Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [2]
- Immunohistochemistry [2]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-14981 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- NEFL Monoclonal Antibody (T.400.5)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- It is not recommended to aliquot this antibody.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Antibody clone number
- T.400.5
- Vial size
- 100 µL
- Concentration
- 24 µg/mL
- Storage
- -20°C
Submitted references Targeted neural differentiation of murine mesenchymal stem cells by a protocol simulating the inflammatory site of neural injury.
Bioengineered constructs combined with exercise enhance stem cell-mediated treatment of volumetric muscle loss.
CDKL5 and Shootin1 Interact and Concur in Regulating Neuronal Polarization.
Chudickova M, Bruza P, Zajicova A, Trosan P, Svobodova L, Javorkova E, Kubinova S, Holan V
Journal of tissue engineering and regenerative medicine 2017 May;11(5):1588-1597
Journal of tissue engineering and regenerative medicine 2017 May;11(5):1588-1597
Bioengineered constructs combined with exercise enhance stem cell-mediated treatment of volumetric muscle loss.
Quarta M, Cromie M, Chacon R, Blonigan J, Garcia V, Akimenko I, Hamer M, Paine P, Stok M, Shrager JB, Rando TA
Nature communications 2017 Jun 20;8:15613
Nature communications 2017 Jun 20;8:15613
CDKL5 and Shootin1 Interact and Concur in Regulating Neuronal Polarization.
Nawaz MS, Giarda E, Bedogni F, La Montanara P, Ricciardi S, Ciceri D, Alberio T, Landsberger N, Rusconi L, Kilstrup-Nielsen C
PloS one 2016;11(2):e0148634
PloS one 2016;11(2):e0148634
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
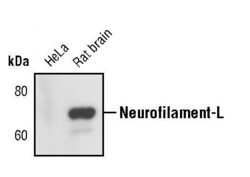
- Experimental details
- Western blot analysis of Neurofilament-L in extracts from HeLa cells and rat brain using Neurofilament-L monoclonal antibody (Product # MA5-14981).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Knockout of NEFL was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042) (Assay ID CRISPR1108488_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Western blot analysis of NEFL was performed by loading 30 µg of HEK-293 wild type (Lane 1) and HEK-293 NEFL KO (Lane 2) whole cell extracts. The blot was probed with Anti-NEFL Monoclonal Antibody (T.400.5)(Product # MA5-14981) using 1:1000 dilution and Goat anti-Rabbit IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036). Loss of signal upon CRISPR mediated knockout (KO) using the LentiArray™ CRISPR product line confirms that antibody is specific to NEFL.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-NEFL Monoclonal Antibody (T.400.5) (Product # MA5-14981) and a 68 kDa band corresponding to NEFL was observed in PC-12 differentiated neurons, HEK-293, Mouse Cerebellum, Mouse Brain and Rat Brain but not in PC-12, HeLa, Mouse Heart and Mouse Lung. Membrane enriched extracts (30 µg lysate) of PC-12 (Lane 1) and PC-12 differentiated neurons (Lane 2), Mouse Cerebellum (Lane 3), Mouse Brain (Lane 4), Rat Brain (Lane 5), Mouse Heart (Lane 6), Mouse Lung (Lane 7), HEK-293 (Lane 8) and HeLa (Lane 9) were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1,000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
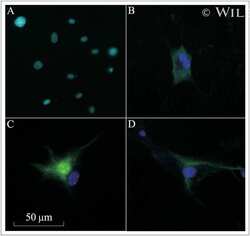
- Immunofluorescence analysis of NF-L was performed using 70% confluent log phase PC-12 and PC-12 differentiated to neuronal lineage. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. Cells were labeled with NF-L Rabbit Monoclonal Antibody (Product # MA5-14981) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415). Panel d represents the merged image of PC-12 cells differentiated to neuronal lineage showing localization in cytoskeleton (intermediate filaments) in the cell body and in neurite extensions. Panel e represents the merged image of undifferentiated PC-12 cells showing no expression for NF-L protein. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
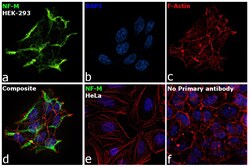
- Experimental details
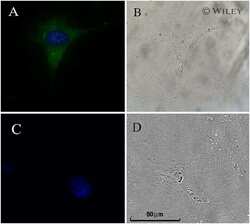
- Immunofluorescence analysis of NF-L was performed using 70% confluent log phase HEK-293 and HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 1 hour at room temperature. HEK-293 cells were labeled with NF-L Rabbit Monoclonal Antibody (Product # MA5-14981) at 1:100 dilution in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415). Panel d represents the merged image of HEK-293, which is a cell model positive for NF-L expression showing cytoskeletal (intermediate filaments) localization. Panel e represents the merged image of HeLa cells, which is null for NF-L protein expression. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
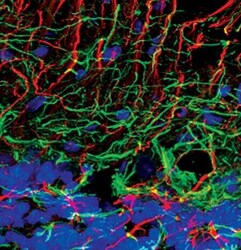
- Experimental details
- Immunofluorescent analysis of Neurofilament-L in normal rat cerebellum using a Neurofilament-L monoclonal antibody (Product # MA5-14981) (green) and a GFAP monoclonal antibody (red). DNA is labeled using a fluorescent blue dye.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
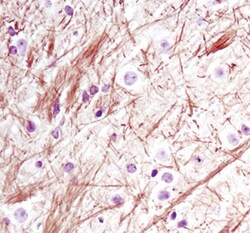
- Experimental details
- Immunohistochemical analysis of Neurofilament-L in paraffin-embedded mouse brain using a Neurofilament-L monoclonal antibody (Product # MA5-14981).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
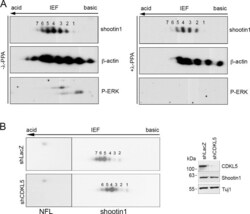
- Fig 6 CDKL5 influences shootin1 phosphorylation in primary cortical neurons. (A) Total cell extracts of DIV7 cortical neurons were treated with or without lambda phosphatase (lamda-PPase) and analyzed by two-dimensional gel electrophoresis and immunoblotting with antibodies against shootin1, beta-actin and, as control for the lamda-PPase treatment, phopho-ERK1/2. (B) Primary cortical neurons were infected with shLacZ- or shCDKL5#1-expressing viral particles at DIV0 and total cell lysates were prepared at DIV7 and subjected to two-dimensional gel electrophoresis. Shootin1 and NFL were detected by immunoblotting; the single NFL-spot was used as internal control for alignment. Silencing of CDKL5 was confirmed by western blot (right panel). (n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
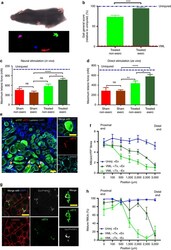
- Figure 6 Exercise improves innervation of de novo myofibres and improves forces in vivo. ( a ) Representative image of a mouse during a gait analysis (top) and the gait footprints collected during the analysis (bottom). Mice were positioned in a transparent treadmill and a camera was positioned underneath to record the gait. ( b ) Quantification of the gait 'disability score' resulting from the analysis of 47 parameters (see Methods) ( n =6). ( c ) In vivo force production measurements of TA muscles treated with bioconstructs following VML injury in non-exercised or exercised mice. After 30 days, the distal tendons were attached to a force transducer and contractions were induced through sciatic nerve stimulation ( n =6). ( d ) Ex vivo force production measurements from non-exercised or exercised mice. The same muscles measured in c were then dissected and cultured in a chamber. The distal tendons were attached to a transducer and contractions were induced electrically in the culture bath ( n =6). ( e ) (Left) Representative IF image of transplanted bioconstruct. Yellow arrows indicate donor-derived (eYFP + ) myofibres with NMJs (alphaBTX + ) within regions of the transplanted bioconstruct. (Right) Higher magnification of an NMJ associated with a donor-derived myofibre (scale bars=50 mum). ( f ) Quantification of NMJs in whole cross-sections of transplanted bioconstructs along 3 mm lengths of TA muscles. Muscles were either uninjured and exercised ('-VML, +Ex&a
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot