Antibody data
- Antibody Data
- Antigen structure
- References [10]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- ABIN5611572 - Provider product page

- Provider
- antibodies-online
- Product name
- anti-Heat Shock 70kDa Protein 9 (Mortalin) (HSPA9) (C-Term) antibody
- Antibody type
- Polyclonal
- Description
- Immunoaffinity Chromatography
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Epitope
- C-Term
- Isotype
- IgG
- Vial size
- 50 μL
- Storage
- Store the antibody (in aliquots) at -20°C or lower. Avoid freeze-thaw cycles.
Submitted references Upregulation of mortalin/mthsp70/Grp75 contributes to human carcinogenesis.
Overexpressed mortalin (mot-2)/mthsp70/GRP75 and hTERT cooperate to extend the in vitro lifespan of human fibroblasts.
Inactivation of tumor suppressor p53 by mot-2, a hsp70 family member.
Mitochondrial Hsp70/MIM44 complex facilitates protein import.
On the cytosolic and perinuclear mortalin: an insight by heat shock.
Identification of a novel member of mouse hsp70 family. Its association with cellular mortal phenotype.
Sequential action of mitochondrial chaperones in protein import into the matrix.
The two mammalian mitochondrial stress proteins, grp 75 and hsp 58, transiently interact with newly synthesized mitochondrial proteins.
The Escherichia coli DnaK chaperone, the 70-kDa heat shock protein eukaryotic equivalent, changes conformation upon ATP hydrolysis, thus triggering its dissociation from a bound target protein.
A member of the Hsp70 family is localized in mitochondria and resembles Escherichia coli DnaK.
Wadhwa R, Takano S, Kaur K, Deocaris CC, Pereira-Smith OM, Reddel RR, Kaul SC
International journal of cancer 2006 Jun 15;118(12):2973-80
International journal of cancer 2006 Jun 15;118(12):2973-80
Overexpressed mortalin (mot-2)/mthsp70/GRP75 and hTERT cooperate to extend the in vitro lifespan of human fibroblasts.
Kaul SC, Yaguchi T, Taira K, Reddel RR, Wadhwa R
Experimental cell research 2003 May 15;286(1):96-101
Experimental cell research 2003 May 15;286(1):96-101
Inactivation of tumor suppressor p53 by mot-2, a hsp70 family member.
Wadhwa R, Takano S, Robert M, Yoshida A, Nomura H, Reddel RR, Mitsui Y, Kaul SC
The Journal of biological chemistry 1998 Nov 6;273(45):29586-91
The Journal of biological chemistry 1998 Nov 6;273(45):29586-91
Mitochondrial Hsp70/MIM44 complex facilitates protein import.
Schneider HC, Berthold J, Bauer MF, Dietmeier K, Guiard B, Brunner M, Neupert W
Nature 1994 Oct 27;371(6500):768-74
Nature 1994 Oct 27;371(6500):768-74
On the cytosolic and perinuclear mortalin: an insight by heat shock.
Kaul SC, Wadhwa R, Komatsu Y, Sugimoto Y, Mitsui Y
Biochemical and biophysical research communications 1993 May 28;193(1):348-55
Biochemical and biophysical research communications 1993 May 28;193(1):348-55
Identification of a novel member of mouse hsp70 family. Its association with cellular mortal phenotype.
Wadhwa R, Kaul SC, Ikawa Y, Sugimoto Y
The Journal of biological chemistry 1993 Mar 25;268(9):6615-21
The Journal of biological chemistry 1993 Mar 25;268(9):6615-21
Sequential action of mitochondrial chaperones in protein import into the matrix.
Manning-Krieg UC, Scherer PE, Schatz G
The EMBO journal 1991 Nov;10(11):3273-80
The EMBO journal 1991 Nov;10(11):3273-80
The two mammalian mitochondrial stress proteins, grp 75 and hsp 58, transiently interact with newly synthesized mitochondrial proteins.
Mizzen LA, Kabiling AN, Welch WJ
Cell regulation 1991 Feb;2(2):165-79
Cell regulation 1991 Feb;2(2):165-79
The Escherichia coli DnaK chaperone, the 70-kDa heat shock protein eukaryotic equivalent, changes conformation upon ATP hydrolysis, thus triggering its dissociation from a bound target protein.
Liberek K, Skowyra D, Zylicz M, Johnson C, Georgopoulos C
The Journal of biological chemistry 1991 Aug 5;266(22):14491-6
The Journal of biological chemistry 1991 Aug 5;266(22):14491-6
A member of the Hsp70 family is localized in mitochondria and resembles Escherichia coli DnaK.
Leustek T, Dalie B, Amir-Shapira D, Brot N, Weissbach H
Proceedings of the National Academy of Sciences of the United States of America 1989 Oct;86(20):7805-8
Proceedings of the National Academy of Sciences of the United States of America 1989 Oct;86(20):7805-8
No comments: Submit comment
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
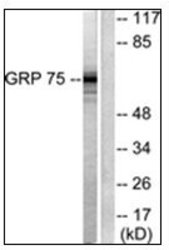
- Experimental details
- WB
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
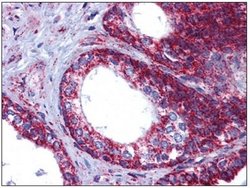
- Experimental details
- IHC
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry