Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 48-1239-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD123 Monoclonal Antibody (6H6), eFluor™ 450, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 6H6 monoclonal antibody reacts with human CD123, the alpha chain of the IL-3 receptor. This 60-70 kDa transmembrane protein binds to IL-3 with low affinity by itself, and when associated with CD131 (common beta chain) binds IL-3 with high affinity. CD123 is expressed by myeloid precursors, macrophages, dendritic cells, mast cells, basophils, and megakaryocytes. Applications Reported: This 6H6 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 6H6 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. eFluor® 450 is an alternative to Pacific Blue®. eFluor® 450 emits at 445 nm and is excited with the Violet laser (405 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 405 nm; Emission: 445 nm; Laser: Violet Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 6H6
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Unsupervised Analysis of Flow Cytometry Data in a Clinical Setting Captures Cell Diversity and Allows Population Discovery.
Neutrophils Inhibit Synthesis of Mineralized Extracellular Matrix by Human Bone Marrow-Derived Stromal Cells In Vitro.
IL-15 Promotes Polyfunctional NK Cell Responses to Influenza by Boosting IL-12 Production.
Enhancement of cytokine-driven NK cell IFN-γ production after vaccination of HCMV infected Africans.
Communication between Human Dendritic Cell Subsets in Tuberculosis: Requirements for Naive CD4(+) T Cell Stimulation.
Crosstalk between human DC subsets promotes antibacterial activity and CD8+ T-cell stimulation in response to bacille Calmette-Guérin.
Malaria-induced NLRP12/NLRP3-dependent caspase-1 activation mediates inflammation and hypersensitivity to bacterial superinfection.
Frailty in old age is associated with decreased interleukin-12/23 production in response to toll-like receptor ligation.
TNF receptor 1 genetic risk mirrors outcome of anti-TNF therapy in multiple sclerosis.
Multiplexed immunophenotyping of human antigen-presenting cells in whole blood by polychromatic flow cytometry.
CD56-positive haematological neoplasms of the skin: a multicentre study of the Cutaneous Lymphoma Project Group of the European Organisation for Research and Treatment of Cancer.
Epithelial cells trigger frontline immunoglobulin class switching through a pathway regulated by the inhibitor SLPI.
Local activation of the innate immune system in Buruli ulcer lesions.
Expression of the plasmacytoid dendritic cell marker BDCA-2 supports a spectrum of maturation among CD4+ CD56+ hematodermic neoplasms.
Baumgaertner P, Sankar M, Herrera F, Benedetti F, Barras D, Thierry AC, Dangaj D, Kandalaft LE, Coukos G, Xenarios I, Guex N, Harari A
Frontiers in immunology 2021;12:633910
Frontiers in immunology 2021;12:633910
Neutrophils Inhibit Synthesis of Mineralized Extracellular Matrix by Human Bone Marrow-Derived Stromal Cells In Vitro.
Bastian OW, Croes M, Alblas J, Koenderman L, Leenen LPH, Blokhuis TJ
Frontiers in immunology 2018;9:945
Frontiers in immunology 2018;9:945
IL-15 Promotes Polyfunctional NK Cell Responses to Influenza by Boosting IL-12 Production.
Wagstaffe HR, Nielsen CM, Riley EM, Goodier MR
Journal of immunology (Baltimore, Md. : 1950) 2018 Apr 15;200(8):2738-2747
Journal of immunology (Baltimore, Md. : 1950) 2018 Apr 15;200(8):2738-2747
Enhancement of cytokine-driven NK cell IFN-γ production after vaccination of HCMV infected Africans.
Darboe A, Danso E, Clarke E, Umesi A, Touray E, Wegmuller R, Moore SE, Riley EM, Goodier MR
European journal of immunology 2017 Jun;47(6):1040-1050
European journal of immunology 2017 Jun;47(6):1040-1050
Communication between Human Dendritic Cell Subsets in Tuberculosis: Requirements for Naive CD4(+) T Cell Stimulation.
