14-4776-80
antibody from Invitrogen Antibodies
Targeting: FOXP3
AIID, DIETER, IPEX, JM2, PIDX, SCURFIN, XPID
Antibody data
- Antibody Data
- Antigen structure
- References [181]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [4]
- Other assay [73]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-4776-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FOXP3 Monoclonal Antibody (PCH101), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: eBioscience offers a panel of monoclonal antibodies to different epitopes of human Foxp3, providing useful tools for investigating the complete expression pattern of Foxp3 at the protein level, and discerning the precise subsets of Foxp3^+ cells. The PCH101 antibody reacts with the amino terminus of human foxp3 protein also known as FORKHEAD BOX P3, SCURFIN, and JM2; cross reactivity of this antibody to other proteins has not been determined. Foxp3, a 49-55 kDa protein, is a member of the forkhead/winged-helix family of transcriptional regulators, and was identified as the gene defective in 'scurfy' (sf) mice. Constitutive high expression of Foxp3 mRNA has been shown in CD4+CD25+ regulatory T cells (Treg cells), and ectopic expression of foxp3 in CD4+CD25- cells imparts a Treg phenotype in these cells. Intracellular staining of human peripheral blood mononuclear cells (PBMCs) with PCH101 antibody using the anti-human Foxp3 Staining Set and protocol reveals approximately 0.5-4% of lymphocytes staining, with the majority of staining occurring in the CD25^bright population. This is subject to donor variability. PCH101 crossreacts with rhesus, chimpanzee and cynomolgus. We recommend the use of CD4 (OKT4, Product # 11-0048-42 , or RPA-T4, Product # 11-0049-42 , depending on the species) and CD25 (BC96, Product # 17-0259-42). Applications Reported: This PCH101 antibody has been reported for use in immunoblotting (WB) and immunohistology on frozen and paraffin-embedded tissue sections. Use of the purified format is not recommended for intracellular staining for flow cytometric analysis. For additional information on IHC, please visit the Foxp3 FAQs. Applications Tested: This PCH101 antibody has been tested by immunoblotting (WB) (1-5 µg/mL) of normal human peripheral blood leukocytes. This PCH101 antibody has been tested by immunohistochemistry of formalin-fixed paraffin embedded tissue using low pH antigen retrieval and can be used at less than or equal to 10 µg/mL. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- PCH101
- Vial size
- 25 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Multiplexed single-cell analysis reveals prognostic and nonprognostic T cell types in human colorectal cancer.
Metabolic Disturbance and Th17/Treg Imbalance Are Associated With Progression of Gingivitis.
VX-765 reduces neuroinflammation after spinal cord injury in mice.
Ex vivo expansion of dysfunctional regulatory T lymphocytes restores suppressive function in Parkinson's disease.
Unmasking the immune microecology of ductal carcinoma in situ with deep learning.
SARS-CoV-2 infection paralyzes cytotoxic and metabolic functions of the immune cells.
FABP4 facilitates inflammasome activation to induce the Treg/Th17 imbalance in preeclampsia via forming a positive feedback with IL-17A.
Deep spatial profiling of human COVID-19 brains reveals neuroinflammation with distinct microanatomical microglia-T-cell interactions.
Soluble fibrinogen‑like protein 2 levels are decreased in patients with ischemic heart failure and associated with cardiac function.
Imbalance between T helper 1 and regulatory T cells plays a detrimental role in experimental Parkinson's disease in mice.
HDAC Inhibitor, CG-745, Enhances the Anti-Cancer Effect of Anti-PD-1 Immune Checkpoint Inhibitor by Modulation of the Immune Microenvironment.
Th17 reprogramming of T cells in systemic juvenile idiopathic arthritis.
Human Tumor-Infiltrating MAIT Cells Display Hallmarks of Bacterial Antigen Recognition in Colorectal Cancer.
Phase I Study of Ficlatuzumab and Cetuximab in Cetuximab-Resistant, Recurrent/Metastatic Head and Neck Cancer.
Safety and pharmacodynamics of anti-CD2 monoclonal antibody treatment in cynomolgus macaques - an experimental study.
CRID3, a blocker of apoptosis associated speck like protein containing a card, ameliorates murine spinal cord injury by improving local immune microenvironment.
Viable bacterial colonization is highly limited in the human intestine in utero.
Depleting T regulatory cells by targeting intracellular Foxp3 with a TCR mimic antibody.
HER2 signaling regulates the tumor immune microenvironment and trastuzumab efficacy.
Disruption of FOXP3-EZH2 Interaction Represents a Pathobiological Mechanism in Intestinal Inflammation.
Lymphoid Aggregates in the CNS of Progressive Multiple Sclerosis Patients Lack Regulatory T Cells.
Tbet Expression in Regulatory T Cells Is Required to Initiate Th1-Mediated Colitis.
Double negative T cells mediate Lag3-dependent antigen-specific protection in allergic asthma.
Umbilical cord blood‑derived Helios‑positive regulatory T cells promote angiogenesis in acute lymphoblastic leukemia in mice via CCL22 and the VEGFA‑VEGFR2 pathway.
Systematic testing and specificity mapping of alloantigen-specific chimeric antigen receptors in regulatory T cells.
T-Lymphocyte Subset Distribution and Activity in Patients With Glaucoma.
Niraparib activates interferon signaling and potentiates anti-PD-1 antibody efficacy in tumor models.
Long-term outcomes of a phase I study of agonist CD40 antibody and CTLA-4 blockade in patients with metastatic melanoma.
Apolipoprotein AI prevents regulatory to follicular helper T cell switching during atherosclerosis.
CD8+ T cells with high TGF‑β1 expression cause lymph node fibrosis following HIV infection.
Intradermal injection of low dose human regulatory T cells inhibits skin inflammation in a humanized mouse model.
Dendritic cell phenotype in severe asthma reflects clinical responsiveness to glucocorticoids.
Induction of porcine-specific regulatory T cells with high specificity and expression of IL-10 and TGF-β1 using baboon-derived tolerogenic dendritic cells.
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Immune rebound associates with a favorable clinical response to autologous HSCT in systemic sclerosis patients.
OX40L/OX40 axis impairs follicular and natural Treg function in human SLE.
Effect of TGF-β1 on blood CD4(+)CD25(high) regulatory T cell proliferation and Foxp3 expression during non-small cell lung cancer blood metastasis.
Analysis of immunobiologic markers in primary and recurrent glioblastoma.
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
A Unique Cellular and Molecular Microenvironment Is Present in Tertiary Lymphoid Organs of Patients with Spontaneous Prostate Cancer Regression.
Differences in Peripheral Blood Lymphocytes between Brand-Name and Generic Tacrolimus Used in Stable Liver Transplant Recipients.
Follicular Regulatory T Cells Are Highly Permissive to R5-Tropic HIV-1.
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Cutting Edge: Increased Autoimmunity Risk in Glycogen Storage Disease Type 1b Is Associated with a Reduced Engagement of Glycolysis in T Cells and an Impaired Regulatory T Cell Function.
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Serial immunomonitoring of cancer patients receiving combined antagonistic anti-CD40 and chemotherapy reveals consistent and cyclical modulation of T cell and dendritic cell parameters.
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Therapeutic immune monitoring of CD4(+)CD25(+) T cells in chronic myeloid leukemia patients treated with tyrosine kinase inhibitors.
Alteration of Th17 and Foxp3(+) regulatory T cells in patients with unexplained recurrent spontaneous abortion before and after the therapy of hCG combined with immunoglobulin.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Expression of a Chimeric Antigen Receptor Specific for Donor HLA Class I Enhances the Potency of Human Regulatory T Cells in Preventing Human Skin Transplant Rejection.
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
Regulatory T Cells and Vitamin D Status in Children with Chronic Autoimmune Thyroiditis.
Loss of ABCG1 influences regulatory T cell differentiation and atherosclerosis.
Equilibrium of Treg/Th17 cells of peripheral blood in syphilitic patients with sero-resistance.
Activation of myeloid dendritic cells, effector cells and regulatory T cells in lichen planus.
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
The prognostic effects of tumor infiltrating regulatory T cells and myeloid derived suppressor cells assessed by multicolor flow cytometry in gastric cancer patients.
Human Head and Neck Squamous Cell Carcinoma-Associated Semaphorin 4D Induces Expansion of Myeloid-Derived Suppressor Cells.
Tissue distribution and clonal diversity of the T and B cell repertoire in type 1 diabetes.
Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche.
Development of Type 2, But Not Type 1, Leprosy Reactions is Associated with a Severe Reduction of Circulating and In situ Regulatory T-Cells.
PD-1 marks dysfunctional regulatory T cells in malignant gliomas.
CD4(+)CD25(High) Treg cells in HIV/HTLV co-infected patients with neuropathy: high expression of Alpha4 integrin and lower expression of Foxp3 transcription factor.
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
Expression of TIM-3, Human β-defensin-2, and FOXP3 and Correlation with Disease Activity in Pediatric Crohn's Disease with Infliximab Therapy.
Tadalafil reduces myeloid-derived suppressor cells and regulatory T cells and promotes tumor immunity in patients with head and neck squamous cell carcinoma.
Role of dendritic cells in the pathogenesis of Whipple's disease.
Repeated Injections of IL-2 Break Renal Allograft Tolerance Induced via Mixed Hematopoietic Chimerism in Monkeys.
Impaired Th17 polarization of phenotypically naive CD4(+) T-cells during chronic HIV-1 infection and potential restoration with early ART.
The effects of CAMPATH-1H on cell viability do not correlate to the CD52 density on the cell surface.
An increased abundance of tumor-infiltrating regulatory T cells is correlated with the progression and prognosis of pancreatic ductal adenocarcinoma.
Immune responses induced by T-cell vaccination in patients with rheumatoid arthritis.
Isolation of human antigen-specific regulatory T cells with high suppressive function.
A case of conventional treatment failure in visceral leishmaniasis: leukocyte distribution and cytokine expression in splenic compartments.
Tumor-infiltrating immune cell profiles and their change after neoadjuvant chemotherapy predict response and prognosis of breast cancer.
Immune correlates of HIV exposure without infection in foreskins of men from Rakai, Uganda.
In vivo induction of cutaneous inflammation results in the accumulation of extracellular trap-forming neutrophils expressing RORγt and IL-17.
CD20+ B cell depletion alters T cell homing.
Memory regulatory T cells reside in human skin.
Response to BRAF inhibition in melanoma is enhanced when combined with immune checkpoint blockade.
Hemin controls T cell polarization in sickle cell alloimmunization.
Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations.
Superiority of rapamycin over tacrolimus in preserving nonhuman primate Treg half-life and phenotype after adoptive transfer.
Selective targeting of Toll-like receptors and OX40 inhibit regulatory T-cell function in follicular lymphoma.
HTLV-1 induces a Th1-like state in CD4+CCR4+ T cells.
Inhibition of CD4+CD25+ regulatory T cell function and conversion into Th1-like effectors by a Toll-like receptor-activated dendritic cell vaccine.
Immunologic targeting of FOXP3 in inflammatory breast cancer cells.
Human T cells upregulate CD69 after coculture with xenogeneic genetically-modified pig mesenchymal stromal cells.
Human secondary lymphoid organs typically contain polyclonally-activated proliferating regulatory T cells.
Human regulatory T cells do not suppress the antitumor immunity in the bone marrow: a role for bone marrow stromal cells in neutralizing regulatory T cells.
Reduced immunosuppressive properties of axitinib in comparison with other tyrosine kinase inhibitors.
Tumor-specific T cells in human Merkel cell carcinomas: a possible role for Tregs and T-cell exhaustion in reducing T-cell responses.
Short communication: HIV+ viremic slow progressors maintain low regulatory T cell numbers in rectal mucosa but exhibit high T cell activation.
High PD-1 expression and suppressed cytokine signaling distinguish T cells infiltrating follicular lymphoma tumors from peripheral T cells.
T regulatory cells and related immunoregulatory factors in polymorphic light eruption following ultraviolet A1 challenge.
Human Treg responses allow sustained recombinant adeno-associated virus-mediated transgene expression.
Enhanced production of IL-17A in patients with severe asthma is inhibited by 1α,25-dihydroxyvitamin D3 in a glucocorticoid-independent fashion.
Low-dose interleukin-2 therapy restores regulatory T cell homeostasis in patients with chronic graft-versus-host disease.
In vitro HIV infection impairs the capacity of myeloid dendritic cells to induce regulatory T cells.
Novel serial positive enrichment technology enables clinical multiparameter cell sorting.
Impaired function of regulatory T cells in cord blood of children of allergic mothers.
Detecting T-cell reactivity to whole cell vaccines: Proof of concept analysis of T-cell response to K562 cell antigens in CML patients.
Increased expression of regulatory T cells and down-regulatory molecules in lepromatous leprosy.
Expression of Forkhead box P3 in tumour cells causes immunoregulatory function of signet ring cell carcinoma of the stomach.
A gynecologic oncology group phase II trial of two p53 peptide vaccine approaches: subcutaneous injection and intravenous pulsed dendritic cells in high recurrence risk ovarian cancer patients.
Induction of endometriosis alters the peripheral and endometrial regulatory T cell population in the non-human primate.
Regulatory T cells exhibit decreased proliferation but enhanced suppression after pulsing with sirolimus.
The vaccine-site microenvironment induced by injection of incomplete Freund's adjuvant, with or without melanoma peptides.
PI16 is expressed by a subset of human memory Treg with enhanced migration to CCL17 and CCL20.
Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients.
A converse 4-1BB and CD40 ligand expression pattern delineates activated regulatory T cells (Treg) and conventional T cells enabling direct isolation of alloantigen-reactive natural Foxp3+ Treg.
Suppression of tumour-specific CD4⁺ T cells by regulatory T cells is associated with progression of human colorectal cancer.
OMIP-006: phenotypic subset analysis of human T regulatory cells via polychromatic flow cytometry.
CD4-like immunological function by CD4- T cells in multiple natural hosts of simian immunodeficiency virus.
Decreased AIRE expression and global thymic hypofunction in Down syndrome.
Upregulated expression of indoleamine 2, 3-dioxygenase in CHO cells induces apoptosis of competent T cells and increases proportion of Treg cells.
HLA-G level on monocytoid dendritic cells correlates with regulatory T-cell Foxp3 expression in liver transplant tolerance.
Engagement of TLR2 reverses the suppressor function of conjunctiva CD4+CD25+ regulatory T cells and promotes herpes simplex virus epitope-specific CD4+CD25- effector T cell responses.
Clinical implications and characteristics of factor forkhead box protein 3 in gastric cancer.
High-scatter T cells: a reliable biomarker for malignant T cells in cutaneous T-cell lymphoma.
Immunovirological analyses of chronically simian immunodeficiency virus SIVmnd-1- and SIVmnd-2-infected mandrills (Mandrillus sphinx).
Significant mobilization of both conventional and regulatory T cells with AMD3100.
T cell subpopulations in lymph nodes may not be predictive of patient outcome in colorectal cancer.
Rapid temporal control of Foxp3 protein degradation by sirtuin-1.
An MHC-defined primate model reveals significant rejection of bone marrow after mixed chimerism induction despite full MHC matching.
Effects of pegylated G-CSF on immune cell number and function in patients with gynecological malignancies.
CCR6 is expressed on an IL-10-producing, autoreactive memory T cell population with context-dependent regulatory function.
Glioblastoma cancer-initiating cells inhibit T-cell proliferation and effector responses by the signal transducers and activators of transcription 3 pathway.
CD40 signalling induces IL-10-producing, tolerogenic dendritic cells.
Regulation of Treg functionality by acetylation-mediated Foxp3 protein stabilization.
Safety and T cell modulating effects of high dose vitamin D3 supplementation in multiple sclerosis.
Dynamic changes in cellular infiltrates with repeated cutaneous vaccination: a histologic and immunophenotypic analysis.
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
CD30 discriminates heat shock protein 60-induced FOXP3+ CD4+ T cells with a regulatory phenotype.
Cutting Edge: Responder T cells regulate human DR+ effector regulatory T cell activity via granzyme B.
Increased ectonucleotidase expression and activity in regulatory T cells of patients with head and neck cancer.
Therapeutic effect of CD137 immunomodulation in lymphoma and its enhancement by Treg depletion.
Foxp3 regulates megakaryopoiesis and platelet function.
HIV-1 binding to CD4 on CD4+CD25+ regulatory T cells enhances their suppressive function and induces them to home to, and accumulate in, peripheral and mucosal lymphoid tissues: an additional mechanism of immunosuppression.
Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells.
Selective reduction of graft-versus-host disease-mediating human T cells by ex vivo treatment with soluble Fas ligand.
CD49d provides access to "untouched" human Foxp3+ Treg free of contaminating effector cells.
Quantitative DNA methylation analysis of FOXP3 as a new method for counting regulatory T cells in peripheral blood and solid tissue.
The importance of Foxp3 antibody and fixation/permeabilization buffer combinations in identifying CD4+CD25+Foxp3+ regulatory T cells.
Human peripheral gammadelta T cells possess regulatory potential.
Abnormally high levels of virus-infected IFN-gamma+ CCR4+ CD4+ CD25+ T cells in a retrovirus-associated neuroinflammatory disorder.
FOXP3 expression and overall survival in breast cancer.
Human spleen contains different subsets of dendritic cells and regulatory T lymphocytes.
Phenotypic characterisation of T-lymphocytes in COPD: abnormal CD4+CD25+ regulatory T-lymphocyte response to tobacco smoking.
Fusions of dendritic cells with breast carcinoma stimulate the expansion of regulatory T cells while concomitant exposure to IL-12, CpG oligodeoxynucleotides, and anti-CD3/CD28 promotes the expansion of activated tumor reactive cells.
Fusions of dendritic cells with breast carcinoma stimulate the expansion of regulatory T cells while concomitant exposure to IL-12, CpG oligodeoxynucleotides, and anti-CD3/CD28 promotes the expansion of activated tumor reactive cells.
High density of FOXP3-positive T cells infiltrating colorectal cancers with microsatellite instability.
FOXP3 expression accurately defines the population of intratumoral regulatory T cells that selectively accumulate in metastatic melanoma lesions.
Epigenetic inheritance of DNA methylation limits activation-induced expression of FOXP3 in conventional human CD25-CD4+ T cells.
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Foxp3 expression in human cancer cells.
The regulatory T cell-associated transcription factor FoxP3 is expressed by tumor cells.
Administration of a CD25-directed immunotoxin, LMB-2, to patients with metastatic melanoma induces a selective partial reduction in regulatory T cells in vivo.
Increased frequencies of CD4+ CD25(high) regulatory T cells in acute dengue infection.
IL-15 and dermal fibroblasts induce proliferation of natural regulatory T cells isolated from human skin.
Association of CD4+CD25+Foxp3+ regulatory T cells with chronic activity and viral clearance in patients with hepatitis B.
Wiskott-Aldrich syndrome protein is required for regulatory T cell homeostasis.
Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression.
FOXP3 regulates TLR10 expression in human T regulatory cells.
FOXP3 regulates TLR10 expression in human T regulatory cells.
Mucosal but not peripheral FOXP3+ regulatory T cells are highly increased in untreated HIV infection and normalize after suppressive HAART.
Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma.
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Foxp3+CD4+CD25+ T cells control virus-specific memory T cells in chimpanzees that recovered from hepatitis C.
Depletion of alloreactive T cells via CD69: implications on antiviral, antileukemic and immunoregulatory T lymphocytes.
In situ analysis of FOXP3+ regulatory T cells in human colorectal cancer.
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Human CD4+ T cells express TLR5 and its ligand flagellin enhances the suppressive capacity and expression of FOXP3 in CD4+CD25+ T regulatory cells.
CD25+CD4+ T cells in human cord blood: an immunoregulatory subset with naive phenotype and specific expression of forkhead box p3 (Foxp3) gene.
Alloantigen specific CD8+CD28- FOXP3+ T suppressor cells induce ILT3+ ILT4+ tolerogenic endothelial cells, inhibiting alloreactivity.
The origin of FOXP3-expressing CD4+ regulatory T cells: thymus or periphery.
Control of regulatory T cell development by the transcription factor Foxp3.
Masuda K, Kornberg A, Miller J, Lin S, Suek N, Botella T, Secener KA, Bacarella AM, Cheng L, Ingham M, Rosario V, Al-Mazrou AM, Lee-Kong SA, Kiran RP, Stoeckius M, Smibert P, Del Portillo A, Oberstein PE, Sims PA, Yan KS, Han A
JCI insight 2022 Apr 8;7(7)
JCI insight 2022 Apr 8;7(7)
Metabolic Disturbance and Th17/Treg Imbalance Are Associated With Progression of Gingivitis.
Wang W, Wang X, Lu S, Lv H, Zhao T, Xie G, Du Y, Fan Y, Xu L
Frontiers in immunology 2021;12:670178
Frontiers in immunology 2021;12:670178
VX-765 reduces neuroinflammation after spinal cord injury in mice.
Chen J, Chen YQ, Shi YJ, Ding SQ, Shen L, Wang R, Wang QY, Zha C, Ding H, Hu JG, Lü HZ
Neural regeneration research 2021 Sep;16(9):1836-1847
Neural regeneration research 2021 Sep;16(9):1836-1847
Ex vivo expansion of dysfunctional regulatory T lymphocytes restores suppressive function in Parkinson's disease.
Thome AD, Atassi F, Wang J, Faridar A, Zhao W, Thonhoff JR, Beers DR, Lai EC, Appel SH
NPJ Parkinson's disease 2021 May 13;7(1):41
NPJ Parkinson's disease 2021 May 13;7(1):41
Unmasking the immune microecology of ductal carcinoma in situ with deep learning.
Narayanan PL, Raza SEA, Hall AH, Marks JR, King L, West RB, Hernandez L, Guppy N, Dowsett M, Gusterson B, Maley C, Hwang ES, Yuan Y
NPJ breast cancer 2021 Mar 1;7(1):19
NPJ breast cancer 2021 Mar 1;7(1):19
SARS-CoV-2 infection paralyzes cytotoxic and metabolic functions of the immune cells.
Singh Y, Trautwein C, Fendel R, Krickeberg N, Berezhnoy G, Bissinger R, Ossowski S, Salker MS, Casadei N, Riess O, Deutsche COVID-19 OMICS Initiate (DeCOI)
Heliyon 2021 Jun;7(6):e07147
Heliyon 2021 Jun;7(6):e07147
FABP4 facilitates inflammasome activation to induce the Treg/Th17 imbalance in preeclampsia via forming a positive feedback with IL-17A.
Chang GP, Yang XL, Liu W, Lin S, Yang SL, Zhao MY
Molecular therapy. Nucleic acids 2021 Jun 4;24:743-754
Molecular therapy. Nucleic acids 2021 Jun 4;24:743-754
Deep spatial profiling of human COVID-19 brains reveals neuroinflammation with distinct microanatomical microglia-T-cell interactions.
Schwabenland M, Salié H, Tanevski J, Killmer S, Lago MS, Schlaak AE, Mayer L, Matschke J, Püschel K, Fitzek A, Ondruschka B, Mei HE, Boettler T, Neumann-Haefelin C, Hofmann M, Breithaupt A, Genc N, Stadelmann C, Saez-Rodriguez J, Bronsert P, Knobeloch KP, Blank T, Thimme R, Glatzel M, Prinz M, Bengsch B
Immunity 2021 Jul 13;54(7):1594-1610.e11
Immunity 2021 Jul 13;54(7):1594-1610.e11
Soluble fibrinogen‑like protein 2 levels are decreased in patients with ischemic heart failure and associated with cardiac function.
You Y, Huang S, Liu H, Fan C, Liu K, Wang Z
Molecular medicine reports 2021 Aug;24(2)
Molecular medicine reports 2021 Aug;24(2)
Imbalance between T helper 1 and regulatory T cells plays a detrimental role in experimental Parkinson's disease in mice.
Li W, Luo Y, Xu H, Ma Q, Yao Q
The Journal of international medical research 2021 Apr;49(4):300060521998471
The Journal of international medical research 2021 Apr;49(4):300060521998471
HDAC Inhibitor, CG-745, Enhances the Anti-Cancer Effect of Anti-PD-1 Immune Checkpoint Inhibitor by Modulation of the Immune Microenvironment.
Kim YD, Park SM, Ha HC, Lee AR, Won H, Cha H, Cho S, Cho JM
Journal of Cancer 2020;11(14):4059-4072
Journal of Cancer 2020;11(14):4059-4072
Th17 reprogramming of T cells in systemic juvenile idiopathic arthritis.
Henderson LA, Hoyt KJ, Lee PY, Rao DA, Jonsson AH, Nguyen JP, Rutherford K, Julé AM, Charbonnier LM, Case S, Chang MH, Cohen EM, Dedeoglu F, Fuhlbrigge RC, Halyabar O, Hazen MM, Janssen E, Kim S, Lo J, Lo MS, Meidan E, Son MBF, Sundel RP, Stoll ML, Nusbaum C, Lederer JA, Chatila TA, Nigrovic PA
JCI insight 2020 Mar 26;5(6)
JCI insight 2020 Mar 26;5(6)
Human Tumor-Infiltrating MAIT Cells Display Hallmarks of Bacterial Antigen Recognition in Colorectal Cancer.
Li S, Simoni Y, Becht E, Loh CY, Li N, Lachance D, Koo SL, Lim TP, Tan EKW, Mathew R, Nguyen A, Golovato J, Berkson JD, Prlic M, Lee B, Minot SS, Nagarajan N, Dey N, Tan DSW, Tan IB, Newell EW
Cell reports. Medicine 2020 Jun 23;1(3):100039
Cell reports. Medicine 2020 Jun 23;1(3):100039
Phase I Study of Ficlatuzumab and Cetuximab in Cetuximab-Resistant, Recurrent/Metastatic Head and Neck Cancer.
Bauman JE, Ohr J, Gooding WE, Ferris RL, Duvvuri U, Kim S, Johnson JT, Soloff AC, Wallweber G, Winslow J, Gaither-Davis A, Grandis JR, Stabile LP
Cancers 2020 Jun 11;12(6)
Cancers 2020 Jun 11;12(6)
Safety and pharmacodynamics of anti-CD2 monoclonal antibody treatment in cynomolgus macaques - an experimental study.
Berglund E, Alonso-Guallart P, Danton M, Sellberg F, Binder C, Fröbom R, Berglund D, Llore N, Sakai H, Iuga A, Ekanayake-Alper D, Reimann KA, Sachs DH, Sykes M, Griesemer A
Transplant international : official journal of the European Society for Organ Transplantation 2020 Jan;33(1):98-107
Transplant international : official journal of the European Society for Organ Transplantation 2020 Jan;33(1):98-107
CRID3, a blocker of apoptosis associated speck like protein containing a card, ameliorates murine spinal cord injury by improving local immune microenvironment.
Chen YQ, Wang SN, Shi YJ, Chen J, Ding SQ, Tang J, Shen L, Wang R, Ding H, Hu JG, Lü HZ
Journal of neuroinflammation 2020 Aug 29;17(1):255
Journal of neuroinflammation 2020 Aug 29;17(1):255
Viable bacterial colonization is highly limited in the human intestine in utero.
Rackaityte E, Halkias J, Fukui EM, Mendoza VF, Hayzelden C, Crawford ED, Fujimura KE, Burt TD, Lynch SV
Nature medicine 2020 Apr;26(4):599-607
Nature medicine 2020 Apr;26(4):599-607
Depleting T regulatory cells by targeting intracellular Foxp3 with a TCR mimic antibody.
Dao T, Mun SS, Scott AC, Jarvis CA, Korontsvit T, Yang Z, Liu L, Klatt MG, Guerreiro M, Selvakumar A, Brea EJ, Oh C, Liu C, Scheinberg DA
Oncoimmunology 2019;8(7):1570778
Oncoimmunology 2019;8(7):1570778
HER2 signaling regulates the tumor immune microenvironment and trastuzumab efficacy.
Triulzi T, Forte L, Regondi V, Di Modica M, Ghirelli C, Carcangiu ML, Sfondrini L, Balsari A, Tagliabue E
Oncoimmunology 2019;8(1):e1512942
Oncoimmunology 2019;8(1):e1512942
Disruption of FOXP3-EZH2 Interaction Represents a Pathobiological Mechanism in Intestinal Inflammation.
Bamidele AO, Svingen PA, Sagstetter MR, Sarmento OF, Gonzalez M, Braga Neto MB, Kugathasan S, Lomberk G, Urrutia RA, Faubion WA Jr
Cellular and molecular gastroenterology and hepatology 2019;7(1):55-71
Cellular and molecular gastroenterology and hepatology 2019;7(1):55-71
Lymphoid Aggregates in the CNS of Progressive Multiple Sclerosis Patients Lack Regulatory T Cells.
Bell L, Lenhart A, Rosenwald A, Monoranu CM, Berberich-Siebelt F
Frontiers in immunology 2019;10:3090
Frontiers in immunology 2019;10:3090
Tbet Expression in Regulatory T Cells Is Required to Initiate Th1-Mediated Colitis.
Di Giovangiulio M, Rizzo A, Franzè E, Caprioli F, Facciotti F, Onali S, Favale A, Stolfi C, Fehling HJ, Monteleone G, Fantini MC
Frontiers in immunology 2019;10:2158
Frontiers in immunology 2019;10:2158
Double negative T cells mediate Lag3-dependent antigen-specific protection in allergic asthma.
Tian D, Yang L, Wang S, Zhu Y, Shi W, Zhang C, Jin H, Tian Y, Xu H, Sun G, Liu K, Zhang Z, Zhang D
Nature communications 2019 Sep 18;10(1):4246
Nature communications 2019 Sep 18;10(1):4246
Umbilical cord blood‑derived Helios‑positive regulatory T cells promote angiogenesis in acute lymphoblastic leukemia in mice via CCL22 and the VEGFA‑VEGFR2 pathway.
Li X, Li D, Shi Q, Huang X, Ju X
Molecular medicine reports 2019 May;19(5):4195-4204
Molecular medicine reports 2019 May;19(5):4195-4204
Systematic testing and specificity mapping of alloantigen-specific chimeric antigen receptors in regulatory T cells.
Dawson NA, Lamarche C, Hoeppli RE, Bergqvist P, Fung VC, McIver E, Huang Q, Gillies J, Speck M, Orban PC, Bush JW, Mojibian M, Levings MK
JCI insight 2019 Mar 21;4(6)
JCI insight 2019 Mar 21;4(6)
T-Lymphocyte Subset Distribution and Activity in Patients With Glaucoma.
Yang X, Zeng Q, Göktas E, Gopal K, Al-Aswad L, Blumberg DM, Cioffi GA, Liebmann JM, Tezel G
Investigative ophthalmology & visual science 2019 Mar 1;60(4):877-888
Investigative ophthalmology & visual science 2019 Mar 1;60(4):877-888
Niraparib activates interferon signaling and potentiates anti-PD-1 antibody efficacy in tumor models.
Wang Z, Sun K, Xiao Y, Feng B, Mikule K, Ma X, Feng N, Vellano CP, Federico L, Marszalek JR, Mills GB, Hanke J, Ramaswamy S, Wang J
Scientific reports 2019 Feb 12;9(1):1853
Scientific reports 2019 Feb 12;9(1):1853
Long-term outcomes of a phase I study of agonist CD40 antibody and CTLA-4 blockade in patients with metastatic melanoma.
Bajor DL, Mick R, Riese MJ, Huang AC, Sullivan B, Richman LP, Torigian DA, George SM, Stelekati E, Chen F, Melenhorst JJ, Lacey SF, Xu X, Wherry EJ, Gangadhar TC, Amaravadi RK, Schuchter LM, Vonderheide RH
Oncoimmunology 2018;7(10):e1468956
Oncoimmunology 2018;7(10):e1468956
Apolipoprotein AI prevents regulatory to follicular helper T cell switching during atherosclerosis.
Gaddis DE, Padgett LE, Wu R, McSkimming C, Romines V, Taylor AM, McNamara CA, Kronenberg M, Crotty S, Thomas MJ, Sorci-Thomas MG, Hedrick CC
Nature communications 2018 Mar 15;9(1):1095
Nature communications 2018 Mar 15;9(1):1095
CD8+ T cells with high TGF‑β1 expression cause lymph node fibrosis following HIV infection.
Huang L, Deng J, Xu W, Wang H, Shi L, Wu F, Wu D, Nei W, Zhao M, Mao P, Zhou X
Molecular medicine reports 2018 Jul;18(1):77-86
Molecular medicine reports 2018 Jul;18(1):77-86
Intradermal injection of low dose human regulatory T cells inhibits skin inflammation in a humanized mouse model.
Landman S, de Oliveira VL, van Erp PEJ, Fasse E, Bauland SCG, Joosten I, Koenen HJPM
Scientific reports 2018 Jul 3;8(1):10044
Scientific reports 2018 Jul 3;8(1):10044
Dendritic cell phenotype in severe asthma reflects clinical responsiveness to glucocorticoids.
Chambers ES, Nanzer AM, Pfeffer PE, Richards DF, Martineau AR, Griffiths CJ, Corrigan CJ, Hawrylowicz CM
Clinical and experimental allergy : journal of the British Society for Allergy and Clinical Immunology 2018 Jan;48(1):13-22
Clinical and experimental allergy : journal of the British Society for Allergy and Clinical Immunology 2018 Jan;48(1):13-22
Induction of porcine-specific regulatory T cells with high specificity and expression of IL-10 and TGF-β1 using baboon-derived tolerogenic dendritic cells.
Li M, Eckl J, Abicht JM, Mayr T, Reichart B, Schendel DJ, Pohla H
Xenotransplantation 2018 Jan;25(1)
Xenotransplantation 2018 Jan;25(1)
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Hsieh WC, Hsu TS, Chang YJ, Lai MZ
Nature communications 2018 Jan 31;9(1):463
Nature communications 2018 Jan 31;9(1):463
Immune rebound associates with a favorable clinical response to autologous HSCT in systemic sclerosis patients.
Arruda LCM, Malmegrim KCR, Lima-Júnior JR, Clave E, Dias JBE, Moraes DA, Douay C, Fournier I, Moins-Teisserenc H, Alberdi AJ, Covas DT, Simões BP, Lansiaux P, Toubert A, Oliveira MC
Blood advances 2018 Jan 23;2(2):126-141
Blood advances 2018 Jan 23;2(2):126-141
OX40L/OX40 axis impairs follicular and natural Treg function in human SLE.
Jacquemin C, Augusto JF, Scherlinger M, Gensous N, Forcade E, Douchet I, Levionnois E, Richez C, Lazaro E, Duffau P, Truchetet ME, Seneschal J, Couzi L, Pellegrin JL, Viallard JF, Schaeverbeke T, Pascual V, Contin-Bordes C, Blanco P
JCI insight 2018 Dec 20;3(24)
JCI insight 2018 Dec 20;3(24)
Effect of TGF-β1 on blood CD4(+)CD25(high) regulatory T cell proliferation and Foxp3 expression during non-small cell lung cancer blood metastasis.
Hu Y, Qi W, Sun L, Zhou H, Zhou B, Yang Z
Experimental and therapeutic medicine 2018 Aug;16(2):1403-1410
Experimental and therapeutic medicine 2018 Aug;16(2):1403-1410
Analysis of immunobiologic markers in primary and recurrent glioblastoma.
Rahman M, Kresak J, Yang C, Huang J, Hiser W, Kubilis P, Mitchell D
Journal of neuro-oncology 2018 Apr;137(2):249-257
Journal of neuro-oncology 2018 Apr;137(2):249-257
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
Arce Vargas F, Furness AJS, Litchfield K, Joshi K, Rosenthal R, Ghorani E, Solomon I, Lesko MH, Ruef N, Roddie C, Henry JY, Spain L, Ben Aissa A, Georgiou A, Wong YNS, Smith M, Strauss D, Hayes A, Nicol D, O'Brien T, Mårtensson L, Ljungars A, Teige I, Frendéus B, TRACERx Melanoma, TRACERx Renal, TRACERx Lung consortia, Pule M, Marafioti T, Gore M, Larkin J, Turajlic S, Swanton C, Peggs KS, Quezada SA
Cancer cell 2018 Apr 9;33(4):649-663.e4
Cancer cell 2018 Apr 9;33(4):649-663.e4
A Unique Cellular and Molecular Microenvironment Is Present in Tertiary Lymphoid Organs of Patients with Spontaneous Prostate Cancer Regression.
García-Hernández ML, Uribe-Uribe NO, Espinosa-González R, Kast WM, Khader SA, Rangel-Moreno J
Frontiers in immunology 2017;8:563
Frontiers in immunology 2017;8:563
Differences in Peripheral Blood Lymphocytes between Brand-Name and Generic Tacrolimus Used in Stable Liver Transplant Recipients.
Kim JM, Kwon CHD, Joh JW, Sinn DH, Choi GS, Park JB, Kang ES, Lee SK
Medical principles and practice : international journal of the Kuwait University, Health Science Centre 2017;26(3):221-228
Medical principles and practice : international journal of the Kuwait University, Health Science Centre 2017;26(3):221-228
Follicular Regulatory T Cells Are Highly Permissive to R5-Tropic HIV-1.
Miller SM, Miles B, Guo K, Folkvord J, Meditz AL, McCarter MD, Levy DN, MaWhinney S, Santiago ML, Connick E
Journal of virology 2017 Sep 1;91(17)
Journal of virology 2017 Sep 1;91(17)
Preferential accumulation of regulatory T cells with highly immunosuppressive characteristics in breast tumor microenvironment.
Syed Khaja AS, Toor SM, El Salhat H, Faour I, Ul Haq N, Ali BR, Elkord E
Oncotarget 2017 May 16;8(20):33159-33171
Oncotarget 2017 May 16;8(20):33159-33171
Cutting Edge: Increased Autoimmunity Risk in Glycogen Storage Disease Type 1b Is Associated with a Reduced Engagement of Glycolysis in T Cells and an Impaired Regulatory T Cell Function.
Melis D, Carbone F, Minopoli G, La Rocca C, Perna F, De Rosa V, Galgani M, Andria G, Parenti G, Matarese G
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3803-3808
Journal of immunology (Baltimore, Md. : 1950) 2017 May 15;198(10):3803-3808
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Jeffery HC, Jeffery LE, Lutz P, Corrigan M, Webb GJ, Hirschfield GM, Adams DH, Oo YH
Clinical and experimental immunology 2017 Jun;188(3):394-411
Clinical and experimental immunology 2017 Jun;188(3):394-411
Serial immunomonitoring of cancer patients receiving combined antagonistic anti-CD40 and chemotherapy reveals consistent and cyclical modulation of T cell and dendritic cell parameters.
McDonnell AM, Cook A, Robinson BWS, Lake RA, Nowak AK
BMC cancer 2017 Jun 15;17(1):417
BMC cancer 2017 Jun 15;17(1):417
HDAC inhibition potentiates immunotherapy in triple negative breast cancer.
Terranova-Barberio M, Thomas S, Ali N, Pawlowska N, Park J, Krings G, Rosenblum MD, Budillon A, Munster PN
Oncotarget 2017 Dec 26;8(69):114156-114172
Oncotarget 2017 Dec 26;8(69):114156-114172
Therapeutic immune monitoring of CD4(+)CD25(+) T cells in chronic myeloid leukemia patients treated with tyrosine kinase inhibitors.
Lu Z, Xu N, Zhou X, Gao G, Li L, Huang J, Li Y, Lu Q, He B, Pan C, Liu X
Oncology letters 2017 Aug;14(2):1363-1372
Oncology letters 2017 Aug;14(2):1363-1372
Alteration of Th17 and Foxp3(+) regulatory T cells in patients with unexplained recurrent spontaneous abortion before and after the therapy of hCG combined with immunoglobulin.
Sha J, Liu F, Zhai J, Liu X, Zhang Q, Zhang B
Experimental and therapeutic medicine 2017 Aug;14(2):1114-1118
Experimental and therapeutic medicine 2017 Aug;14(2):1114-1118
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
Expression of a Chimeric Antigen Receptor Specific for Donor HLA Class I Enhances the Potency of Human Regulatory T Cells in Preventing Human Skin Transplant Rejection.
Boardman DA, Philippeos C, Fruhwirth GO, Ibrahim MA, Hannen RF, Cooper D, Marelli-Berg FM, Watt FM, Lechler RI, Maher J, Smyth LA, Lombardi G
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2017 Apr;17(4):931-943
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2017 Apr;17(4):931-943
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
Seebauer CT, Brunner S, Glockzin G, Piso P, Ruemmele P, Schlitt HJ, Geissler EK, Fichtner-Feigl S, Kesselring R
Oncoimmunology 2016;5(12):e1242543
Oncoimmunology 2016;5(12):e1242543
Regulatory T Cells and Vitamin D Status in Children with Chronic Autoimmune Thyroiditis.
Şıklar Z, Karataş D, Doğu F, Hacıhamdioğlu B, İkincioğulları A, Berberoğlu M
Journal of clinical research in pediatric endocrinology 2016 Sep 1;8(3):276-81
Journal of clinical research in pediatric endocrinology 2016 Sep 1;8(3):276-81
Loss of ABCG1 influences regulatory T cell differentiation and atherosclerosis.
Cheng HY, Gaddis DE, Wu R, McSkimming C, Haynes LD, Taylor AM, McNamara CA, Sorci-Thomas M, Hedrick CC
The Journal of clinical investigation 2016 Sep 1;126(9):3236-46
The Journal of clinical investigation 2016 Sep 1;126(9):3236-46
Equilibrium of Treg/Th17 cells of peripheral blood in syphilitic patients with sero-resistance.
Zhao J, Ma J, Zhang X, Li Q, Yang X
Experimental and therapeutic medicine 2016 Jun;11(6):2300-2304
Experimental and therapeutic medicine 2016 Jun;11(6):2300-2304
Activation of myeloid dendritic cells, effector cells and regulatory T cells in lichen planus.
Domingues R, de Carvalho GC, Aoki V, da Silva Duarte AJ, Sato MN
Journal of translational medicine 2016 Jun 10;14(1):171
Journal of translational medicine 2016 Jun 10;14(1):171
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
Legorreta-Haquet MV, Chávez-Rueda K, Chávez-Sánchez L, Cervera-Castillo H, Zenteno-Galindo E, Barile-Fabris L, Burgos-Vargas R, Álvarez-Hernández E, Blanco-Favela F
Medicine 2016 Feb;95(5):e2384
Medicine 2016 Feb;95(5):e2384
Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells--possible implications for the ICOS/ICOS-ligand axis in disease course and resolution.
Sakthivel P, Grunewald J, Eklund A, Bruder D, Wahlström J
Clinical and experimental immunology 2016 Feb;183(2):294-306
Clinical and experimental immunology 2016 Feb;183(2):294-306
The prognostic effects of tumor infiltrating regulatory T cells and myeloid derived suppressor cells assessed by multicolor flow cytometry in gastric cancer patients.
Choi HS, Ha SY, Kim HM, Ahn SM, Kang MS, Kim KM, Choi MG, Lee JH, Sohn TS, Bae JM, Kim S, Kang ES
Oncotarget 2016 Feb 16;7(7):7940-51
Oncotarget 2016 Feb 16;7(7):7940-51
Human Head and Neck Squamous Cell Carcinoma-Associated Semaphorin 4D Induces Expansion of Myeloid-Derived Suppressor Cells.
Younis RH, Han KL, Webb TJ
Journal of immunology (Baltimore, Md. : 1950) 2016 Feb 1;196(3):1419-29
Journal of immunology (Baltimore, Md. : 1950) 2016 Feb 1;196(3):1419-29
Tissue distribution and clonal diversity of the T and B cell repertoire in type 1 diabetes.
Seay HR, Yusko E, Rothweiler SJ, Zhang L, Posgai AL, Campbell-Thompson M, Vignali M, Emerson RO, Kaddis JS, Ko D, Nakayama M, Smith MJ, Cambier JC, Pugliese A, Atkinson MA, Robins HS, Brusko TM
JCI insight 2016 Dec 8;1(20):e88242
JCI insight 2016 Dec 8;1(20):e88242
Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche.
Clever D, Roychoudhuri R, Constantinides MG, Askenase MH, Sukumar M, Klebanoff CA, Eil RL, Hickman HD, Yu Z, Pan JH, Palmer DC, Phan AT, Goulding J, Gattinoni L, Goldrath AW, Belkaid Y, Restifo NP
Cell 2016 Aug 25;166(5):1117-1131.e14
Cell 2016 Aug 25;166(5):1117-1131.e14
Development of Type 2, But Not Type 1, Leprosy Reactions is Associated with a Severe Reduction of Circulating and In situ Regulatory T-Cells.
Vieira AP, Trindade MÂ, Pagliari C, Avancini J, Sakai-Valente NY, Duarte AJ, Benard G
The American journal of tropical medicine and hygiene 2016 Apr;94(4):721-7
The American journal of tropical medicine and hygiene 2016 Apr;94(4):721-7
PD-1 marks dysfunctional regulatory T cells in malignant gliomas.
Lowther DE, Goods BA, Lucca LE, Lerner BA, Raddassi K, van Dijk D, Hernandez AL, Duan X, Gunel M, Coric V, Krishnaswamy S, Love JC, Hafler DA
JCI insight 2016 Apr 21;1(5)
JCI insight 2016 Apr 21;1(5)
CD4(+)CD25(High) Treg cells in HIV/HTLV co-infected patients with neuropathy: high expression of Alpha4 integrin and lower expression of Foxp3 transcription factor.
Matavele Chissumba R, Silva-Barbosa SD, Augusto Â, Maueia C, Mabunda N, Gudo ES Jr, Bhatt N, Jani I, Savino W
BMC immunology 2015 Sep 2;16:52
BMC immunology 2015 Sep 2;16:52
Glycolysis controls the induction of human regulatory T cells by modulating the expression of FOXP3 exon 2 splicing variants.
De Rosa V, Galgani M, Porcellini A, Colamatteo A, Santopaolo M, Zuchegna C, Romano A, De Simone S, Procaccini C, La Rocca C, Carrieri PB, Maniscalco GT, Salvetti M, Buscarinu MC, Franzese A, Mozzillo E, La Cava A, Matarese G
Nature immunology 2015 Nov;16(11):1174-84
Nature immunology 2015 Nov;16(11):1174-84
Expression of TIM-3, Human β-defensin-2, and FOXP3 and Correlation with Disease Activity in Pediatric Crohn's Disease with Infliximab Therapy.
Kim MJ, Lee WY, Choe YH
Gut and liver 2015 May 23;9(3):370-80
Gut and liver 2015 May 23;9(3):370-80
Tadalafil reduces myeloid-derived suppressor cells and regulatory T cells and promotes tumor immunity in patients with head and neck squamous cell carcinoma.
Weed DT, Vella JL, Reis IM, De la Fuente AC, Gomez C, Sargi Z, Nazarian R, Califano J, Borrello I, Serafini P
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Jan 1;21(1):39-48
Clinical cancer research : an official journal of the American Association for Cancer Research 2015 Jan 1;21(1):39-48
Role of dendritic cells in the pathogenesis of Whipple's disease.
Schinnerling K, Geelhaar-Karsch A, Allers K, Friebel J, Conrad K, Loddenkemper C, Kühl AA, Erben U, Ignatius R, Moos V, Schneider T
Infection and immunity 2015 Feb;83(2):482-91
Infection and immunity 2015 Feb;83(2):482-91
Repeated Injections of IL-2 Break Renal Allograft Tolerance Induced via Mixed Hematopoietic Chimerism in Monkeys.
Yamada Y, Nadazdin O, Boskovic S, Lee S, Zorn E, Smith RN, Colvin RB, Madsen JC, Cosimi AB, Kawai T, Benichou G
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2015 Dec;15(12):3055-66
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2015 Dec;15(12):3055-66
Impaired Th17 polarization of phenotypically naive CD4(+) T-cells during chronic HIV-1 infection and potential restoration with early ART.
DaFonseca S, Niessl J, Pouvreau S, Wacleche VS, Gosselin A, Cleret-Buhot A, Bernard N, Tremblay C, Jenabian MA, Routy JP, Ancuta P
Retrovirology 2015 Apr 30;12:38
Retrovirology 2015 Apr 30;12:38
The effects of CAMPATH-1H on cell viability do not correlate to the CD52 density on the cell surface.
Lee F, Luevano M, Veys P, Yong K, Madrigal A, Shaw BE, Saudemont A
PloS one 2014;9(7):e103254
PloS one 2014;9(7):e103254
An increased abundance of tumor-infiltrating regulatory T cells is correlated with the progression and prognosis of pancreatic ductal adenocarcinoma.
Tang Y, Xu X, Guo S, Zhang C, Tang Y, Tian Y, Ni B, Lu B, Wang H
PloS one 2014;9(3):e91551
PloS one 2014;9(3):e91551
Immune responses induced by T-cell vaccination in patients with rheumatoid arthritis.
Ivanova I, Seledtsova G, Mamaev S, Shishkov A, Seledtsov V
Human vaccines & immunotherapeutics 2014;10(5):1221-7
Human vaccines & immunotherapeutics 2014;10(5):1221-7
Isolation of human antigen-specific regulatory T cells with high suppressive function.
Noyan F, Lee YS, Zimmermann K, Hardtke-Wolenski M, Taubert R, Warnecke G, Knoefel AK, Schulde E, Olek S, Manns MP, Jaeckel E
European journal of immunology 2014 Sep;44(9):2592-602
European journal of immunology 2014 Sep;44(9):2592-602
A case of conventional treatment failure in visceral leishmaniasis: leukocyte distribution and cytokine expression in splenic compartments.
Dos-Santos WL, Pagliari C, Santos LG, Almeida VA, e Silva TL, Coutinho Jde J Jr, Souza T, Duarte MI, de Freitas LA, Costa CH
BMC infectious diseases 2014 Sep 9;14:491
BMC infectious diseases 2014 Sep 9;14:491
Tumor-infiltrating immune cell profiles and their change after neoadjuvant chemotherapy predict response and prognosis of breast cancer.
García-Martínez E, Gil GL, Benito AC, González-Billalabeitia E, Conesa MA, García García T, García-Garre E, Vicente V, Ayala de la Peña F
Breast cancer research : BCR 2014 Nov 29;16(6):488
Breast cancer research : BCR 2014 Nov 29;16(6):488
Immune correlates of HIV exposure without infection in foreskins of men from Rakai, Uganda.
Prodger JL, Hirbod T, Kigozi G, Nalugoda F, Reynolds SJ, Galiwango R, Shahabi K, Serwadda D, Wawer MJ, Gray RH, Kaul R, Rakai Genital Immunology Research Group
Mucosal immunology 2014 May;7(3):634-44
Mucosal immunology 2014 May;7(3):634-44
In vivo induction of cutaneous inflammation results in the accumulation of extracellular trap-forming neutrophils expressing RORγt and IL-17.
Keijsers RRMC, Hendriks AGM, van Erp PEJ, van Cranenbroek B, van de Kerkhof PCM, Koenen HJPM, Joosten I
The Journal of investigative dermatology 2014 May;134(5):1276-1284
The Journal of investigative dermatology 2014 May;134(5):1276-1284
CD20+ B cell depletion alters T cell homing.
Kap YS, van Driel N, Laman JD, Tak PP, 't Hart BA
Journal of immunology (Baltimore, Md. : 1950) 2014 May 1;192(9):4242-53
Journal of immunology (Baltimore, Md. : 1950) 2014 May 1;192(9):4242-53
Memory regulatory T cells reside in human skin.
Sanchez Rodriguez R, Pauli ML, Neuhaus IM, Yu SS, Arron ST, Harris HW, Yang SH, Anthony BA, Sverdrup FM, Krow-Lucal E, MacKenzie TC, Johnson DS, Meyer EH, Löhr A, Hsu A, Koo J, Liao W, Gupta R, Debbaneh MG, Butler D, Huynh M, Levin EC, Leon A, Hoffman WY, McGrath MH, Alvarado MD, Ludwig CH, Truong HA, Maurano MM, Gratz IK, Abbas AK, Rosenblum MD
The Journal of clinical investigation 2014 Mar;124(3):1027-36
The Journal of clinical investigation 2014 Mar;124(3):1027-36
Response to BRAF inhibition in melanoma is enhanced when combined with immune checkpoint blockade.
Cooper ZA, Juneja VR, Sage PT, Frederick DT, Piris A, Mitra D, Lo JA, Hodi FS, Freeman GJ, Bosenberg MW, McMahon M, Flaherty KT, Fisher DE, Sharpe AH, Wargo JA
Cancer immunology research 2014 Jul;2(7):643-54
Cancer immunology research 2014 Jul;2(7):643-54
Hemin controls T cell polarization in sickle cell alloimmunization.
Zhong H, Bao W, Friedman D, Yazdanbakhsh K
Journal of immunology (Baltimore, Md. : 1950) 2014 Jul 1;193(1):102-10
Journal of immunology (Baltimore, Md. : 1950) 2014 Jul 1;193(1):102-10
Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations.
Schubert D, Bode C, Kenefeck R, Hou TZ, Wing JB, Kennedy A, Bulashevska A, Petersen BS, Schäffer AA, Grüning BA, Unger S, Frede N, Baumann U, Witte T, Schmidt RE, Dueckers G, Niehues T, Seneviratne S, Kanariou M, Speckmann C, Ehl S, Rensing-Ehl A, Warnatz K, Rakhmanov M, Thimme R, Hasselblatt P, Emmerich F, Cathomen T, Backofen R, Fisch P, Seidl M, May A, Schmitt-Graeff A, Ikemizu S, Salzer U, Franke A, Sakaguchi S, Walker LSK, Sansom DM, Grimbacher B
Nature medicine 2014 Dec;20(12):1410-1416
Nature medicine 2014 Dec;20(12):1410-1416
Superiority of rapamycin over tacrolimus in preserving nonhuman primate Treg half-life and phenotype after adoptive transfer.
Singh K, Stempora L, Harvey RD, Kirk AD, Larsen CP, Blazar BR, Kean LS
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2014 Dec;14(12):2691-703
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2014 Dec;14(12):2691-703
Selective targeting of Toll-like receptors and OX40 inhibit regulatory T-cell function in follicular lymphoma.
Voo KS, Foglietta M, Percivalle E, Chu F, Nattamai D, Harline M, Lee ST, Bover L, Lin HY, Baladandayuthapani V, Delgado D, Luong A, Davis RE, Kwak LW, Liu YJ, Neelapu SS
International journal of cancer 2014 Dec 15;135(12):2834-46
International journal of cancer 2014 Dec 15;135(12):2834-46
HTLV-1 induces a Th1-like state in CD4+CCR4+ T cells.
Araya N, Sato T, Ando H, Tomaru U, Yoshida M, Coler-Reilly A, Yagishita N, Yamauchi J, Hasegawa A, Kannagi M, Hasegawa Y, Takahashi K, Kunitomo Y, Tanaka Y, Nakajima T, Nishioka K, Utsunomiya A, Jacobson S, Yamano Y
The Journal of clinical investigation 2014 Aug;124(8):3431-42
The Journal of clinical investigation 2014 Aug;124(8):3431-42
Inhibition of CD4+CD25+ regulatory T cell function and conversion into Th1-like effectors by a Toll-like receptor-activated dendritic cell vaccine.
Lee MK 4th, Xu S, Fitzpatrick EH, Sharma A, Graves HL, Czerniecki BJ
PloS one 2013;8(11):e74698
PloS one 2013;8(11):e74698
Immunologic targeting of FOXP3 in inflammatory breast cancer cells.
Nair S, Aldrich AJ, McDonnell E, Cheng Q, Aggarwal A, Patel P, Williams MM, Boczkowski D, Lyerly HK, Morse MA, Devi GR
PloS one 2013;8(1):e53150
PloS one 2013;8(1):e53150
Human T cells upregulate CD69 after coculture with xenogeneic genetically-modified pig mesenchymal stromal cells.
Li J, Andreyev O, Chen M, Marco M, Iwase H, Long C, Ayares D, Shen Z, Cooper DK, Ezzelarab MB
Cellular immunology 2013 Sep-Oct;285(1-2):23-30
Cellular immunology 2013 Sep-Oct;285(1-2):23-30
Human secondary lymphoid organs typically contain polyclonally-activated proliferating regulatory T cells.
Peters JH, Koenen HJ, Fasse E, Tijssen HJ, Ijzermans JN, Groenen PJ, Schaap NP, Kwekkeboom J, Joosten I
Blood 2013 Sep 26;122(13):2213-23
Blood 2013 Sep 26;122(13):2213-23
Human regulatory T cells do not suppress the antitumor immunity in the bone marrow: a role for bone marrow stromal cells in neutralizing regulatory T cells.
Guichelaar T, Emmelot ME, Rozemuller H, Martini B, Groen RW, Storm G, Lokhorst HM, Martens AC, Mutis T
Clinical cancer research : an official journal of the American Association for Cancer Research 2013 Mar 15;19(6):1467-75
Clinical cancer research : an official journal of the American Association for Cancer Research 2013 Mar 15;19(6):1467-75
Reduced immunosuppressive properties of axitinib in comparison with other tyrosine kinase inhibitors.
Stehle F, Schulz K, Fahldieck C, Kalich J, Lichtenfels R, Riemann D, Seliger B
The Journal of biological chemistry 2013 Jun 7;288(23):16334-16347
The Journal of biological chemistry 2013 Jun 7;288(23):16334-16347
Tumor-specific T cells in human Merkel cell carcinomas: a possible role for Tregs and T-cell exhaustion in reducing T-cell responses.
Dowlatshahi M, Huang V, Gehad AE, Jiang Y, Calarese A, Teague JE, Dorosario AA, Cheng J, Nghiem P, Schanbacher CF, Thakuria M, Schmults CD, Wang LC, Clark RA
The Journal of investigative dermatology 2013 Jul;133(7):1879-89
The Journal of investigative dermatology 2013 Jul;133(7):1879-89
Short communication: HIV+ viremic slow progressors maintain low regulatory T cell numbers in rectal mucosa but exhibit high T cell activation.
Shaw JM, Hunt PW, Critchfield JW, McConnell DH, Garcia JC, Pollard RB, Somsouk M, Deeks SG, Shacklett BL
AIDS research and human retroviruses 2013 Jan;29(1):172-7
AIDS research and human retroviruses 2013 Jan;29(1):172-7
High PD-1 expression and suppressed cytokine signaling distinguish T cells infiltrating follicular lymphoma tumors from peripheral T cells.
Myklebust JH, Irish JM, Brody J, Czerwinski DK, Houot R, Kohrt HE, Timmerman J, Said J, Green MR, Delabie J, Kolstad A, Alizadeh AA, Levy R
Blood 2013 Feb 21;121(8):1367-76
Blood 2013 Feb 21;121(8):1367-76
T regulatory cells and related immunoregulatory factors in polymorphic light eruption following ultraviolet A1 challenge.
Gambichler T, Terras S, Kampilafkos P, Kreuter A, Skrygan M
The British journal of dermatology 2013 Dec;169(6):1288-94
The British journal of dermatology 2013 Dec;169(6):1288-94
Human Treg responses allow sustained recombinant adeno-associated virus-mediated transgene expression.
Mueller C, Chulay JD, Trapnell BC, Humphries M, Carey B, Sandhaus RA, McElvaney NG, Messina L, Tang Q, Rouhani FN, Campbell-Thompson M, Fu AD, Yachnis A, Knop DR, Ye GJ, Brantly M, Calcedo R, Somanathan S, Richman LP, Vonderheide RH, Hulme MA, Brusko TM, Wilson JM, Flotte TR
The Journal of clinical investigation 2013 Dec;123(12):5310-8
The Journal of clinical investigation 2013 Dec;123(12):5310-8
Enhanced production of IL-17A in patients with severe asthma is inhibited by 1α,25-dihydroxyvitamin D3 in a glucocorticoid-independent fashion.
Nanzer AM, Chambers ES, Ryanna K, Richards DF, Black C, Timms PM, Martineau AR, Griffiths CJ, Corrigan CJ, Hawrylowicz CM
The Journal of allergy and clinical immunology 2013 Aug;132(2):297-304.e3
The Journal of allergy and clinical immunology 2013 Aug;132(2):297-304.e3
Low-dose interleukin-2 therapy restores regulatory T cell homeostasis in patients with chronic graft-versus-host disease.
Matsuoka K, Koreth J, Kim HT, Bascug G, McDonough S, Kawano Y, Murase K, Cutler C, Ho VT, Alyea EP, Armand P, Blazar BR, Antin JH, Soiffer RJ, Ritz J
Science translational medicine 2013 Apr 3;5(179):179ra43
Science translational medicine 2013 Apr 3;5(179):179ra43
In vitro HIV infection impairs the capacity of myeloid dendritic cells to induce regulatory T cells.
Presicce P, Moreno-Fernandez ME, Rusie LK, Fichtenbaum C, Chougnet CA
PloS one 2012;7(8):e42802
PloS one 2012;7(8):e42802
Novel serial positive enrichment technology enables clinical multiparameter cell sorting.
Stemberger C, Dreher S, Tschulik C, Piossek C, Bet J, Yamamoto TN, Schiemann M, Neuenhahn M, Martin K, Schlapschy M, Skerra A, Schmidt T, Edinger M, Riddell SR, Germeroth L, Busch DH
PloS one 2012;7(4):e35798
PloS one 2012;7(4):e35798
Impaired function of regulatory T cells in cord blood of children of allergic mothers.
Hrdý J, Kocourková I, Prokešová L
Clinical and experimental immunology 2012 Oct;170(1):10-7
Clinical and experimental immunology 2012 Oct;170(1):10-7
Detecting T-cell reactivity to whole cell vaccines: Proof of concept analysis of T-cell response to K562 cell antigens in CML patients.
Brusic A, Hainz U, Wadleigh M, Neuberg D, Su M, Canning CM, Deangelo DJ, Stone RM, Lee JS, Mulligan RC, Ritz J, Dranoff G, Sasada T, Wu CJ
Oncoimmunology 2012 Oct 1;1(7):1095-1103
Oncoimmunology 2012 Oct 1;1(7):1095-1103
Increased expression of regulatory T cells and down-regulatory molecules in lepromatous leprosy.
Palermo ML, Pagliari C, Trindade MA, Yamashitafuji TM, Duarte AJ, Cacere CR, Benard G
The American journal of tropical medicine and hygiene 2012 May;86(5):878-83
The American journal of tropical medicine and hygiene 2012 May;86(5):878-83
Expression of Forkhead box P3 in tumour cells causes immunoregulatory function of signet ring cell carcinoma of the stomach.
Yoshii M, Tanaka H, Ohira M, Muguruma K, Iwauchi T, Lee T, Sakurai K, Kubo N, Yashiro M, Sawada T, Hirakawa K
British journal of cancer 2012 May 8;106(10):1668-74
British journal of cancer 2012 May 8;106(10):1668-74
A gynecologic oncology group phase II trial of two p53 peptide vaccine approaches: subcutaneous injection and intravenous pulsed dendritic cells in high recurrence risk ovarian cancer patients.
Rahma OE, Ashtar E, Czystowska M, Szajnik ME, Wieckowski E, Bernstein S, Herrin VE, Shams MA, Steinberg SM, Merino M, Gooding W, Visus C, Deleo AB, Wolf JK, Bell JG, Berzofsky JA, Whiteside TL, Khleif SN
Cancer immunology, immunotherapy : CII 2012 Mar;61(3):373-84
Cancer immunology, immunotherapy : CII 2012 Mar;61(3):373-84
Induction of endometriosis alters the peripheral and endometrial regulatory T cell population in the non-human primate.
Braundmeier A, Jackson K, Hastings J, Koehler J, Nowak R, Fazleabas A
Human reproduction (Oxford, England) 2012 Jun;27(6):1712-22
Human reproduction (Oxford, England) 2012 Jun;27(6):1712-22
Regulatory T cells exhibit decreased proliferation but enhanced suppression after pulsing with sirolimus.
Singh K, Kozyr N, Stempora L, Kirk AD, Larsen CP, Blazar BR, Kean LS
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2012 Jun;12(6):1441-57
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2012 Jun;12(6):1441-57
The vaccine-site microenvironment induced by injection of incomplete Freund's adjuvant, with or without melanoma peptides.
Harris RC, Chianese-Bullock KA, Petroni GR, Schaefer JT, Brill LB 2nd, Molhoek KR, Deacon DH, Patterson JW, Slingluff CL Jr
Journal of immunotherapy (Hagerstown, Md. : 1997) 2012 Jan;35(1):78-88
Journal of immunotherapy (Hagerstown, Md. : 1997) 2012 Jan;35(1):78-88
PI16 is expressed by a subset of human memory Treg with enhanced migration to CCL17 and CCL20.
Nicholson IC, Mavrangelos C, Bird DR, Bresatz-Atkins S, Eastaff-Leung NG, Grose RH, Gundsambuu B, Hill D, Millard DJ, Sadlon TJ, To S, Zola H, Barry SC, Krumbiegel D
Cellular immunology 2012 Jan-Feb;275(1-2):12-8
Cellular immunology 2012 Jan-Feb;275(1-2):12-8
Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients.
Clark RA, Watanabe R, Teague JE, Schlapbach C, Tawa MC, Adams N, Dorosario AA, Chaney KS, Cutler CS, Leboeuf NR, Carter JB, Fisher DC, Kupper TS
Science translational medicine 2012 Jan 18;4(117):117ra7
Science translational medicine 2012 Jan 18;4(117):117ra7
A converse 4-1BB and CD40 ligand expression pattern delineates activated regulatory T cells (Treg) and conventional T cells enabling direct isolation of alloantigen-reactive natural Foxp3+ Treg.
Schoenbrunn A, Frentsch M, Kohler S, Keye J, Dooms H, Moewes B, Dong J, Loddenkemper C, Sieper J, Wu P, Romagnani C, Matzmohr N, Thiel A
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 15;189(12):5985-94
Journal of immunology (Baltimore, Md. : 1950) 2012 Dec 15;189(12):5985-94
Suppression of tumour-specific CD4⁺ T cells by regulatory T cells is associated with progression of human colorectal cancer.
Betts G, Jones E, Junaid S, El-Shanawany T, Scurr M, Mizen P, Kumar M, Jones S, Rees B, Williams G, Gallimore A, Godkin A
Gut 2012 Aug;61(8):1163-71
Gut 2012 Aug;61(8):1163-71
OMIP-006: phenotypic subset analysis of human T regulatory cells via polychromatic flow cytometry.
Murdoch DM, Staats JS, Weinhold KJ
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 Apr;81(4):281-3
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2012 Apr;81(4):281-3
CD4-like immunological function by CD4- T cells in multiple natural hosts of simian immunodeficiency virus.
Vinton C, Klatt NR, Harris LD, Briant JA, Sanders-Beer BE, Herbert R, Woodward R, Silvestri G, Pandrea I, Apetrei C, Hirsch VM, Brenchley JM
Journal of virology 2011 Sep;85(17):8702-8
Journal of virology 2011 Sep;85(17):8702-8
Decreased AIRE expression and global thymic hypofunction in Down syndrome.
Lima FA, Moreira-Filho CA, Ramos PL, Brentani H, Lima Lde A, Arrais M, Bento-de-Souza LC, Bento-de-Souza L, Duarte MI, Coutinho A, Carneiro-Sampaio M
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 15;187(6):3422-30
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 15;187(6):3422-30
Upregulated expression of indoleamine 2, 3-dioxygenase in CHO cells induces apoptosis of competent T cells and increases proportion of Treg cells.
Sun J, Yu J, Li H, Yang L, Wei F, Yu W, Liu J, Ren X
Journal of experimental & clinical cancer research : CR 2011 Sep 14;30(1):82
Journal of experimental & clinical cancer research : CR 2011 Sep 14;30(1):82
HLA-G level on monocytoid dendritic cells correlates with regulatory T-cell Foxp3 expression in liver transplant tolerance.
Castellaneta A, Mazariegos GV, Nayyar N, Zeevi A, Thomson AW
Transplantation 2011 May 27;91(10):1132-40
Transplantation 2011 May 27;91(10):1132-40
Engagement of TLR2 reverses the suppressor function of conjunctiva CD4+CD25+ regulatory T cells and promotes herpes simplex virus epitope-specific CD4+CD25- effector T cell responses.
Dasgupta G, Chentoufi AA, You S, Falatoonzadeh P, Urbano LA, Akhtarmalik A, Nguyen K, Ablabutyan L, Nesburn AB, BenMohamed L
Investigative ophthalmology & visual science 2011 May 17;52(6):3321-33
Investigative ophthalmology & visual science 2011 May 17;52(6):3321-33
Clinical implications and characteristics of factor forkhead box protein 3 in gastric cancer.
Jiang C, Wang W, Yan W, Zhang Y, Yang J, Zhang S, Zhang C, Zhang W, Han W, Wang J, Zhang YQ
Experimental and therapeutic medicine 2011 Jul;2(4):667-673
Experimental and therapeutic medicine 2011 Jul;2(4):667-673
High-scatter T cells: a reliable biomarker for malignant T cells in cutaneous T-cell lymphoma.
Clark RA, Shackelton JB, Watanabe R, Calarese A, Yamanaka K, Campbell JJ, Teague JE, Kuo HP, Hijnen D, Kupper TS
Blood 2011 Feb 10;117(6):1966-76
Blood 2011 Feb 10;117(6):1966-76
Immunovirological analyses of chronically simian immunodeficiency virus SIVmnd-1- and SIVmnd-2-infected mandrills (Mandrillus sphinx).
Apetrei C, Sumpter B, Souquiere S, Chahroudi A, Makuwa M, Reed P, Ribeiro RM, Pandrea I, Roques P, Silvestri G
Journal of virology 2011 Dec;85(24):13077-87
Journal of virology 2011 Dec;85(24):13077-87
Significant mobilization of both conventional and regulatory T cells with AMD3100.
Kean LS, Sen S, Onabajo O, Singh K, Robertson J, Stempora L, Bonifacino AC, Metzger ME, Promislow DE, Mattapallil JJ, Donahue RE
Blood 2011 Dec 15;118(25):6580-90
Blood 2011 Dec 15;118(25):6580-90
T cell subpopulations in lymph nodes may not be predictive of patient outcome in colorectal cancer.
Kemp RA, Black MA, McCall J, Yoon HS, Phillips V, Anjomshoaa A, Reeve AE
Journal of experimental & clinical cancer research : CR 2011 Aug 24;30(1):78
Journal of experimental & clinical cancer research : CR 2011 Aug 24;30(1):78
Rapid temporal control of Foxp3 protein degradation by sirtuin-1.
van Loosdregt J, Brunen D, Fleskens V, Pals CE, Lam EW, Coffer PJ
PloS one 2011 Apr 20;6(4):e19047
PloS one 2011 Apr 20;6(4):e19047
An MHC-defined primate model reveals significant rejection of bone marrow after mixed chimerism induction despite full MHC matching.
Larsen CP, Page A, Linzie KH, Russell M, Deane T, Stempora L, Strobert E, Penedo MC, Ward T, Wiseman R, O'Connor D, Miller W, Sen S, Singh K, Kean LS
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2010 Nov;10(11):2396-409
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2010 Nov;10(11):2396-409
Effects of pegylated G-CSF on immune cell number and function in patients with gynecological malignancies.
Bonanno G, Procoli A, Mariotti A, Corallo M, Perillo A, Danese S, De Cristofaro R, Scambia G, Rutella S
Journal of translational medicine 2010 Nov 9;8:114
Journal of translational medicine 2010 Nov 9;8:114
CCR6 is expressed on an IL-10-producing, autoreactive memory T cell population with context-dependent regulatory function.
Rivino L, Gruarin P, Häringer B, Steinfelder S, Lozza L, Steckel B, Weick A, Sugliano E, Jarrossay D, Kühl AA, Loddenkemper C, Abrignani S, Sallusto F, Lanzavecchia A, Geginat J
The Journal of experimental medicine 2010 Mar 15;207(3):565-77
The Journal of experimental medicine 2010 Mar 15;207(3):565-77
Glioblastoma cancer-initiating cells inhibit T-cell proliferation and effector responses by the signal transducers and activators of transcription 3 pathway.
Wei J, Barr J, Kong LY, Wang Y, Wu A, Sharma AK, Gumin J, Henry V, Colman H, Priebe W, Sawaya R, Lang FF, Heimberger AB
Molecular cancer therapeutics 2010 Jan;9(1):67-78
Molecular cancer therapeutics 2010 Jan;9(1):67-78
CD40 signalling induces IL-10-producing, tolerogenic dendritic cells.
Tuettenberg A, Fondel S, Steinbrink K, Enk AH, Jonuleit H
Experimental dermatology 2010 Jan;19(1):44-53
Experimental dermatology 2010 Jan;19(1):44-53
Regulation of Treg functionality by acetylation-mediated Foxp3 protein stabilization.
van Loosdregt J, Vercoulen Y, Guichelaar T, Gent YY, Beekman JM, van Beekum O, Brenkman AB, Hijnen DJ, Mutis T, Kalkhoven E, Prakken BJ, Coffer PJ
Blood 2010 Feb 4;115(5):965-74
Blood 2010 Feb 4;115(5):965-74
Safety and T cell modulating effects of high dose vitamin D3 supplementation in multiple sclerosis.
Smolders J, Peelen E, Thewissen M, Cohen Tervaert JW, Menheere P, Hupperts R, Damoiseaux J
PloS one 2010 Dec 13;5(12):e15235
PloS one 2010 Dec 13;5(12):e15235
Dynamic changes in cellular infiltrates with repeated cutaneous vaccination: a histologic and immunophenotypic analysis.
Schaefer JT, Patterson JW, Deacon DH, Smolkin ME, Petroni GR, Jackson EM, Slingluff CL Jr
Journal of translational medicine 2010 Aug 20;8:79
Journal of translational medicine 2010 Aug 20;8:79
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Green AM, Mattila JT, Bigbee CL, Bongers KS, Lin PL, Flynn JL
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
CD30 discriminates heat shock protein 60-induced FOXP3+ CD4+ T cells with a regulatory phenotype.
de Kleer I, Vercoulen Y, Klein M, Meerding J, Albani S, van der Zee R, Sawitzki B, Hamann A, Kuis W, Prakken B
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 15;185(4):2071-9
Journal of immunology (Baltimore, Md. : 1950) 2010 Aug 15;185(4):2071-9
Cutting Edge: Responder T cells regulate human DR+ effector regulatory T cell activity via granzyme B.
Ashley CW, Baecher-Allan C
Journal of immunology (Baltimore, Md. : 1950) 2009 Oct 15;183(8):4843-7
Journal of immunology (Baltimore, Md. : 1950) 2009 Oct 15;183(8):4843-7
Increased ectonucleotidase expression and activity in regulatory T cells of patients with head and neck cancer.
Mandapathil M, Szczepanski MJ, Szajnik M, Ren J, Lenzner DE, Jackson EK, Gorelik E, Lang S, Johnson JT, Whiteside TL
Clinical cancer research : an official journal of the American Association for Cancer Research 2009 Oct 15;15(20):6348-57
Clinical cancer research : an official journal of the American Association for Cancer Research 2009 Oct 15;15(20):6348-57
Therapeutic effect of CD137 immunomodulation in lymphoma and its enhancement by Treg depletion.
Houot R, Goldstein MJ, Kohrt HE, Myklebust JH, Alizadeh AA, Lin JT, Irish JM, Torchia JA, Kolstad A, Chen L, Levy R
Blood 2009 Oct 15;114(16):3431-8
Blood 2009 Oct 15;114(16):3431-8
Foxp3 regulates megakaryopoiesis and platelet function.
Bernard JJ, Seweryniak KE, Koniski AD, Spinelli SL, Blumberg N, Francis CW, Taubman MB, Palis J, Phipps RP
Arteriosclerosis, thrombosis, and vascular biology 2009 Nov;29(11):1874-82
Arteriosclerosis, thrombosis, and vascular biology 2009 Nov;29(11):1874-82
HIV-1 binding to CD4 on CD4+CD25+ regulatory T cells enhances their suppressive function and induces them to home to, and accumulate in, peripheral and mucosal lymphoid tissues: an additional mechanism of immunosuppression.
Ji J, Cloyd MW
International immunology 2009 Mar;21(3):283-94
International immunology 2009 Mar;21(3):283-94
Fc receptor-like 3 protein expressed on IL-2 nonresponsive subset of human regulatory T cells.
Nagata S, Ise T, Pastan I
Journal of immunology (Baltimore, Md. : 1950) 2009 Jun 15;182(12):7518-26
Journal of immunology (Baltimore, Md. : 1950) 2009 Jun 15;182(12):7518-26
Selective reduction of graft-versus-host disease-mediating human T cells by ex vivo treatment with soluble Fas ligand.
Bohana-Kashtan O, Morisot S, Hildreth R, Brayton C, Levitsky HI, Civin CI
Journal of immunology (Baltimore, Md. : 1950) 2009 Jul 1;183(1):696-705
Journal of immunology (Baltimore, Md. : 1950) 2009 Jul 1;183(1):696-705
CD49d provides access to "untouched" human Foxp3+ Treg free of contaminating effector cells.
Kleinewietfeld M, Starke M, Di Mitri D, Borsellino G, Battistini L, Rötzschke O, Falk K
Blood 2009 Jan 22;113(4):827-36
Blood 2009 Jan 22;113(4):827-36
Quantitative DNA methylation analysis of FOXP3 as a new method for counting regulatory T cells in peripheral blood and solid tissue.
Wieczorek G, Asemissen A, Model F, Turbachova I, Floess S, Liebenberg V, Baron U, Stauch D, Kotsch K, Pratschke J, Hamann A, Loddenkemper C, Stein H, Volk HD, Hoffmüller U, Grützkau A, Mustea A, Huehn J, Scheibenbogen C, Olek S
Cancer research 2009 Jan 15;69(2):599-608
Cancer research 2009 Jan 15;69(2):599-608
The importance of Foxp3 antibody and fixation/permeabilization buffer combinations in identifying CD4+CD25+Foxp3+ regulatory T cells.
Law JP, Hirschkorn DF, Owen RE, Biswas HH, Norris PJ, Lanteri MC
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2009 Dec;75(12):1040-50
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2009 Dec;75(12):1040-50
Human peripheral gammadelta T cells possess regulatory potential.
Kühl AA, Pawlowski NN, Grollich K, Blessenohl M, Westermann J, Zeitz M, Loddenkemper C, Hoffmann JC
Immunology 2009 Dec;128(4):580-8
Immunology 2009 Dec;128(4):580-8
Abnormally high levels of virus-infected IFN-gamma+ CCR4+ CD4+ CD25+ T cells in a retrovirus-associated neuroinflammatory disorder.
Yamano Y, Araya N, Sato T, Utsunomiya A, Azakami K, Hasegawa D, Izumi T, Fujita H, Aratani S, Yagishita N, Fujii R, Nishioka K, Jacobson S, Nakajima T
PloS one 2009 Aug 5;4(8):e6517
PloS one 2009 Aug 5;4(8):e6517
FOXP3 expression and overall survival in breast cancer.
Merlo A, Casalini P, Carcangiu ML, Malventano C, Triulzi T, Mènard S, Tagliabue E, Balsari A
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2009 Apr 10;27(11):1746-52
Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2009 Apr 10;27(11):1746-52
Human spleen contains different subsets of dendritic cells and regulatory T lymphocytes.
Velásquez-Lopera MM, Correa LA, García LF
Clinical and experimental immunology 2008 Oct;154(1):107-14
Clinical and experimental immunology 2008 Oct;154(1):107-14
Phenotypic characterisation of T-lymphocytes in COPD: abnormal CD4+CD25+ regulatory T-lymphocyte response to tobacco smoking.
Barceló B, Pons J, Ferrer JM, Sauleda J, Fuster A, Agustí AG
The European respiratory journal 2008 Mar;31(3):555-62
The European respiratory journal 2008 Mar;31(3):555-62
Fusions of dendritic cells with breast carcinoma stimulate the expansion of regulatory T cells while concomitant exposure to IL-12, CpG oligodeoxynucleotides, and anti-CD3/CD28 promotes the expansion of activated tumor reactive cells.
Vasir B, Wu Z, Crawford K, Rosenblatt J, Zarwan C, Bissonnette A, Kufe D, Avigan D
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):808-21
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):808-21
Fusions of dendritic cells with breast carcinoma stimulate the expansion of regulatory T cells while concomitant exposure to IL-12, CpG oligodeoxynucleotides, and anti-CD3/CD28 promotes the expansion of activated tumor reactive cells.
Vasir B, Wu Z, Crawford K, Rosenblatt J, Zarwan C, Bissonnette A, Kufe D, Avigan D
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):808-21
Journal of immunology (Baltimore, Md. : 1950) 2008 Jul 1;181(1):808-21
High density of FOXP3-positive T cells infiltrating colorectal cancers with microsatellite instability.
Michel S, Benner A, Tariverdian M, Wentzensen N, Hoefler P, Pommerencke T, Grabe N, von Knebel Doeberitz M, Kloor M
British journal of cancer 2008 Dec 2;99(11):1867-73
British journal of cancer 2008 Dec 2;99(11):1867-73
FOXP3 expression accurately defines the population of intratumoral regulatory T cells that selectively accumulate in metastatic melanoma lesions.
Ahmadzadeh M, Felipe-Silva A, Heemskerk B, Powell DJ Jr, Wunderlich JR, Merino MJ, Rosenberg SA
Blood 2008 Dec 15;112(13):4953-60
Blood 2008 Dec 15;112(13):4953-60
Epigenetic inheritance of DNA methylation limits activation-induced expression of FOXP3 in conventional human CD25-CD4+ T cells.
Nagar M, Vernitsky H, Cohen Y, Dominissini D, Berkun Y, Rechavi G, Amariglio N, Goldstein I
International immunology 2008 Aug;20(8):1041-55
International immunology 2008 Aug;20(8):1041-55
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Kasprzycka M, Zhang Q, Witkiewicz A, Marzec M, Potoczek M, Liu X, Wang HY, Milone M, Basu S, Mauger J, Choi JK, Abrams JT, Hou JS, Rook AH, Vonderheid E, Woetmann A, Odum N, Wasik MA
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Gamma c-signaling cytokines induce a regulatory T cell phenotype in malignant CD4+ T lymphocytes.
Kasprzycka M, Zhang Q, Witkiewicz A, Marzec M, Potoczek M, Liu X, Wang HY, Milone M, Basu S, Mauger J, Choi JK, Abrams JT, Hou JS, Rook AH, Vonderheid E, Woetmann A, Odum N, Wasik MA
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Journal of immunology (Baltimore, Md. : 1950) 2008 Aug 15;181(4):2506-12
Foxp3 expression in human cancer cells.
Karanikas V, Speletas M, Zamanakou M, Kalala F, Loules G, Kerenidi T, Barda AK, Gourgoulianis KI, Germenis AE
Journal of translational medicine 2008 Apr 22;6:19
Journal of translational medicine 2008 Apr 22;6:19
The regulatory T cell-associated transcription factor FoxP3 is expressed by tumor cells.
Ebert LM, Tan BS, Browning J, Svobodova S, Russell SE, Kirkpatrick N, Gedye C, Moss D, Ng SP, MacGregor D, Davis ID, Cebon J, Chen W
Cancer research 2008 Apr 15;68(8):3001-9
Cancer research 2008 Apr 15;68(8):3001-9
Administration of a CD25-directed immunotoxin, LMB-2, to patients with metastatic melanoma induces a selective partial reduction in regulatory T cells in vivo.
Powell DJ Jr, Felipe-Silva A, Merino MJ, Ahmadzadeh M, Allen T, Levy C, White DE, Mavroukakis S, Kreitman RJ, Rosenberg SA, Pastan I
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 1;179(7):4919-28
Journal of immunology (Baltimore, Md. : 1950) 2007 Oct 1;179(7):4919-28
Increased frequencies of CD4+ CD25(high) regulatory T cells in acute dengue infection.
Lühn K, Simmons CP, Moran E, Dung NT, Chau TN, Quyen NT, Thao le TT, Van Ngoc T, Dung NM, Wills B, Farrar J, McMichael AJ, Dong T, Rowland-Jones S
The Journal of experimental medicine 2007 May 14;204(5):979-85
The Journal of experimental medicine 2007 May 14;204(5):979-85
IL-15 and dermal fibroblasts induce proliferation of natural regulatory T cells isolated from human skin.
Clark RA, Kupper TS
Blood 2007 Jan 1;109(1):194-202
Blood 2007 Jan 1;109(1):194-202
Association of CD4+CD25+Foxp3+ regulatory T cells with chronic activity and viral clearance in patients with hepatitis B.
Yang G, Liu A, Xie Q, Guo TB, Wan B, Zhou B, Zhang JZ
International immunology 2007 Feb;19(2):133-40
International immunology 2007 Feb;19(2):133-40
Wiskott-Aldrich syndrome protein is required for regulatory T cell homeostasis.
Humblet-Baron S, Sather B, Anover S, Becker-Herman S, Kasprowicz DJ, Khim S, Nguyen T, Hudkins-Loya K, Alpers CE, Ziegler SF, Ochs H, Torgerson T, Campbell DJ, Rawlings DJ
The Journal of clinical investigation 2007 Feb;117(2):407-18
The Journal of clinical investigation 2007 Feb;117(2):407-18
Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression.
Borsellino G, Kleinewietfeld M, Di Mitri D, Sternjak A, Diamantini A, Giometto R, Höpner S, Centonze D, Bernardi G, Dell'Acqua ML, Rossini PM, Battistini L, Rötzschke O, Falk K
Blood 2007 Aug 15;110(4):1225-32
Blood 2007 Aug 15;110(4):1225-32
FOXP3 regulates TLR10 expression in human T regulatory cells.
Bell MP, Svingen PA, Rahman MK, Xiong Y, Faubion WA Jr
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
FOXP3 regulates TLR10 expression in human T regulatory cells.
Bell MP, Svingen PA, Rahman MK, Xiong Y, Faubion WA Jr
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
Journal of immunology (Baltimore, Md. : 1950) 2007 Aug 1;179(3):1893-900
Mucosal but not peripheral FOXP3+ regulatory T cells are highly increased in untreated HIV infection and normalize after suppressive HAART.
Epple HJ, Loddenkemper C, Kunkel D, Tröger H, Maul J, Moos V, Berg E, Ullrich R, Schulzke JD, Stein H, Duchmann R, Zeitz M, Schneider T
Blood 2006 Nov 1;108(9):3072-8
Blood 2006 Nov 1;108(9):3072-8
Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma.
Fecci PE, Mitchell DA, Whitesides JF, Xie W, Friedman AH, Archer GE, Herndon JE 2nd, Bigner DD, Dranoff G, Sampson JH
Cancer research 2006 Mar 15;66(6):3294-302
Cancer research 2006 Mar 15;66(6):3294-302
IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients.
Ahmadzadeh M, Rosenberg SA
Blood 2006 Mar 15;107(6):2409-14
Blood 2006 Mar 15;107(6):2409-14
Foxp3+CD4+CD25+ T cells control virus-specific memory T cells in chimpanzees that recovered from hepatitis C.
Manigold T, Shin EC, Mizukoshi E, Mihalik K, Murthy KK, Rice CM, Piccirillo CA, Rehermann B
Blood 2006 Jun 1;107(11):4424-32
Blood 2006 Jun 1;107(11):4424-32
Depletion of alloreactive T cells via CD69: implications on antiviral, antileukemic and immunoregulatory T lymphocytes.
Hartwig UF, Nonn M, Khan S, Meyer RG, Huber C, Herr W
Bone marrow transplantation 2006 Feb;37(3):297-305
Bone marrow transplantation 2006 Feb;37(3):297-305
In situ analysis of FOXP3+ regulatory T cells in human colorectal cancer.
Loddenkemper C, Schernus M, Noutsias M, Stein H, Thiel E, Nagorsen D
Journal of translational medicine 2006 Dec 13;4:52
Journal of translational medicine 2006 Dec 13;4:52
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Lim HW, Hillsamer P, Banham AH, Kim CH
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells.
Lim HW, Hillsamer P, Banham AH, Kim CH
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Journal of immunology (Baltimore, Md. : 1950) 2005 Oct 1;175(7):4180-3
Human CD4+ T cells express TLR5 and its ligand flagellin enhances the suppressive capacity and expression of FOXP3 in CD4+CD25+ T regulatory cells.
Crellin NK, Garcia RV, Hadisfar O, Allan SE, Steiner TS, Levings MK
Journal of immunology (Baltimore, Md. : 1950) 2005 Dec 15;175(12):8051-9
Journal of immunology (Baltimore, Md. : 1950) 2005 Dec 15;175(12):8051-9
CD25+CD4+ T cells in human cord blood: an immunoregulatory subset with naive phenotype and specific expression of forkhead box p3 (Foxp3) gene.
Takahata Y, Nomura A, Takada H, Ohga S, Furuno K, Hikino S, Nakayama H, Sakaguchi S, Hara T
Experimental hematology 2004 Jul;32(7):622-9
Experimental hematology 2004 Jul;32(7):622-9
Alloantigen specific CD8+CD28- FOXP3+ T suppressor cells induce ILT3+ ILT4+ tolerogenic endothelial cells, inhibiting alloreactivity.
Manavalan JS, Kim-Schulze S, Scotto L, Naiyer AJ, Vlad G, Colombo PC, Marboe C, Mancini D, Cortesini R, Suciu-Foca N
International immunology 2004 Aug;16(8):1055-68
International immunology 2004 Aug;16(8):1055-68
The origin of FOXP3-expressing CD4+ regulatory T cells: thymus or periphery.
Sakaguchi S
The Journal of clinical investigation 2003 Nov;112(9):1310-2
The Journal of clinical investigation 2003 Nov;112(9):1310-2
Control of regulatory T cell development by the transcription factor Foxp3.
Hori S, Nomura T, Sakaguchi S
Science (New York, N.Y.) 2003 Feb 14;299(5609):1057-61
Science (New York, N.Y.) 2003 Feb 14;299(5609):1057-61
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoblotting of lysate from normal human peripheral blood cells (lane A), BALB/c splenocytes (lane B), or recombinant human Foxp3 fusion protein (lane C); probed with 2 µg/mL of Anti-Human Foxp3 Purified. Bands were revealed with Anti-Rat IgG HRP.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of FOXP3 was performed using formalin-fixed paraffin-embedded Lymph node [Follicular Grade: I (small cell)] tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without FOXP3 Monoclonal Antibody (PCH101), eBioscience™ (Product # 14-4776-82) at 5 µg/mL dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 (Product # A-11006) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of FOXP3 was performed using formalin-fixed paraffin-embedded Lymph node [Follicular Grade: I (small cell)] tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without FOXP3 Monoclonal Antibody (PCH101), eBioscience™ (Product # 14-4776-82) at 5 µg/mL dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 (Product # A-11006) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of FOXP3 was performed using formalin-fixed paraffin-embedded Lymph node (Hodgkin lymphoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - Low pH (10X) (Product # 00-4955-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without FOXP3 Monoclonal Antibody (PCH101), eBioscience™ (Product # 14-4776-82) at 5 µg/mL dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 (Product # A-11006) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of FOXP3 was performed using formalin-fixed paraffin-embedded Lymph node (Hodgkin lymphoma) tissue sections. To expose the target protein, heat-induced epitope retrieval was performed on de-paraffinized sections using eBioscience™ IHC Antigen Retrieval Solution - High pH (10X) (Product # 00-4956-58) diluted to 1X solution in water in a decloaking chamber at 110 degree Celsius for 15 minutes. Following antigen retrieval, the sections were blocked with 2% normal goat serum in 1X PBS for 45 minutes at room temperature and then probed with or without FOXP3 Monoclonal Antibody (PCH101), eBioscience™ (Product # 14-4776-82) at 5 µg/mL dilution in 0.1% normal goat serum overnight at 4 degree Celsius in a humidified chamber. Detection was performed using Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 (Product # A-11006) at a dilution of 1:2000 in 0.1% normal goat serum for 45 minutes at room temperature. ReadyProbes™ Tissue Autofluorescence Quenching Kit (Product # R37630) was used to quench autofluorescence from the tissues. Nuclei were stained with DAPI (Product # D1306) and the sections were mounted using ProLong™ Glass Antifade Mountant (Product # P36984). The images were captured on EVOS™ M7000 Imaging System (Product # AMF7000) at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
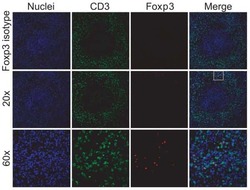
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
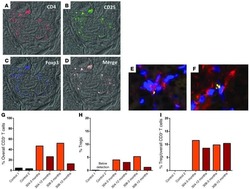
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
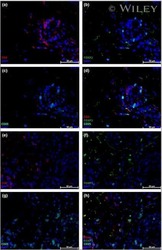
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
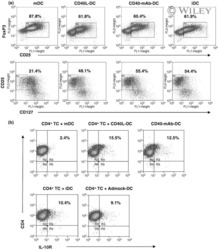
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
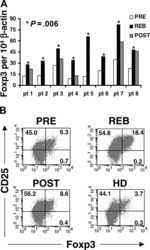
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
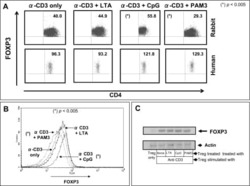
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
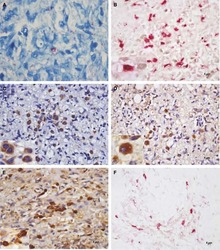
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
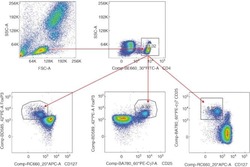
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
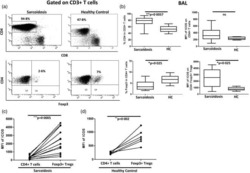
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
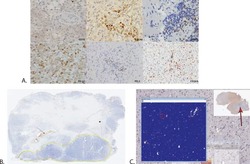
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
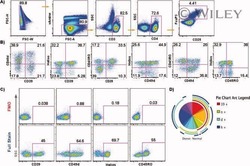
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
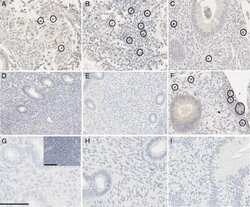
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
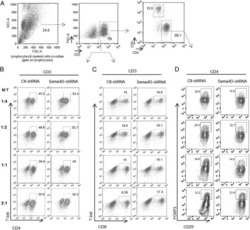
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
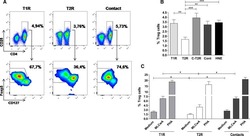
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
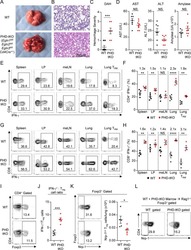
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
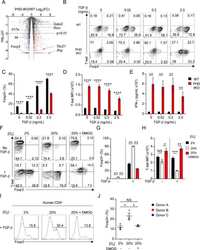
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
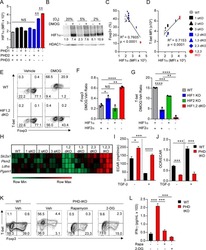
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
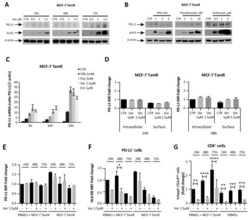
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
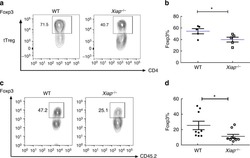
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
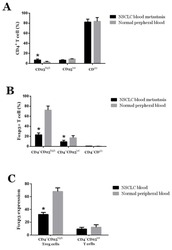
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
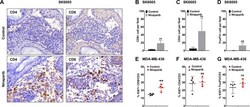
- Experimental details
- Figure 2 Niraparib promoted tumor immune cell infiltration in both the BRCA -proficient SK6005 syngeneic and BRCA -deficient MDA-MB-436 NOG-EXL humanized tumor models ( A ) Representative images of CD4 and CD8 immunohistochemical staining in control- and niraparib-treated BRCA -proficient SK6005 tumors. ( B - D ) Quantification of the number of CD4 + cells, CD8 + cells and FoxP3 + cells per field upon niraparib treatment in BRCA -proficient SK6005 tumors. ( E-G ) Percentage of Ki67-positive CD4 + cells, CD8 + cells and FoxP3 + cells among the total CD3 + population by flow cytometry in humanized NOG-EXL MDA-MB-436 tumors. **p-value is less than 0.05 by Student''s t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Number and suppressive function of Tregs isolated and expanded from UCB and PB. (A) Total numbers of CD4 + CD25 + FoxP3 + Helios + Tregs over time in the UCB and PB. (B) Proliferation indices of Tregs between days 7 and 28 in the UCB and PB, determined using a CFSE-based proliferation assay. (C) Proportions of Helios + FoxP3 + cells in CD4 + CD25 + cell populations from UCB and PB determined by flow cytometry on days 7, 14 and 21. (D) Proliferation indices of Th cells in a CFSE-based suppression assay between days 7 and 28 of culture in UCB and PB. (**P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
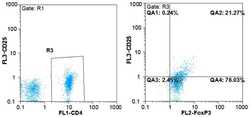
- Experimental details
- Figure 1. Flow cytometric mapping of the CD4 + CD25 + Foxp3 + cells in peripheral blood withdrawn from the syphilitic patients with sero-resistance. Foxp3, forkhead box P3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
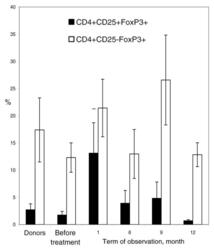
- Experimental details
- Figure 5. Percentage of regulatory T-cells in the course of T-cell vaccination in RA patients. PBMC from RA patients (n = 11) were analyzed by flow cytometry for ""naive"" regulatory CD4 + CD25 + FoxP3 + T-cells (closed bars) and induced regulatory CD4 + CD25 - FoxP3 + T-cells (open bars) before the onset and in course of T-cell vaccination (observation period 12 mo), as compared with PBMC from healthy donors (n = 10). Percentage of positive cells from the total lymphoid cell population ( y -axis) was determined by flow cytometry. ** P < 0.01, as compared with the levels before therapy (U criterion). Error bars denote means +- SEM (percentage of positive cells).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
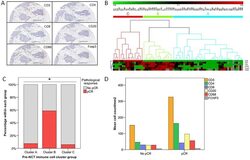
- Experimental details
- Figure 1 Pre-treatment lymphocyte subpopulations profile of breast cancer and pathological response to neoadjuvant chemotherapy. A) representative digital images (100x) of ductal invasive carcinoma of the breast showing CD3, CD4, CD8, CD20, CD68 and Foxp3 staining (scale bar, 100 mum). B) unsupervised hierarchical clustering analysis of pre-chemotherapy TIL subpopulations yielded three groups or immune clusters (arbitrarily named A, B and C); each column represents a patient and each row represents an immunohistochemical marker. C) pathologic complete response distribution according to pre-chemotherapy immune cluster group. D) differential distribution of lymphocyte populations (CD3, CD4, CD8, CD20, Foxp3, CD68) according to pathological response to neoadjuvant chemotherapy; differences were statistically significant for CD3, CD4 and CD20 and non significant for CD8, Foxp3 and CD68. Statistical analysis: Mann-Whitney U-test. *, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
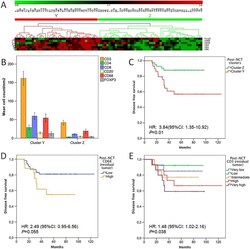
- Experimental details
- Figure 3 Post-chemotherapy immune cell subpopulation profile and breast cancer prognosis. A) unsupervised hierarchical clustering analysis of post-chemotherapy immune cell populations generated two groups (named as Y and Z); each column represents a patient and each row represents an immunohistochemical marker. B) distribution of immune cell populations in post-chemotherapy cluster groups Y and Z; statistically significant higher infiltration was shown in cluster Y for all populations (CD3, CD4, CD8, CD20, Foxp3, CD68), Mann-Whitney U-test. C) Kaplan-Meier curves showed a worse disease-free survival for patients with tumors belonging to post-chemotherapy immune cluster Y ( P = 0.01). D) disease free survival curves according to post-treatment CD68 infiltration in residual tumor ( P = 0.055). E) Kaplan-Meier disease free survival curves according to level of post-treatment CD3 infiltration in residual tumor, showing an increasingly worse prognosis as CD3 infiltration increases ( P = 0.038). Hazard ratios (HR) and 95%CI calculated according to Cox proportional hazard regression models.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Additional file 1: Figure S1.: Distribution of individual immune cell populations in pre- and post-treatment breast cancer biopsies. A) pre-chemotherapy distribution of immune cell populations (absolute counts/mm 2 ). B) post-chemotherapy distribution of immune cell populations (absolute counts/mm 2 ). C) percentage distribution of immune cell populations in pre- and post-treatment biopsies, showing a chemotherapy-induced inversion of CD4/CD8 ratio and a decrease of CD20; percentages calculated over total immune cell counts (CD4 + CD8 + CD20 + Foxp3 + CD68). (TIFF 663 KB)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Gating strategy for frequency of Treg cells in the study population. Three color flow cytometry was performed in whole blood. Three subsets of CD4+T cells were defined according to CD25 staining cells expressing CD25 high were chosen and gated for the detection of FOXP3+T cells
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Flow cytometric analysis of FOXP3, GITR and CTLA-4 in CD4 + CD25 + T cells in three TKI treatment groups. ''Resting'' is a negative control without antibodies. Percentage of FOXP3-positive cells in (A) imatinib, (B) dasatinib and (C) nilotinib treatment groups. Percentage of GITR-positive cells in (D) imatinib, (E) dasatinib and (F) nilotinib treatment groups. Percentage of CTLA-4-positive cells in (G) imatinib, (H) dasatinib and (I) nilotinib treatment groups. FOXP3, forkhead box P3; GITR, tumor necrosis factor receptor; CTLA-4, cytotoxic T-lymphocyte-associated antigen 4; CD, cluster of differentiation; TKI, tyrosine kinase inhibitor. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Proportion of Treg cells and TGF-beta1-positive Treg cells in the peripheral superficial LNs of the Asy, AIDS and control groups. (A) Representative prevalence of Treg cells from individual subjects in the three studied groups. Foxp3, an X chromosome-encoded fork head transcription factor family member is indispensable for the differentiation of Treg cells. Treg cells were gated from Foxp3 + subsets of CD3 + CD4 + T cells. (B) Representative prevalence of TGF-beta1-positive Treg cells from individual subjects in three studied groups. (C) Double staining of Foxp3 and TGF-beta1 from individual subjects in the three studied groups. Blue, Foxp3 (nucleus expression; BCIP/NBT, magnification, x400); Red, TGF-beta1 (cell membrane expression; aminoethyl carbazole; x400). Double positive cells for Foxp3 and TGF-beta1 are indicated by arrows. (D) Statistical analysis of the frequency of CD3 + CD4 + Foxp3 + Treg and (E) CD3 + CD4 + Foxp3 + TGF-beta1 + Tregs in peripheral superficial LNs of patients with AIDS, asy and controls. Data in D and E are expressed as box plots, in which the horizontal lines illustrate the 25, 50, 75th percentiles. Vertical lines represent the 10 and 90th percentiles. Tissue samples, n=10 Control; n=10 Asy; n=22 AIDS. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
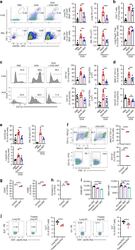
- Experimental details
- Fig. 4 OVA DNT treatment selectively inhibited Tfh cells and CD11b + DCs. OVA-sensitized mice were treated with an intravenous transfer of OVA DNTs after the first OVA challenge. The mice were challenged daily for the next two days and sacrificed 48 h after the last aerosol challenge. a The lung and BALF Tfh cell (CD4 + B220 - CXCR5 + PD-1 + ), b CD4 + T cell (CD4 + B220 - ) and Treg cell (CD4 + B220 - Foxp3 + ) proportions were measured by flow cytometry. c The lung and mLN CD11b + DC (CD11c + MHC-II + CD11b + ), d DC (CD11c + MHC-9II + ) and CD103 + DC (CD11c + MHC-II + CD103 + CD11b - ) proportions were measured by flow cytometry. e The proportions of B cells (B220 + CD4 - ) in the mLN (mediastinum lymph node), BALF and lungs were measured by flow cytometry. OVA-stimulated bone marrow cells were cocultured with OVA DNTs and stimulated with GM-CSF (20 ng/ml) for 3 days to test the direct effect of OVA DNTs on OVA DC proliferation and differentiation. f The proportions of bone marrow-derived DCs and CD11b+ DCs were measured by flow cytometry. The direct effects of OVA DNTs on g costimulatory molecule expression and h apoptosis in DCs were measured by flow cytometry. i OVA tetramer + and tetramer - DNT cells were sorted by flow cytometry from OVA-primed DNT cells. Tetramer + or tetramer - DNT cells were cocultured with lung DCs from allergic asthma mice for 3 days. The MFIs of CD86 and MHC-II in DCs were measured by flow cytometry. j Lung DCs (2.5 x 10 4 ) from OVA DNT cell-t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
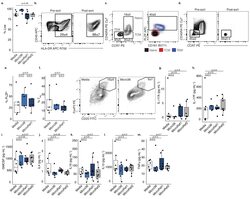
- Experimental details
- Extended Data 9. Fetal Micrococcus isolate promotes distinct APC and T cell phenotypes. a. Proportion of live cells after treatment with media (n=9) or Micrococcus (Micro36 n=6, MicroRef1 n=9, MicroRef2 n=3) strains, where n represents biologically independent fetal specimens for the indicated treatment. ANOVA test for significance. b. HLA-DR + CD45 + lin - cells pre- (left) and post- (right) fluorescence activated cell sorting (FACS). c. Proportion of naive (CD45RA + CCR7 + ), central memory (TCM, CD45RA - CCR7 + ), and effector memory T cells (TEM, CD45RA - CCR7 - ) among live, TCRbeta + , CD4 + cells (left panel) and PLZF and CD161 expression among memory subsets, numbers indicate proportion in TEM (right panel). d. Pre- (left) and post- (right) FACS of effector memory T cells. e. Proportion of PLZF + T cells or f. left, proportion of CD25 hi FoxP3 + regulatory T cells (T regs ) and right, representative flow plots of FoxP3 and CD25 expression among intestinal live, TCRbeta + , CD4 + , Valpha7.2 - , cells after exposure to splenic APCs pretreated with media or Micrococcus (Micro36, MicroRef1) strains for n=5 biologically independent fetal specimens. Concentration of g. IL-17A, h. IL-17F, i. GM-CSF, j. IL-4, k. IL-10, l. IL-13, m. TNFalpha in culture supernatants of lamina propria T cell co-cultures with splenic antigen presenting cells pre-exposed to media (n=7) or Micrococcus (Micro36 n=6, MicroRef1 n=7, MicroRef2 n=7) strains, where n represents biologically independent
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
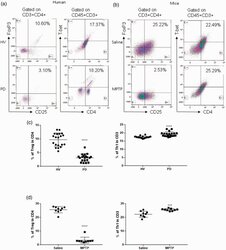
- Experimental details
- Figure 1. Patients with Parkinson's disease (PD) and mice with experimental PD exhibit decreased regulatory T (Treg) and increased T helper 1 (Th1) cell numbers in the blood. Treg cells were defined as CD3+CD4+CD25+FoxP3+ cells, whereas Th1 cells were identified as CD3+CD4+T-bet+ cells. (a) Representative plots of Treg and Th1 cells in patients with PD and healthy volunteers (HVs). (b) Representative plots of Treg and Th1 cells in control mice (Saline) and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated mice. (c) Patients with PD exhibited lower Treg cell levels and greater Th1 cell levels in the blood than HVs. n = 20. ****, p < 0.0001 by a two-tailed Student's t tests. (d) MPTP-induced experimental PD mice had fewer Treg and more Th1 cells in the circulation than saline-treated mice. n = 9/group in each experiment performed in triplicate. ***, p < 0.001; ****, p < 0.0001, according to Student's t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
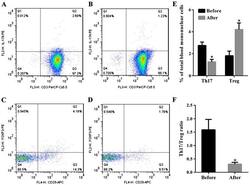
- Experimental details
- Figure 1. The percentage of T helper 17 (Th17) and regulatory T (Treg) cells in peripheral blood mononuclear cells (PBMCs) and the Th17/Treg ratio varies in patients with a successful pregnancy after immunotherapy. The percentage of Th17 and Treg cells in PBMCs were detected by flow cytometry in patients with unexplained recurrent spontaneous abortion (URSA) before and after therapy. Representative CD3 + CD8 + IL-17A + flow cytometry plots from patients with URSA are shown (A) before and (B) after therapy. Representative CD4 + CD25 + Foxp3 + flow cytometry plots from patients with URSA are shown (C) before and (D) after therapy. (E) The percentage of Th17 cells in PBMCs significantly decreased after therapy. The percentage of Treg cells in PBMCs significantly increased after therapy (*P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
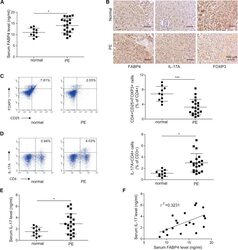
- Experimental details
- Figure 1 Elevation of FABP4 is accompanied with Treg/Th17 imbalance in PE (A) The serum level of FABP4 was determined by ELISA (normal pregnant women, n = 10; PE, n = 20). (B) The immunoreactivities of FABP4, IL-17A, and FOXP3 in placental tissues were monitored by IHC analysis. (C and D) The percentages of Treg (C) and Th17 cells (D) in peripheral blood were assessed by FACS (normal pregnant women, n = 10; PE, n = 20). (E) The serum level of IL-17A was determined by ELISA (normal pregnant women, n = 10; PE, n = 20). (F) Pearson's correlation analysis between IL-17A and FABP4 expression. Data were representative images. *p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
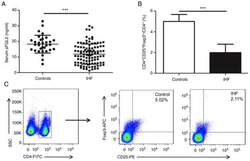
- Experimental details
- Figure 1. Serum sFGL2 levels and circulating Tregs frequencies are decreased in patients with IHF. (A) Serum sFGL2 levels were analyzed by ELISA. (B) The frequency of Tregs was compared in each group. (C) Representative FSC/SSC pseudo-color density image shows the gated CD4 + T cells and representative fluorescence-activated cell sorting plots of CD4 + CD25 + Foxp3 + Tregs from a single patient in each group. ***P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
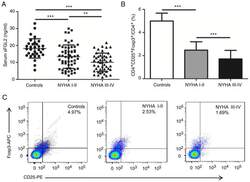
- Experimental details
- Figure 2. Serum sFGL2 levels and circulating Tregs frequencies are decreased with the elevation of NYHA classification. (A) sFGL2 levels were analyzed by ELISA. (B) The proportion of Tregs was compared in each subgroup. (C) Representative fluorescence-activated cell sorting plots of CD4 + CD25 + Foxp3 + Tregs from a single person in each subgroup. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3. Serum sFGL2 levels and circulating Tregs frequencies are decreased with the reduction of LVEF. (A) sFGL2 levels were analyzed by ELISA. (B) The proportion of Tregs was compared in each subgroup. (C) Representative fluorescence-activated cell sorting plots of CD4 + CD25 + Foxp3 + Tregs from a single person in each subgroup. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 3 Investigation into PD Treg populations shows reduced numbers and impaired function. The number of Tregs in PD patients are decreased via a flow cytometric analysis of CD4 + CD25 + FOXP3 + cells as a percent of total CD4 + population and b counting of viable CD4 + CD25 + immune cells following bead/column-based isolation methods from peripheral blood (C n = 28, PD n = 34). Treg health and function markers were deceased in PD patients compared to controls when examining c CD25 protein MFI and d FOXP3 protein MFI from CD4 + CD25 + FOXP3 + cells during flow cytometry (C n = 28, PD n = 34). e Treg suppression of Tresp proliferation is impaired in PD patients compared to controls at ratios (Tresp:Treg) of 1:1, 1:1/2, and 1:1/4 (C n = 25, PD n = 30). f The suppressive capacity of PD Tregs on Tresp proliferation decreases with increasing PD disease burden using the H&Y disease scale (C n = 25, H&Y1 n = 4, H&Y2 n = 17, H&Y3 n = 5, H&Y4 n = 4). Numbers shown as averages +- SEM with Student's t test or one-way ANOVA with appropriate post hoc testing for multiple comparisons as appropriate. P -values are * p < 0.05, ** p < 0.01, and *** p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 Kinetics of regulatory T cells is not affected significantly in mild, moderate and convalescent patients. Foxp3 + expression on CD19 - CD3 + CD4 + CD45RA - T cells to identify the regulatory T cells in HC, outpatient, outpatient and convalescent (upper FACS panel). There was a statistically significant difference among HC, mild, moderate and convalescent (upper FACS panel). Figure 5
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 9 Effects of VX-765 on Treg in the injured spinal cord at 7 days post-injury: immunofluorescence and flow cytometry assay. (A-C) CD4 (red, stained by rhodamine) and FoxP3 (green, stained by FITC) for Treg. Nuclei are blue, stained by Hoechst 33342. Scale bars: 400 um, 20 um (enlarged parts). (D) CD4 + FoxP3 + Treg cell counts in the sham, DMSO, and VX-765 groups. (E) Flow cytometry images of cells derived from spinal cord homogenate in the different groups. (F) Proportional analysis of CD3 + CD4 + CD25 + CD127 - Treg. The original data for Figure 9D and F are shown in Additional file 9 . Data are represented as the mean +- SD ( n = 6). ** P < 0.01 (non-parametric Kruskal-Wallis analysis of variance). DMSO: Dimethyl sulfoxide; FITC: fluoresceine isothiocyanate; FoxP3: forkhead box P3; SCI: spinal cord injury; Treg: regulatory T cells; VX-765: caspase-1 selective inhibitor.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
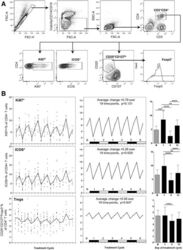
- Experimental details
- Fig. 6 a Representative flow cytometry data, demonstrating the gating strategy used on PBMC for Treg identification and analysis. FSC-area vs. FSC-height was used for doublet discrimination. A ""dump"" channel was used to gate out dead cells (LIVE/DEAD fixable viability stain), CD14 + monocytes and CD19 + B cells, and lymphocytes were subsequently selected by FSC vs. SSC. CD4 + T cells were gated on the basis of CD4 vs. CD3 staining, then examined for expression of Ki67 and ICOS. Tregs were identified within the CD4 + T cell population as CD25 hi CD127 lo and Foxp3 + . b Longitudinal empirical data, linear mixed models and estimated means (left, centre and right-hand panels respectively) for Ki67+ and ICOS+ expression on CD4+ T cells, and the Treg proportion of CD4 cells ( P -values: *
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
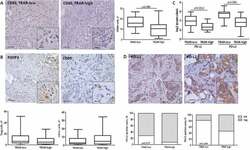
- Experimental details
- Figure 2. Characterization of immune TME of HER2+ BCs. A-B) IHC evaluation of CD68+ cells (A), FOXP3+ Treg, and CD33+ cells (B) in tissues of the GHEA cohort. The average number of positive cells in three-fields is shown. Scale bars: 50 mum in the main images and 20 mum in the zoomed images. p-value by unpaired t-test (n = 47) C) PDL1 and PDL2 mRNA expression in tumors of the GHEA cohort according to TRAR classification. p-values by unpaired t-test. D) Representative image of PD-L1 and PD-L2 positive tumors. Scale bars: 50 mum and 20 mum in the zoomed image. Lower panel shows the percentages of negative cases (grey boxes) and positive cases (white boxes) according to TRAR classification. p-values by Fisher's test (PD-L1: n = 51, PD-L2: n = 25).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Proportions of CD4+CD25+FoxP3+ Treg and CD4+IL-17+ Th17 cells in gingivitis patients and healthy controls. (A) Dot plot int the upperright quadrant represents CD4+ CD25 + T Cells. (B) Isotype control staining of FoxP3. (C) Dot plot represents Treg cells from a healthy control subject (D) Treg cells from a representative patient with gingivitis. (E) CD4+CD25+Foxp3+Treg percentages. (F) Non stimulation of Leukocyte Activation Cocktail of the Th17 cells in a healthy control . (G) Dot plot in the upper right quadrant represents Th17 cells from a healthy control subject. (H) Th17 cells from a representative patient with gingivitis. (I) CD4+IL-17+Th17 percentages. (J) the ratio of Th17/Tre. Data presented are means +- SD. ** P < 0.01, *** P < 0.001 versus healthy control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
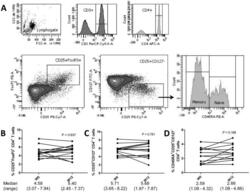
- Experimental details
- Figure 2 Phenotypic analysis of circulating regulatory T cells (Treg). A) Isolated PBMC were analyzed directly ex-vivo by flow cytometry. In the lymphogate, CD3 + CD4 + cells were assessed for the proportions of CD25 + FoxP3 + and CD25 + CD127 - Treg cells. Expression of CD45RA was analyzed to phenotype naive (CD45RA + ) and memory (CD45RA - ) Treg. B-D) The proportions of circulating CD25 + FoxP3 + (B) and CD25 + CD127 - (C)Treg, and of naive Treg (D) before and after vitamin D 3 supplementation (week 0 and 12). Significance was assessed with the Wilcoxon signed ranks comparison test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
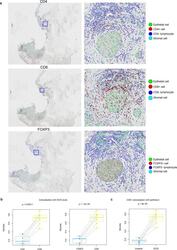
- Experimental details
- Fig. 7 Spatial colocalisation patterns of TIL subset in adjacent DCIS samples in the IHC dataset. a Low power CD4, CD8, FOXP3 IHC images of a sample and high-resolution images of a region of DCIS showing single-cell detection and classification using DRDIN and SCCNN. b Boxplots showing differences in DCIS immune colocalisation score between CD8 + , CD4 + and FOXP3 + cells using paired tests. c Boxplots showing the difference in CD8 + cell colocalisation between DCIS and invasive epithelium indicating differential immune response within the same sample in different compartments. DCIS immune colocalisation score is referred to as Morisita to conserve space in the representation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
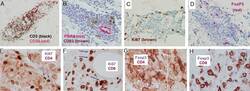
- Experimental details
- FIGURE 5. Cellular changes after repeated vaccination with multipeptide vaccine in IFA, without GM-CSF (Mel44). A patient on the Mel44 vaccine trial underwent vaccine site biopsy 2 weeks after the third vaccine; the formalin-fixed paraffin-embedded biopsy was evaluated by immunohistochemistry, with the stated chromogens: (A) CD3 (DAB-nickel, black) and CD20 (NovaRED), (B) PNAd (Vector-Red) and CD83 (DAB, brown), (C) Ki67 (DAB, brown), (D) FoxP3 (NovaRED); hematoxylin counter-stain is used. Dermal cellular aggregates also were double-stained, without counterstain, for (E) CD4 and Ki67, (F) CD8 and Ki67, (G) CD4 and Fox P3, or (H) CD8 and Foxp3. CD4 and CD8 are stained orange-red (Vector NovaRED) at the cell surface; Ki67 and FoxP3 are nuclear-stained blue-gray (Vector SG). In (E), red arrows indicate CD4 &plus Ki67 &plus cells, and blue dotted arrows indicate CD4-negative Ki67 &plus cells. In (F), blue arrows indicate CD8 &plus Ki67 &plus cells. In (G), 2 separate areas are shown. There is significant diffuse background staining with the CD4 antibody, which is common with this antibody; CD4 &plus cells staining strongly at the cell surface are evident. Double staining CD4 &plus FoxP3 &plus cells are strongly staining for CD4. In (H), Foxp3&plus cells are CD8 negative. These results are representative of 8 patients studied in this way.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 In vivo knockdown of PITPNM3 in CD4 + T cells reverses immunosuppression and inhibits tumor progression in humanized mice. (A) Humanized mice bearing palpable MDA-MB-231 orthotopic xenografts were intraperitoneally injected daily for 14 days with PBS, 1 nmol CD4-aptamer-control siRNA (AsiC-con) or CD4-aptamer-siRNA targeting PITPNM3 (sequence in A , AsiC-PI) to assess the role of PITPNM3 in TI Tregs, and other T cells and tumor control. Experimental schematic is provided in Supplementary information, Figure S9A . (B) Representative immunoblots showing selective knockdown of PITPNM3 protein in PB CD4 + T cells, but not tumor xenografts ( n = 3). (C) PITPNM3 knockdown did not affect the distribution of human CD45 + hematopoietic cells, CD4 + and CD8 + T cells, and CD14 + monocytes in the peripheral blood of humanized mice. Representative flow plots are shown ( n = 3). (D , E) Effect of PITPNM3 knockdown on TI naive CD4 + , Tregs and CD8 + T cell numbers, and apoptosis by TUNEL assay in xenografts. D shows representative immunofluorescence microscopy images. Top row indicates CD4 + naive T cells by arrows; the second row indicates CD4 + CD45RO + Foxp3 - CD4 + memory T cells (yellow arrows) and Foxp3 + Tregs (white arrows). Scale bar, 50 mum. E shows number of cells of each subtype/high power field in eight mice ( ** P < 0.01, *** P < 0.001 compared to PBS group by Student's t -test). (F) Flow cytometry analysis of gated human CD3 + CD4 + cells isolated from xenogra
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Enhanced conversion of XIAP-deficient Treg cells into Foxp3 + IFN-gamma + cells. a , b Increased transition of Xiap -/- tTreg cells into IFN-gamma-producing cells. WT and Xiap -/- tTreg cells (Foxp3-GFP tagged) were isolated by GFP-expression and re-stimulated with plate-bound anti-CD3/CD28 and IL-2 (CD3/CD28), with additional IL-12 ( + IL-12, 50 ng ml -1 ), for 4 days. tTreg cells were then reactivated with TPA/A23187 for 6 h, and the expressions of IFN-gamma in the gated GFP + (representing Foxp3 + ) fraction were determined by intracellular staining ( a ). Quantitation of Foxp3 + IFN-gamma + cells from tTreg cells isolated by either Foxp3-GFP or CD4 + CD25 + expression ( b ), n = 10. *** P < 0.001 for unpaired t -test. c Increased secretion of IFN-gamma by Xiap -/- tTreg cells. Purified WT and Xiap -/- tTreg cells were stimulated as in ( a ), or with additional IL-6 plus IL-1alpha + IL-1beta ( + IL-6/IL-1), and re-activated with TPA/A23187 for 24 h and the levels of IFN-gamma in supernatants was analyzed by ELISA. d , e Enhanced expression and secretion of IFN-gamma by human XIAP-knockdown iTreg cells. Human control and XIAP-knockdown iTreg cells were activated by anti-CD3/CD28 and IL-2, with the addition of IL-12 or IL-6, as indicated, for 4 days. Secretion of IFN-gamma was quantitated ( d ) and intracellular expressions of Foxp3 and IFN-gamma were determined ( e ). f Elevated secretion of IL-17A by Xiap -/- tTreg cells. WT and Xiap -/- tTreg cells were stimulated
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 Anti-IL-6R reduces the expression of IFN-gamma in activated Xiap -/- Treg cells. a , b Anti-IL-6R decreases IFN-gamma expression in re-stimulated Xiap -/- Treg cells. WT and Xiap -/- iTreg cells were stimulated with anti-CD3/CD28 and IL-2, or with additional IL-12 as indicated, in the absence or presence of anti-IL-6R (50 mug ml -1 ) for 4 days. iTreg cells were re-stimulated with TPA/A23187 for 24 h and IFN-gamma production was determined by ELISA ( a ), or reactivated with TPA/A23187 for 5 h and the expressions of Foxp3 and IFN-gamma were determined by intracellular staining ( b ). c , d Anti-IL-6R inhibits IFN-gamma expression in human Treg cells. Control and human XIAP-knockdown iTreg cells were stimulated as in ( a , b ), with additional IL-6 as indicated, and secretion ( c ) or intracellular expression ( d ) of IFN-gamma was determined. e Inability of anti-TNF or anti-IL-1R to inhibit the production of IFN-gamma in activated Xiap -/- Treg cells. WT and Xiap -/- iTreg cells were stimulated, as described in ( a ), in the presence or absence of anti-TNF or anti-IL-1R (50 mug ml -1 each) for 4 days. Treg cells were re-stimulated with TPA/A23187 for 24 h and the secreted IFN-gamma was determined by ELISA. f Anti-IL-6R rescues the impaired suppressive activity of Xiap -/- tTreg cells in vivo. CD45.2 + WT or Xiap -/- tTreg cells were co-transferred with CD45.1 + CD4 + CD25 - effector T cells into male CD45.1 + RagI -/- mice. Anti-IL-6R antibody (500 mug per mouse) was i
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 Combination anti-IL-6R and Xiap -/- Treg cells rescue mice from infection-induced inflammation. a , b Xiap -/- iTreg cells plus anti-IL-6R rescue Xiap -/- mice from C. albicans infection. Male Xiap -/- mice (KO) were intravenously administered with C. albicans and Xiap -/- iTreg cells (1 x 10 6 ) or anti-IL-6R antibody (500 mug) at day 2, or both, as indicated. Survival of mice is presented as a Kaplan-Meier survival curve ( a ). n = 11 for KO, n = 10 for KO + KO iTreg, n = 6 for KO + Ab, n = 9 for KO + KO iTreg + Ab. *** P < 0.001 for Log-rank (Mantel-Cox) Test ( a ). n.s., not significant. The serum level of IL-6 was determined on day 14 after infection ( b ). * P < 0.05 for unpaired t -test ( b ). c , d Anti-IL-6R prevents the conversion of the transferred Xiap -/- Treg cells. CD45.1 + Xiap -/- iTreg cells were transferred into CD45.2 + Xiap -/- mice infected with C. albicans as in ( a ). CD45.1 + T cells were recovered at day 10 post-infection, and the expression of Foxp3 and IFN-gamma was determined ( c ). The Foxp3 + IFN-gamma + fractions in the CD45.1 + Treg cells were quantitated ( d ), n = 5. ** P < 0.01 for unpaired t -test ( d ). e , f Anti-IL-6R reduces kidney neutrophil infiltration and fungal load in C. albicans -infected Xiap -/- mice with KO iTreg cells transfer. Male Xiap -/- mice were infected with C. albicans , as described in ( a ). Kidneys were isolated 14 days post-infection and neutrophils ( e ) and C. albicans titres ( f ) were quantitated. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4 Intra-tumoral Treg Cell Depletion Is Required for the Anti-tumor Activity of Anti-CTLA-4 Mice were treated with 200 mug of anti-CTLA-4 on days 6 and 9 after s.c. inoculation of MCA205 tumor cells (n = 9-21). TILs, LNs, and PBMCs were processed on day 11 and stained for flow cytometry analysis. (A) Percentage of FoxP3 + CD4 + Treg cells from total CD4 + T cells. (B) CD8 + /Treg cell ratio in the indicated sites. Horizontal bars represent the mean. (C) Percentage of Ki67-expressing CD4 + FoxP3 - and CD8 + T cells. (D) Percentage of CD4 + FoxP3 - and CD8 + T cells expressing IFNgamma following re-stimulation with phorbol 12-myristate 13-acetate (PMA) and ionomycin; cumulative data of three separate experiments. Error bars show +-SEM. (E and F) hFcgammaR mice were treated with anti-CTLA-4 on days 6, 9, and 12 after s.c. inoculation of MCA205 (50 mug/dose), MC38 (100 mug/dose) or B16 (200 mug/dose) tumor cells. (E) MCA205 tumor growth in individual hFcgammaR mice in each treatment group. Inset numbers show the fraction of mice with complete long-term response. (F) Kaplan-Meier curves demonstrating survival of hFcgammaR mice for each tumor model. The total number of mice in each treatment group is shown at the right. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001. See also Figure S4 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
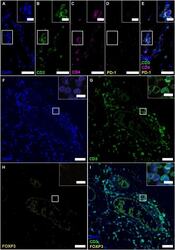
- Experimental details
- Figure 4 Follicle-like structures of SPMS brains exhibit CD3 + CD4 + T cells, which neither express PD-1 nor FOXP3. (A-E) IF staining of CD3, CD4 and PD-1 reveal CD3 + CD4 + PD-1 - T-helper cells in progressive MS. Inserts in the upper right corners show magnification of the white box. (F-I) IF staining of CD3 and FOXP3 on serial sections of a representative meningeal follicle-like structure in SPMS (same region as Figure 3 ). CD3 + T cells, but no FOXP3 + cells were detected. Inserts show magnification of the white box. Scale bars indicate 100 mum, inserts 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
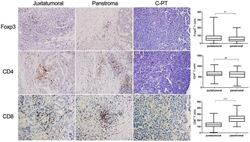
- Experimental details
- Figure 3 IHC analysis of paraffin-embedded tumor sections from patients with PDA or control pancreatic tissue. Magnification: 200x. Comparisons of FoxP3 + cell, CD8 + cell and CD4 + cell infiltrates between the juxtatumoral stroma and the panstroma. IHC analyzed the expression of Foxp3, CD4 and CD8 and statistical analysis the frequencies in juxtatumoral stroma and in panstroma. Comparisons between the two groups were assessed using Student''s t test. NS, not significant; ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
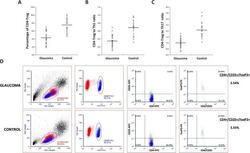
- Experimental details
- Figure 2 T-lymphocyte subset distribution in glaucoma. Mononuclear cells isolated from peripheral blood samples from glaucoma (n = 32) and control (n = 21) groups were analyzed by multicolor flow cytometry for T-cell subset markers. (A) The percentage of CD4+/CD25+/FoxP3+ Tregs (CD4-Tregs) within the entire CD4+ T-cell population was significantly lower in glaucomatous samples than nonglaucomatous controls (ANOVA, P < 0.001). (B, C) The Treg to Th1 or Treg to Th17 ratios were also significantly lower in the glaucoma group than control (ANOVA, P < 0.001). Bars on univariate scatterplots represent the group mean. (D) Shown are representative flow cytometry images after CD4/CD25/FoxP3 immunostaining for Tregs.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
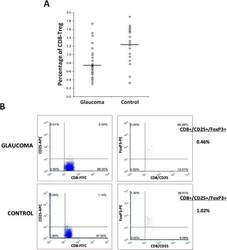
- Experimental details
- Figure 3 T-lymphocyte subset distribution in glaucoma. Mononuclear cells isolated from peripheral blood samples from glaucoma (n = 32) and control (n = 21) groups were analyzed by multicolor flow cytometry for T-cell subset markers. (A) Similar to CD4+ Tregs, the percentage of CD8+/CD25+/FoxP3+ Tregs (CD8-Tregs) was also significantly lower in glaucomatous samples than nonglaucomatous controls (ANOVA, P < 0.001). (B) Shown are representative flow cytometry images after CD8/CD25/FoxP3 immunostaining for Tregs.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 CG-745 increases helper T cells, cytotoxic T cells and natural killer T cells, and decreases Treg: (A) hPBMCs were incubated with CG (CG-745) for 36 hours and a subset of hPBMCs was analyzed using the antibodies indicated in the text; (B) hPBMCs were co-cultured with Huh7, Hep3B, HepG2 or PLC/PRF/5 cells for 36 hours with or without CG, and a subset of hPBMCs was analyzed by Attune Nxt (Invitrogen, USA).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
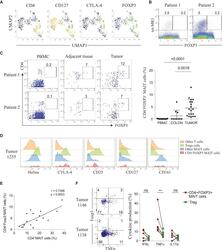
- Experimental details
- Figure 2 Identification of a CD4 + Foxp3 + Subset in Tumor-Infiltrating MAIT Cells (A) UMAP plots showing expression of selected markers on MAIT cells; intensities are red (high), yellow/green (intermediate), blue (low). (B) Tet-MR1 staining plotted against Foxp3 on total T cells from two CRC tumor samples. (C) Representative staining of CD4 + Foxp3 + MAIT cells from PBMC, adjacent tissue, and tumors of two patients, gated on total MAIT cells. Shown are frequencies of Foxp3 expression among total MAIT cells (PBMC = 13, colon = 10, tumor = 19). Data are mean with SD from at least 7 experiments. Mann-Whitney U test. (D) Expression intensities of Treg-related markers and CD161 on different T cells compared with the CD4 + Foxp3 + MAIT subset; one representative tumor sample. (E) Correlation of Foxp3 expression on MAIT cells with CD4 + MAIT cell frequency; n = 20, two-tailed paired t test, Pearson''s correlation. (F) Co-expression of Foxp3 and TNF-alpha gated on total MAIT cells (left) and cytokine production (IFNgamma, TNF-alpha, and IL-17) by CD4 + Foxp3 + tumor-infiltrating MAIT cells compared with Tregs upon 4 h of PMA/ionomycin stimulation (right); n = 6. Data are mean with SD from 2 experiments; two-tailed paired t test. See also Figure S2 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
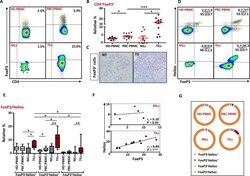
- Experimental details
- Figure 6 FoxP3 and Helios expression in CD4 + T cells PBMC from HD and PBC patients, NILs and TILs were stained for CD3 and CD4 antibodies followed by intracellular staining for FoxP3 and Helios. Live cells were first gated using Fixable Viability Dye 660. ( A ). Representative flow cytometric plots of FoxP3 staining from one cancer patient are shown. ( B ). Scatter plot showing the differences in frequencies of FoxP3 + Tregs between different samples. ( C ). IHC staining of FoxP3 + expression in one NT and TT samples. ( D ). Flow cytometric plots of FoxP3 and Helios co-expression in CD4 + T cells from different samples and whisker plots ( E ) showing differences in various FoxP3 and Helios-expressing Treg subsets. ( F ). Non-parametric Spearman''s test showing correlations between FoxP3 and Helios expressions in NILs and TILs. ( G ). Pie charts show the relative percentages of different FoxP3 and Helios Treg subsets.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot