Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [1]
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 36-9700 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-EGFR (Tyr1086) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 50 µg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Spatial Metabolomics Identifies Distinct Tumor-Specific Subtypes in Gastric Cancer Patients.
Overcoming EGFR(G724S)-mediated osimertinib resistance through unique binding characteristics of second-generation EGFR inhibitors.
Afatinib restrains K-RAS-driven lung tumorigenesis.
Effects of general anesthesia with or without epidural block on tumor metastasis and mechanisms.
Cytoplasmic p53 couples oncogene-driven glucose metabolism to apoptosis and is a therapeutic target in glioblastoma.
Tumor-penetration and antitumor efficacy of cetuximab are enhanced by co-administered iRGD in a murine model of human NSCLC.
Preclinical evaluation of afatinib (BIBW2992) in esophageal squamous cell carcinoma (ESCC).
Resistance to EGF receptor inhibitors in glioblastoma mediated by phosphorylation of the PTEN tumor suppressor at tyrosine 240.
Biomarker analysis of cetuximab plus oxaliplatin/leucovorin/5-fluorouracil in first-line metastatic gastric and oesophago-gastric junction cancer: results from a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie (AIO).
Epithelial-to-mesenchymal transition in the development and progression of adenocarcinoma and squamous cell carcinoma of the lung.
Chronic pharmacologic inhibition of EGFR leads to cardiac dysfunction in C57BL/6J mice.
Enhanced activation of epidermal growth factor receptor caused by tumor-derived E-cadherin mutations.
Clinicopathologic characteristics of the EGFR gene mutation in non-small cell lung cancer.
Wang J, Kunzke T, Prade VM, Shen J, Buck A, Feuchtinger A, Haffner I, Luber B, Liu DHW, Langer R, Lordick F, Sun N, Walch A
Clinical cancer research : an official journal of the American Association for Cancer Research 2022 Jul 1;28(13):2865-2877
Clinical cancer research : an official journal of the American Association for Cancer Research 2022 Jul 1;28(13):2865-2877
Overcoming EGFR(G724S)-mediated osimertinib resistance through unique binding characteristics of second-generation EGFR inhibitors.
Fassunke J, Müller F, Keul M, Michels S, Dammert MA, Schmitt A, Plenker D, Lategahn J, Heydt C, Brägelmann J, Tumbrink HL, Alber Y, Klein S, Heimsoeth A, Dahmen I, Fischer RN, Scheffler M, Ihle MA, Priesner V, Scheel AH, Wagener S, Kron A, Frank K, Garbert K, Persigehl T, Püsken M, Haneder S, Schaaf B, Rodermann E, Engel-Riedel W, Felip E, Smit EF, Merkelbach-Bruse S, Reinhardt HC, Kast SM, Wolf J, Rauh D, Büttner R, Sos ML
Nature communications 2018 Nov 7;9(1):4655
Nature communications 2018 Nov 7;9(1):4655
Afatinib restrains K-RAS-driven lung tumorigenesis.
Moll HP, Pranz K, Musteanu M, Grabner B, Hruschka N, Mohrherr J, Aigner P, Stiedl P, Brcic L, Laszlo V, Schramek D, Moriggl R, Eferl R, Moldvay J, Dezso K, Lopez-Casas PP, Stoiber D, Hidalgo M, Penninger J, Sibilia M, Győrffy B, Barbacid M, Dome B, Popper H, Casanova E
Science translational medicine 2018 Jun 20;10(446)
Science translational medicine 2018 Jun 20;10(446)
Effects of general anesthesia with or without epidural block on tumor metastasis and mechanisms.
Yang B, Qian F, Li W, Li Y, Han Y
Oncology letters 2018 Apr;15(4):4662-4668
Oncology letters 2018 Apr;15(4):4662-4668
Cytoplasmic p53 couples oncogene-driven glucose metabolism to apoptosis and is a therapeutic target in glioblastoma.
Mai WX, Gosa L, Daniels VW, Ta L, Tsang JE, Higgins B, Gilmore WB, Bayley NA, Harati MD, Lee JT, Yong WH, Kornblum HI, Bensinger SJ, Mischel PS, Rao PN, Clark PM, Cloughesy TF, Letai A, Nathanson DA
Nature medicine 2017 Nov;23(11):1342-1351
Nature medicine 2017 Nov;23(11):1342-1351
Tumor-penetration and antitumor efficacy of cetuximab are enhanced by co-administered iRGD in a murine model of human NSCLC.
Zhang Y, Yang J, Ding M, Li L, Lu Z, Zhang Q, Zheng J
Oncology letters 2016 Nov;12(5):3241-3249
Oncology letters 2016 Nov;12(5):3241-3249
Preclinical evaluation of afatinib (BIBW2992) in esophageal squamous cell carcinoma (ESCC).
Wong CH, Ma BB, Hui CW, Tao Q, Chan AT
American journal of cancer research 2015;5(12):3588-99
American journal of cancer research 2015;5(12):3588-99
Resistance to EGF receptor inhibitors in glioblastoma mediated by phosphorylation of the PTEN tumor suppressor at tyrosine 240.
Fenton TR, Nathanson D, Ponte de Albuquerque C, Kuga D, Iwanami A, Dang J, Yang H, Tanaka K, Oba-Shinjo SM, Uno M, Inda MM, Wykosky J, Bachoo RM, James CD, DePinho RA, Vandenberg SR, Zhou H, Marie SK, Mischel PS, Cavenee WK, Furnari FB
Proceedings of the National Academy of Sciences of the United States of America 2012 Aug 28;109(35):14164-9
Proceedings of the National Academy of Sciences of the United States of America 2012 Aug 28;109(35):14164-9
Biomarker analysis of cetuximab plus oxaliplatin/leucovorin/5-fluorouracil in first-line metastatic gastric and oesophago-gastric junction cancer: results from a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie (AIO).
Luber B, Deplazes J, Keller G, Walch A, Rauser S, Eichmann M, Langer R, Höfler H, Hegewisch-Becker S, Folprecht G, Wöll E, Decker T, Endlicher E, Lorenzen S, Fend F, Peschel C, Lordick F
BMC cancer 2011 Dec 7;11:509
BMC cancer 2011 Dec 7;11:509
Epithelial-to-mesenchymal transition in the development and progression of adenocarcinoma and squamous cell carcinoma of the lung.
Prudkin L, Liu DD, Ozburn NC, Sun M, Behrens C, Tang X, Brown KC, Bekele BN, Moran C, Wistuba II
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2009 May;22(5):668-78
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2009 May;22(5):668-78
Chronic pharmacologic inhibition of EGFR leads to cardiac dysfunction in C57BL/6J mice.
Barrick CJ, Yu M, Chao HH, Threadgill DW
Toxicology and applied pharmacology 2008 May 1;228(3):315-25
Toxicology and applied pharmacology 2008 May 1;228(3):315-25
Enhanced activation of epidermal growth factor receptor caused by tumor-derived E-cadherin mutations.
Bremm A, Walch A, Fuchs M, Mages J, Duyster J, Keller G, Hermannstädter C, Becker KF, Rauser S, Langer R, von Weyhern CH, Höfler H, Luber B
Cancer research 2008 Feb 1;68(3):707-14
Cancer research 2008 Feb 1;68(3):707-14
Clinicopathologic characteristics of the EGFR gene mutation in non-small cell lung cancer.
Tsao AS, Tang XM, Sabloff B, Xiao L, Shigematsu H, Roth J, Spitz M, Hong WK, Gazdar A, Wistuba I
Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer 2006 Mar;1(3):231-9
Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer 2006 Mar;1(3):231-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on membrane enriched extracts (30 µg lysate) of A-431 (1), A-431 treated with EGF (200 ng/mL for 10 minutes) (2), A-431 treated with Gefitinib followed by EGF (1uM for 16 hours, 200 ng/mL for 10 minutes) (3) and A-431 treated with Afatinib followed by EGF (0.5 uM for 6 hours, 200 ng/mL for 10 minutes) (4). The blot was probed with Anti-Phospho-EGFR (Tyr1086) Rabbit Polyclonal Antibody (Product # 36-9700, 0.5 µg/mL) and detected by chemiluminescence using Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). A 170 kDa band corresponding to Phospho-EGFR (Tyr1086) was detected and observed to increase upon EGF treatment across cell lines tested. Pre-treatment with EGFR-antagonists, Gefitinib and Afatinib, resulted in inhibition of Phospho-EGFR in A-431 cell line. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane using the wet transfer system. The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western Blot analysis of (A) serum-starved, unstimulated A431 and (B) serum Starved, EGF-stimulated A431 cells using Rabbit anti phospho EGFR (pY1086) polyclonal antibody (Product # 36-9700)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of EGFR (pY1086) was performed by loading 30 µg of A-549 (lane1) Serum Starved A-549 (lane2), A-549 treated for 10 minutes with 200 ng/mL of EGF (lane3), NIH\3T3 (lane4) and NIH\3T3 treated for 10 minutes with 200 ng/mL of EGF (lane5) cell tissue lysate using Novex® NuPAGE® 10 % Bis-Tris gel (Product # NP0301BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and Proteins were transferred to a nitrocellulose membrane with overnight wet transfer setup at 4°C and blocked with 5 % skim milk for 1 hour at room temperature. EGFR (pY1086) was detected at ~ 180 kDa using EGFR (pY1086) Rabbit Polyclonal Antibody (Product # 36-9700) at 0.1-0.5 µg/mL in 5 % skim milk for 3 hour at room temperature on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
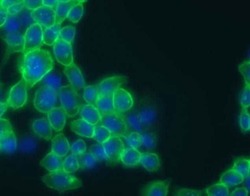
- Experimental details
- Immunofluorescent staining of EGF-stimulated A431 cells using our Rabbit anti-phospho-EGFr (Tyr1086) polyclonal antibody (Product # 36-9700).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Detection of EGFR gene amplification and survival impact of EGFR copy number gain . ( a ) EGFR gene amplification (8.2 signals per nucleus) was observed in one patient with OGJ cancer by image-based three-dimensional dual colour FISH analysis for EGFR (red) and chromosome 7 (green). ( b ) Overall survival in metastatic gastric or OGJ cancer patients treated with cetuximab plus FUFOX was analysed with the Kaplan-Meier method after stratification of patients according to EGFR gene copy numbers (cut-off 4.0). The log-rank test statistical analysis indicates a statistically significant relationship ( P = 0.011).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunoprecipitation of EGFR (pY1086) was performed with 5 µg of the EGFR (pY1086) Rabbit Polyclonal Antibody (Product # 36-9700) on cell extract from NIH/3T3 treated for 10 minutes with 200 ng/mL of EGF using the Dynabeads® Protein A Immunoprecipitation Kit (10006D). Normal Rabbit IgG was used as a negative IP control. Subsequently, western blot analysis was performed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800). Proteins were transferred using Pierce™ G2 Fast Blotter (22834SPCL) to a nitrocellulose membrane and blocked with 5% skim milk for 1 hour at room temperature on a rocking platform. EGFR (pY1086) was detected at ~ 180 kDa using EGFR (pY1086) Rabbit Polyclonal Antibody (Product # 36-9700) at 0.1-0.5 µg/mL in 5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106). Lane 1 represents 10% of the total cell extract (input), Lane 2 represents Rabbit IgG as Isotype control and Lane 3 represents IP performed with EGFR (pY1086) Rabbit Polyclonal Antibody (Product # 36-9700).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 EGFR G724S mediates resistance to third-generation EGFR inhibitors in vitro and in vivo. a Immunoblotting results of NIH-3T3 cells (empty vector, EGFR 19del , EGFR G724S , or EGFR 19del+G724S ) monitoring phospho-EGFR and total EGFR under erlotinib treatment (24 h). HSP90 was used as loading control. b Immunoblotting of NIH-3T3 cells under osimertinib treatment (24 h) is shown. Dose-response measurement of Ba/F3 cells expressing EGFR 19del , EGFR G724S , or EGFR 19del+G724S treated for 72 h with c erlotinib or d osimertinib. Experiments were performed at least three times. e , f Graphs show tumor volume of mice injected with NIH-3T3 EGFR 19del and EGFR 19del+G724S cells treated with osimertinib (5 mg/kg, i.p., once daily). e Tumor volumes ( EGFR 19del vehicle, n = 7 mice; EGFR 19del osimertinib, n = 8 mice; EGFR 19del+G724S vehicle, n = 7 mice; EGFR 19del+G724S osimertinib, n =10 mice) were assessed for 20 days by longitudinal caliper measurements every second day following treatment initiation. f Tumor volumes were quantified after 8 days of treatment. Volume changes in the osimertinib treatment cohort (dark gray and green) were compared with the vehicle-treated control group (light gray and green). Each dot represents a single tumor per mouse. Significance is calculated by two-tailed Student's t test, n.s.: non-significant. g Representative images of Cleaved Caspase-3 stainings. Tumors of mice bearing NIH-3T3 EGFR 19del
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Biochemical profiling of EGFR G724S . a Homogeneous time-resolved fluorescence (HTRF) is used for IC 50 -determination for EGFR 19del and EGFR 19del +G724S with representative inhibitors. Representative dose-response curves of a single measurement in duplicates are shown. b Comparison of biochemical IC 50 -values with HTRF for the three generations of EGFR TKIs against EGFR 19del+G724S . Values are the mean of three independent measurements in duplicates. c Immunoblotting results of NIH-3T3 cells (empty vector, EGFR 19del , EGFR G724S or EGFR 19del+G724S ) showing phospho-EGFR and total EGFR under afatinib treatment (24 h). HSP90 was used as loading control ( n = 3). d Dose-response measurement of Ba/F3 cells expressing EGFR 19del , EGFR G724S , or EGFR 19del+G724S treated for 72 h with afatinib. Experiments were accomplished for at least three times. e Structure of exon 20 mutant EGFR , bound to 4-anilinoquinazoline based TKI PD168393, is shown
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Kinetic evaluation of second- and third-generation EGFR TKIs against EGFR mutant proteins. a Schematic overview of two-step binding mechanism of covalent inhibitors to kinases with K i (quotient of k off and k on ) describing the reversible binding affinity and k inact describing the rate of inactivation. b Time-dependent IC 50 -determination of afatinib and osimertinib on EGFR mutant proteins. Representative curves of single measurements in duplicates are shown. c Heatmap of biochemical IC 50 -, K i -, and k inact determination of second- and third-generation EGFR TKIs against EGFR mutant proteins. Values are the mean of three independent measurements in duplicates. d Immunoblotting results of NIH-3T3 cells (EGFR 19del or EGFR 19del+G724S ) monitoring phospho-EGFR and total EGFR. Cells were treated for indicated times (0, 1, 3, 6, and 24 h) with osimertinib (1 uM) or afatinib (1 uM). HSP90 was used as loading control ( n = 3)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. The inhibition of EGFR phosphorylation following co-administration of cetuximab and iRGD. (A) Immunohistochemical staining analysis. EGFR and p-EGFR positive cells in the sections are stained brown. The nuclei are stained blue. Representative figures from each group are shown; n=6; magnification, x400; Scale bars=50 um. Quantitative analysis results of (B) EGFR and (C) p-EGFR for part (A). Five fields of each tumor tissue section were randomly selected for the calculation of the IOD value of the positive region through Image-Pro Plus software, which indicated the amount of antigen expression. (D) Western blot analysis. Total protein from the tumor tissues was extracted. EGFR and p-EGFR were detected with beta-actin as an internal control. n=3. Quantitative analysis results of (E) EGFR and (F) p-EGFR of part (D). The intensity of each strip was analyzed by ImageJ software. The average intensities of EGFR and p-EGFR were standardized to beta-actin. Error bars, mean +- standard deviation; ns, not significant; *P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Immunoprecipitation
Immunoprecipitation