Lozza L, Farinacci M, Bechtle M, Stäber M, Zedler U, Baiocchini A, Del Nonno F, Kaufmann SH
Frontiers in immunology 2014;5:324
Frontiers in immunology 2014;5:324
Crosstalk between human DC subsets promotes antibacterial activity and CD8+ T-cell stimulation in response to bacille Calmette-Guérin.
Lozza L, Farinacci M, Faé K, Bechtle M, Stäber M, Dorhoi A, Bauer M, Ganoza C, Weber S, Kaufmann SH
European journal of immunology 2014 Jan;44(1):80-92
European journal of immunology 2014 Jan;44(1):80-92
Malaria-induced NLRP12/NLRP3-dependent caspase-1 activation mediates inflammation and hypersensitivity to bacterial superinfection.
Ataide MA, Andrade WA, Zamboni DS, Wang D, Souza Mdo C, Franklin BS, Elian S, Martins FS, Pereira D, Reed G, Fitzgerald KA, Golenbock DT, Gazzinelli RT
PLoS pathogens 2014 Jan;10(1):e1003885
PLoS pathogens 2014 Jan;10(1):e1003885
Frailty in old age is associated with decreased interleukin-12/23 production in response to toll-like receptor ligation.
Compté N, Zouaoui Boudjeltia K, Vanhaeverbeek M, De Breucker S, Tassignon J, Trelcat A, Pepersack T, Goriely S
PloS one 2013;8(6):e65325
PloS one 2013;8(6):e65325
TNF receptor 1 genetic risk mirrors outcome of anti-TNF therapy in multiple sclerosis.
Gregory AP, Dendrou CA, Attfield KE, Haghikia A, Xifara DK, Butter F, Poschmann G, Kaur G, Lambert L, Leach OA, Prömel S, Punwani D, Felce JH, Davis SJ, Gold R, Nielsen FC, Siegel RM, Mann M, Bell JI, McVean G, Fugger L
Nature 2012 Aug 23;488(7412):508-511
Nature 2012 Aug 23;488(7412):508-511
Multiplexed immunophenotyping of human antigen-presenting cells in whole blood by polychromatic flow cytometry.
Fung E, Esposito L, Todd JA, Wicker LS
Nature protocols 2010 Feb;5(2):357-70
Nature protocols 2010 Feb;5(2):357-70
CD56-positive haematological neoplasms of the skin: a multicentre study of the Cutaneous Lymphoma Project Group of the European Organisation for Research and Treatment of Cancer.
Assaf C, Gellrich S, Whittaker S, Robson A, Cerroni L, Massone C, Kerl H, Rose C, Chott A, Chimenti S, Hallermann C, Petrella T, Wechsler J, Bagot M, Hummel M, Bullani-Kerl K, Bekkenk MW, Kempf W, Meijer CJ, Willemze R, Sterry W
Journal of clinical pathology 2007 Sep;60(9):981-9
Journal of clinical pathology 2007 Sep;60(9):981-9
Epithelial cells trigger frontline immunoglobulin class switching through a pathway regulated by the inhibitor SLPI.
Xu W, He B, Chiu A, Chadburn A, Shan M, Buldys M, Ding A, Knowles DM, Santini PA, Cerutti A
Nature immunology 2007 Mar;8(3):294-303
Nature immunology 2007 Mar;8(3):294-303
Local activation of the innate immune system in Buruli ulcer lesions.
Peduzzi E, Groeper C, Schütte D, Zajac P, Rondini S, Mensah-Quainoo E, Spagnoli GC, Pluschke G, Daubenberger CA
The Journal of investigative dermatology 2007 Mar;127(3):638-45
The Journal of investigative dermatology 2007 Mar;127(3):638-45
Expression of the plasmacytoid dendritic cell marker BDCA-2 supports a spectrum of maturation among CD4+ CD56+ hematodermic neoplasms.
Jaye DL, Geigerman CM, Herling M, Eastburn K, Waller EK, Jones D
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2006 Dec;19(12):1555-62
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2006 Dec;19(12):1555-62
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD85g (ILT7) PE (Product # 12-5179-42) and Mouse IgG1 kappa Isotype Control eFluor® 450 (Product # 48-4714-82) (left) or Anti-Human CD123 eFluor® 450 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
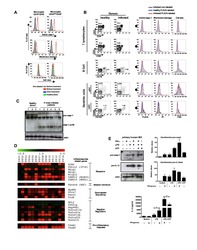
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
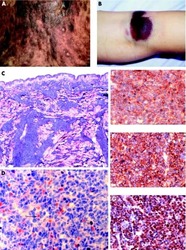
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
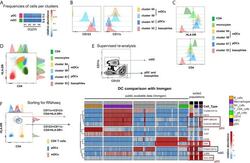
- Experimental details
- Figure 5 Identification of multiple mDC and pDC subsets by MegaClust. (A) Comparison of the different clusters identified by MegaClust as mDCs and pDCs (example for one patient for four time points). (B) Expression level of CD123 and CD11c of the two mDC clusters and the two pDC clusters identified by MegaClust. (C) Expression levels of HLA-DR and CD4 for the two pDC clusters and the two mDC clusters identified by MegaClust. Cluster 53 could be assigned as basophils due to the lack of expression of CD4 and HLA-DR (). The expression of CD4 is shown for CD4 T cells and CD14 + monocyte clusters for comparison. (D) Overlay of CD4 and HLA-DR of MegaClust identified clusters for mDC, pDC, basophils, CD4 and monocytes. (E) mDCs and pDCs were discriminated according to CD11c and CD123 expression in the supervised flow cytometry re-analysis according to the new gating strategy described in Supplementary Figure 5 (one representative patient 0QZW). (F) Representative illustration of HLA-DR and CD4 co-expression on mDCs and pDCs prior to FACS sorting CD11c+CD123 - CD4 + HLA-DR+ mDCs and CD123 + CD11c - CD4 + HLA-DR + were FACS sorted (dotted lines). (G) Gene expression profiles of flow cytometry sorted CD11c + CD123 - CD4 + HLA-DR + mDCs, CD123 + CD11c - CD4 + HLA-DR + CD4 + HLA-DR + pDC and CD14 + CD16 - monocytes from 3 healthy donors are shown in comparison to the RNA expression profile of public available RNAseq datasets (Immgen) indicating the gene expression profile of B cells, mac
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
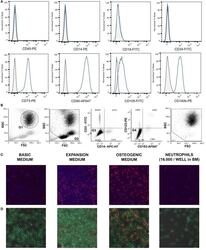
- Experimental details
- Figure 1 (A) Surface antigen expression of bone marrow stromal cells (BMSCs) isolated from the talus bone marrow using flow cytometry. >95% of cells were negative for CD45 and CD14, and >99% of cells were negative for CD19 and CD34. In addition, >95% were positive for CD73, CD90, CD105, and CD140b. Since plastic adherence is a well-established and validated technique to isolate multipotent stromal cells (MSCs), we have only characterized one BMSC donor using flowcytometry instead of all donors. The blue lines are stained cells and the gray lines are negative (unstained) controls. Adapted from Croes et al. ( 25 ). (B) Fluorescence-activated cell sorting (FACS) gating strategy used to isolate granulocytes/neutrophils from peripheral blood leukocytes. Granulocytes were either isolated from unlabeled leukocytes using gate 1 (G1) within the forward/sideward scatter (FSC/SSC). Alternatively, leukocytes were stained using CD3, CD14, CD193, and CD123. Within the FSC/SSC of these labeled cells, debris was first excluded [gate 2 (G2)]. Subsequently, CD3+ cells (lymphocytes) and CD14+ (monocytes) were excluded [gate 3 (G3)]. In addition, CD193+ cells (eosinophils) and CD123+ cells (basophils) were excluded [gate 4 (G4)]. The remaining CD3- CD14- CD193- CD123- cells were defined FACS-sorted neutrophils (G2+, G3+, G4+ sorted neutrophils). Re-analysis of FACS-sorted neutrophils shows adequate exclusion of lymphocytes and monocytes based on their FSC/SSC. (C) Images of BMSCs obtained by arr
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry