Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- A-6474 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NMDAR2B Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Other
- Description
- This antibody is specific for the ~180 kDa 2B subunit from rat, mouse and human. Storage and reconstitution: when stored at -20ºC or below, the lyophilized antibody should retain full activity for over one year. Reconstitute using 50 µL PBS, pH 7.4, to yield a 0.2 mg/mL solution. If storing at 0-4ºC, add sodium azide to a final concentration of 2mM. For longer storage, divide the solution into aliquots and freeze at -20ºC, avoiding freeze/thaw cycles.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 10 µg
- Storage
- -20°C
Submitted references Effects of RORγt overexpression on the murine central nervous system.
Exo70 intracellular redistribution after repeated mild traumatic brain injury.
Interfering of the Reelin/ApoER2/PSD95 Signaling Axis Reactivates Dendritogenesis of Mature Hippocampal Neurons.
Mechanisms of Functional Hypoconnectivity in the Medial Prefrontal Cortex of Mecp2 Null Mice.
Chronic copper exposure causes spatial memory impairment, selective loss of hippocampal synaptic proteins, and activation of PKR/eIF2α pathway in mice.
PSD95 suppresses dendritic arbor development in mature hippocampal neurons by occluding the clustering of NR2B-NMDA receptors.
Pre- and postsynaptic localization of NMDA receptor subunits at hippocampal mossy fibre synapses.
Subunit-specific modulation of [(3)H]MK-801 binding to NMDA receptors mediated by dopamine receptor ligands in rodent brain.
Cholinergic dysfunction alters synaptic integration between thalamostriatal and corticostriatal inputs in DYT1 dystonia.
Neurotoxic lupus autoantibodies alter brain function through two distinct mechanisms.
Regulation of NMDA receptor subunits and nitric oxide synthase expression during cocaine withdrawal.
Sasaki T, Nagata R, Takahashi S, Takei Y
Neuropsychopharmacology reports 2021 Mar;41(1):102-110
Neuropsychopharmacology reports 2021 Mar;41(1):102-110
Exo70 intracellular redistribution after repeated mild traumatic brain injury.
Lira M, Zamorano P, Cerpa W
Biological research 2021 Feb 16;54(1):5
Biological research 2021 Feb 16;54(1):5
Interfering of the Reelin/ApoER2/PSD95 Signaling Axis Reactivates Dendritogenesis of Mature Hippocampal Neurons.
Ampuero E, Jury N, Härtel S, Marzolo MP, van Zundert B
Journal of cellular physiology 2017 May;232(5):1187-1199
Journal of cellular physiology 2017 May;232(5):1187-1199
Mechanisms of Functional Hypoconnectivity in the Medial Prefrontal Cortex of Mecp2 Null Mice.
Sceniak MP, Lang M, Enomoto AC, James Howell C, Hermes DJ, Katz DM
Cerebral cortex (New York, N.Y. : 1991) 2016 May;26(5):1938-1956
Cerebral cortex (New York, N.Y. : 1991) 2016 May;26(5):1938-1956
Chronic copper exposure causes spatial memory impairment, selective loss of hippocampal synaptic proteins, and activation of PKR/eIF2α pathway in mice.
Ma Q, Ying M, Sui X, Zhang H, Huang H, Yang L, Huang X, Zhuang Z, Liu J, Yang X
Journal of Alzheimer's disease : JAD 2015;43(4):1413-27
Journal of Alzheimer's disease : JAD 2015;43(4):1413-27
PSD95 suppresses dendritic arbor development in mature hippocampal neurons by occluding the clustering of NR2B-NMDA receptors.
Bustos FJ, Varela-Nallar L, Campos M, Henriquez B, Phillips M, Opazo C, Aguayo LG, Montecino M, Constantine-Paton M, Inestrosa NC, van Zundert B
PloS one 2014;9(4):e94037
PloS one 2014;9(4):e94037
Pre- and postsynaptic localization of NMDA receptor subunits at hippocampal mossy fibre synapses.
Berg LK, Larsson M, Morland C, Gundersen V
Neuroscience 2013 Jan 29;230:139-50
Neuroscience 2013 Jan 29;230:139-50
Subunit-specific modulation of [(3)H]MK-801 binding to NMDA receptors mediated by dopamine receptor ligands in rodent brain.
Wigestrand MB, Fonnum F, Ivar Walaas S
Neurochemistry international 2012 Jul;61(2):266-76
Neurochemistry international 2012 Jul;61(2):266-76
Cholinergic dysfunction alters synaptic integration between thalamostriatal and corticostriatal inputs in DYT1 dystonia.
Sciamanna G, Tassone A, Mandolesi G, Puglisi F, Ponterio G, Martella G, Madeo G, Bernardi G, Standaert DG, Bonsi P, Pisani A
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Aug 29;32(35):11991-2004
The Journal of neuroscience : the official journal of the Society for Neuroscience 2012 Aug 29;32(35):11991-2004
Neurotoxic lupus autoantibodies alter brain function through two distinct mechanisms.
Faust TW, Chang EH, Kowal C, Berlin R, Gazaryan IG, Bertini E, Zhang J, Sanchez-Guerrero J, Fragoso-Loyo HE, Volpe BT, Diamond B, Huerta PT
Proceedings of the National Academy of Sciences of the United States of America 2010 Oct 26;107(43):18569-74
Proceedings of the National Academy of Sciences of the United States of America 2010 Oct 26;107(43):18569-74
Regulation of NMDA receptor subunits and nitric oxide synthase expression during cocaine withdrawal.
Loftis JM, Janowsky A
Journal of neurochemistry 2000 Nov;75(5):2040-50
Journal of neurochemistry 2000 Nov;75(5):2040-50
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
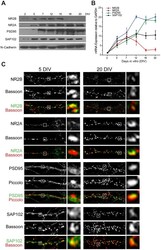
- Figure 3 Expression of NMDAR subunits NR2A and NR2B, and MAGUKs PSD95 and SAP102, at different stages of development. A , Total protein extracts were obtained from hippocampal neurons at 2, 5, 7, 12, 15, 18, and 20 DIV and immunoblotted with antibodies against NR2B, NR2A, PSD95, SAP102, or N-cadherin as loading control. B , Results of qRT-PCR run with primers designed to determine mRNA levels for NR2B, NR2A, PSD95, or SAP102 at the same developmental stages; results were normalized against levels of GAPDH mRNA. Note that NR2B expression decreases with development, while NR2A and PSD95 increases. Expression of SAP102 remains relatively stable at these developmental stages. C , Immunodetection of endogenous NR2B, NR2A, PSD95, or SAP102 (green) and co-localization with presynaptic marker Bassoon or Piccolo (red) at 5 and 20 DIV. Insets show magnified views of boxed areas showing examples of synaptic clusters located in close apposition to presynaptic clusters.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Knockdown of PSD95 promote NR2B clustering in mature hippocampal neurons. A , Cultured hippocampal neurons were transfected with a magnetofection-based method at 15 DIV with GFP plus shRNA-PSD95. At 20 DIV, cultures were fixed and stained with anti-PSD95 antibody. Confocal images (one optical section of 1 mum) show transfected cell (green) and PSD95-IR (red). Scale bar: 20 mum. B , Representative images of neuronal branches expressing GFP alone (Control; left images) or GFP plus shRNA-PSD95 (shRNA-PSD95; right images) and immunostained against PSD95 (red). Note that shRNA-PSD95 causes a significant loss of mature spines and an increase in the number of filopodia-like structures. Images were constructed from a stack of optical section (total 1.5 mum). Scale bar: 5 mum. C , Representative images of neuronal branches expressing GFP plus NR2B (NR2B; left images) or GFP plus NR2B and shRNA-PSD95 (NR2B+shRNA-PSD95; right images) and immunostained against NR2B (red). Images were constructed from a stack of optical section (total 1.5 mum). Scale bar: 5 mum. D , Quantification of total NR2B cluster number per 100 mum of neurite length, in NR2B neurons or those expressing both NR2B and shRNA-PSD95. For each condition at least 9 neurons were analyzed. Figures show Mean +- SEM. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 mTBI increases exocyst complex assembly and interaction between Exo70 and GluN2B in the PSD fraction. Two-month-old male mice were subjected to mTBI and hippocampal PSD/nonPSD fractions were obtained. 50 and 300 µg of PSD (a) and nonPSD (c) protein samples, respectively, were immunoprecipitated using a specific Exo70 polyclonal antibody. Beads were recovered by brief centrifugation. Samples were resolved in 6% SDS-PAGE and transferred to PVDF membranes. Signal intensity was normalized with Exo70 band intensity. Both hippocampi of each mouse were used. Membranes were stripped and tested again with the indicated antibodies. b , d GluN2A, GluN2B, GluR1, GluR2, Sec6, and Sec10 densitometric analysis from PSD and nonPSD immunoprecipitations. The analysis shows the assembly level of the exocyst complex and Exo70-GluN2B increased interaction after mTBI. Values represent means +- SEM, n = 3 mice per experimental group. Statistical differences were determined by an unpaired t-test comparing Sham and mTBI. * p < 0.05
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 2 (A) Representative images of immunohistochemistry of Iba1 in the DG (left) and CA1 (right) regions of WT (upper panel) and RORgammat Tg (lower panel) mice. (B) Semi-quantitative results of Iba1 immunofluorescence in DG (left) and CA1 (right) of WT and RORgammat Tg mice (n = 3 per group, * P < 0.05). (C) Density of Iba1 + microglia in the DG (left) and CA1 (right) of WT and RORgammat Tg mice (n = 3 per group, * P < 0.05). Microglial activity and density were significantly lower in the DG of RORgammat Tg compared with WT mice. (D) Representative images of immunohistochemistry for GFAP in DG (left) and CA1 (right) regions of WT (upper panel) and RORgammat Tg (lower panel) mice. (E) Semi-quantitative results of GFAP immunofluorescence in the DG (left) and CA1 (right) of WT and RORgammat Tg mice (n = 3 per group). (F) Density of GFAP + astrocytes in the DG (left) and CA1 (right) of WT and RORgammat Tg mice (n = 3 per group). There were no significant differences in astrocyte activity or density between RORgammat Tg and WT mice. (G) Representative images of immunohistochemistry of DCX (green) and DAPI (blue) in DG regions of WT (upper panel) and RORgammat Tg (lower panel) mice. (H) Density of DCX + immature neurons in the DG of WT and RORgammat Tg mice (n = 4 per group). (I) Western blot of synaptic molecules (NR2A, NR2B, PSD-93, and PSD-95) in the hippocampi of WT (left) and RORgammat Tg (right) mice. For normalization of proteins in each sample, alpha-tubulin was used as
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Other assay
Other assay