Antibody data
- Antibody Data
- Antigen structure
- References [0]
- Comments [0]
- Validations
- Western blot [5]
- Immunocytochemistry [5]
- Immunohistochemistry [3]
- Flow cytometry [3]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-16308-1MG - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- beta Tubulin Loading Control Monoclonal Antibody (BT7R)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat, Canine, Chicken/Avian, Rabbit
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- BT7R
- Vial size
- 1 mg
- Concentration
- 1 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30µg lysate) of A-431 (Lane 1), COS-7 (Lane 2), MDCK (Lane 3), NIH/3T3 (Lane 4), HT-29 (Lane 5), PC-12 (Lane 6), Neuro-2a (Lane 7) and tissue extract of Rat Brain (Lane 8). The blot was probed with Anti- beta Tubulin Mouse Monoclonal Antibody (Product # MA5-16308-1MG, 1:2000 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25µg/mL, 1:4000 dilution). A 52 kDa band corresponding to beta Tubulin was observed across the cell lines and tissue tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
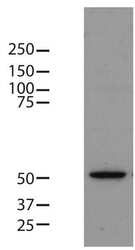
- Experimental details
- Western blot analysis of beta-tubulin was performed by loading 8 µg of mouse embryonic stem cell lysate onto an SDS-PAGE gel. Proteins were transferred to a PVDF membrane and blocked with 5% milk in TBST for one hour at room temperature. The membrane was probed with a beta-tubulin loading control monoclonal antibody (Product # MA5-16308) at a dilution of 1:3000 for two hours at room temperature, washed in TBST, and probed with an HRP-conjugated anti-mouse IgG secondary antibody at a dilution of 1:25,000 for one hour at room temperature. Detection was performed using a chemiluminescent substrate. Data courtesy of the Innovators Program.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of beta-Tubulin was performed by loading 20 µg of THP-1 whole cell lysate per well onto a SDS-PAGE gel. Proteins were transferred to a membrane and blocked with 5% non-fat dry milk in TBST. The membrane was probed with a beta-Tubulin loading control monoclonal antibody (Product # MA5-16308) at a dilution of 1:5000 followed by a HRP-conjugated anti-mouse IgG secondary antibody. Chemiluminescent detection was performed using ECL substrate. Data courtesy of the Innovators Program.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
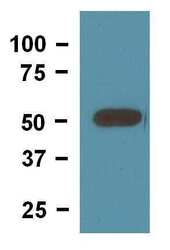
- Experimental details
- Western blot of mouse brain tissue lysate loaded at 10 µg/lane and detected with 0.25 µg/mL (Product # MA5-16308).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30µg lysate) of A-431 (Lane 1), COS-7 (Lane 2), MDCK (Lane 3), NIH/3T3 (Lane 4), HT-29 (Lane 5), PC-12 (Lane 6), Neuro-2a (Lane 7) and tissue extract of Rat Brain (Lane 8). The blot was probed with Anti- beta Tubulin Mouse Monoclonal Antibody (Product # MA5-16308-1MG, 1:2000 dilution) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25µg/mL, 1:4000 dilution). A 52 kDa band corresponding to beta Tubulin was observed across the cell lines and tissue tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
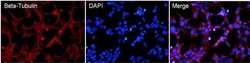
- Experimental details
- Immunofluorescent analysis of Beta-Tubulin (red) in HEK293T cells. Cells fixed in 4% formaldehyde were permeabilized and blocked with 1X PBS containing 5% BSA and 0.3% Triton X-100 for 1 hour at room temperature. Cells were probed with a Beta-Tubulin monoclonal antibody (Product # MA5-16308) at a dilution of 1:100 overnight at 4°C in 1X PBS containing 1% BSA and 0.3% Triton X-100, washed with 1X PBS, and incubated with a fluorophore-conjugated goat anti-mouse IgG secondary antibody at a dilution of 1:200 for 1 hour at room temperature. Nuclei (blue) were stained with DAPI. Images were taken on a Leica DM1000 microscope at 40X magnification. Data courtesy of the Innovators Program.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
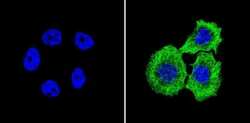
- Experimental details
- Immunofluorescent analysis of Beta-Tubulin (green) showing staining in the in the cytoskeleton of A431 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) in 3% BSA-PBS at a dilution of 1:150 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
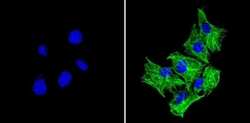
- Experimental details
- Immunofluorescent analysis of Beta-Tubulin (green) showing staining in the in the cytoskeleton of C2C12 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) in 3% BSA-PBS at a dilution of 1:150 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
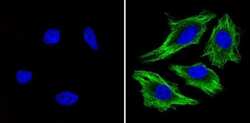
- Experimental details
- Immunofluorescent analysis of Beta-Tubulin (green) showing staining in the in the cytoskeleton of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) in 3% BSA-PBS at a dilution of 1:150 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
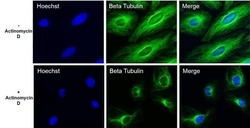
- Experimental details
- Immunofluorescent analysis of Beta- Tubulin (green) in HeLa cells untreated or treated with 1uM Actinomycin D for 19 hours. The cells were fixed with 4% Paraformaldehyde for 15 minutes, permeabilized with 0.1% Triton X-100 for 15 minutes, and blocked with 3% BSA for 30 minutes at room temperature. Cells were stained with a Beta-Tubulin mouse monoclonal antibody (Product # MA5-16308) at a concentration of 2.5 µg/mL in blocking buffer for 1 hour at room temperature, and then incubated with a Goat anti-Mouse IgG (H+L) Secondary Antibody, Alexa Fluor Plus 488 conjugate (Product # A32723) at a dilution of 1:500 for at least 30 minutes at a room temperature in the dark (green). Nuclei (blue) were stained with Hoechst 33342 (Product # 62249). Images were taken on a Thermo Scientific ToxInsight Instrument at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
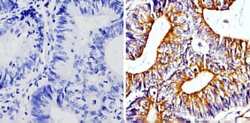
- Experimental details
- Immunohistochemistry analysis of Beta-Tubulin showing staining in the cytoskeleton of paraffin-embedded human colon carcinoma (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
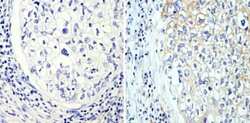
- Experimental details
- Immunohistochemistry analysis of Beta-Tubulin showing staining in the cytoskeleton of paraffin-embedded human lung adenocarcinoma (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
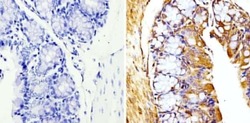
- Experimental details
- Immunohistochemistry analysis of Beta-Tubulin showing staining in the cytoskeleton of paraffin-embedded mouse colon tissue (right) compared with a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Beta-Tubulin loading control antibody (Product # MA5-16308) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
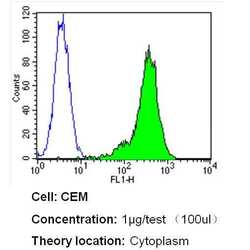
- Experimental details
- Flow cytometry analysis of Beta Tubulin in CEM cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a Beta Tubulin loading control antibody (Product # MA5-16308) at a dilution of 1 µg/test for 40 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
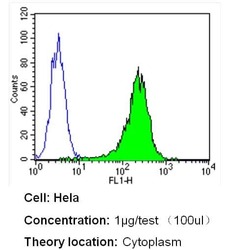
- Experimental details
- Flow cytometry analysis of Beta Tubulin in Hela cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a Beta Tubulin loading control antibody (Product # MA5-16308) at a dilution of 1 µg/test for 40 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
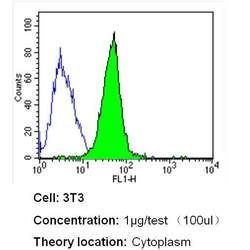
- Experimental details
- Flow cytometry analysis of Beta Tubulin in NIH-3T3 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a Beta Tubulin loading control antibody (Product # MA5-16308) at a dilution of 1 µg/test for 40 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
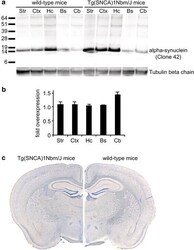
- Experimental details
- Fig. 3 Biochemical and histological analysis of alpha-synuclein expression in wild-type and Tg(SNCA)1Nbm/J mice. Biochemical analysis with an antibody against alpha-synuclein (clone 42) that equally reacts with human as well as mouse alpha-synuclein shows that human alpha-synuclein is only very moderately overexpressed in Tg(SNCA)1Nbm/J mice in comparison to alpha-synuclein expression in wild-type mice. Equal amounts of total protein (5 mug) were loaded onto each lane of the SDS-polyacrylamide gel and show expression of alpha-synuclein in the striatum (Str), cortex (Ctx), hippocampus (Hc), brainstem (Bs), and cerebellum (Cb). Molecular sizes are shown in kilodalton a . The signal for alpha-synuclein was quantified by densitometry from western blots and normalized against tubulin and is shown as fold overexpression of alpha-synuclein in Tg(SNCA)1Nbm/J mice (n = 3) versus wild-type mice (n = 3) b . Immunohistochemistry of mouse brain sections with the same antibody (clone 42) against alpha-synuclein reveals comparable localization of alpha-synuclein in Tg(SNCA)1Nbm/J and wild-type mice c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 RT-PCR (A and C) and Western blot (B and D) analyses of CNTF (A and B) and CNTFRalpha (C and D) expression in the hypothalamus of HFD, control (Ctrl) and CR mice. For Western blots the densitometric analysis was normalized to beta-tubulin expression. In (B) recombinant CNTF (P, 30 ng) loaded together with hypothalamic protein extracts served as a positive control. Mean +- SEM. * P < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
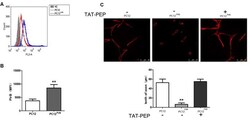
- Experimental details
- Figure 2 Overexpression of PirB inhibited neurites outgrowth in PC12 cells, which was reversed by TAT-PEP treatment ( n = 4). Flow cytometry diagram (A) and quantification (B) of PirB expression in PC12 PirB cells (NC means normal PC12 cells without any treatment). (C) Representative images of PC12 cells, PC12 PirB cells with and without treatment of TAT-PEP. Cells were stained with anti-beta-tubulin antibody. The length of axons was measured using fluorescence microscope. ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2. RJ feeding decreases TORC1 activity in the nprl3 mutant . yw and nprl3 1 /Df flies were collected from normal food or RJ food. (A) Phosphorylated S6K (top), total S6K (middle), and Tubulin (bottom) in male flies were detected using western blot. (B) The relative intensity of phosphorylated S6K band to total S6K band. (C) Phosphorylated 4E-BP (top), total 4E-BP (middle), and Tubulin (bottom) in male flies were detected using western blot. (D) The relative intensity of phosphorylated 4E-BP band to total 4E-BP band. The ratio of p4E-BP/4E-BP in the yw was set as 1. Data are presented as mean+-s.d. ; values are from four independent experiments; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
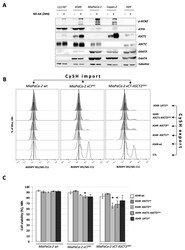
- Experimental details
