, Poc M, Fuller A, Filby A, Kraft S, Kurowski K, Sörensen K, Huber J, Pfeiffer J, Proietti M, Stehl V, Werner M, Seidl M
International journal of molecular sciences 2021 Dec 25;23(1)
International journal of molecular sciences 2021 Dec 25;23(1)
Increased CD3(+), CD8(+), or FoxP3(+) T Lymphocyte Infiltrations Are Associated with the Pathogenesis of Colorectal Cancer but Not with the Overall Survival of Patients.
Barbosa AM, Martinho O, Nogueira R, Campos J, Lobo L, Pinto H, Longatto-Filho A, Castro AG, Martins SF, Torrado E
Biology 2021 Aug 20;10(8)
Biology 2021 Aug 20;10(8)
Digital Pathology Analysis Quantifies Spatial Heterogeneity of CD3, CD4, CD8, CD20, and FoxP3 Immune Markers in Triple-Negative Breast Cancer.
Mi H, Gong C, Sulam J, Fertig EJ, Szalay AS, Jaffee EM, Stearns V, Emens LA, Cimino-Mathews AM, Popel AS
Frontiers in physiology 2020;11:583333
Frontiers in physiology 2020;11:583333
Distinct Tumor Microenvironments Are a Defining Feature of Strain-Specific CRISPR/Cas9-Induced MPNSTs.
Scherer A, Stephens VR, McGivney GR, Gutierrez WR, Laverty EA, Knepper-Adrian V, Dodd RD
Genes 2020 May 23;11(5)
Genes 2020 May 23;11(5)
Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response.
Court AC, Le-Gatt A, Luz-Crawford P, Parra E, Aliaga-Tobar V, Bátiz LF, Contreras RA, Ortúzar MI, Kurte M, Elizondo-Vega R, Maracaja-Coutinho V, Pino-Lagos K, Figueroa FE, Khoury M
EMBO reports 2020 Feb 5;21(2):e48052
EMBO reports 2020 Feb 5;21(2):e48052
hESC-derived immune suppressive dendritic cells induce immune tolerance of parental hESC-derived allografts.
Todorova D, Zhang Y, Chen Q, Liu J, He J, Fu X, Xu Y
EBioMedicine 2020 Dec;62:103120
EBioMedicine 2020 Dec;62:103120
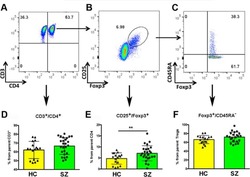
Single-cell multi-omics analysis presents the landscape of peripheral blood T-cell subsets in human chronic prostatitis/chronic pelvic pain syndrome.
Zhang M, Liu Y, Chen J, Chen L, Meng J, Yang C, Yin S, Zhang X, Zhang L, Hao Z, Chen X, Liang C
Journal of cellular and molecular medicine 2020 Dec;24(23):14099-14109
Journal of cellular and molecular medicine 2020 Dec;24(23):14099-14109
Inhibition of RANK signaling in breast cancer induces an anti-tumor immune response orchestrated by CD8+ T cells.
Gómez-Aleza C, Nguyen B, Yoldi G, Ciscar M, Barranco A, Hernández-Jiménez E, Maetens M, Salgado R, Zafeiroglou M, Pellegrini P, Venet D, Garaud S, Trinidad EM, Benítez S, Vuylsteke P, Polastro L, Wildiers H, Simon P, Lindeman G, Larsimont D, Van den Eynden G, Velghe C, Rothé F, Willard-Gallo K, Michiels S, Muñoz P, Walzer T, Planelles L, Penninger J, Azim HA Jr, Loi S, Piccart M, Sotiriou C, González-Suárez E
Nature communications 2020 Dec 10;11(1):6335
Nature communications 2020 Dec 10;11(1):6335
Biomarker recommendation for PD-1/PD-L1 immunotherapy development in pediatric cancer based on digital image analysis of PD-L1 and immune cells.
Silva MA, Triltsch N, Leis S, Kanchev I, Tan TH, Van Peel B, Van Kerckhoven M, Deschoolmeester V, Zimmermann J
The journal of pathology. Clinical research 2020 Apr;6(2):124-137
The journal of pathology. Clinical research 2020 Apr;6(2):124-137
Molecular profiling of primary uveal melanomas with tumor-infiltrating lymphocytes.
Triozzi PL, Schoenfield L, Plesec T, Saunthararajah Y, Tubbs RR, Singh AD
Oncoimmunology 2019;8(10):e947169
Oncoimmunology 2019;8(10):e947169
Highly multiplexed immunofluorescence images and single-cell data of immune markers in tonsil and lung cancer.
Rashid R, Gaglia G, Chen YA, Lin JR, Du Z, Maliga Z, Schapiro D, Yapp C, Muhlich J, Sokolov A, Sorger P, Santagata S
Scientific data 2019 Dec 17;6(1):323
Scientific data 2019 Dec 17;6(1):323
Hijacking antibody-induced CTLA-4 lysosomal degradation for safer and more effective cancer immunotherapy.
Zhang Y, Du X, Liu M, Tang F, Zhang P, Ai C, Fields JK, Sundberg EJ, Latinovic OS, Devenport M, Zheng P, Liu Y
Cell research 2019 Aug;29(8):609-627
Cell research 2019 Aug;29(8):609-627
Spontaneous Regression of Ovarian Carcinoma After Septic Peritonitis; A Unique Case Report.
Roelofsen T, Wefers C, Gorris MAJ, Textor JC, Massuger LFAG, de Vries IJM, van Altena AM
Frontiers in oncology 2018;8:562
Frontiers in oncology 2018;8:562
Increased circulating regulatory T cells in medicated people with schizophrenia.
Kelly DL, Li X, Kilday C, Feldman S, Clark S, Liu F, Buchanan RW, Tonelli LH
Psychiatry research 2018 Nov;269:517-523
Psychiatry research 2018 Nov;269:517-523
Multicomponent analysis of the tumour microenvironment reveals low CD8 T cell number, low stromal caveolin-1 and high tenascin-C and their combination as significant prognostic markers in non-small cell lung cancer.
Onion D, Isherwood M, Shridhar N, Xenophontos M, Craze ML, Day LJ, García-Márquez MA, Pineda RG, Reece-Smith AM, Saunders JH, Duffy JP, Argent RH, Grabowska AM
Oncotarget 2018 Jan 5;9(2):1760-1771
Oncotarget 2018 Jan 5;9(2):1760-1771
STAT3 Inhibition Combined with CpG Immunostimulation Activates Antitumor Immunity to Eradicate Genetically Distinct Castration-Resistant Prostate Cancers.
Moreira D, Adamus T, Zhao X, Su YL, Zhang Z, White SV, Swiderski P, Lu X, DePinho RA, Pal SK, Kortylewski M
Clinical cancer research : an official journal of the American Association for Cancer Research 2018 Dec 1;24(23):5948-5962
Clinical cancer research : an official journal of the American Association for Cancer Research 2018 Dec 1;24(23):5948-5962
Long-lived pancreatic ductal adenocarcinoma slice cultures enable precise study of the immune microenvironment.
Jiang X, Seo YD, Chang JH, Coveler A, Nigjeh EN, Pan S, Jalikis F, Yeung RS, Crispe IN, Pillarisetty VG
Oncoimmunology 2017;6(7):e1333210
Oncoimmunology 2017;6(7):e1333210
Regulatory T Cell Migration Is Dependent on Glucokinase-Mediated Glycolysis.
Kishore M, Cheung KCP, Fu H, Bonacina F, Wang G, Coe D, Ward EJ, Colamatteo A, Jangani M, Baragetti A, Matarese G, Smith DM, Haas R, Mauro C, Wraith DC, Okkenhaug K, Catapano AL, De Rosa V, Norata GD, Marelli-Berg FM
Immunity 2017 Nov 21;47(5):875-889.e10
Immunity 2017 Nov 21;47(5):875-889.e10
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Syed Khaja AS, Toor SM, El Salhat H, Faour I, Ul Haq N, Ali BR, Elkord E
Oncotarget 2017 May 16;8(20):33159-33171
Oncotarget 2017 May 16;8(20):33159-33171
Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10.
Zanin-Zhorov A, Weiss JM, Trzeciak A, Chen W, Zhang J, Nyuydzefe MS, Arencibia C, Polimera S, Schueller O, Fuentes-Duculan J, Bonifacio KM, Kunjravia N, Cueto I, Soung J, Fleischmann RM, Kivitz A, Lebwohl M, Nunez M, Woodson J, Smith SL, West RF, Berger M, Krueger JG, Ryan JL, Waksal SD
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3809-3814
A novel method for assaying human regulatory T cell direct suppression of B cell effector function.
Weingartner E, Courneya JP, Keegan A, Golding A
Journal of immunological methods 2017 Feb;441:1-7
Journal of immunological methods 2017 Feb;441:1-7
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Terranova-Barberio M, Thomas S, Ali N, Pawlowska N, Park J, Krings G, Rosenblum MD, Budillon A, Munster PN
Oncotarget 2017 Dec 26;8(69):114156-114172
Oncotarget 2017 Dec 26;8(69):114156-114172
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
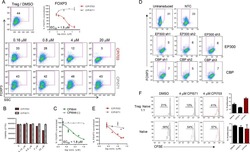
Inhibition of the JAK/STAT Signaling Pathway in Regulatory T Cells Reveals a Very Dynamic Regulation of Foxp3 Expression.
Goldstein JD, Burlion A, Zaragoza B, Sendeyo K, Polansky JK, Huehn J, Piaggio E, Salomon BL, Marodon G
PloS one 2016;11(4):e0153682
PloS one 2016;11(4):e0153682
The ratio of CD8 to Treg tumor-infiltrating lymphocytes is associated with response to cisplatin-based neoadjuvant chemotherapy in patients with muscle invasive urothelial carcinoma of the bladder.
Baras AS, Drake C, Liu JJ, Gandhi N, Kates M, Hoque MO, Meeker A, Hahn N, Taube JM, Schoenberg MP, Netto G, Bivalacqua TJ
Oncoimmunology 2016 May;5(5):e1134412
Oncoimmunology 2016 May;5(5):e1134412
The immune microenvironment of breast ductal carcinoma in situ.
Thompson E, Taube JM, Elwood H, Sharma R, Meeker A, Warzecha HN, Argani P, Cimino-Mathews A, Emens LA
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2016 Mar;29(3):249-58
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2016 Mar;29(3):249-58
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Ghosh S, Taylor A, Chin M, Huang HR, Conery AR, Mertz JA, Salmeron A, Dakle PJ, Mele D, Cote A, Jayaram H, Setser JW, Poy F, Hatzivassiliou G, DeAlmeida-Nagata D, Sandy P, Hatton C, Romero FA, Chiang E, Reimer T, Crawford T, Pardo E, Watson VG, Tsui V, Cochran AG, Zawadzke L, Harmange JC, Audia JE, Bryant BM, Cummings RT, Magnuson SR, Grogan JL, Bellon SF, Albrecht BK, Sims RJ 3rd, Lora JM
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Liang DY, Hou YQ, Luo LJ, Ao L
International journal of molecular medicine 2016 Jul;38(1):131-8
International journal of molecular medicine 2016 Jul;38(1):131-8
Differential tumor infiltration by T-cells characterizes intrinsic molecular subtypes in breast cancer.
Miyan M, Schmidt-Mende J, Kiessling R, Poschke I, de Boniface J
Journal of translational medicine 2016 Jul 29;14(1):227
Journal of translational medicine 2016 Jul 29;14(1):227
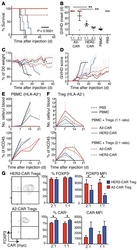
Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.
MacDonald KG, Hoeppli RE, Huang Q, Gillies J, Luciani DS, Orban PC, Broady R, Levings MK
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
The Journal of clinical investigation 2016 Apr 1;126(4):1413-24
Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2(V617F) allelic burden.
Kovacsovics-Bankowski M, Kelley TW, Efimova O, Kim SJ, Wilson A, Swierczek S, Prchal J
Experimental hematology & oncology 2015;5:28
Experimental hematology & oncology 2015;5:28
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
Altered expression of autoimmune regulator in infant down syndrome thymus, a possible contributor to an autoimmune phenotype.
Skogberg G, Lundberg V, Lindgren S, Gudmundsdottir J, Sandström K, Kämpe O, Annerén G, Gustafsson J, Sunnegårdh J, van der Post S, Telemo E, Berglund M, Ekwall O
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 1;193(5):2187-95
Journal of immunology (Baltimore, Md. : 1950) 2014 Sep 1;193(5):2187-95
Tumor-derived exosomes promote tumor progression and T-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma.
Ye SB, Li ZL, Luo DH, Huang BJ, Chen YS, Zhang XS, Cui J, Zeng YX, Li J
Oncotarget 2014 Jul 30;5(14):5439-52
Oncotarget 2014 Jul 30;5(14):5439-52
HLA Class II tetramers reveal tissue-specific regulatory T cells that suppress T-cell responses in breast carcinoma patients.
Schmidt HH, Ge Y, Hartmann FJ, Conrad H, Klug F, Nittel S, Bernhard H, Domschke C, Schuetz F, Sohn C, Beckhove P
Oncoimmunology 2013 Jun 1;2(6):e24962
Oncoimmunology 2013 Jun 1;2(6):e24962
IDO and regulatory T cell support are critical for cytotoxic T lymphocyte-associated Ag-4 Ig-mediated long-term solid organ allograft survival.
Sucher R, Fischler K, Oberhuber R, Kronberger I, Margreiter C, Ollinger R, Schneeberger S, Fuchs D, Werner ER, Watschinger K, Zelger B, Tellides G, Pilat N, Pratschke J, Margreiter R, Wekerle T, Brandacher G
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 1;188(1):37-46
Journal of immunology (Baltimore, Md. : 1950) 2012 Jan 1;188(1):37-46
Expansion of CD4+CD25+FOXP3+ regulatory T cells in infants of mothers with type 1 diabetes.
Luopajärvi K, Nieminen JK, Ilonen J, Akerblom HK, Knip M, Vaarala O
Pediatric diabetes 2012 Aug;13(5):400-7
Pediatric diabetes 2012 Aug;13(5):400-7
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Green AM, Mattila JT, Bigbee CL, Bongers KS, Lin PL, Flynn JL
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
Selective expression of latency-associated peptide (LAP) and IL-1 receptor type I/II (CD121a/CD121b) on activated human FOXP3+ regulatory T cells allows for their purification from expansion cultures.
Tran DQ, Andersson J, Hardwick D, Bebris L, Illei GG, Shevach EM
Blood 2009 May 21;113(21):5125-33
Blood 2009 May 21;113(21):5125-33
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Early resolution of acute immune activation and induction of PD-1 in SIV-infected sooty mangabeys distinguishes nonpathogenic from pathogenic infection in rhesus macaques.
Estes JD, Gordon SN, Zeng M, Chahroudi AM, Dunham RM, Staprans SI, Reilly CS, Silvestri G, Haase AT
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Journal of immunology (Baltimore, Md. : 1950) 2008 May 15;180(10):6798-807
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Kasprzycka M, Zhang Q, Witkiewicz A, Marzec M, Potoczek M, Liu X, Wang HY, Milone M, Basu S, Mauger J, Choi JK, Abrams JT, Hou JS, Rook AH, Vonderheid E, Woetmann A, Odum N, Wasik MA
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Ahmadzadeh M, Rosenberg SA
Blood 2006 Mar 15;107(6):2409-14
Blood 2006 Mar 15;107(6):2409-14
Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level.
Roncador G, Brown PJ, Maestre L, Hue S, Martínez-Torrecuadrada JL, Ling KL, Pratap S, Toms C, Fox BC, Cerundolo V, Powrie F, Banham AH
European journal of immunology 2005 Jun;35(6):1681-91
European journal of immunology 2005 Jun;35(6):1681-91
Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells.
Alvaro T, Lejeune M, Salvadó MT, Bosch R, García JF, Jaén J, Banham AH, Roncador G, Montalbán C, Piris MA
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Feb 15;11(4):1467-73
FOXP3, a selective marker for a subset of adult T-cell leukaemia/lymphoma.
Roncador G, Garcia JF, Garcia JF, Maestre L, Lucas E, Menarguez J, Ohshima K, Nakamura S, Banham AH, Piris MA
Leukemia 2005 Dec;19(12):2247-53
Leukemia 2005 Dec;19(12):2247-53
The expression of the regulatory T cell-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer.
Wolf D, Wolf AM, Rumpold H, Fiegl H, Zeimet AG, Muller-Holzner E, Deibl M, Gastl G, Gunsilius E, Marth C
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
Clinical cancer research : an official journal of the American Association for Cancer Research 2005 Dec 1;11(23):8326-31
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
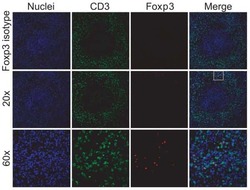
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
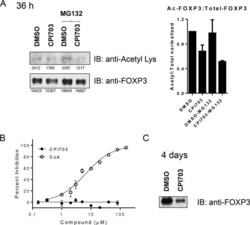
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
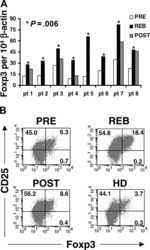
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
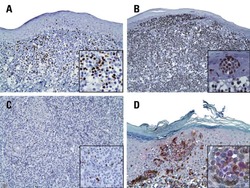
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
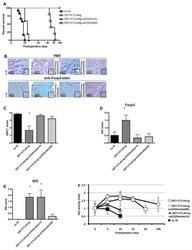
- Figure 1 T FR expansion in human lymph nodes (LNs) during HIV infection. ( a ) Representative images of immunofluorescently stained LN sections from a subject with chronic, untreated HIV infection ( n =17) and an uninfected control subject ( n =8). LNs were stained with fluorescently labelled antibodies to CD20 (white), IgD (blue), Foxp3 (green) and CD4 (red). Follicle (F) was defined as CD20 + (white line) and germinal centre (GC) was defined as CD20 + IgD - (yellow line). Images were scanned at x 60 magnification and scale bars equal 20 mum. ( b ) Foxp3 + CD4 + cells were quantified in different regions of immunofluorescently stained LN shown in a from uninfected ( n =8) and HIV-infected ( n =17) subjects using visual inspection and quantitative image analysis to determine areas. ( c ) The average areas of total (LN), follicular and GC regions per LN cross-section were determined by quantitative image analysis. ( d ) The average number of CD4 + Foxp3 + cells per LN, F and GC cross-section was calculated by multiplying the frequency of CD4 + Foxp3 + cells per mm 2 ( b ) by the average area of each region ( c ) for each subject. The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Mann-Whitney (Wilcoxon) tests to compare unpaired, nonparametric values and significance is denoted by asterisks where * P
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
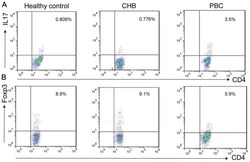
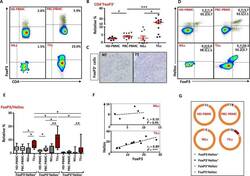
- Figure 6 FoxP3 and Helios expression in CD4 + T cells PBMC from HD and PBC patients, NILs and TILs were stained for CD3 and CD4 antibodies followed by intracellular staining for FoxP3 and Helios. Live cells were first gated using Fixable Viability Dye 660. ( A ). Representative flow cytometric plots of FoxP3 staining from one cancer patient are shown. ( B ). Scatter plot showing the differences in frequencies of FoxP3 + Tregs between different samples. ( C ). IHC staining of FoxP3 + expression in one NT and TT samples. ( D ). Flow cytometric plots of FoxP3 and Helios co-expression in CD4 + T cells from different samples and whisker plots ( E ) showing differences in various FoxP3 and Helios-expressing Treg subsets. ( F ). Non-parametric Spearman's test showing correlations between FoxP3 and Helios expressions in NILs and TILs. ( G ). Pie charts show the relative percentages of different FoxP3 and Helios Treg subsets.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
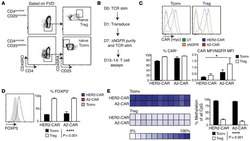
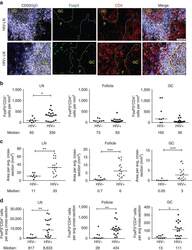
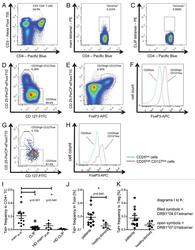
- Figure 3. Tetramer staining of conventional and regulatory T cells from breast cancer patients. ( A-G ) Peripheral blood mononuclear cells (PBMCs) of a breast cancer patient were analyzed, upon gating on living CD3 + CD4 + T cells. The patient sample was stained with tetramers presenting either mam 34-48 ( B ) or the CLIP peptide ( C ). Numbers indicate the percentage of cells in the respective gate, referring to lymphocytes ( A ) or CD3 + CD4 + T cells ( B ) and ( C ). Within CD3 + CD4 + T cells, regulatory T cells (Tregs) were identified as CD25 high CD127 low ( D ) and ( G ) and CD25 high CD127 low FOXP3 + ( E ), ( F ) and ( H ). ( G ) reports the percentage of Tregs within tetramer-positive cells in a representative patient. ( I ) Frequency of mam 34-48 - and CLIP-presenting tetramer-positive cells among CD3 + CD4 + T cells of breast cancer patients and healthy donors (HD) (p values as per Mann-Whitney U tests are indicated). ( J and K ) Frequency of mam 34-48 -specific Tcon ( J ) cells and Tregs ( K ) of breast cancer patients and HDs.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
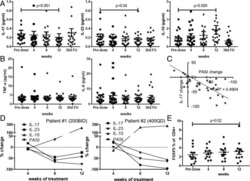
- Fig. 1 PegINFalpha increases Treg and highly suppressive Treg cells in peripheral blood of PV and ET patients. PBMC were collected from patients with PV or ET treated with PegINFalpha or HU for at least 70 days (range 70-616, median 112 for PegINFalpha and range 113-2422, median 175 for HU treated patients). Lymphocytes were analyzed by flow cytometry using surface markers, CD3, CD4, CD25, CD39, HLA-DR and intracellular markers Foxp3, Ki-67 and Helios. a represents CD4 + CD25 + Foxp3 + Treg cells. In b the frequency of Treg was analyzed at different time points after initiation of PegINFalpha treatment. c-e show frequency of Ki-67 + Treg, Helios + Treg, and CD39 + /HLA-DR + ) Treg
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
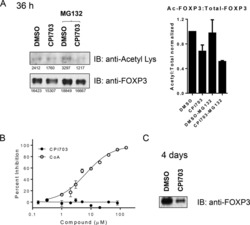
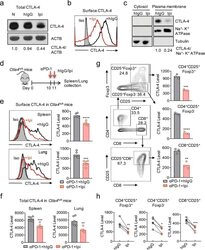
- Fig. 1 Ipilimumab downregulates cell CTLA-4. a CHO cells stably expressing human CTLA-4 were treated with either control human IgG or Ipilimumab (Ipi) for 4 h at 37 degC, then the CTLA-4 protein levels were analyzed by western blot. b Cell surface CTLA-4 on CHO cells used in ( a ) was measured by flow cytometry. To avoid the caveat associated with CTLA-4 masking by residual antibodies from the treatment phase, the cells were incubated with Ipilimumab (10 mug/mL) for 30 min at 4 degC before the staining with the non-competing anti-CTLA-4 antibody, clone BNI3. c Plasma membrane and cytosolic proteins purified from ( b ) were detected for CTLA-4 by immunoblot. Na + -K + ATPase and Tubulin were used as controls for purity of cellular fractionation. d Ten-days old Ctla4 h/h mice (body weight: 4.5-5.3 g; n = 6) were treated with anti-mouse PD-1 antibody intraperitoneally (i.p.) (100 mug/mouse). 24 h later, mice were further treated (i.p.) with 100 mug of control hIgG Fc or Ipilimumab for 4 h. e , f Cell surface ( e ) and total CTLA-4 ( f ) in Tregs isolated from spleen or lung were evaluated by flow cytometry. g , h Human PBMCs from healthy donors were stimulated with anti-CD3/anti-CD28 for 2 days and then treated with either control hIgGFc or Ipilimumab for 4 h at 37 degC. Cell surface CTLA-4 on CD4 + CD25 + FOXP3 + Tregs, CD4 + CD25 + FOXP3 - non-Tregs and CD8 + CD25 + cells were measured by flow cytometry. Again, for all the flow cytometry data of CTLA-4-positive cells treated w
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
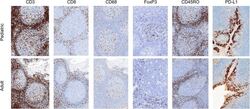
- Figure 2 Representative micrographs of CD3, CD8, CD45RO, CD68 IHC (x10) as well as FoxP3 and PD-L1 IHC (x20) in pediatric and adult tonsils, respectively, from the bridging study.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
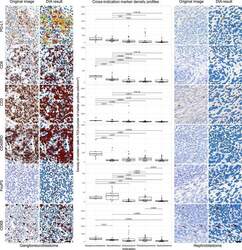
- Figure 3 DIA results from IHC. Representative IHC stained images and their corresponding DIA segmentation overlays of CD3, CD8, CD45RO, CD68, FoxP3, and PD-L1 IHC (x20) from ganglioneuroblastoma, the indication showing the highest PD-L1 expression, far left, and from nephroblastoma, the indication showing the lowest PD-L1 expression, far right. Same cases and similar regions from serial sections were selected. Classification colors indicate blue for negative nuclei, brown for CD3, CD8, CD45RO, CD68, and FoxP3 positive cells; and yellow, orange, and red for PD-L1 positive cells of increasing stain intensity. The boxplots in the middle panel show distribution profiles of investigated biomarkers across investigated indications. Indications are sorted by descending order of mean PD-L1 positive cell densities. Horizontal lines mark indication pairs where the differences in distribution are statistically significant and corresponding adjusted P values are indicated above.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
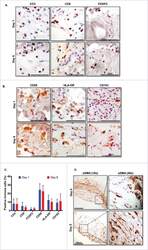
- Figure 5. Immune and stromal cells survive in slice culture. IHC for (A) T cell markers (CD3, CD8 + , and FOXP3) (N = 3) and (B) macrophage markers (CD68, HLA-DR and CD163) was performed on days 1 and 6 of PDA slice cultures (all Bar = 50 mum). (N = 3) (C) Quantitation of cells staining for each marker demonstrates stability of immune cell populations. (N = 3) Error bars represent STDEV. (D) Day 1 and 6 slices were stained with alpha smooth muscle actin (alphaSMA) (Bar = 100 mum). (N = 4) The right panels are the expanded views of the region of interest indicated by the black boxes in the left panels.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
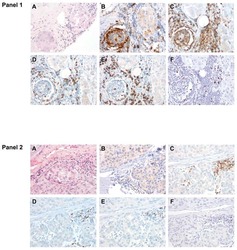
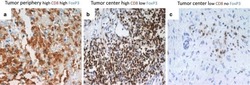
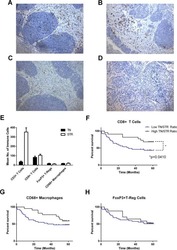
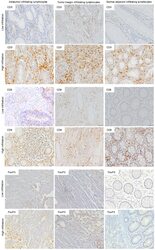
- Figure 1 Immunohistochemistry (IHC) of CD3, CD8, and FoxP3 T lymphocytes in CRC. Representative IHC showing low and high CD3 + , CD8 + , and FoxP3 + T lymphocytes densities. Magnification: 200x.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
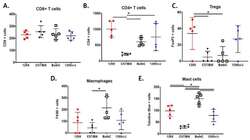
- Figure 3 The MPNST immune landscape is determined by genetic background. ( A ) Levels of CD8+ T cells in terminally-harvested MPNSTs are similar across all host strains. ( B ) Infiltration of CD4+ T cells are significantly lower in tumors from C57BL/6 mice compared to MPNSTs in mice from 129X1, BALB/c, and 129/SvJae backgrounds. ( C ) Foxp3+ Tregs are detected at higher levels in tumors from 129X1 mice compared to C57BL/6 and BALB/c mice. ( D ) MPNSTs from BALB/c mice have significantly higher levels of infiltrating F4/80+ macrophages compared to C57BL/6 mice. ( E ) Mast cell infiltration is higher in tumors from BALB/c mice compared to 129/SvJae, C57BL/6, and 129X1 mice. Mast cell levels are lowest in MPNSTs from C57BL/6 mice. 129X1 (red circles), C57BL/6 (black triangles), BABL/c (white squares), and 129/SvJae (blue triangles). Analyzed by one-way ANOVA with Tukey's multiple comparison test. A p -value of less than 0.05 is considered statistically significant and is denoted by ""*"" ( n = 5 tumors per strain).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
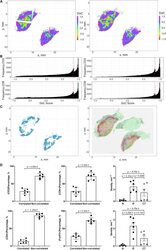
- FIGURE 9 Results of positive correlation analysis. In this study, we analyze two pairs of markers: CD3+, CD8+ and CD4+, FoxP3+. Each marker within the correlation pair is defined as a ""channel"". (A) DoC scores distributions for Cases 1A (left island) and 1B (right island). (B) Histograms of CD3+ and CD8+ DoC scores for Case 1A (left) and 1B (right). Red line is the predetermined threshold to select potential correlated cells. (C) Selected cells for both channels are mapped for clustering analysis (left) using HDBSCAN algorithm. We then define those clusters that both channels are involved as correlation hotspots (right, each cluster is represented by a colored outline). Green: normal tissue (N); Red: invasive front (IF); Yellow: central tumor (CT). (D) Statistical analysis. Top row: proportions of correlated CD3+ and CD8+ cells and density of correlated hotspots in different regions; bottom row: same metrics for CD4+ and FoxP3+.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
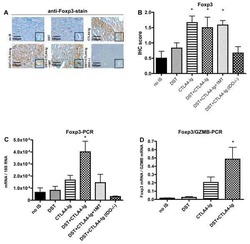
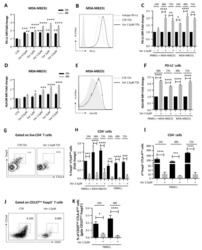
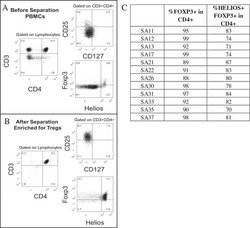
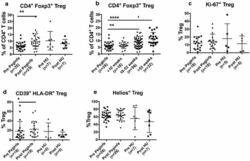
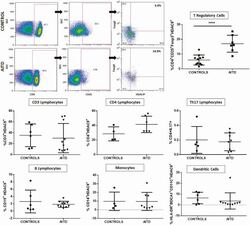
- Figure 5. Flow cytometry analysis of HDAC9 expression by different cell subsets from patients with AITD. Peripheral blood mononuclear cells from patients with AITD and healthy controls were immunostained for CD3 + (T lymphocytes), CD4 + CD25 + Foxp3 + (T regulatory cells), CD19 + (B lymphocytes), CD14 + (monocytes) and HLA-DR + CD11c + BDCA1 + (myeloid dendritic cells), and HDAC9, and the cells were analyzed by flow cytometry, as stated in ""Methods."" Analysis strategy is shown in the dot plot left panel. A significant increase in the percent of HDAC9 + Treg (CD4 + CD25 + Foxp3 + ) cells was detected in patients with AITD compared with healthy controls. (right panel). * P < 0.05; **** P < 0.001.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
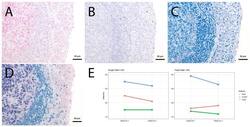
- Figure 4 Single vs. triple stainings, immunohistochemistry on tonsil sections. ( A - C ) Single stainings: ( A ) Bcl6 red after 15 min TRIS pH 9, ( B ) FoxP3 brown after 20 min TRIS pH 6.1, ( C ) PAX5 blue after 20 min TRIS pH 6.1. ( D ) Human tonsil with triple staining IHC, according to the FOLGAS protocol: Bcl6 red, FoxP3 brown, PAX5 blue; antigen retrievals equivalent to the corresponding single staining. Magnifications indicated by bar. ( E ) Sum of H-scores of the respective stainings under single and triple staining conditions, from two independent observers.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
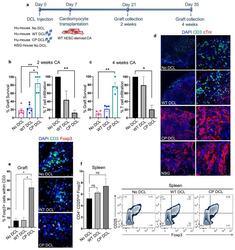
- Fig. 4 CP DCL cells inhibit the allogeneic immune rejection of the same hESC line-derived cardiomyocytes in Hu-mice. (a) Schematic description of the experiments. DCL cells derived from WT and CP hESCs were delivered via the tail vein into Hu-mice. The experiments were performed using 2 different hESCs lines, H1 and HUES3. Seven days after intravenous injection of DCLs, the same hESC line-derived cardiomyocytes were transplanted into the skeletal muscle of Hu-mice and NSG control mice. (b) Cardiomyocyte survival and T cell infiltration into the cardiomyocyte graft 2 weeks after transplantation ( n = 5, H1 n = 3, HUES3 n = 2). (c) Cardiomyocyte survival and T cell infiltration into the cardiomyocyte graft 4 weeks after transplantation ( n = 4, H1 n = 2, HUES3 n = 2). Sections were stained with anti-CD3 (shown in green) and anti-CTnI (shown in red) antibodies. For each section, the numbers of CD3 + T cells and CTnI cells were counted, 3 sections per graft were analyzed. The graft survival rate was calculated in comparison to the graft survival in NSG mice. The T cell infiltration rate was calculated in comparison to that of grafts in Hu-mice without any DCL cell injection. Data are presented as mean values +- SEM. Data from different groups were compared using one-way ANOVA test. * P
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
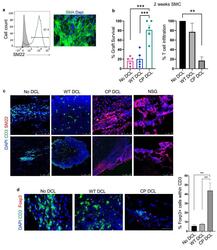
- Fig. 5 CP DCL cells inhibit the allogeneic immune rejection of the same hESC line-derived smooth muscle cells in Hu-mice. (a) Characterization of hESC-derived smooth muscle cells (SMCs). The expression of SM22 was analyzed by Flow cytometry and immunofluorescence microscopy, scale bar 50 mum. (b) SMC survival and T cell infiltration in the grafts 2 weeks after transplantation. Seven days after intravenous injection of DCLs, the same hESC line-derived SMCs were transplanted into the skeletal muscle of Hu-mice and NSG control mice ( n = 5). Data are presented as mean values +- SEM. Data from different groups were compared using one-way ANOVA test. ** p
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
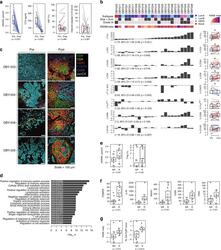
- Fig. 5 The immunomodulatory role of anti-RANKL in BC. a Change from baseline in serum levels of free-sRANKL ( n = 23, p = 2.384e-07) and CTX ( n = 17, p = 1.526e-05) (significance assessed by the two-tailed sign test), the percentage of Ki-67-positive cells ( p = 0.485) and the staining of activated caspase-3 ( p = 0.391) (H-score) ( n = 24) (significance assessed by two-tailed paired t -tests). Boxplots display median line, IQR boxes, 1.5 x IQR whiskers, and data points. b Each bar plot shows the change from baseline (Delta; post- minus pretreatment values) of the immune parameters assessed using HE (TILs) and IHC (CD3, CD20, CD8, FOXP3, proliferative TILs (TILs Ki67+ ), CD68, and CD163). Each bar represents one patient, which are ranked by their increase in stromal TIL levels. Geometric mean changes, 95% CIs, and p -values are shown below each bar plot. For each measured parameter, the corresponding boxplot is displayed on the right-hand side. Boxplots display median line, IQR boxes, 1.5 x IQR whiskers, and data points. Tumor characteristics and tumor RANK metagene expression at baseline are shown above. p; p -values derived from two-tailed paired t -tests (* p < 0.05) # . c Representative micrographs of multiplex IHC of pre- and posttreatment tumor sections from the four patients with the highest immunomodulatory response. White scale bar, 100 mum. d Top 20 significantly enriched pathways after denosumab treatment, identified by GAGE. e Comparison of baseline serum levels
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
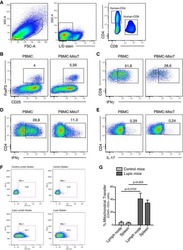
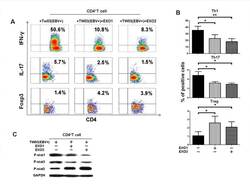
- Figure 3 NPC tumor-released exosomes inhibited Th1 and Th17 cell induction but promoted Tregs by altering STAT protein phosphorylation A. Differentiation of naive CD4 + T cells into Th1 and Th17 cells and Tregs after stimulation with tumor cells with or without EXOs. Purified naive CD4 + T cells from healthy donors were co-cultured with the irradiated NPC cell line TW03 (EBV + ) and then treated with EXOs or left or untreated in the presence of OKT3 in IL-2 medium for 5 days. INFgamma, IL-17, and Foxp3 staining following by FACS analysis was performed after stimulation with PMA/ionomycin. Representative data of five experiments are shown. B. Numerous data showing the mean percentage +- S.E.M. of positive cell subsets in three independent experiments. * means P < 0.05. C. Western blot analysis for the expression of P-STAT1, P-STAT3, and P-STAT5 proteins. Stimulation with EXOs decreased the expression of P-STAT1 and P-STAT3 but increased the expression of P-STAT5 in OKT3 and NPC cells stimulated by CD4 + T cells; the GAPDH gene was included as a control. Representative data of three experiments are shown.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
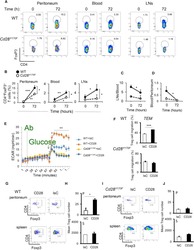
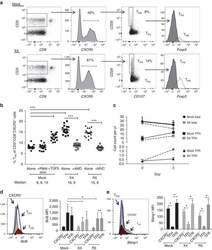
- Figure 3 HIV entry and replication promote T FR expansion. Disaggregated tonsil cells were spinoculated with X4 or R5 HIV and T FR populations were analysed by flow cytometry ( n =15). ( a ) A representative example of tonsil cell flow gating. From viable CD3 + CD8 - cells, T FR are defined as CXCR5 + and CD25 hi CD127 - . T FR cells contain Foxp3 + cells, whereas remaining T FH (CXCR5 + CD25 lo/- ) cells are Foxp3 - . ( b ) Percentages of T FR determined by gating strategies in a are shown. Experimental conditions include mock-spinoculated cells cultured with PMA (50 ng ml -1 ) and ionomycin (1 mug ml -1 ) or exogenous TGF-beta (100 ng ml -1 ) for 24 h and cells pretreated to block CXCR4 (AMD, 200 muM) and CCR5 (MVC, 2 muM). ( c ) Using flow cytometry counting beads, the number of cells per mul were determined for total (CD3 + CD8 - ), T FH (CXCR5 + CD25 lo/- ) and T FR (CXCR5 + CD25 hi CD127 - ) subsets in mock- and X4-spinoculated samples ( n =3). ( d ) Bcl-6 expression is shown in CXCR5- (grey), T FH (blue) and T FR (red) populations after mock-, X4- or R5-spinoculation ( n =5). ( e ) Blimp-1 expression was also determined as in d . The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman nonparametric tests ( b , d , e ) and significance is denoted by asterisks where * P
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
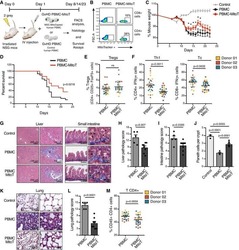
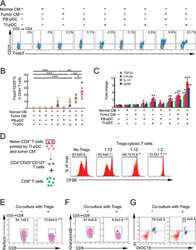
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
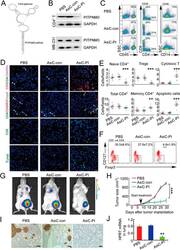
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
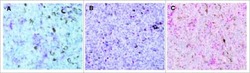
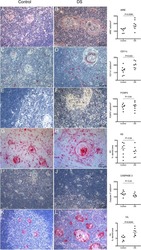
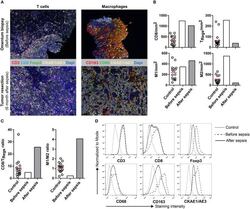
- Figure 1 Multiplex IHC on tissue before and after sepsis. (A) Images showing T cell infiltration and macrophage infiltration in tissue before (omentum biopsy) and after (tumor resection) sepsis. CKAE1/AE3 was used to visualize tumor cells. Effector T cells were characterized as CD3 + CD8 + , regulatory T cells as CD3 + Foxp3 + , M1 macrophages as CD68 + , and M2 macrophages as CD163 + . (B) CD8 T cells, T Regs , M1 and M2 macrophages per mm 2 . Immune cells were quantified in control tissue, and in tissue obtained before and after sepsis. (C) CD8 effector T cell/T Reg ratio and M1/M2 ratio before and after sepsis. (D) Average staining intensity of immune cell markers before and after sepsis. Unstained tissue was used as control. Untreated primary tumor from stage III and IV ovarian cancer patient were used as control ( n = 16). Red line indicates mean.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
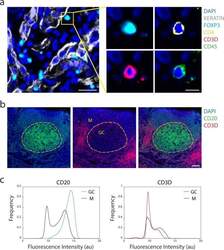
- Fig. 3 Antibody staining quality. ( a ) Immunofluorescence image from LUNG-3-PR showing epithelial tumor cells marked by Keratin (white) and a regulatory T cell marked by FOXP3 (cyan), CD4 (yellow), CD3D (red), and CD45 (green) (scale bar: 25 um; inset scale bar: 10 um). ( b ) A region of TONSIL-1 showing CD20 (green) and CD3D (red) expression. Area inside yellow dashed circle denotes germinal center (GC), and area outside denotes the mantle (M) region (scale bar: 100 um). ( c ) Probability density function of fluorescence signal intensity of every pixel in the germinal center (n = 1,446,450 pixels) and mantle (n = 4,369,358 pixels) for CD20 and CD3D within the region shown in ( b ). X-axis is fluorescence intensity (log2 au) and y-axis is frequency of pixels.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Heatmaps of cell populations from lung cancer and tonsil tissues using k-means clustering demonstrates distinct cell immune populations with expected patterns of biomarker expression. ( a ) Heatmap of the expression of Keratin, alphaSMA, and CD45 in all cells that were collected from LUNG-1-LN, LUNG-2-BR, LUNG-3-PR, and TONSIL-1 using k-means clustering. Each row is a cluster. The last column in each heat map shows the probability density function (pdf) plot showing the fit of each cell within the cluster, with the x-axis denoting the frequency of cells and y-axis denoting the Euclidean distance of the cell from the centroid of the cluster. The black vertical bars mark the immune cluster with high CD45 expression. ( b , c ) Heatmaps showing the expression of seven lymphocyte markers (CD45, CD3D, CD8A, CD4, CD20, PD1, FOXP3) from the cells within the CD45 high cluster from panel (a). ( b ) Each row represents protein marker expression data from a single cell or ( c ) each row represents a cluster. Note that fluorescence intensity values were log transformed and normalized between -1 to 1 as indicated by the color bar. ( d ) Galleries of immunofluorescence images of representative cells from each cluster in ( c ). (Scale bar: 5 um).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Spectrum of tumor-infiltrating lymphocytes characterized by MIBC PD-L1 staining. (A) Normalized histograms are shown with the TIL densities (TIL counts per 100 tumors cells) binned into the designated intervals on the x -axes and the sample proportions shown on the y -axes. In each case (overall, CD8, and FOXP3 + TILs) a significant (*Goodman-Kruskal p < 0.01) increase in the amount of TILs is observed with increased MIBC PD-L1 staining. (B) The percentage of cases with MIBC PD-L1 positivity (defined as > 0%) is shown on the x -axes and the per sample CD8/Treg ratio on the y -axes. No statistically significant (NS) association to MIBC PD-L1 expression status was observed.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
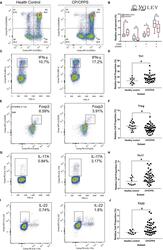
- 5 FIGURE Flow cytometry revealed central memory T cell, Th1, Th17, Th22 and Treg proportions increased in PBMCs derived from CP/CPPS patients than healthy controls. PBMCs were incubated with various fluorescein-labelled antigens for surface staining. PerCP/Cyanine5.5-conjugated CD3 and FITC-conjugated CD4 were used for the central memory T cell, Th1, Th17 and Th22 cell staining, and PE-conjugated CD25 and FITC-conjugated CD4 were incubated for staining Treg cells. After fixing and permeabilizing with cell fixation/permeabilization kit, (A) for samples staining central memory T cells were incubated with PE-conjugated CD45RA and APC-conjugated CD62L; (C) for samples staining, Th1 cells were incubated with PE-conjugated IFN-gamma; (E) for samples staining, Treg cells were incubated with eFluor 660-conjugated FoxP3; (G) for samples staining, Th17 cells were incubated with PE-conjugated IL-17A; and (I) for samples staining, Th22 cells were incubated with PE-conjugated IL-17A and APC-conjugated IL-22. The quantification data were presented in B, D, F, H and J. * P < 0.05; PBMC, peripheral blood mononuclear cell
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
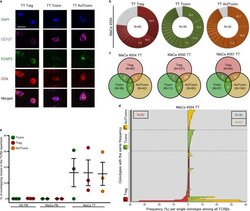
- Fig. 6 Major overlap between Treg and Tconv subsets in breast tumors. a Representative immunostaining of FFPE breast tumor tissue (TT) from n = 3 biologically independent replicates. CD4 (red), CD127 (purple), FOXP3 (green), DAPI (blue). TT Treg (CD4 + FOXP3 + CD127 - ), TT Tconv (CD4 + FOXP3 - CD127 + ), TT ActTconv (CD4 + FOXP3 + CD127 + ). Scale bar denotes 2.32 um. b Pie chart of all clonotypes (unique TCRbeta nucleotide sequences) detected among TT Treg (red), TT Tconv (green), and TT ActTconv (orange) in order of decreasing percentage (%). Gray slices correspond to the TCRbeta repertoire fraction that contains clonotypes with a frequency below 1%. Representative data from breast cancer patient with mammary gland adenocarcinoma MaCa 4554 from n = 3 biologically independent replicates. c Venn diagram of shared clonotypes (white) between TT Treg, TT Tconv, and TT ActTconv within each patient. d Frequency of common clonotypes between TT Treg, TT Tconv, and TT ActTconv in relation to the frequency distribution of all clonotypes per subset. N , total number of clonotypes recovered from TT Treg (red), TT Tconv (green), and TT ActTconv (orange). e Total frequency of Treg clonotypes overlapping with Tconv among Tconv (green), with Tconv and ActTconv among Treg (red) or with ActTconv among ActTconv (orange) either in peripheral blood (PB) or within TT in n = 3 MaCa patients and n = 3 healthy individuals (HD) (cumulative data, mean +- s.e.m.). n refers to biologically independent
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Immunohistochemistry
Immunohistochemistry Other assay
Other assay