- Figure 4 Genetic disruption of the ASCT2 cysteine transporter affects cooperation between xCT-expressing and xCT KO cells. ( A ). A variety of xCT wt cells (LS174T, A549, MiaPaCa-2, Capan-2 and HDF) were grown in DMEM media +/- amino acids during 24 h and protein content of different cysteine transporters (ASCT1 (SLC1A4), ASCT2 (SLC1A5), EAAT3 (SLC1A1), EAAT4 (SLC1A6) were analyzed by Western blotting. ( B , C ). Lipid hydroperoxide accumulation and cell viability of MiaPaCa-2 xCT KO and xCT-ASCT2 DKO cells (guest cells--CySH import) in control conditions or co-cultured with 6% A549 wt, ASCT1 KO , ASCT2 KO , ASCT1-ASCT2 DKO , or LAT1 KO (host cells--CySH export). All experiments have been performed in triplicate and the representative blots and histograms are shown. Bar graph shows mean +- SEM; n = 3; *, p < 0.05, comparison with WT control group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2. Hypoxia decreases ACE2 and NRP1 protein levels on Vero E6 cells. ( a ) Relative ACE2 gene expression on Vero E6 and NCI-H460 measured by Q-PCR ( n = 3, unpaired t test). ( b ) (Left) Western blot of HIF-1alpha, ACE2 and beta-actin on NCI-H460 and Vero E6 cells cultured under normoxia (21% O 2 ) or hypoxia (1% O 2 ) for the indicated time points. (Right) Relative quantification of ACE2 protein expression by densitometry ( n = 2, one-way ANOVA). ( c ) Relative gene expression of PGK1 (left) and ACE2 (right) on Vero E6 cells cultured under normoxia or hypoxia for 24 h ( n = 3, unpaired t test). ( d ) (Left) Western blot of HIF-1alpha, NRP1, TMPRSS2 and beta-tubulin on Vero E6 and NCI-H460 cells cultured under normoxia or hypoxia for 48 h. (Right) Relative quantification of NRP1 and TMPRSS2 proteins by densitometry ( n = 3, 2-way ANOVA). Error bars represent SEM and asterisks represent p values (*
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
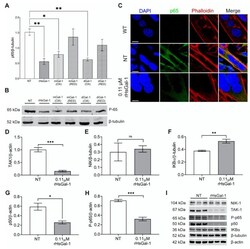
- Figure 2 In vitro treatment with rHsGal-1 modulates inflammatory response through the NF-kappaB pathway. ( A ) Quantification of expression levels of p65 (normalized to beta-tubulin) in 48 h A/J -/- NT or 0.11 muM rHsGal-1 treated myotubes. ( B ) Western blot images showing the p65 expression in NT or 0.11 muM rHsGal-1 48 h A/J -/- treated myotubes. ( C ) Representative images of WT and NT or 0.11 muM rHsGal-1 48 h A/J -/- treated myotubes cultured and immunostained with p65 (green), Phalloidin (red), and DAPI (blue). ( D - H ) Western blot quantification of 48 h NT or 0.11 muM rHsGal-1treated myotubes expressing levels of TAK1 ( D ), NIK ( E ), IKBalpha ( F ), p50 ( G ), and P-p65 ( H ). ( I ) Western blot images of 48 h NT or 0.11 muM rHsGal-1treated myotubes expressing NF-kappaB inflammatory subunits quantified in D-H. n = 3 in each group. A. * p < 0.05 and ** p < 0.01 NT vs. all forms of Gal-1. D-H. * p < 0.05, ** p < 0.01, *** p < 0.001 NT vs. rHsGal-1. Data are represented as SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
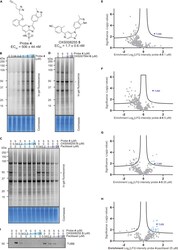
- Target identification using chemical probe and chemoproteomics (A) Chemical structures of probe 4 and OXS008255 5 . EC 50 values for CD11b upregulation are represented as means +- SEM. In-gel fluorescence showing: (B) dose-dependent labelling by probe 4 ; (C) competition of OXS008255 5 and paclitaxel with probe 4 ; (D) competition of inactive analogue OXS007564 6 with probe 4 . Coomassie stain shows equal protein loading on each gel. Volcano plots showing significantly enriched proteins in the pull-down experiment by probe 4 compared to competition with: (E) 1 muM of OXS008255 5 ; (F) 5 muM of OXS008255 5 ; (G) 25 muM of OXS008255 5 ; (H) 25 muM of paclitaxel. For (E)-(H) a student's t-test (FDR = 0.05; S0 = 0.1) was performed between the active probe sample and each DMSO control, and between active probe sample and probe/parent competition samples. Full list of proteins for each volcano plot is available in the supplementary information; (I) Confirmation of tubulin beta chain enrichment by pull-down and immunoblotting. Western blot is cropped, full western blot available in Figure S5 . See also Figure S5 and Tables S3 , S4-S7 , and S8 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
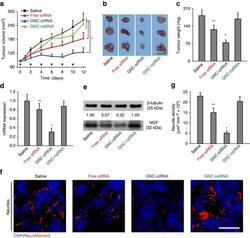
- Figure 6 The antitumour and gene knockdown effects of GNC-siRNA complex in subcutaneous pancreatic tumours. Panc-1 cells were injected into the flank of Balb/c nude mice to form subcutaneous tumours. When the tumours reached about 5 mm in diameter, the animals received peritumoral injections of various formulations every 2 days for six injections. ( a ) Tumour growth curve during the treatments. The black arrows indicated the days of injection. ( b ) Ex vivo tumour image and ( c ) tumour weight at the end of experiment. Scale bar, 1 cm. ( d ) Expression level of NGF mRNA and ( e ) NGF protein level in subcutaneous tumours. ( f ) IF staining of neurites in tumour tissues with various siRNA treatments. Neurites were stained with neurofilament antibody (red), the cell nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI; blue). Scale bars, 20 mum. ( g ) Quantification of neurite density in the subcutaneous tumours by counting the neurite area positive to neurofilament antibody per unit area. Mean+-s.d. ( n =6 per group). Significant difference was from the saline control, * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
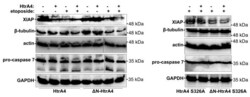
- Figure 9 HtrA4 promotes degradation of XIAP and, less efficiently, of beta-tubulin, actin, and pro-caspase 7. The A549 cells with exogenous HtrA4, DeltaN-HtrA4, or their inactive variants (+) induced by adding doxycycline to the medium were treated with 15 uM etoposide for 48 h. Control cells (-) were incubated with etoposide but without doxycycline. The cells and medium were collected and probed with specific antibodies. Representative blots are presented. Densitometric analyses of the immunoblotting results are shown in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 4 EIPR-1 interacts with members of the GARP and EARP complexes. (A) The rat ortholog of EIPR-1 is expressed in 832/13 cells. Left and middle: RT-PCR shows that rat orthologs of CCCP-1, RAB-2, EIPR-1 and RUND-1 are expressed in 832/13 cells. All bands are of the predicted size for the full-length cDNA except for CCCP1, which is truncated and corresponds to the N-terminal half of the gene. (B) EIPR1 interacts with members of the GARP and EARP complexes. List of top hits from mass spectrometry of a pulldown of rEIPR1::GFP in 832/13 cells after subtracting hits found in GFP control pulldowns. # seq = number of unique peptides from each protein. All proteins with more than 5 unique peptides are shown from one of two independent experiments. More complete data tables for both experiments are shown in S5A Fig . (C) EIPR1 interacts with VPS51 and VPS50. EGFP-tagged rat EIPR1 or EGFP was coexpressed with mCherry-tagged rat VPS51 in 832/13 cells. Immunoprecipitation of EIPR1::EGFP pulled down VPS51::mCherry and endogenous VPS50. Immunoprecipitation of untagged EGFP did not pull down VPS51::mCherry or VPS50. IN: input; IP: immunoprecipitation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
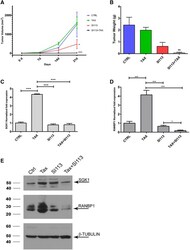
- Figure 5 SI113 sensitizes A2780TC xenografts in immunocompromised mice to paclitaxel. (A) Tumor growth curves of animals treated with vehicle alone (control) SI113, paclitaxel, or both agents together. The data are expressed as mm 3 +- standard error (SE) and were evaluated by one-way ANOVA. (B) The mice were sacrificed 21 days after the beginning of the treatment, and their tumors were excised and weighed. The histogram shows the tumor weight (in grams) of each experimental arm expressed in means +- SEs. The differences among the groups were analyzed by one-way ANOVA. (C, D) Quantitative RT-PCR analysis of SGK1 (left) and RANBP1 (right) mRNA levels in tumors from the above-mentioned four experimental arms. The results (normalized to the HPRT1 levels) are expressed as the average relative fold expression values +- SDs from triplicate experiments and were evaluated by one-way ANOVA. (E) Western blot of proteins extracted from A2780TC-derived xenografts treated as indicated. Cell extracts were separated by SDS-PAGE and detected with SGK1 and RANBP1 antibodies. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
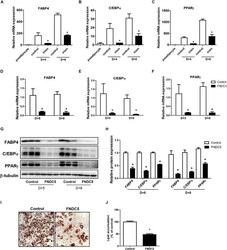
- FIGURE 2 Effect of recombinant irisin and FNDC5 overexpression on adipogenesis. (A-C) 3T3-L1 cells were treated with or without 100 ng/mL irisin from D-2 to D+6 of adipogenesis. Gene expressions of transcription factors were measured by real-time PCR and normalized to 18S rRNA. (D-J) 3T3-L1 preadipocytes were transfected with control or plasmid expressing FNDC5. Two days confluent cells were induced to differentiate and were harvested at day + 5 and day + 8. (D-F) Gene expressions of PPARgamma, CEBPalpha, and FABP4 were analyzed by real-time PCR and normalized to 18S. (G,H) Western blot analysis of transcription factors. (I,J) Representative pictures and quantification of Oil red O staining in FNDC5-overexpressed cells at day 8. * P < 0.05 compared with control at each time-point.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 3 Effect of recombinant irisin treatment on Wnt expression. (A-C) Gene expression analysis of Wnt ligands during 3T3-L1 adipocyte differentiation by real-time PCR. Quantification were normalized to 18S rRNA * P < 0.05 compared with D-0. (D-F) Gene expression of analysis of Wnt ligands at early stage of adipogenesis by real-time PCR. * P < 0.05 compared with D-2, + P < 0.05 compared with D-0. (G-J) 3T3-L1 cells were treated with 100 ng/mL recombinant irisin at D-2. (G-I) Wnt gene expressions were measured by real-time PCR and normalized to 18S rRNA. (J) Protein expression of Wnt10a with or without recombinant irisin treatment were analyzed by Western blot, * P < 0.05 compared with control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
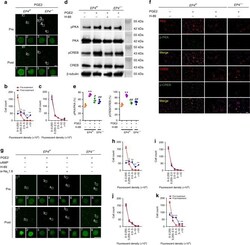
- Fig. 6 PGE2 stimulates PKA/CREB signaling through EP4 to induce sodium influx. a Representative images of sodium indicator (green) analysis pre- and post-PGE2 (20 muM) stimulation for 5 min in primary DRG neurons from EP4 f/f or EP4 -/- mice, indicating sodium influx. Scale bar, 100 mum. Magnification, scale bar, 20 mum. b , c Quantitative analysis of the fluorescent density distribution of the 1st ( b ) and 2nd ( c ) column in ( a ). * p < 0.05, ** p < 0.01 compared with the corresponding pre-treatment group. n = 3 per group. d Western blots of the phosphorylation of PKA and CREB in primary DRG neurons treated with PGE2 (20 muM) for 30 min and PKA inhibitor (H-89, 10 muM) for 60 min. e Quantitative analysis of ( d ). ** p < 0.01 compared with the negative control group from EP4 f/f mice. # p < 0.05, ## p < 0.01 compared with only PGE2 treatment group from EP4 f/f mice n = 3 per group. f First to third row, representative images of immunofluorescent analysis of PKA (red), p-PKA (green) staining, and DAPI (blue) staining of nuclei; 4th to 6th row, representative images of immunofluorescent analysis of CREB (red), p-CREB (green) staining, and DAPI (blue) staining of nuclei pre- and post-PGE2 (20 muM) stimulation combined with H-89 (10 muM) in primary DRG neurons from EP4 f/f or EP4 -/- mice. Scale bar, 100 mum. g Representative images of sodium indicator (green) analysis pre- and post-PGE2 (20 muM) stimulation combined with cAMP, PKA inhibitor (H-89), or siRNA for Na v 1.8 (si-
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA