Antibody data
- Antibody Data
- Antigen structure
- References [73]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [79]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 48-5698-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Ki-67 Monoclonal Antibody (SolA15), eFluor™ 450, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody SolA15 recognizes mouse and rat Ki-67, a 300 kDa nuclear protein. Ki-67 is present during all active phases of the cell cycle (G1, S, G2, and mitosis), but is absent from resting cells (G0). Ki-67 is detected within the nucleus during interphase but redistributes to the chromosomes during mitosis. Ki-67 is used as a marker for determining the growth fraction of a given population of cells. In studies of tumor cells, the "Ki-67 labeling index" refers to the number of Ki-67 positive cells within the population and this is used to predict outcome of particular cancer types. Ki-67 has been shown to interact with the DNA-bound protein chromobox protein homolog 3 (CBX3) (heterochromatin). The SolA15 antibody also recognizes human, non-human primate and canine Ki-67. Applications Reported: This SolA15 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This SolA15 antibody has been tested by intracellular staining and flow cytometric analysis of stimulated mouse splenocytes using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523-00). This can be used at less than or equal to 0.125 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. eFluor® 450 is an alternative to Pacific Blue®. eFluor® 450 emits at 445 nm and is excited with the Violet laser (405 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 405 nm; Emission: 445 nm; Laser: Violet Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse, Rat, Canine
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- SolA15
- Vial size
- 25 µg
- Concentration
- 0.2 mg/mL
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Maternal-fetal conflict averted by progesterone- induced FOXP3+ regulatory T cells.
Endogenous retroviruses promote homeostatic and inflammatory responses to the microbiota.
Lymphatic PD-L1 Expression Restricts Tumor-Specific CD8(+) T-cell Responses.
Coordinated Viral Control by Cytotoxic Lymphocytes Ensures Optimal Adaptive NK Cell Responses.
Mucosal-associated invariant T cells promote inflammation and intestinal dysbiosis leading to metabolic dysfunction during obesity.
Thymic iNKT single cell analyses unmask the common developmental program of mouse innate T cells.
Genetic ablation of adipocyte PD-L1 reduces tumor growth but accentuates obesity-associated inflammation.
Targeting ANXA1 abrogates Treg-mediated immune suppression in triple-negative breast cancer.
TLR9 Sensing of Self-DNA Controls Cell-Mediated Immunity to Listeria Infection via Rapid Conversion of Conventional CD4(+) T Cells to T(reg).
MicroRNA-127-3p controls murine hematopoietic stem cell maintenance by limiting differentiation.
Pulsatile MEK Inhibition Improves Anti-tumor Immunity and T Cell Function in Murine Kras Mutant Lung Cancer.
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
C-Kit Cardiac Progenitor Cell Based Cell Sheet Improves Vascularization and Attenuates Cardiac Remodeling following Myocardial Infarction in Rats.
Factors Within the Endoneurial Microenvironment Act to Suppress Tumorigenesis of MPNST.
ARTS mediates apoptosis and regeneration of the intestinal stem cell niche.
Lymphotoxin α fine-tunes T cell clonal deletion by regulating thymic entry of antigen-presenting cells.
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Stem cell factor is selectively secreted by arterial endothelial cells in bone marrow.
The organic ester O,O'-diethyl-(S,S)-ethylenediamine-N,N'-di-2-(3-cyclohexyl)propanoate dihydrochloride attenuates murine breast cancer growth and metastasis.
Selective pharmacological inhibition of DDR1 prevents experimentally-induced glomerulonephritis in prevention and therapeutic regime.
Vitreous Cytokine Expression and a Murine Model Suggest a Key Role of Microglia in the Inflammatory Response to Retinal Detachment.
Inhibition of histone deacetylase 1 ameliorates renal tubulointerstitial fibrosis via modulation of inflammation and extracellular matrix gene transcription in mice.
Modulation of Myelopoiesis Progenitors Is an Integral Component of Trained Immunity.
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
1810011o10 Rik Inhibits the Antitumor Effect of Intratumoral CD8(+) T Cells through Suppression of Notch2 Pathway in a Murine Hepatocellular Carcinoma Model.
A somatic mutation in erythro-myeloid progenitors causes neurodegenerative disease.
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Neonatal pancreatic pericytes support β-cell proliferation.
Resident macrophages of pancreatic islets have a seminal role in the initiation of autoimmune diabetes of NOD mice.
Intestinal Batf3-dependent dendritic cells are required for optimal antiviral T-cell responses in adult and neonatal mice.
Histone variant H2A.J accumulates in senescent cells and promotes inflammatory gene expression.
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Egr2 and 3 control adaptive immune responses by temporally uncoupling expansion from T cell differentiation.
IL-4 as a Repurposed Biological Drug for Myocardial Infarction through Augmentation of Reparative Cardiac Macrophages: Proof-of-Concept Data in Mice.
Paracrine Activin-A Signaling Promotes Melanoma Growth and Metastasis through Immune Evasion.
Mutations in 5-methylcytosine oxidase TET2 and RhoA cooperatively disrupt T cell homeostasis.
Myeloid progenitor cluster formation drives emergency and leukaemic myelopoiesis.
The stress kinase GCN2 does not mediate suppression of antitumor T cell responses by tryptophan catabolism in experimental melanomas.
Growth and metastasis of lung adenocarcinoma is potentiated by BMP4-mediated immunosuppression.
Schwann cell proliferation and differentiation that is induced by ferulic acid through MEK1/ERK1/2 signalling promotes peripheral nerve remyelination following crush injury in rats.
Host-Protozoan Interactions Protect from Mucosal Infections through Activation of the Inflammasome.
Whole Chromosome Instability induces senescence and promotes SASP.
Hyaluronan and TLR4 promote surfactant-protein-C-positive alveolar progenitor cell renewal and prevent severe pulmonary fibrosis in mice.
Immune response modulation by Galectin-1 in a transgenic model of neuroblastoma.
Maternal-Derived Hepatitis B Virus e Antigen Alters Macrophage Function in Offspring to Drive Viral Persistence after Vertical Transmission.
The cell proliferation antigen Ki-67 organises heterochromatin.
A Human Trypanosome Suppresses CD8+ T Cell Priming by Dendritic Cells through the Induction of Immune Regulatory CD4+ Foxp3+ T Cells.
Image-based detection and targeting of therapy resistance in pancreatic adenocarcinoma.
RANKL/RANK control Brca1 mutation- .
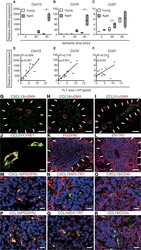
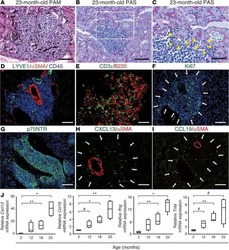
Heterogeneous fibroblasts underlie age-dependent tertiary lymphoid tissues in the kidney.
Detection of Cell Proliferation Markers by Immunofluorescence Staining and Microscopy Imaging in Paraffin-Embedded Tissue Sections.
Oligodendrocyte death results in immune-mediated CNS demyelination.
Lineage-specific enhancers activate self-renewal genes in macrophages and embryonic stem cells.
Foxp3 and Toll-like receptor signaling balance T(reg) cell anabolic metabolism for suppression.
Suppression of ischemia in arterial occlusive disease by JNK-promoted native collateral artery development.
Targeting myeloid-derived suppressor cells with colony stimulating factor-1 receptor blockade can reverse immune resistance to immunotherapy in indoleamine 2,3-dioxygenase-expressing tumors.
Epithelial-to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis.
Mammary Stem Cells and Tumor-Initiating Cells Are More Resistant to Apoptosis and Exhibit Increased DNA Repair Activity in Response to DNA Damage.
Low levels of endogenous or X-ray-induced DNA double-strand breaks activate apoptosis in adult neural stem cells.
NF-κB-induced microRNA-31 promotes epidermal hyperplasia by repressing protein phosphatase 6 in psoriasis.
Beta-Chemokine CCL15 Affects the Adhesion and Migration of Hematopoietic Progenitor Cells.
The adhesion G protein-coupled receptor GPR56 is a cell-autonomous regulator of oligodendrocyte development.
Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression.
Autophagy is a critical regulator of memory CD8(+) T cell formation.
Dynamic changes in macrophage activation and proliferation during the development and resolution of intestinal inflammation.
Alveolar progenitor and stem cells in lung development, renewal and cancer.
E and Id proteins influence invariant NKT cell sublineage differentiation and proliferation.
Myeloid cells expressing VEGF and arginase-1 following uptake of damaged retinal pigment epithelium suggests potential mechanism that drives the onset of choroidal angiogenesis in mice.
Small intestine inflammation in Roquin-mutant and Roquin-deficient mice.
Dedifferentiation of committed epithelial cells into stem cells in vivo.
IL-1R signaling in dendritic cells replaces pattern-recognition receptors in promoting CD8⁺ T cell responses to influenza A virus.
Primate B-1 cells generate antigen-specific B cell responses to T cell-independent type 2 antigens.
Severance AL, Kinder JM, Xin L, Burg AR, Shao TY, Pham G, Tilburgs T, Goodman WA, Mesiano S, Way SS
iScience 2022 Jun 17;25(6):104400
iScience 2022 Jun 17;25(6):104400
Endogenous retroviruses promote homeostatic and inflammatory responses to the microbiota.
Lima-Junior DS, Krishnamurthy SR, Bouladoux N, Collins N, Han SJ, Chen EY, Constantinides MG, Link VM, Lim AI, Enamorado M, Cataisson C, Gil L, Rao I, Farley TK, Koroleva G, Attig J, Yuspa SH, Fischbach MA, Kassiotis G, Belkaid Y
Cell 2021 Jul 8;184(14):3794-3811.e19
Cell 2021 Jul 8;184(14):3794-3811.e19
Lymphatic PD-L1 Expression Restricts Tumor-Specific CD8(+) T-cell Responses.
Cousin N, Cap S, Dihr M, Tacconi C, Detmar M, Dieterich LC
Cancer research 2021 Aug 1;81(15):4133-4144
Cancer research 2021 Aug 1;81(15):4133-4144
Coordinated Viral Control by Cytotoxic Lymphocytes Ensures Optimal Adaptive NK Cell Responses.
Diaz-Salazar C, Sun JC
Cell reports 2020 Sep 22;32(12):108186
Cell reports 2020 Sep 22;32(12):108186
Mucosal-associated invariant T cells promote inflammation and intestinal dysbiosis leading to metabolic dysfunction during obesity.
Toubal A, Kiaf B, Beaudoin L, Cagninacci L, Rhimi M, Fruchet B, da Silva J, Corbett AJ, Simoni Y, Lantz O, Rossjohn J, McCluskey J, Lesnik P, Maguin E, Lehuen A
Nature communications 2020 Jul 24;11(1):3755
Nature communications 2020 Jul 24;11(1):3755
Thymic iNKT single cell analyses unmask the common developmental program of mouse innate T cells.
Harsha Krovi S, Zhang J, Michaels-Foster MJ, Brunetti T, Loh L, Scott-Browne J, Gapin L
Nature communications 2020 Dec 7;11(1):6238
Nature communications 2020 Dec 7;11(1):6238
Genetic ablation of adipocyte PD-L1 reduces tumor growth but accentuates obesity-associated inflammation.
Wu B, Chiang HC, Sun X, Yuan B, Mitra P, Hu Y, Curiel TJ, Li R
Journal for immunotherapy of cancer 2020 Aug;8(2)
Journal for immunotherapy of cancer 2020 Aug;8(2)
Targeting ANXA1 abrogates Treg-mediated immune suppression in triple-negative breast cancer.
Bai F, Zhang P, Fu Y, Chen H, Zhang M, Huang Q, Li D, Li B, Wu K
Journal for immunotherapy of cancer 2020 Apr;8(1)
Journal for immunotherapy of cancer 2020 Apr;8(1)
TLR9 Sensing of Self-DNA Controls Cell-Mediated Immunity to Listeria Infection via Rapid Conversion of Conventional CD4(+) T Cells to T(reg).
Dolina JS, Lee J, Griswold RQ, Labarta-Bajo L, Kannan S, Greenbaum JA, Bahia El Idrissi N, Pont MJ, Croft M, Schoenberger SP
Cell reports 2020 Apr 7;31(1):107249
Cell reports 2020 Apr 7;31(1):107249
MicroRNA-127-3p controls murine hematopoietic stem cell maintenance by limiting differentiation.
Crisafulli L, Muggeo S, Uva P, Wang Y, Iwasaki M, Locatelli S, Anselmo A, Colombo FS, Carlo-Stella C, Cleary ML, Villa A, Gentner B, Ficara F
Haematologica 2019 Sep;104(9):1744-1755
Haematologica 2019 Sep;104(9):1744-1755
Pulsatile MEK Inhibition Improves Anti-tumor Immunity and T Cell Function in Murine Kras Mutant Lung Cancer.
Choi H, Deng J, Li S, Silk T, Dong L, Brea EJ, Houghton S, Redmond D, Zhong H, Boiarsky J, Akbay EA, Smith PD, Merghoub T, Wong KK, Wolchok JD
Cell reports 2019 Apr 16;27(3):806-819.e5
Cell reports 2019 Apr 16;27(3):806-819.e5
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
Glal D, Sudhakar JN, Lu HH, Liu MC, Chiang HY, Liu YC, Cheng CF, Shui JW
Frontiers in immunology 2018;9:2522
Frontiers in immunology 2018;9:2522
C-Kit Cardiac Progenitor Cell Based Cell Sheet Improves Vascularization and Attenuates Cardiac Remodeling following Myocardial Infarction in Rats.
Dergilev K, Tsokolaeva Z, Makarevich P, Beloglazova I, Zubkova E, Boldyreva M, Ratner E, Dyikanov D, Menshikov M, Ovchinnikov A, Ageev F, Parfyonova Y
BioMed research international 2018;2018:3536854
BioMed research international 2018;2018:3536854
Factors Within the Endoneurial Microenvironment Act to Suppress Tumorigenesis of MPNST.
Stratton JA, Assinck P, Sinha S, Kumar R, Moulson A, Patrick N, Raharjo E, Chan JA, Midha R, Tetzlaff W, Biernaskie J
Frontiers in cellular neuroscience 2018;12:356
Frontiers in cellular neuroscience 2018;12:356
ARTS mediates apoptosis and regeneration of the intestinal stem cell niche.
Koren E, Yosefzon Y, Ankawa R, Soteriou D, Jacob A, Nevelsky A, Ben-Yosef R, Bar-Sela G, Fuchs Y
Nature communications 2018 Nov 2;9(1):4582
Nature communications 2018 Nov 2;9(1):4582
Lymphotoxin α fine-tunes T cell clonal deletion by regulating thymic entry of antigen-presenting cells.
Lopes N, Charaix J, Cédile O, Sergé A, Irla M
Nature communications 2018 Mar 28;9(1):1262
Nature communications 2018 Mar 28;9(1):1262
Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche.
Maryanovich M, Zahalka AH, Pierce H, Pinho S, Nakahara F, Asada N, Wei Q, Wang X, Ciero P, Xu J, Leftin A, Frenette PS
Nature medicine 2018 Jun;24(6):782-791
Nature medicine 2018 Jun;24(6):782-791
Stem cell factor is selectively secreted by arterial endothelial cells in bone marrow.
Xu C, Gao X, Wei Q, Nakahara F, Zimmerman SE, Mar J, Frenette PS
Nature communications 2018 Jun 22;9(1):2449
Nature communications 2018 Jun 22;9(1):2449
The organic ester O,O'-diethyl-(S,S)-ethylenediamine-N,N'-di-2-(3-cyclohexyl)propanoate dihydrochloride attenuates murine breast cancer growth and metastasis.
Jurisevic M, Arsenijevic A, Pantic J, Gajovic N, Milovanovic J, Milovanovic M, Poljarevic J, Sabo T, Vojvodic D, Radosavljevic GD, Arsenijevic N
Oncotarget 2018 Jun 15;9(46):28195-28212
Oncotarget 2018 Jun 15;9(46):28195-28212
Selective pharmacological inhibition of DDR1 prevents experimentally-induced glomerulonephritis in prevention and therapeutic regime.
Moll S, Yasui Y, Abed A, Murata T, Shimada H, Maeda A, Fukushima N, Kanamori M, Uhles S, Badi L, Cagarelli T, Formentini I, Drawnel F, Georges G, Bergauer T, Gasser R, Bonfil RD, Fridman R, Richter H, Funk J, Moeller MJ, Chatziantoniou C, Prunotto M
Journal of translational medicine 2018 Jun 1;16(1):148
Journal of translational medicine 2018 Jun 1;16(1):148
Vitreous Cytokine Expression and a Murine Model Suggest a Key Role of Microglia in the Inflammatory Response to Retinal Detachment.
Kiang L, Ross BX, Yao J, Shanmugam S, Andrews CA, Hansen S, Besirli CG, Zacks DN, Abcouwer SF
Investigative ophthalmology & visual science 2018 Jul 2;59(8):3767-3778
Investigative ophthalmology & visual science 2018 Jul 2;59(8):3767-3778
Inhibition of histone deacetylase 1 ameliorates renal tubulointerstitial fibrosis via modulation of inflammation and extracellular matrix gene transcription in mice.
Nguyễn-Thanh T, Kim D, Lee S, Kim W, Park SK, Kang KP
International journal of molecular medicine 2018 Jan;41(1):95-106
International journal of molecular medicine 2018 Jan;41(1):95-106
Modulation of Myelopoiesis Progenitors Is an Integral Component of Trained Immunity.
Mitroulis I, Ruppova K, Wang B, Chen LS, Grzybek M, Grinenko T, Eugster A, Troullinaki M, Palladini A, Kourtzelis I, Chatzigeorgiou A, Schlitzer A, Beyer M, Joosten LAB, Isermann B, Lesche M, Petzold A, Simons K, Henry I, Dahl A, Schultze JL, Wielockx B, Zamboni N, Mirtschink P, Coskun Ü, Hajishengallis G, Netea MG, Chavakis T
Cell 2018 Jan 11;172(1-2):147-161.e12
Cell 2018 Jan 11;172(1-2):147-161.e12
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Chu F, Hu Y, Zhou Y, Guo M, Lu J, Zheng W, Xu H, Zhao J, Xu L
Clinical and experimental immunology 2018 Feb;191(2):166-179
Clinical and experimental immunology 2018 Feb;191(2):166-179
Fc Effector Function Contributes to the Activity of Human Anti-CTLA-4 Antibodies.
Arce Vargas F, Furness AJS, Litchfield K, Joshi K, Rosenthal R, Ghorani E, Solomon I, Lesko MH, Ruef N, Roddie C, Henry JY, Spain L, Ben Aissa A, Georgiou A, Wong YNS, Smith M, Strauss D, Hayes A, Nicol D, O'Brien T, Mårtensson L, Ljungars A, Teige I, Frendéus B, TRACERx Melanoma, TRACERx Renal, TRACERx Lung consortia, Pule M, Marafioti T, Gore M, Larkin J, Turajlic S, Swanton C, Peggs KS, Quezada SA
Cancer cell 2018 Apr 9;33(4):649-663.e4
Cancer cell 2018 Apr 9;33(4):649-663.e4
1810011o10 Rik Inhibits the Antitumor Effect of Intratumoral CD8(+) T Cells through Suppression of Notch2 Pathway in a Murine Hepatocellular Carcinoma Model.
Dai K, Huang L, Huang YB, Chen ZB, Yang LH, Jiang YA
Frontiers in immunology 2017;8:320
Frontiers in immunology 2017;8:320
A somatic mutation in erythro-myeloid progenitors causes neurodegenerative disease.
Mass E, Jacome-Galarza CE, Blank T, Lazarov T, Durham BH, Ozkaya N, Pastore A, Schwabenland M, Chung YR, Rosenblum MK, Prinz M, Abdel-Wahab O, Geissmann F
Nature 2017 Sep 21;549(7672):389-393
Nature 2017 Sep 21;549(7672):389-393
Regulatory Innate Lymphoid Cells Control Innate Intestinal Inflammation.
Wang S, Xia P, Chen Y, Qu Y, Xiong Z, Ye B, Du Y, Tian Y, Yin Z, Xu Z, Fan Z
Cell 2017 Sep 21;171(1):201-216.e18
Cell 2017 Sep 21;171(1):201-216.e18
Neonatal pancreatic pericytes support β-cell proliferation.
Epshtein A, Rachi E, Sakhneny L, Mizrachi S, Baer D, Landsman L
Molecular metabolism 2017 Oct;6(10):1330-1338
Molecular metabolism 2017 Oct;6(10):1330-1338
Resident macrophages of pancreatic islets have a seminal role in the initiation of autoimmune diabetes of NOD mice.
Carrero JA, McCarthy DP, Ferris ST, Wan X, Hu H, Zinselmeyer BH, Vomund AN, Unanue ER
Proceedings of the National Academy of Sciences of the United States of America 2017 Nov 28;114(48):E10418-E10427
Proceedings of the National Academy of Sciences of the United States of America 2017 Nov 28;114(48):E10418-E10427
Intestinal Batf3-dependent dendritic cells are required for optimal antiviral T-cell responses in adult and neonatal mice.
Sun T, Rojas OL, Li C, Ward LA, Philpott DJ, Gommerman JL
Mucosal immunology 2017 May;10(3):775-788
Mucosal immunology 2017 May;10(3):775-788
Histone variant H2A.J accumulates in senescent cells and promotes inflammatory gene expression.
Contrepois K, Coudereau C, Benayoun BA, Schuler N, Roux PF, Bischof O, Courbeyrette R, Carvalho C, Thuret JY, Ma Z, Derbois C, Nevers MC, Volland H, Redon CE, Bonner WM, Deleuze JF, Wiel C, Bernard D, Snyder MP, Rübe CE, Olaso R, Fenaille F, Mann C
Nature communications 2017 May 10;8:14995
Nature communications 2017 May 10;8:14995
Differential cytokine contributions of perivascular haematopoietic stem cell niches.
Asada N, Kunisaki Y, Pierce H, Wang Z, Fernandez NF, Birbrair A, Ma'ayan A, Frenette PS
Nature cell biology 2017 Mar;19(3):214-223
Nature cell biology 2017 Mar;19(3):214-223
Egr2 and 3 control adaptive immune responses by temporally uncoupling expansion from T cell differentiation.
Miao T, Symonds ALJ, Singh R, Symonds JD, Ogbe A, Omodho B, Zhu B, Li S, Wang P
The Journal of experimental medicine 2017 Jun 5;214(6):1787-1808
The Journal of experimental medicine 2017 Jun 5;214(6):1787-1808
IL-4 as a Repurposed Biological Drug for Myocardial Infarction through Augmentation of Reparative Cardiac Macrophages: Proof-of-Concept Data in Mice.
Shintani Y, Ito T, Fields L, Shiraishi M, Ichihara Y, Sato N, Podaru M, Kainuma S, Tanaka H, Suzuki K
Scientific reports 2017 Jul 31;7(1):6877
Scientific reports 2017 Jul 31;7(1):6877
Paracrine Activin-A Signaling Promotes Melanoma Growth and Metastasis through Immune Evasion.
Donovan P, Dubey OA, Kallioinen S, Rogers KW, Muehlethaler K, Müller P, Rimoldi D, Constam DB
The Journal of investigative dermatology 2017 Dec;137(12):2578-2587
The Journal of investigative dermatology 2017 Dec;137(12):2578-2587
Mutations in 5-methylcytosine oxidase TET2 and RhoA cooperatively disrupt T cell homeostasis.
Zang S, Li J, Yang H, Zeng H, Han W, Zhang J, Lee M, Moczygemba M, Isgandarova S, Yang Y, Zhou Y, Rao A, You MJ, Sun D, Huang Y
The Journal of clinical investigation 2017 Aug 1;127(8):2998-3012
The Journal of clinical investigation 2017 Aug 1;127(8):2998-3012
Myeloid progenitor cluster formation drives emergency and leukaemic myelopoiesis.
Hérault A, Binnewies M, Leong S, Calero-Nieto FJ, Zhang SY, Kang YA, Wang X, Pietras EM, Chu SH, Barry-Holson K, Armstrong S, Göttgens B, Passegué E
Nature 2017 Apr 6;544(7648):53-58
Nature 2017 Apr 6;544(7648):53-58
The stress kinase GCN2 does not mediate suppression of antitumor T cell responses by tryptophan catabolism in experimental melanomas.
Sonner JK, Deumelandt K, Ott M, Thomé CM, Rauschenbach KJ, Schulz S, Munteanu B, Mohapatra S, Adam I, Hofer AC, Feuerer M, Opitz CA, Hopf C, Wick W, Platten M
Oncoimmunology 2016;5(12):e1240858
Oncoimmunology 2016;5(12):e1240858
Growth and metastasis of lung adenocarcinoma is potentiated by BMP4-mediated immunosuppression.
Chen L, Yi X, Goswami S, Ahn YH, Roybal JD, Yang Y, Diao L, Peng D, Peng D, Fradette JJ, Wang J, Byers LA, Kurie JM, Ullrich SE, Qin FX, Gibbons DL
Oncoimmunology 2016;5(11):e1234570
Oncoimmunology 2016;5(11):e1234570
Schwann cell proliferation and differentiation that is induced by ferulic acid through MEK1/ERK1/2 signalling promotes peripheral nerve remyelination following crush injury in rats.
Zhu X, Li K, Guo X, Wang J, Xiang Y
Experimental and therapeutic medicine 2016 Sep;12(3):1915-1921
Experimental and therapeutic medicine 2016 Sep;12(3):1915-1921
Host-Protozoan Interactions Protect from Mucosal Infections through Activation of the Inflammasome.
Chudnovskiy A, Mortha A, Kana V, Kennard A, Ramirez JD, Rahman A, Remark R, Mogno I, Ng R, Gnjatic S, Amir ED, Solovyov A, Greenbaum B, Clemente J, Faith J, Belkaid Y, Grigg ME, Merad M
Cell 2016 Oct 6;167(2):444-456.e14
Cell 2016 Oct 6;167(2):444-456.e14
Whole Chromosome Instability induces senescence and promotes SASP.
Andriani GA, Almeida VP, Faggioli F, Mauro M, Tsai WL, Santambrogio L, Maslov A, Gadina M, Campisi J, Vijg J, Montagna C
Scientific reports 2016 Oct 12;6:35218
Scientific reports 2016 Oct 12;6:35218
Hyaluronan and TLR4 promote surfactant-protein-C-positive alveolar progenitor cell renewal and prevent severe pulmonary fibrosis in mice.
Liang J, Zhang Y, Xie T, Liu N, Chen H, Geng Y, Kurkciyan A, Mena JM, Stripp BR, Jiang D, Noble PW
Nature medicine 2016 Nov;22(11):1285-1293
Nature medicine 2016 Nov;22(11):1285-1293
Immune response modulation by Galectin-1 in a transgenic model of neuroblastoma.
Büchel G, Schulte JH, Harrison L, Batzke K, Schüller U, Hansen W, Schramm A
Oncoimmunology 2016 May;5(5):e1131378
Oncoimmunology 2016 May;5(5):e1131378
Maternal-Derived Hepatitis B Virus e Antigen Alters Macrophage Function in Offspring to Drive Viral Persistence after Vertical Transmission.
Tian Y, Kuo CF, Akbari O, Ou JH
Immunity 2016 May 17;44(5):1204-14
Immunity 2016 May 17;44(5):1204-14
The cell proliferation antigen Ki-67 organises heterochromatin.
Sobecki M, Mrouj K, Camasses A, Parisis N, Nicolas E, Llères D, Gerbe F, Prieto S, Krasinska L, David A, Eguren M, Birling MC, Urbach S, Hem S, Déjardin J, Malumbres M, Jay P, Dulic V, Lafontaine DLj, Feil R, Fisher D
eLife 2016 Mar 7;5:e13722
eLife 2016 Mar 7;5:e13722
A Human Trypanosome Suppresses CD8+ T Cell Priming by Dendritic Cells through the Induction of Immune Regulatory CD4+ Foxp3+ T Cells.
Ersching J, Basso AS, Kalich VL, Bortoluci KR, Rodrigues MM
PLoS pathogens 2016 Jun;12(6):e1005698
PLoS pathogens 2016 Jun;12(6):e1005698
Image-based detection and targeting of therapy resistance in pancreatic adenocarcinoma.
Fox RG, Lytle NK, Jaquish DV, Park FD, Ito T, Bajaj J, Koechlein CS, Zimdahl B, Yano M, Kopp J, Kritzik M, Sicklick J, Sander M, Grandgenett PM, Hollingsworth MA, Shibata S, Pizzo D, Valasek M, Sasik R, Scadeng M, Okano H, Kim Y, MacLeod AR, Lowy AM, Reya T
Nature 2016 Jun 16;534(7607):407-411
Nature 2016 Jun 16;534(7607):407-411
RANKL/RANK control Brca1 mutation- .
Sigl V, Owusu-Boaitey K, Joshi PA, Kavirayani A, Wirnsberger G, Novatchkova M, Kozieradzki I, Schramek D, Edokobi N, Hersl J, Sampson A, Odai-Afotey A, Lazaro C, Gonzalez-Suarez E, Pujana MA, Cimba F, Heyn H, Vidal E, Cruickshank J, Berman H, Sarao R, Ticevic M, Uribesalgo I, Tortola L, Rao S, Tan Y, Pfeiler G, Lee EY, Bago-Horvath Z, Kenner L, Popper H, Singer C, Khokha R, Jones LP, Penninger JM
Cell research 2016 Jul;26(7):761-74
Cell research 2016 Jul;26(7):761-74
Heterogeneous fibroblasts underlie age-dependent tertiary lymphoid tissues in the kidney.
Sato Y, Mii A, Hamazaki Y, Fujita H, Nakata H, Masuda K, Nishiyama S, Shibuya S, Haga H, Ogawa O, Shimizu A, Narumiya S, Kaisho T, Arita M, Yanagisawa M, Miyasaka M, Sharma K, Minato N, Kawamoto H, Yanagita M
JCI insight 2016 Jul 21;1(11):e87680
JCI insight 2016 Jul 21;1(11):e87680
Detection of Cell Proliferation Markers by Immunofluorescence Staining and Microscopy Imaging in Paraffin-Embedded Tissue Sections.
Eminaga S, Teekakirikul P, Seidman CE, Seidman JG
Current protocols in molecular biology 2016 Jul 1;115:14.25.1-14.25.14
Current protocols in molecular biology 2016 Jul 1;115:14.25.1-14.25.14
Oligodendrocyte death results in immune-mediated CNS demyelination.
Traka M, Podojil JR, McCarthy DP, Miller SD, Popko B
Nature neuroscience 2016 Jan;19(1):65-74
Nature neuroscience 2016 Jan;19(1):65-74
Lineage-specific enhancers activate self-renewal genes in macrophages and embryonic stem cells.
Soucie EL, Weng Z, Geirsdóttir L, Molawi K, Maurizio J, Fenouil R, Mossadegh-Keller N, Gimenez G, VanHille L, Beniazza M, Favret J, Berruyer C, Perrin P, Hacohen N, Andrau JC, Ferrier P, Dubreuil P, Sidow A, Sieweke MH
Science (New York, N.Y.) 2016 Feb 12;351(6274):aad5510
Science (New York, N.Y.) 2016 Feb 12;351(6274):aad5510
Foxp3 and Toll-like receptor signaling balance T(reg) cell anabolic metabolism for suppression.
Gerriets VA, Kishton RJ, Johnson MO, Cohen S, Siska PJ, Nichols AG, Warmoes MO, de Cubas AA, MacIver NJ, Locasale JW, Turka LA, Wells AD, Rathmell JC
Nature immunology 2016 Dec;17(12):1459-1466
Nature immunology 2016 Dec;17(12):1459-1466
Suppression of ischemia in arterial occlusive disease by JNK-promoted native collateral artery development.
Ramo K, Sugamura K, Craige S, Keaney JF, Davis RJ
eLife 2016 Aug 9;5
eLife 2016 Aug 9;5
Targeting myeloid-derived suppressor cells with colony stimulating factor-1 receptor blockade can reverse immune resistance to immunotherapy in indoleamine 2,3-dioxygenase-expressing tumors.
Holmgaard RB, Zamarin D, Lesokhin A, Merghoub T, Wolchok JD
EBioMedicine 2016 Apr;6:50-58
EBioMedicine 2016 Apr;6:50-58
Epithelial-to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis.
Lovisa S, LeBleu VS, Tampe B, Sugimoto H, Vadnagara K, Carstens JL, Wu CC, Hagos Y, Burckhardt BC, Pentcheva-Hoang T, Nischal H, Allison JP, Zeisberg M, Kalluri R
Nature medicine 2015 Sep;21(9):998-1009
Nature medicine 2015 Sep;21(9):998-1009
Mammary Stem Cells and Tumor-Initiating Cells Are More Resistant to Apoptosis and Exhibit Increased DNA Repair Activity in Response to DNA Damage.
Chang CH, Zhang M, Rajapakshe K, Coarfa C, Edwards D, Huang S, Rosen JM
Stem cell reports 2015 Sep 8;5(3):378-91
Stem cell reports 2015 Sep 8;5(3):378-91
Low levels of endogenous or X-ray-induced DNA double-strand breaks activate apoptosis in adult neural stem cells.
Barazzuol L, Rickett N, Ju L, Jeggo PA
Journal of cell science 2015 Oct 1;128(19):3597-606
Journal of cell science 2015 Oct 1;128(19):3597-606
NF-κB-induced microRNA-31 promotes epidermal hyperplasia by repressing protein phosphatase 6 in psoriasis.
Yan S, Xu Z, Lou F, Zhang L, Ke F, Bai J, Liu Z, Liu J, Wang H, Zhu H, Sun Y, Cai W, Gao Y, Su B, Li Q, Yang X, Yu J, Lai Y, Yu XZ, Zheng Y, Shen N, Chin YE, Wang H
Nature communications 2015 Jul 3;6:7652
Nature communications 2015 Jul 3;6:7652
Beta-Chemokine CCL15 Affects the Adhesion and Migration of Hematopoietic Progenitor Cells.
Richter R, Rüster B, Bistrian R, Forssmann WG, Seifried E, Henschler R
Transfusion medicine and hemotherapy : offizielles Organ der Deutschen Gesellschaft fur Transfusionsmedizin und Immunhamatologie 2015 Jan;42(1):29-37
Transfusion medicine and hemotherapy : offizielles Organ der Deutschen Gesellschaft fur Transfusionsmedizin und Immunhamatologie 2015 Jan;42(1):29-37
The adhesion G protein-coupled receptor GPR56 is a cell-autonomous regulator of oligodendrocyte development.
Giera S, Deng Y, Luo R, Ackerman SD, Mogha A, Monk KR, Ying Y, Jeong SJ, Makinodan M, Bialas AR, Chang BS, Stevens B, Corfas G, Piao X
Nature communications 2015 Jan 21;6:6121
Nature communications 2015 Jan 21;6:6121
Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression.
Chen L, Gibbons DL, Goswami S, Cortez MA, Ahn YH, Byers LA, Zhang X, Yi X, Dwyer D, Lin W, Diao L, Wang J, Roybal J, Patel M, Ungewiss C, Peng D, Antonia S, Mediavilla-Varela M, Robertson G, Suraokar M, Welsh JW, Erez B, Wistuba II, Chen L, Peng D, Wang S, Ullrich SE, Heymach JV, Kurie JM, Qin FX
Nature communications 2014 Oct 28;5:5241
Nature communications 2014 Oct 28;5:5241
Autophagy is a critical regulator of memory CD8(+) T cell formation.
Puleston DJ, Zhang H, Powell TJ, Lipina E, Sims S, Panse I, Watson AS, Cerundolo V, Townsend AR, Klenerman P, Simon AK
eLife 2014 Nov 11;3
eLife 2014 Nov 11;3
Dynamic changes in macrophage activation and proliferation during the development and resolution of intestinal inflammation.
Little MC, Hurst RJ, Else KJ
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4684-95
Journal of immunology (Baltimore, Md. : 1950) 2014 Nov 1;193(9):4684-95
Alveolar progenitor and stem cells in lung development, renewal and cancer.
Desai TJ, Brownfield DG, Krasnow MA
Nature 2014 Mar 13;507(7491):190-4
Nature 2014 Mar 13;507(7491):190-4
E and Id proteins influence invariant NKT cell sublineage differentiation and proliferation.
D'Cruz LM, Stradner MH, Yang CY, Goldrath AW
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2227-36
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2227-36
Myeloid cells expressing VEGF and arginase-1 following uptake of damaged retinal pigment epithelium suggests potential mechanism that drives the onset of choroidal angiogenesis in mice.
Liu J, Copland DA, Horie S, Wu WK, Chen M, Xu Y, Paul Morgan B, Mack M, Xu H, Nicholson LB, Dick AD
PloS one 2013;8(8):e72935
PloS one 2013;8(8):e72935
Small intestine inflammation in Roquin-mutant and Roquin-deficient mice.
Schaefer JS, Montufar-Solis D, Nakra N, Vigneswaran N, Klein JR
PloS one 2013;8(2):e56436
PloS one 2013;8(2):e56436
Dedifferentiation of committed epithelial cells into stem cells in vivo.
Tata PR, Mou H, Pardo-Saganta A, Zhao R, Prabhu M, Law BM, Vinarsky V, Cho JL, Breton S, Sahay A, Medoff BD, Rajagopal J
Nature 2013 Nov 14;503(7475):218-23
Nature 2013 Nov 14;503(7475):218-23
IL-1R signaling in dendritic cells replaces pattern-recognition receptors in promoting CD8⁺ T cell responses to influenza A virus.
Pang IK, Ichinohe T, Iwasaki A
Nature immunology 2013 Mar;14(3):246-53
Nature immunology 2013 Mar;14(3):246-53
Primate B-1 cells generate antigen-specific B cell responses to T cell-independent type 2 antigens.
Yammani RD, Haas KM
Journal of immunology (Baltimore, Md. : 1950) 2013 Apr 1;190(7):3100-8
Journal of immunology (Baltimore, Md. : 1950) 2013 Apr 1;190(7):3100-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
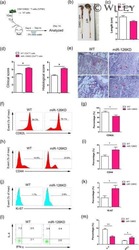
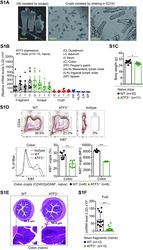
- C57Bl/6 splenocytes were unstimulated (left) or stimulated for 2 days with Anti-Mouse CD3 Functional Grade Purified (Product # 16-0031-82) (right). Cells were surface stained with Anti-Mouse CD19 FITC (Product # 11-0193-82) then fixed and permeabilized with the Foxp3 Staining Buffer Set (Product # 00-5523-00) and intracellularly stained with 0.06 µg of Anti-Mouse/Rat Ki-67 eFluor® 450. Total viable cells, as determined by Fixable Viability Dye eFluor® 780 (Product # 65-0865-14), were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
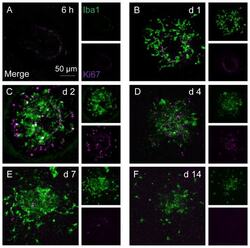
- Figure 4 Microglia/monocyte accumulation precedes appearance of non-myeloid cell proliferation. Iba1 and Ki67 dual-staining on RPE/choroid whole-mounts during the time course of CNV development. ( A ) Six hours post laser induction, neither macrophages nor cell proliferation are detected at lesions. ( B ) On day 1, accumulating Iba1 + cells are Ki67-negative and no proliferating cells are observed. By day 2 (C) and 4 (D), majority of macrophages are not co-localised with Ki67 immuno-reactivity, while abundant Iba1 - Ki67 + cells are seen at lesions. Between day 7 (E) and 14 (F), both Iba1 + and Ki67 + cell numbers subside. Images are representative of at least 12 lesions for each time point. Smaller images display separate stains for Iba1 and Ki67, respectively; bigger images are two channels merged.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
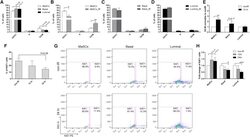
- Figure 2 Wild-Type MaSCs Exhibit Increased G2 Arrest and Evade Damage-Induced Quiescence after IR (A) Cell-cycle distributions of different subpopulations from MECs before IR were examined using PI staining (data are represented as mean +- SEM; n = 5). (B) A significant increase of cells in G2/M was observed in MaSCs 12 hr after IR (data are shown as mean +- SEM; n = 3; ** p < 0.01; * p < 0.05). (C and D) The cell-cycle profiles of basal and luminal compartments before and 12 hr after IR (data are shown as mean +- SEM; n = 3). (E) The fold change of cells in G2/M phase 12 hr after IR as compared to non-IR samples (data are shown as mean +- SEM; n = 3; ** p < 0.01). (F) Percentage of Ki67-positive MECs before and after IR (data are shown as mean +- SEM; n = 3). (G) Representative FACS plots of Ki67 in different subpopulations before and after IR. (H) Quantification of Ki67 positivity shows that basal and luminal cells became significantly more quiescent after IR (data are shown as mean +- SEM; n = 3; *** p < 0.001; * p < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
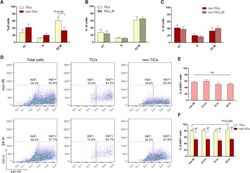
- Figure 6 p53- Tumor Cells Are Highly Proliferative and Fail to Exhibit Proper Cell-cycle Regulation after IR (A) Cell-cycle distribution of TICs and non-TICs before IR (data are shown as mean +- SEM; n = 4). (B and C) Twelve hours after IR, both subpopulations exhibit similar cell-cycle profiles as compared to their non-IR counterparts (data are shown as mean +- SEM; n = 4). (D) Representative FACS plots of Ki67 staining in total tumor cells, TICs, and non-TICs before and after IR. (E) Percentage of Ki67-positive cells in total tumor cells before and after IR (data are shown as mean +- SEM; n = 3). (F) Percentage of Ki67-positive cells in TICs and non-TICs before and after IR (data are shown as mean +- SEM; n = 3; * p < 0.05; ** p < 0.01).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
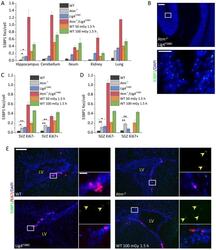
- Fig. 3. Similar levels of endogenous DSB formation in differentiated neuronal tissues and adult stem cell compartments. (A) Quantification of 53BP1 foci per cell in different tissues of untreated adult mice of the genotypes indicated and WT mice exposed to 50 or 100 mGy X-rays. (B) Representative images of untreated Atm - / - / Lig4 Y288C cerebellum stained for 53BP1 (green) and DAPI (blue). The lower image represents the boxed area shown in the upper image. Scale bars: 100 um (top), 20 um (bottom). (C,D) 53BP1 foci per cell in the adult SVZ and SGZ regions comparing proliferating (Ki67 + ) and non-proliferating (Ki67 - ) cells. Note that 53BP1 foci were not scored in the Atm - / - / Lig4 Y288C SGZ due to the low number of Ki67 + cells. (E) Representative images of the adult SVZ of untreated WT, Atm -/- and Lig4 Y288C mice, and WT SVZ 1.5 h after 100 mGy X-rays stained for Ki67 (red), 53BP1 (green) and DAPI (blue). Scale bars: 150 um (left), 20 um (right). The position of the lateral ventricle (LV) is shown for orientation. Yellow arrowheads show 53BP1 foci. Data represent mean+-s.e.m. (WT, n =4; Atm - / - , n =3; Lig4 Y288C , n =5; Atm - / - / Lig4 Y288C , n =3; WT 50 mGy 1.5 h, n =2; WT 100 mGy 1.5 h, n =3). ns, not significant; * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
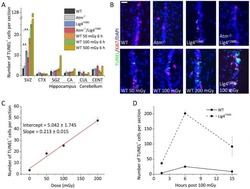
- Fig. 5. Endogenous and ionising radiation-induced apoptosis is sensitively activated in the adult SVZ. (A) TUNEL + cells per section in untreated WT, Atm - / - , Lig4 Y288C , Atm - / - / Lig4 Y288C adult mice, and following irradiation with 50, 100 or 500 mGy X-rays. The region scored in each section encompassed the entire area of the tissue under analysis. CTX, Isocortex ; CA, Ammon's Horn; CUL, culmen; CENT, central lobule. (B) Representative images of a portion of the ventral SVZ stained for Ki67 (red), TUNEL (green) and DAPI (blue). Scale bar: 25 um. The upper panel shows untreated mice and the lower panel figures were from mice killed at 6 h following irradiation with the indicated doses. (C) Dose-response and linear fitting of radiation-induced apoptosis in WT adult SVZ. (D) Timecourse of radiation-induced apoptosis in WT and Lig4 Y288C adult SVZ exposed to 100 mGy X-rays. Data represent mean+-s.e.m. (WT 50 mGy 6 h, n =3; WT 100 mGy 6 h, n =5; WT 500 mGy 6 h, n =1; WT 100 mGy 15 h, n =2; Lig4 Y288C 100 mGy 1.5 h, n =2; Lig4 Y288C 100 mGy 6 h, n =2; Lig4 Y288C 100 mGy 15 h, n =2). ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
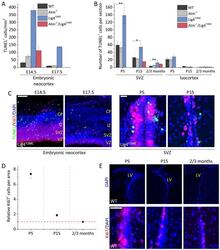
- Experimental details
- Fig. 6. Developmentally regulated ATM-independent apoptosis coupled with ATM-dependent DSB-induced apoptosis. (A) TUNEL + cells per mm 2 in the embryonal neocortex at E14.5 and E17.5. (B) TUNEL + cells per section in the postnatal (P5 and P15) and adult (2-3 months) SVZ and isocortex. Atm - / - / Lig4 Y288C marked P15 represents P20. To allow direct comparison, both the number of TUNEL + cells per section and per mm 2 area were scored in the SVZ region demonstrating a similar profile ( supplementary material Fig. S2B ). (C) Representative images of the embryonal neocortex and postnatal SVZ of untreated Lig4 Y288C mice stained for Ki67 (red), TUNEL (green) and DAPI (blue). Scale bars: 100 um (left), 25 um (right). (D) Quantification of Ki67 + cells per unit area (0.07 mm 2 ) in the SVZ of P5, P15 and 2-3-month-old WT mice relative to the number of Ki67 + cells in the same area in 2-3-month-old WT mice. (E) Representative images of the lateral ventricle (LV) (top) of P5, P15 and 2-3-month-old WT mice stained for DAPI (blue) and a portion of the SVZ region (bottom) stained for Ki67 (red) and DAPI (blue). CP, cortical plate; IZ, intermediate zone; VZ, ventricular zone. Data represent mean+-s.e.m. ( n as described in Fig. 4 ). * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
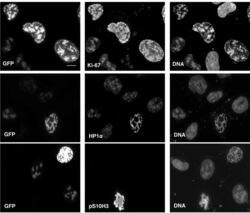
- Experimental details
- Figure 11--figure supplement 1. Overexpression of Ki-67 induces ectopic heterochromatin. Overexpression of full length Ki-67 N-terminal fusion with eGFP (GFP, left panels) in U2OS cells induces ectopic heterochromatin, as visualised by DAPI staining (DNA, right panels) or immunofluorescence of HP1alpha (centre, middle), whereas cells with lower Ki-67 expression levels have normal chromatin. Immunofluorescence of Ki-67 (top, middle) shows colocalisation of fusion protein with overall Ki-67 pattern. Immunofluorescence of phospho-histone H3S10 shows expected staining of mitotic metaphase (bottom, middle panel) but no staining in a cell with ectopic heterochromatin (top right cell, same panel) due to Ki-67 overexpression (GFP, bottom left panel). Bar, 10 um DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
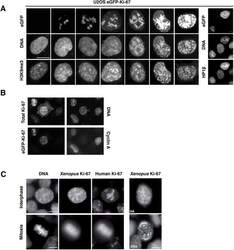
- Main image
- Experimental details
- Figure 11. Overexpression of Ki-67 induces ectopic heterochromatin. ( A ) Overexpression of full length Ki-67 N-terminal fusion with eGFP in U2OS cells induces ectopic heterochromatin, as visualised by DAPI staining (middle) and immunofluorescence of H3K9Me3 (left) or HP1beta (right). Eight representative cells that have different levels of Ki-67 expression, as determined by eGFP fluorescence intensity, are shown. Bar, 10 um. ( B ) U2OS cells expressing high levels of exogenous eGFP-Ki-67 and showing ectopic chromatin condensation are negative for cyclin A staining by immunofluorescence. ( C ) Left: Immunofluorescence analysis of the localisation of endogenous human and ectopically expressed Xenopus Ki-67 in U2OS cells, showing colocalisation in metaphase at the perichromosomal region. Right: DNA condensation caused by high overexpression of Xenopus Ki-67 in U2OS cells. Bars, 10 um. DOI: http://dx.doi.org/ Figure 11--figure supplement 1. Overexpression of Ki-67 induces ectopic heterochromatin. Overexpression of full length Ki-67 N-terminal fusion with eGFP (GFP, left panels) in U2OS cells induces ectopic heterochromatin, as visualised by DAPI staining (DNA, right panels) or immunofluorescence of HP1alpha (centre, middle), whereas cells with lower Ki-67 expression levels have normal chromatin. Immunofluorescence of Ki-67 (top, middle) shows colocalisation of fusion protein with overall Ki-67 pattern. Immunofluorescence of phospho-histone H3S10 shows expected staining of mitoti
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
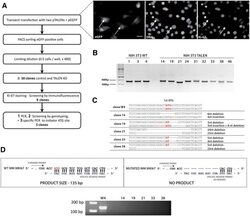
- Experimental details
- Figure 3--figure supplement 8. Generation of Ki-67-mutant NIH-3T3 cells. ( A ) Top and left, schematic representation of TALEN-mediated generation of NIH-3T3 biallelic Ki-67 mutant cells. Top right, immunofluorescence of Ki-67 and eGFP staining in NIH 3T3 cells transfected with two plasmids encoding TALEN pair and plasmid pEGFP. White arrows show Ki-67-negative pEGFP cotransfected cells. Scale bar 25 um. ( B ) PCR analysis of Mki67 initiator ATG surrounding sequence in genomic DNA prepared from three WT clones and nine Ki-67 immunofluorescence-negative clones selected for further analysis. ( C ) sequencing of Mki67 initiator ATG area from selected clones (14, 19, 21, 33, 38). ( D ), PCR analysis targeted to the initiator ATG in Mki67 gene of genomic DNA purified from NIH 3T3 WT clone W4 and Ki-67 KO clones 14, 19, 21, 33, 38. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
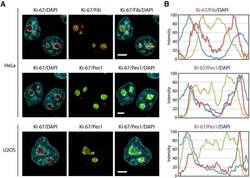
- Experimental details
- Figure 7--figure supplement 1. Ki-67 is a nucleolar protein localizing in the cortical side of the GC. ( A ) Localisation of Ki-67 protein by immunofluorescence in HeLa and U2OS cells. Fibrillarin (DFC) or Pes1 (GC) were used as nucleolar markers. Images were captured in confocal mode with a spinning-disk microscope. Objective 100 x. Scale bar: 5 mum. ( B ) Line scans showing the distribution of fluorescence signals within specific nucleoli (see dotted lines in panel A). DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
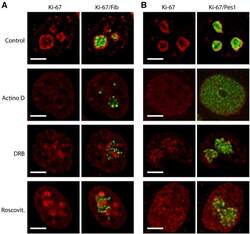
- Experimental details
- Figure 7--figure supplement 2. Ki-67 follows GC components upon drug-induced nucleolar disruption. Actinomycin D or the kinase inhibitors DRB and Roscovitine were added to cells during 90 min and Ki-67 was located within these cells by immunofluorescence. Fibrillarin ( A ) and PES1 ( B ) proteins were also localised by immunofluorescence. Objective 100 x. Scale bar: 5 mum. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
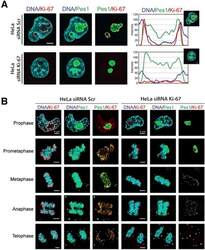
- Experimental details
- Figure 7. Ki-67 localises PES1 to mitotic chromosomes. ( A ) Analysis of the interphase localisation of PES1 and Ki-67 proteins by immunofluorescence in HeLa cells 72 hr after transfection with control siRNA (scramble; Scr) or Ki-67 RNAi. Right, line scans showing the distribution of fluorescence signals within indicated nucleoli (dashed line). Images were captured in confocal mode with a spinning-disk microscope. Bar, 5 mum. ( B ) Analysis of the mitotic localisation of PES1 and Ki-67 proteins by immunofluorescence in HeLa cells 72 hr after transfection with control siRNA (scramble; Scr) or Ki-67 RNAi. Bar, 5 mum. DOI: http://dx.doi.org/ Figure 7--figure supplement 1. Ki-67 is a nucleolar protein localizing in the cortical side of the GC. ( A ) Localisation of Ki-67 protein by immunofluorescence in HeLa and U2OS cells. Fibrillarin (DFC) or Pes1 (GC) were used as nucleolar markers. Images were captured in confocal mode with a spinning-disk microscope. Objective 100 x. Scale bar: 5 mum. ( B ) Line scans showing the distribution of fluorescence signals within specific nucleoli (see dotted lines in panel A). DOI: http://dx.doi.org/ Figure 7--figure supplement 2. Ki-67 follows GC components upon drug-induced nucleolar disruption. Actinomycin D or the kinase inhibitors DRB and Roscovitine were added to cells during 90 min and Ki-67 was located within these cells by immunofluorescence. Fibrillarin ( A ) and PES1 ( B ) proteins were also localised by immunofluorescence. Objective 10
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
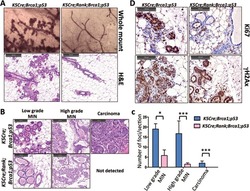
- Experimental details
- Figure 1 Ablation of Rank in mammary epithelial cells markedly decreases tumor formation in Brca1/p53 mutant female mice. (A) Representative whole mount images (haematoxylin staining, magnification 52x) and paraffin sections (H&E staining, scale bar, 200 mum) of mammary glands from 4-month-old K5Cre;Brca1;p53 double- and K5Cre;Rank;Brca1;p53 triple-knockout littermate mice. (B) Representative images (H&E staining, scale bar, 100 mum) and (C) quantification of low-grade MINs, high-grade MINs and adenocarcinomas in mammary glands from 4-month-old K5Cre;Brca1;p53 and K5Cre;Rank;Brca1;p53 mutant littermates. Data are shown as average number of foci/section of 1 inguinal and 2 thoracic mammary glands per mouse +/- SEM. n >= 4 mice/group. * P < 0.05, *** P < 0.001 (2-way ANOVA). (D) Representative images of Ki67 and gammaH2AX immunostaining of mammary glands from 4-month-old K5Cre;Brca1;p53 double- and K5Cre;Rank;Brca1;p53 triple-knockout littermates. Scale bar, 100 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
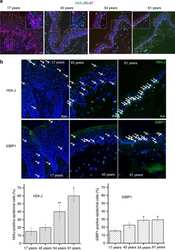
- Figure 10 H2A.J accumulates in the aging human epidermis. ( a ) H2A.J and Ki-67 co-localization in sections of human skin of the indicated ages by immunofluorescent staining. H2A.J showed mutually exclusive staining with the Ki-67 proliferation marker. ( b ) H2A.J and 53BP1 foci increase in aging human epidermis. Arrows indicate positively-stained nuclei. A quantification of positive cells for three biological replicates is shown below the immunofluorescence images, and the one-sided Mann-Whitney U -test was used to determine the statistical significance (* P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
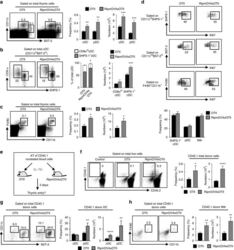
- Fig. 1 Ag-specific interactions between mTECs and CD4 + T cells increase the thymic entry of circulating DCs and macrophages. a - c Flow cytometry profiles, frequencies and numbers of cDCs (CD11c hi BST-2 lo ), pDCs (CD11c int BST-2 hi ) ( a ), resident cDCs (CD8alpha hi SHPS-1 - ), migratory cDCs (CD8alpha lo SHPS-1 + ) ( b ) and macrophages (F4/80 + CD11b + ) ( c ) in the thymus from OTII- Rag2 -/- and RipmOVAxOTII- Rag2 -/- mice. Data are representative of three independent experiments ( n = 3 mice per group and per experiment). d Flow cytometry profiles and frequencies of proliferating Ki-67 + thymic DC subsets and macrophages. Data are representative of two independent experiments ( n = 3 mice per group and per experiment). e Experimental setup: nucleated blood cells from CD45.1 WT congenic mice were adoptively transferred into sublethally irradiated CD45.2 OTII- Rag2 -/- and RipmOVAxOTII- Rag2 -/- recipients. Three days after i.v . adoptive transfer (AT), the thymic entry of DCs and macrophages of CD45.1 donor origin was analysed. SL-TBI: sublethal total body irradiation. f - h Flow cytometry profiles, frequencies and numbers of CD45.1 total donor cells ( f ) as well as cDCs, pDCs ( g ) and macrophages ( h ) of CD45.1 donor origin in the thymus from OTII- Rag2 -/- and RipmOVAxOTII- Rag2 -/- recipients. Control: non-injected irradiated OTII- Rag2 -/- mice. Data are representative of three independent experiments ( n = 3-4 mice per group and per experiment). d , h MPhi: m
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
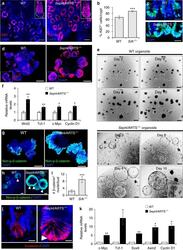
- Fig. 4 Deletion of Sept4 /ARTS leads to enhanced activity of the Wnt/beta-catenin pathway. a Intestinal tissue wholemount stained for Ki67, viewed from the base of the crypt, shows enhanced crypt base proliferation in Sept4 /ARTS -/- ( S /A -/- ) mice. Inset shows isolated and stained crypts, which display greater proliferation in the S /A -/- crypt base (dotted white line). b Quantification of percentage of Ki67 + proliferating cells per crypt. c WT and S /A -/- intestinal sections stained for PCNA. d WT and Sept4 /ARTS -/- intestinal organoids stained against Ki67. e S /A -/- organoids often display cystic-like morphology that continue to expand up to 10 days in culture. Inset shows S /A -/- cystic organoid with characteristic organoid ""buds"". f Real-time (RT)-PCR analysis shows increased relative mRNA transcripts of Wnt3, Tcf-1, c-Myc and Cyclin D1 in control and S /A -/- organoids. g WT and S /A -/- intestinal organoids stained against non-phosphorylated beta-catenin. h Zoom-in of intestinal organoids shows nuclear beta-catenin + cells (white arrowheads) within the S /A -/- organoid crypt. i Quantifications for number of nuclear beta-catenin + cells per organoid crypt. j Small intestinal crypts in vivo stained for beta-catenin show high nuclear localization (white arrowhead) in the Sept4 /ARTS -/- crypt base. k RT-PCR analysis indicates increased relative mRNA levels of Wnt target genes c-Myc , Tcf-1 , Sox9 , Axin2 and
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 8 ARTS mediates its functions via interaction with XIAP in the intestinal crypt. a Confocal microscopy image of mouse small intestinal crypt base shows co-localization of ARTS and XIAP within the same cell [ n = 4 mice]. b Western blot of XIAP in isolated wild-type (WT) and Sept4 /ARTS -/- ( S /A -/- ) crypts. Signal intensity is normalized to GADPH. c Super-resolution stimulated emission depletion (STED) microscopy shows co-localization of ARTS and XIAP in normal and apoptotic human colonic crypt base cells. d Pearson's coefficient values demonstrate higher co-localization of ARTS and XIAP in both mouse and human apoptotic crypt cells [ n = 3 human colons and n = 4 mice. Error bars represent s.e.m.]. e Co-immunoprecipitation (co-IP) of ARTS and XIAP in isolated XIAP DeltaRING small intestinal crypts. XIAP DeltaRING only interacts mildly with ARTS in the absence of apoptotic stimulation. After staurosporine (STS) treatment, efficient binding between ARTS and XIAP DeltaRING is detected. f Western blot and signal intensity of active caspase-3 (CP3) in STS-treated crypts show that deletion of XIAP or RING domain increases cleaved CP3 levels. g Organoids derived from XIAP -/- , XIAP DeltaRING and S;X -/- crypts display hindered growth and development after 11 days post seeding. h XIAP -/- , XIAP DeltaRING and S;X -/- organoids stained for Ki67. i , j Fold difference in i organoid formation capacity and j number of Ki67 + cells
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
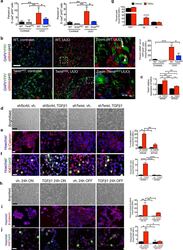
- Experimental details
- Figure 5 EMT program G2 cell cycle arrest. ( a ) Percent E-cadherin + pH3 + cells in kidneys from the indicated experimental groups. WT contralat., n = 4; Twist cKO contralat., n = 5; WT UUO, n = 4; Twist cKO UUO, n = 5. WT contralat., n = 3; Snail cKO contralat., n = 3; WT UUO, n = 4; Snail cKO UUO, n = 3. ( b ) Representative images (5 visual fields for each tissue analyzed) of immunolabeling for YFP, AQP1 and pH3 and percentage of alphaSMA + cells out of the YFP + pH3 + TECs (contralat., n = 6; UUO n = 3). Scale bar, 100 ~m. ( c ) Relative Twist1 expression in indicated cells. ( d ) Representative images of brightfield imaging in indicated cells. ( e ) Representative images (3 visual fields for each tissue analyzed) of immunolabeling for beta-catenin and percent nuclear accumulation. Scale bar, 50 ~m. ( f,g ) Representative images (3 visual fields for each tissue analyzed) of immunolabeling for Ki67 and pH3 ( f ) and percent cells in G2 ( g ). Scale bar, 50 ~m. ( h ) Representative images of brightfield imaging in indicated cells. ( i ) Representative images (3 visual fields for each tissue analyzed) of immunolabeling for beta-catenin and percent nuclear accumulation. Scale bar, 50 ~m. ( j ) Representative images (3 visual fields for each tissue analyzed) of immunolabeling for Ki67 and pH3 ( f) and percent cells in G2 ( g ). Scale bar, 50 ~m. Vehicle (vh) and TGF-beta1 treatment were conducted for 24 hours, followed by vehicle or TGF-beta1 withdrawal for 24 hours, n = 3. D
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
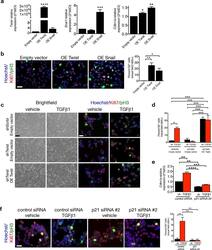
- Experimental details
- Figure 6 p21 controls EMT program G2 cell cycle arrest. ( a ) Relative expression of Twist1 Snai1 and Cdkn1a (p21) in MCT cells transfected with empty vector, Twist or Snail overexpression vectors, 24 hours post transfection, n = 3. ( b ) Representative images (3 visual fields for each tissue analyzed) of vehicle or MCT cells transfected with empty vector, Twist or Snail overexpression vectors, 24 hours post transfection, immunolabeled for Ki67 and pH3 and respective quantification of the percentage of cells in G2 phase, n = 3. Scale bar: 50 ~m. ( c ) Phase contrast light microscopy and immunolabeling for Ki67 and pH3 of MCT shScrbl and MCT shTwist cells transfected with empty or Twist overexpression (OE Twist) plasmids and treated with vehicle or TGF-beta1. ( d ) Quantification of the percentage of empty or Twist OE transfected MCT shScrbl and MCT shTwist cells in G2 phase of the cell cycle comparing cells treated with vehicle or TGF-beta1, n = 3. ( e ) Relative expression of Cdkn1a (p21). ( f ) Representative images (3 visual fields for each tissue analyzed) of immunolabeling for Ki67 and pH3 of control or p21 siRNA-transfected MCT cells and percent of cells in G2 phase. Scale bar, 50 ~m. Data is represented as mean +- SEM. Hoechst: nucleus. One-way ANOVA with Tukey post-hoc analysis. b and e , unpaired one-tailed t-test. * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
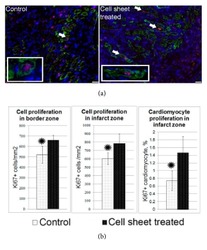
- Experimental details
- Figure 7 Total and cardiomyocyte proliferation in the left ventricle wall 14 days after myocardial infarction and delivery of CPC-based cell sheets. (a) Representative images of cardiomyocyte proliferation in control group and after cell sheet treatment. Two weeks after epicardial delivery of the CPC sheet, the heart sections were costained with the antibodies against the cell-proliferation-associated antigen-Ki67 (red fluorescence) and cardiomyocyte marker, Troponin I (green fluorescence). Combined red and green fluorescence and DAPI-stained nuclei (blue) are shown in merged images. Arrows indicate costained proliferated cardiomyocytes. (b) Bar graphs show quantitative data of the number of total proliferating cells in border or infarct zones and cardiomyocyte proliferation. Data is presented as mean+-SD. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
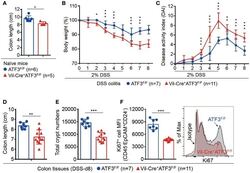
- Experimental details
- Figure 4 Epithelial ATF3 is required for protection against DSS colitis. (A) Comparison of colon length between 3-month-old naive mice as indicated. (B-F) Analysis of colitis severity during DSS treatment. (B) Percentage of body weight loss during DSS colitis. (C) Disease activity index (weight loss percentage, stool consistency, and blood in stools) was indicated in each group of mice during DSS colitis. (D) Colon length, (E) total colon crypt numbers, and (F) Ki67 + proliferating crypt cells by flow cytometry analysis, were measured at day-8 post DSS treatment. Results were from two independent experiments. ""n"" refers to the number of mice analyzed. Statistical analysis was done using Multiple T -test on Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
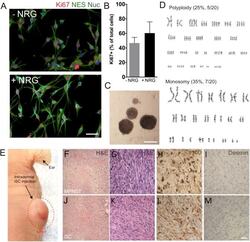
- FIGURE 1 The development of a malignant peripheral nerve sheath tumors (MPNST) model. (A) Representative immunocytochemical images of induced tumor Schwann cells (iSCs) either deprived of growth factors (-NRG) or treated with neuregulin and forskolin (+NRG, 50 ng/ml neuregulin, 5 mM Forskolin). Note the similar numbers of Nestin+ (green, NES), Hoechst+ (blue) iSCs that express Ki67 (red) across both conditions. (B) Quantification of percentage of Ki67 + cells revealed no difference between groups (Student's t -test, n = 4, P = 0.3). (C) Note the presence of sphere formation when iSCs are grown in growth factor deprived conditions. (D) Karyotyping analysis indicates multiple chromosomal abnormalities, including polyploidy and monosomy cells. (E) Intradermal iSC injections into back skin resulted in the development of tumors within 16 weeks. (F-M) Representative histological images of H&E (F,G,J,K) , s100 (H,L) and Desmin (I,M) from a biopsied sample of human MPNSTs (F-I) and from a sample from iSCs (J-M) . Note the presence of necrosis, mitosis and spindle-shaped cells in both samples (F,G and J,K) . Also note the presence of S100 (H,L) immunoreactivity, as well as the lack of Desmin immunoreactivity (I,M) in both samples. Scale bars = A (50 mum), C (200 mum), G-I (20 mum).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
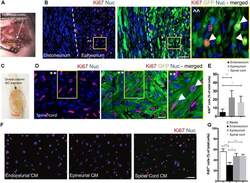
- FIGURE 2 The endoneurial microenvironment suppresses tumorigenesis of MPNST. (A) Intraneural iSC injections into the sciatic nerve resulted in the development of tumors within 2 months. (B) Representative immunohistochemical images of the endoneurial and epineurial compartments. Note the presence of widespread Ki67+ (red), Hoechst+ (blue) proliferative iSC (green) in the epineurial compartment compared to the endoneurial compartment. See inset (^^) for high-resolution example of localization (arrowheads). (C) iSCs injected into the dorsal column of the spinal cord formed tumors within 2 months. (D) Representative immunohistochemical images of the spinal cord. Note the presence of widespread Ki67+ (red), Hoechst+ (blue) proliferative iSC (green) in the spinal cord. See inset ( ** ) for high-resolution example of localization (arrowheads). (E) Quantification of the percentage of Ki67+ iSCs in the endoneurial compartment, epineurial compartment and spinal cord demonstrated a significant decrease in endoneurial compartment compared to other regions (One-way ANOVA, Tukey's posthoc test, n = 6-8, * p < 0.01, ** p < 0.001). (F) Representative immunocytochemical images of iSCs treated with conditioned media (CM). Note there are less Ki67+ (red) Hoechst+ (blue) iSCs in the cultures treated with endoneurial CM compared to epineurial, spinal cord or base media. (G) Quantification of the percentage of Ki67+ iSCs demonstrated a significant decrease in the percenta
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
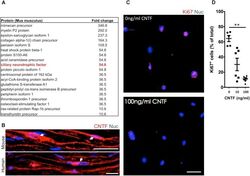
- FIGURE 4 The identification of suppressive factors in the endoneurial compartment. (A) Fold change in candidate proteins (10-60 kDa) enriched in the endoneurium compared to spinal cord tissue. Ciliary neurotrophic factor (CNTF, red box), a growth factor known for its role in promoting differentiation of Schwann cells, was found at levels 5.5-fold higher in endoneurium tissue compared to spinal cord tissue. (B) Representative immunohistochemical images of uninjured mouse and human sciatic nerves demonstrating the presence of CNTF (red) in the cytoplasm of Schwann cells (blue). Note the presence of CNTF around the peri-nuclear area adjacent to nuclei (blue)--a cytoplasmic rich region of the myelinating Schwann cell. (C) Representative immunocytochemical images of iSCs (blue) treated with 0 ng/ml and 100 ng/ml CNTF. Note the reduction in Ki67+ (red) cells in CNTF treated conditions. (D) Quantification of the percentage of Ki67+ cells treated with 0, 10, or 100 ng/ml CNTF revealed a reduction in ki67+ cells at 100 ng/ml compared to 0 ng/ml (One-way ANOVA, Tukey's posthoc test, n = 3, ** p < 0.05). Scale bars = B (25 mum), C (50 mum).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
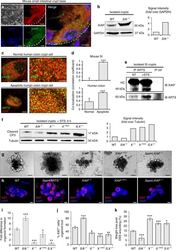
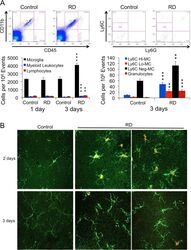
- Figure 3 Effect of RD on mouse retinal microglia and leukocyte populations. (A) Flow cytometry of microglia and leukocyte populations in the mouse retina at 1 day (n = 2/group) and 3 days (n = 4/ group) following experimental RD. Populations were defined by gating on the leukocyte common antigen CD45 and the myeloid lineage marker CD11b. Gating for inflammatory monocyte marker Ly6C and granulocyte/neutrophil marker Ly6G further defined populations of microglia and subpopulations of myeloid leukocytes. (B) Immunofluorescence of flat-mounted mouse retinas at 2 and 3 days after detachment showing the proliferation of microglia. Iba-1 + (green) cells with ramified morphology are microglia; Ki67 (red) is a proliferative cell marker.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
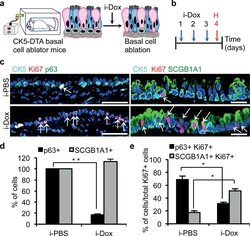
- Figure 1 Secretory cells proliferate after basal cell ablation a, Schematic representation of the ablation of CK5 -expressing basal cells of the trachea. Secretory, ciliated and basal stem cells are shown in pink, blue and grey colors respectively. b, Schematic of the timeline of i-dox or i-PBS administration and tissue harvest. c, Immunostaining for basal (p63 (green) and CK5 (cyan)) and secretory cells (SCGB1A1 (green)) in combination with Ki67 (red) on either i-PBS (upper panels) or i-Dox (lower panels) treated mice (n=6). White arrows, Ki67+ cells. d, Quantification of the percentage of p63+ and SCGB1A1+ cells per total DAPI+ cells in i-PBS or i-Dox n=3. e, Percentage of p63+Ki67+ and SCGB1A1+Ki67+ cells relative to total Ki67+ cells in i-PBS and i-Dox (n=3) treated CK5-DTA mice. i-Dox, inhaled doxycycline; i-PBS, inhaled PBS. Nuclei, DAPI (blue). *- p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
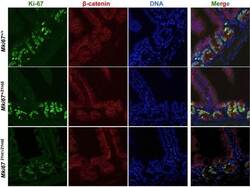
- Figure 3--figure supplement 3. Background Ki-67 levels in Ki-67 mutant mice. Immunofluorescence of Ki-67 and beta-catenin on sagittal sections of the mouse intestinal epithelium from Mki67 +/+ , heterozygous Mki67 +/21nt and homozygous Mki67 21nt/21nt mice. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
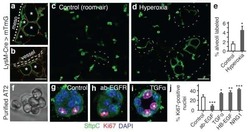
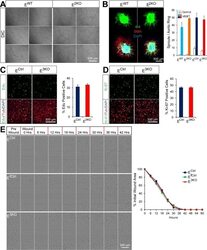
- Figure 1--figure supplement 4. Endothelial JNK is not required for proliferation, migration, and angiogenic responses in vitro. ( A ) JNK-deficient and control primary MLEC form similar tubular networks in matrigel. Images are representative of two experiments performed in triplicate with independent primary MLEC preparations. ( B ) Representative maximum projection confocal images of collagen embedded aortic ring explants. Similar numbers of VEGF-induced iB4 (green) positive microvessels sprouting from aortic rings from control and endothelial JNK-deficient mice were detected. Smooth muscle actin (SMA) immunofluorescence (red) labels supporting cells. DAPI (blue) labels nuclei. Quantitation of microvessel number demonstrated no significant differences between aortic rings from control and JNK-deficient mice (mean +- SEM; n = 8~21 rings per group). The data presented were obtained in one experiment and are representative of three experiments with similar results. Aortas from 2~3 mice per group were used in each experiment. ( C , D ) Representative confocal images and quantitation of the percentage of endothelial cells staining positive for the incorporation of Edu (green, C ) or the proliferation marker Ki-67 (green, D ) (mean +- SEM; n = 10 images per group). The data presented were obtained in one experiment and are representative of three experiments with similar results. alphaTubulin (red) labels cell bodies. DAPI (blue) labels nuclei. ( E ) Endothelial monolayers were ex
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
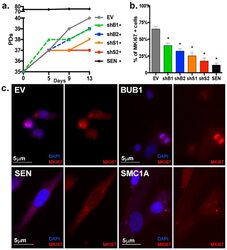
- Figure 2 Proliferation of HPF is impaired upon BUB1 or SMC1A knockdown. ( a ) Growth curve of EV, BUB1 or SMC1-depleted and SEN cultures as assessed by number of PDs. ( b ) Quantification of percentage of MKI67 positively stained cells per cell line. ( c ) Representative IF images for the proliferation marker MKI67 (red) in EV, SEN, BUB1 and SMC1A-depleted cells. (*) Indicates significant differences ( p < 0.05) from EV cells tested by One-way ANOVA. Data are expressed as mean +- SD (n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
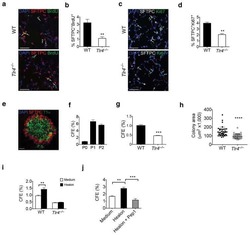
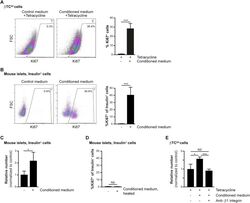
- Figure 2 Increased beta-cell proliferation upon exposure to pericyte-conditioned medium . A ) Tetracycline-treated betaTC-tet cells were cultured in either control (complete DMEM; 'Control medium') or neonatal pericyte-conditioned ('Conditioned medium'; described in Figure 1 B) medium, both supplemented with tetracycline. After incubation for 96 h, cells were fixed and stained for the proliferative marker Ki67. Left , representative dotplots showing flow-cytometry analysis of Ki67 expression by betaTC-tet cells. Gated are Ki67 + cells; the numbers represent the percentage of gated cells out of the analyzed cell population. Right , Bar diagrams (mean +- SD) represent the percentage of Ki67 + cells. N = 3. ***P < 0.005 (Student's t -test), as compared to the control medium. A representative of three independent experiments is shown. B ) Isolated islets from 3-month-old wild-type mice were cultured in either control (complete DMEM; 'Control medium') or neonatal pericyte-conditioned ('Conditioned medium'; described in Figure 1 B) medium for 24 h. Islets were dispersed to single cells, fixed, and stained for insulin and the proliferative marker Ki67. Left , representative dotplots showing flow-cytometry analysis of Ki67 expression by insulin + cells. Gated are Ki67 + cells; the numbers represent the percentage of gated cells out of the total insulin + cell population. Right , Bar diagrams (mean +- SD) represent the percentage of Ki67 +
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
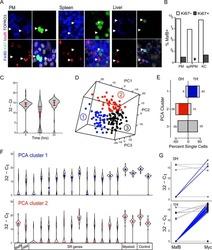
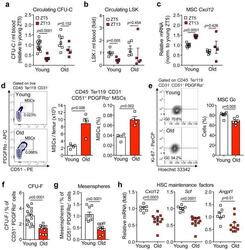
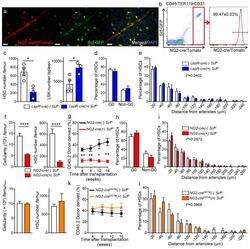
- Figure 3 Aging expands MSCs and reduces their HSC maintenance activity (a, b) Circadian oscillations of circulating CFU-C (normalized to young at ZT5; n=13 young ZT5, 7 old ZT5, 7 young ZT13 and 4 old ZT13 mice) (a) and lineage - Sca-1 + c-Kit + (LSK) progenitors (normalized to young at ZT5; n=5 mice per group) (b) in peripheral blood of young and old C57BL/6 mice. (c) Quantification of Cxcl12 mRNA levels relative to Actb in sorted MSCs from young and old C57BL/6 mice at ZT5 and ZT13 (normalized to young at ZT5; n=8 young ZT5, 5 old ZT5, 5 young ZT13 and 4 old ZT13 mice). (d) Left, representative FACS plots showing the gating strategy for CD45 - Ter119 - CD31 - CD51 + PDGFRalpha + MSCs in young (top) and old (bottom) C57BL/6 mice. Right, absolute numbers and frequency of MSCs in young and old C57BL/6 mice (n=4 mice per group). (e) Left, representative FACS plots showing the gating strategy for MSCs Ki-67 and Hoechst 33342 staining in young (top) and old (bottom) C57BL/6 mice. Right, quantification of Ki-67 - G0 MSCs in young and old C57BL/6 mice (n=6 mice per group). (f, g) Frequency of CFU-F (n=15 cultures per group) (f) and mesenspheres (n=9 young, 11 old cultures) (g) from sorted MSCs plated at equal numbers and clonal densities under CFU-F or mesensphere culture conditions (n=5 mice per group). (h) Quantification of mRNA levels of Cxcl12, Scf and Angpt1 relative to Gapdh in sorted MSCs (normalized to young; n=7 young, 11 old mice)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
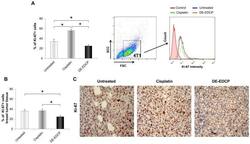
- Figure 6 DE-EDCP treatment attenuates expression of Ki-67 in murine breast cancer (A) Analysis of Ki-67 expression in 4T1 cells exposed to DE-EDCP or cisplatin (31.25 muM) for 24h using flow cytometry by first gaiting out cell debris and cell clumps in forward/side scatter plot. Data are presented as the mean +- SD, ( * DE-EDCP vs. untreated p=0.020; DE-EDCP vs. cisplatin p=0.002; cisplatin vs. untreated p=0.009). Representative histograms of three independent experiments are shown. (B, C) At 36 th day of the experiment, tumors were harvested from tumor-bearing mice treated with DE-EDCP, cisplatin and vehicle and Ki-67 expression was detected using immunohistochemical method. Representative images and quantitative analysis of the percentage of Ki-67- positive cells are shown. Ki-67-positive cells were counted in five random fields (magnification at x 400), and data were summarized as the mean percentage of positive cells (four tumors per group). Data are presented as mean +- SE. ( * DE-EDCP vs. untreated p=0.006; DE-EDCP vs. cisplatin p=0.004)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
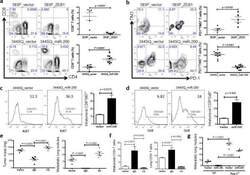
- Figure 4 The miR-200/ZEB1 axis controls tumor metastasis through regulating CD8 + TILs ( a , b ) FACS analysis of ( a ) CD8 + TIL frequency; ( b ) PD1 and TIM3 marker expression on CD8 + T cells from 393P_vector and 393P_ZEB1 (n = 5), as well as 344SQ_vector and 344SQ_miR-200 (n = 10) primary tumors. Analysis was done 2 weeks post-cancer cell injection. ( c , d ) ( c ) Intratumoral Ki67 + CD8 + T cells; ( d ) granzyme B (GzB) + CD8 + T cells in 344SQ_vector or 344SQ_miR-200 primary tumors 6 weeks post-subcutaneous injection of cancer cells into 129/Sv mice. Representative Ki67 or GzB staining in an individual tumor sample is shown on the left, and mean Ki67 + or GzB + populations of gated CD8 + T cells in total T cells are shown on the right (n = 5). ( e ) CD8 + T cell depletion results in tumor growth and metastasis in mice (n = 5) that received subcutaneous tumor cell injections. No treatment (344SQ_vector (Vector)), IgG (344SQ_miR-200 + IgG control), or Ab (344SQ_miR-200 + anti-CD8 Ab). The analysis was done 6 weeks post-injection. ( f ) Relative abundance of CD8 + T cells in the tumor (left) or lung (right) from 129/Sv mice (n =5) with syngeneic control 344SQ tumors (Vector), 344SQ_miR-200 tumors with control IgG treatment (IgG) or anti-CD8 antibody treatment (Ab). ( g ) Lung metastases of 344SQ_vector (Vector) and 344SQ_miR-200 (miR-200) tumors in wild-type (WT) or 129/Sv Rag2 -/- ( Rag2 -/- ) mice (n = 5). The analysis was done 6 weeks post-tumor cell subcutaneous injec
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
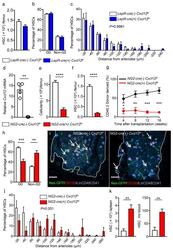
- Figure 3 Cxcl12 from distinct peri-vascular niche cells contributes differentially to HSC functions (a-c) Analyses of LepR-cre/ Cxcl12 fl/- mice. (a) Absolute numbers of HSCs in BM. n=6 mice for each group. (b) FACS analyses of cell cycle of HSCs with Ki-67 and Hoechst 33342 staining. n=5 mice per group. (c) HSC localization relative to arterioles. Error bars: n=3 mice. The p value has been calculated using n=129 HSCs for cre (-), 160 HSCs for cre (+), pooled from 3 mice per group. P =0.9981. (d-k) Analyses of NG2-cre / Cxcl12 flox/-. mice (d) Cxcl12 mRNA expression relative to beta-actin in CD45 - TER119 - CD31 - Nes-GFP + cells from NG2-cre(-) Cxcl12 f/- and NG2-cre(+) Cxcl12 f/- mice. n=4 mice for cre (-), n=3 mice for cre (+), from two independent experiments. (e,f) Bone marrow cellularity (e) and absolute numbers of phenotypic CD150 + CD48 - Lineage - Sca-1 + c-kit + (LSK) HSCs (f) per one femur. n=10 mice. (g) Percentages of donor-derived cells after competitive reconstitution. n=5 mice per group. (h) Quantification of cell cycle of HSCs with Ki-67 and Hoechst 33342 staining. n=5 mice for cre (-), n=7 mice for cre (+). (i) Representative images of whole-mount immunofluorescent staining of the sternal bone marrow from 3 mice. Arrows indicate CD150 + CD48 - CD41 - Lineage - HSCs. Dashed lines depict the border between bone and bone marrow. Scale bars, 100 muH. (j) HSC localization relative to arterioles. Error bars: n=3 mice for cre (-), n=4 mice for cre (+). The p value
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 NG2-cre, but not NG2-cre ERTM , targeted cells are the source of Scf in the bone marrow (a) Whole-mount sternum from NG2-cre/ iTdTomato/ Scf-GFP mice, anti-VE-cadherin. Representative images from 3 mice. Scale bars, 20 mum. (b) Representative FACS plot showing percentage of NG2-cre/ iTdTomato + cells within CD45 - TER119 - CD31 - Scf-GFP + cells. n=3 mice. (c-e) Analyses of LepR-cre/ Scf fl/- mice. (c) Numbers of HSCs (left) in BM and LSK cells in spleen (right). n=4 mice for cre (-), n=3 mice for cre (+). (d) FACS analyses of HSC (CD150 + CD48 - LSK) cell cycle with Ki-67 and Hoechst 33342 staining. n=5 mice for cre (-), n=6 mice for cre (+). (e) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=272 HSCs for cre (-), 293 HSCs for cre (+) pooled from 3 mice per group. P =0.3402. (f-i) Analyses of NG2-cre/ Scf fl/- mice. (f) Numbers of total BM cells (left) and CD150 + CD48 - LSK HSCs (right) in BM. n=5 mice for cre (-), n=7 mice for cre (+). (g) Percentages of donor-derived cells after competitive reconstitution. n=5 mice for cre (-), n=7 mice for cre (+). (h) FACS analyses of HSC cell cycle with Ki-67 and Hoechst 33342 staining. n=6 mice for cre (-), n=7 mice for cre (+). (i) HSC localization relative to arterioles. Error bars: n=3 mice. P value has been calculated using n=224 HSCs for cre (-), 274 HSCs for cre (+) pooled from 3 mice per group. P =0.2872. (j-l) Analyses of NG2-cre ERTM / Scf fl/- mice. (j) Absolute nu
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 MLN cells from Roquin san/san mice are proliferative and have more SLECs. Based on Ki67 staining, (panel B ) a greater proportion of OX40 + cells and ICOS + MLN cells were proliferating T cells compared to MLN cells from (panel A ) normal mice. Representative staining from 1 normal and 2 Roquin san/san mice. Similarly, there was a greater proportion of CD44 hi CD62L lo KLRG1 + SLECs present in MLN cells of (panel D ) Roquin san/san mice compared to (panel C ) normal mice. Representative data from 3 normal and 3 Roquin san/san mice.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
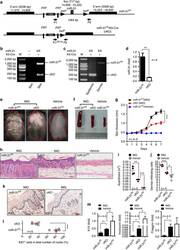
- Experimental details
- Figure 3 Decreased epidermal hyperplasia and dermal cellular infiltrates in miR-31 fl/fl /K5-Cre mice treated with IMQ. ( a ) Schematic representation of primers for genotyping and targeting strategy. ( b ) miR-31 genotyping using P1/P2, 1,064 bp band for miR-31 fl/fl and 235 bp band for miR-31 fl/fl /K5-Cre (cKO). DNA samples were prepared from total skin. ( c ) Cre-mediated tissue-specific deletion of miR-31 in epidermis. DNA samples were prepared from either epidermis or dermis. ( d ) miR-31 expression in epidermis derived from miR-31 fl/fl and cKO mice. ( e ) Phenotypic presentation of mouse back skin for miR-31 fl/fl or cKO mice treated with IMQ or vehicle for 7 days. ( f ) Splenomegaly and lymphadenopathy in miR-31 fl/fl or cKO mice treated with IMQ or vehicle for 7 days. Data are representative of more than five mice. ( g ) Skin thickness was measured on the days indicated. Symbols represent mean skin thickness+-s.e.m. for five to six mice per group. ( h ) H&E staining of the back skin of miR-31 fl/fl or cKO mice treated with IMQ or vehicle. Dotted line indicates the border between the epidermis and the dermis. Scale bar, 100 mum. ( i ) Acanthosis of miR-31 fl/fl or cKO mice treated with IMQ or vehicle. ( j ) Dermal cellular infiltrates of miR-31 fl/fl or cKO mice treated with IMQ. ( k ) Immunostaining of Ki67 in lesional skin derived from miR-31 fl/fl (left panel) or cKO (right panel) mice treated with IMQ. Scale bar, 100 mum. ( l ) Quantitation of Ki67 + cells in epi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
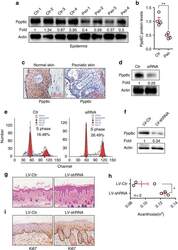
- Experimental details
- Figure 5 Inhibition of ppp6c is functionally important for the biological effects of miR-31 in epidermal hyperplasia. ( a , b ) Western blotting of ppp6c expression in the epidermis of normal skin derived from four healthy individuals (Ctr-1 to 4) or in psoriatic lesions derived from four patients with psoriasis (Pso-1 to 4). ( c ) Immunohistochemical staining of ppp6c in skin sections derived from healthy or psoriatic skin. Scale bar, 100 mum. ( d ) Primary mouse keratinocytes were transfected with scramble siRNA (Ctr) or with ppp6c siRNA (siRNA). Cell lysates were immunoblotted with anti-ppp6c or anti-actin. ( e ) Cell cycle analysis of mouse primary keratinocytes transfected with scrambled siRNA or ppp6c siRNA. ( f ) Western blotting of ppp6c expression in epidermis derived from lentiviral shRNA-control (LV-Ctr) or lentiviral shRNA-ppp6c (LV-shRNA) treated mice. ( g ) H&E staining of the back skin injected with LV-Ctr or LV-shRNA in mice applied with IMQ. Dotted line indicates the border between the epidermis and dermis. Scale bar, 100 mum. ( h ) Acanthosis of the back skin injected with LV-Ctr or LV-shRNA in mice applied with IMQ. ( i ) Immunohistochemical staining of Ki67 in skin sections derived from mice injected with LV-Ctr or LV-shRNA prior to IMQ painting. Dotted line indicates the border between the epidermis and dermis. Scale bar, 50 mum. Values ( a , d , f ) were expressed as fold changes relative to Ctr-1 ( a ), to scramble siRNA ( d ), or to lentiviral shRNA-co
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
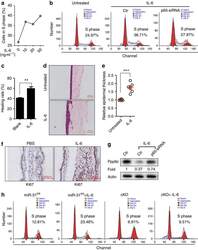
- Experimental details
- Figure 6 NF-kappaB signaling inhibits ppp6c expression by inducing miR-31. ( a ) Undifferentiated NHEK proliferation induced by various concentrations of IL-6 for 24 h, and analysed by BrdU incorporation assay. ( b ) NHEK were transfected with scramble siRNA (Ctr) or with p65 siRNA (p65 siRNA). Cell cycle analysis of NHEK or transfected NHEK treated without or with IL-6 for 24 h. ( c ) In vitro wound healing rate of NHEK treated without or with IL-6 for 16 h. ( d , e ) Three-dimensional organotypic culture of HaCaT keratinocytes treated without or with IL-6. Scale bar, 100 mum. ( f ) 1 mug recombinant mouse IL-6 (in 25 mul PBS) or PBS was injected i.d. in ears of C57BL/6J mice. Ear sections were prepared for Ki67 staining 3 days after IL-6 administration. Scale bar, 100 mum. ( g ) NHEK were transfected with scramble siRNA (Ctr) or with p65 siRNA (p65 siRNA). Western blotting of ppp6c expression in NHEK with or without IL-6 treatment for 24 h. ( h ) Cell cycle analysis of primary mouse keratinocytes derived from miR-31 fl/fl or cKO mice in absence or presence of IL-6. Values were expressed as fold changes relative to non-stimulated HaCaT keratinocytes ( e ) or to non-stimulated NHEK ( g ) and normalized to beta-actin. IL-6 was used at the concentration of 50 ng ml -1 ( b - e , g , h ). ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
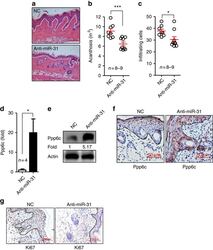
- Experimental details
- Figure 7 Administration of antagomir-31 decreases epidermal hyperplasia and dermal cellular infiltration induced by IMQ. Mice were injected subcutaneously with an irrelevant antagomir (NC) or an antagomir to miR-31 (anti-miR-31). The first injection was administered 3 days before the application of IMQ and thereafter was performed every other day until the end of the experiment. ( a ) H&E staining of the back skin derived from mice injected with NC (upper panel) or anti-miR-31 (lower panel). Scale bar, 100 mum. ( b , c ) Acanthosis and dermal cellular infiltrates were quantitated for mice treated with NC or anti-miR-31. ( d , e ) Ppp6c mRNA and protein levels in NC- or anti-miR-31-treated mice. ( f , g ) Immunohistochemical staining of ppp6c or Ki67 in skin sections derived from NC- or anti-miR-31-treated mice after induction of skin phenotype by IMQ ( n =8-9). Scale bar, 50 mum ( f ) or 100 mum ( g ). For all measurements ( c ), the median number of specifically stained dermal nucleated cells was counted in three high-power fields per section. Results ( d ) are presented as the ratio of mRNA to the beta-actin, relative to that in NC-treated mice. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
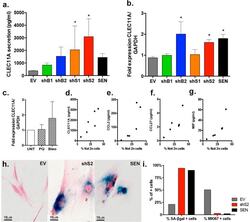
- Experimental details
- Figure 7 W-CIN induced senescent cells secrete CLEC11A. ( a ) Levels of CLEC11A secreted in the CM of each SMC1 and BUB1-depleted cell line. ( b ) Fold expression of CLEC11A mRNA levels normalized to GAPDH in W-CIN-induced senescent and SEN cells and ( c ) Paraquat (PQ, black and white stripe bar) or Bleomycin (Bleo, gray bar)-induced senescence relative to untreated cells (UNT, white bar). ( d-g ) Secretion levels of SASP factors that significantly correlate with W-CIN levels: ( d ) CLEC11A, ( e ) CCL2, ( f ) CCL27 and ( g ) MIF. ( h ) Bright field representative images of SA-betagal staining in EV, shS2 and SEN fibroblasts 45 days after lentiviral transduction. ( i ) Quantification of SA-betagal and MKI67 positively stained fibroblasts 45 days after lentiviral transduction. (*) Indicates significant differences ( p < 0.05) from EV cells tested by One-way ANOVA. Data are expressed as mean +- SD (n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
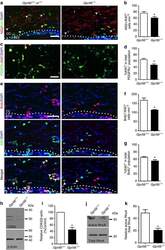
- Experimental details
- Figure 5 Loss of GPR56 leads to fewer proliferating OPCs. ( a ) Representative images of NG2 (red) and Ki67 (green) double IHC in the CC of Gpr56 +/- and Gpr56 -/- P14 mice. Arrowheads indicate double-positive cells. ( b ) Quantification of NG2 and Ki67 dual-positive cells. The asterisks represent significance based on unpaired t -test. P =0.0382; n =6 per genotype. ( c ) Representative images of PDGFRalpha (green) and Ki67 (red) double immunostaining on OPCs after cultured for 4 days in proliferation media. ( d ) Quantification of PDGFRalpha and Ki67 dual-positive OPCs. The asterisks represent significance based on paired t -test. P =0.0156; n =3 per genotype. ( e ) Representative images of BrdU (red) and Ki67 (green) double staining on P14 Gpr56 +/+ and Gpr56 -/- brains that were pulsed with BrdU 24 h before. Arrowheads indicate double-positive cells. ( f ) The number of BrdU and Ki67 double-positive cells was quantified in the CC of Gpr56 -/- mice compared with controls. The asterisks represent significance based on unpaired t -test. P =0.0153; n =3 per genotype. ( g ) The percentage of Ki67 + in the total BrdU + cell population was quantified in the CC of Gpr56 -/- mice compared with the Gpr56 +/+ controls. The asterisks represent significance based on unpaired t -test. P =0.0289; n =4 per genotype. ( h ) Western blot depicting CDK2 protein level in actually isolated OPCs from Gpr56 +/+ and Gpr56 -/- P6 mice. ( i ) The relative CDK2 protein levels were shown. The asterisk
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
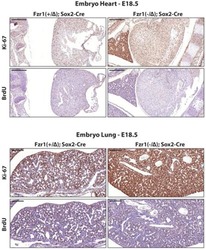
- Experimental details
- Figure 2--figure supplement 1. Ki-67 expression is restricted to proliferating cells by APC/C-Cdh1. Top, IHC staining of Ki-67 and BrdU in sagittal section of embryo heart (E18.5) of Fzr1 (+/Delta) ;Sox2-Cre and Fzr1 (-/Delta) ;Sox2-Cre mice. Scale bar, 500 um. Bottom, IHC staining of Ki-67 and BrdU in sagittal section of embryo lung (E18.5) of Fzr1 (+/Delta) ;Sox2-Cre and Fzr1 (-/Delta) ;Sox2-Cre mice. Scale bar, 200 um. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
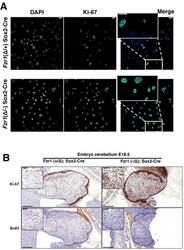
- Experimental details
- Figure 2. Maintenance of Ki-67 expression in quiescent cells in vivo by Cdh1 mutation. ( A ) Immunofluorescence analysis of Ki-67 in MEF cells isolated from embryo (E13.5) of Fzr1 (+/Delta); Sox2 -Cre and Fzr1(-/Delta); Sox2 -Cre mice. Scale bar, 25 um. ( B ) IHC staining of Ki-67 and BrdU in sagittal sections of embryo cerebellum (E18.5) of Fzr1 (+/Delta); Sox2 -Cre and Fzr1 (-/Delta); Sox2 -Cre mice. Bars, 200 um. Bars in zoom, 50 um. DOI: http://dx.doi.org/ Figure 2--figure supplement 1. Ki-67 expression is restricted to proliferating cells by APC/C-Cdh1. Top, IHC staining of Ki-67 and BrdU in sagittal section of embryo heart (E18.5) of Fzr1 (+/Delta) ;Sox2-Cre and Fzr1 (-/Delta) ;Sox2-Cre mice. Scale bar, 500 um. Bottom, IHC staining of Ki-67 and BrdU in sagittal section of embryo lung (E18.5) of Fzr1 (+/Delta) ;Sox2-Cre and Fzr1 (-/Delta) ;Sox2-Cre mice. Scale bar, 200 um. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
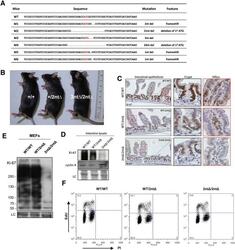
- Experimental details
- Figure 3. Mouse development with a mutated Ki-67 gene. ( A ) Table describing Ki-67 mutant mouse lines resulting from germline transmission of mutations generated by cytoplasmic injection of TALEN-encoding mRNA into zygotes. ( B ) Macroscopic appearance of littermate female mice at 10 weeks of age. Genotypes are specified. ( C ) IHC staining of Ki-67 in sagittal section of intestine from Mki67 WT/WT , Mki67 WT/2nt and Mki67 2nt/2nt mice. ( D ) Western blots of Ki-67 and cyclin A expression from intestine isolated from Mki67 WT/WT , Mki67 WT/2nt and Mki67 2nt/2nt mice. LC, loading control. ( E ) Western blot of Ki-67 in MEFs from WT, Mki67 WT/2nt and Mki67 2nt/2nt mice. LC, loading control. ( F ) Flow cytometry profiles in WT, Mki67 WT/2nt and Mki67 2nt/2nt MEFs showing EdU incorporation upon a 1 hr pulse and DNA content. DOI: http://dx.doi.org/ Figure 3--figure supplement 1. Ki-67 mutant mice develop normally. ( A ) Pair of TALE-nucleases designed to target the initiator ATG of mouse Mki67 gene. ( B ) Sequencing traces of initiator ATG (underlined) of Mki67 gene in WT Mki67 +/+ (WT/WT), heterozygous Mki67 +/2nt (WT/2nt) and homozygous Mki67 2nt/2nt (2nt/2nt) mice. ( C ) Sequencing traces of initiator ATG (underlined) of Mki67 gene in WT Mki67 +/+ (WT/WT), heterozygous Mki67 +/21nt (WT/21nt) and homozygous Mki67 21nt/21nt (21nt/21nt) mice. ( D ) WT Mki67 +/+ (+/+), heterozygous Mki67 +/21nt (+/21nt) and homozygous Mki67 21nt/21nt (21nt/21nt) mice. DOI: http://dx.doi.org/ Figur
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
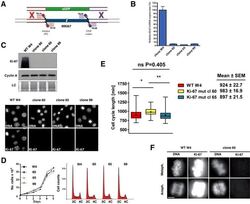
- Experimental details
- Figure 4. Cell proliferation without Ki-67. ( A ) Schematic representation of strategy for TALEN-mediated generation of Mki67 allele. ( B ) qRT-PCR analysis of Ki-67 mRNA levels in NIH-3T3 WT clone W4 and Ki-67-negative 60, 65, 99 clones. ( C ) Top: Western blot of Ki-67 and Cyclin A in NIH-3T3 WT clone W4 and Ki-67-negative mutant clones 60, 65, 99; LC, loading control; below, Ki-67 immunofluorescence; bar, 10 um. ( D ) Left, growth curves of WT and Ki-67 cell lines 60, 65 and 99; right, cell cycle distribution analysed by flow cytometry. ( E ) Cell cycle length of WT clone W4 and Ki-67 clones 60 and 65 as determined by time-lapse videomicroscopy. ( F ) Cells of clone 65 show altered chromosomal periphery in mitosis. The Ki-67 staining is deliberately overexposed to demonstrate absence of detectable Ki-67 in clone 65, even in metaphase. Bar, 5 um. DOI: http://dx.doi.org/ Figure 4--figure supplement 1. Generation of NIH-3T3 cells lacking Ki-67. Top, schematic representation of the wild type (WT), knock-out or eGFP knock-in Mki67 locus and the predicted insertion of tandem repeats of the eGFP insert upstream of Mki67 locus. Bottom, Southern-blot of two NIH-3T3 WT clones (WT, W4) and six NIH-3T3 Ki-67 mutant clones (60,63,65,82,99). Clones were digested with PstI or SphI and probed with 5' or 3' probe, respectively. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
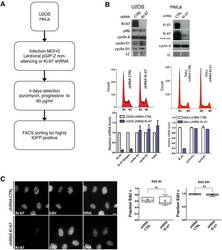
- Experimental details
- Figure 5--figure supplement 2. Cells lacking Ki-67 proliferate efficiently. ( A ) Schematic presentation of generation of Ki-67 shRNA knockdown cell lines. ( B ) Top: Western blot analysis of the indicated proteins in asynchronously growing U2OS and HeLa cells stably expressing non-targeting (CTRL) or Ki-67 shRNA. Lanes separated by lines were from a single exposure of a single SDS-PAGE gel and Western blot. LC, loading control. Middle: Cell cycle distribution of these cells. Bottom: qRT-PCR analysis of the indicated mRNA levels, normalized by mRNA expression of beta-2-microglobulin ( B2m ). ( C ) Immunofluorescence of Ki-67 and EdU in asynchronous U2OS cells stably expressing control or Ki-67 shRNA, incubated with 5-ethynyl-2'-deoxyuridine (EdU) for 3 hr. Bar, 10 um. Right: Ratio of EdU-positive cells to the total cell number for each incubation time; ns: not significant. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
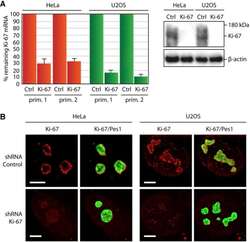
- Experimental details
- Figure 7--figure supplement 3. Depletion of Ki-67 does not affect overall nucleolar structure. ( A ) Histograms showing the level of Ki-67 mRNA remaining in the cell lines expressing constitutively the shRNA against Ki-67. These levels were assessed by RT-qPCR using two different pairs of primers. Right, the Western-blot against Ki-67 shows a disappearance of a bands in the cell lines constitutively expressing the shRNA against Ki-67. ( B ) Immuno-fluorescence against Ki-67 in control HeLa and U20S cell lines in the absence (control) or in the presence (Ki-67) of the shRNA targeting Ki-67 mRNA. The PES1 signal shows that the nucleolar structure is maintained. Objective 100 x. Scale bar: 5 mum. DOI: http://dx.doi.org/
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
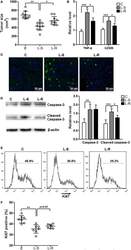
- Experimental details
- Figure 5 Rik expression in CD8 + T cells favors tumor survival . (A) Tumor size. C: control mice receiving phosphate-buffered saline. L-S: mice receiving CD8 + T cells transduced with scramble lentiviruses. L-R: mice receiving CD8 + T cells transduced with Rik-expressing lentiviruses. Each circle represents an individual mouse. (B) mRNA levels of TNF-alpha and granzyme B in tumor tissues. N = 4 per group. (C) Tumor cell apoptosis is indicated by terminal deoxynucleotidyl transferase dUTP nick end labeling. This is a representative of three independent experiments. (D) Activation of caspase-3 in tumor tissues. Left panel: representative Immunoblot image. Right panel: statistics of caspase-3. N = 5 per group. (E,F) Tumor cell proliferation is demonstrated by Ki67 staining. Tumor implants were digested as described in Section "" Materials and Methods ."" Then the whole tissue was pressed through a 70-mum nylon mesh to prepare a single cell suspension, followed by staining with APC anti-CD45 and APC anti-CD31 antibodies. Cells were then stained for Ki67 as described in Section "" Materials and Methods ."" CD45 - CD31 - tumor cells were shown here. Representative histograms are shown in panel (E) , and statistical analysis for Ki67 + cells were shown in panel (F) . N = 7 per group (* p < 0.05; ** p < 0.01; *** p < 0.001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
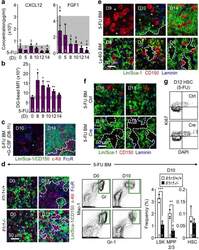
- Experimental details
- Extended Data Fig. 9 Mechanisms controlling GMP cluster formation during regeneration a , ELISA measurement of cytokine levels in BM fluids of 5-FU-treated WT mice at the indicated days post-treatment. b , Quantification of vascular leakage in 5-FU-treated BM at the indicated days post-treatment. Results are expressed as dragon green (DG) microsphere MFI upon masking of laminin + blood vessels. c , Representative IF staining showing GMPs (purple) in 5-FU-treated BM with concomitant daily injections of G-CSF (+G) on d8-11. d , Investigation of 5-FU-treated Il1r1 +/+ and Il1r1 -/- mice at the indicated days post-treatment showing representative IF staining of GMPs (purple), FACS plots of Gr regeneration, and quantification of the indicated BM populations. e , Representative IF staining of CD150 + megakaryocytes (red) in 5-FU- and Ly-6G-treated BM. f , g , Megakaryocyte depletion studies in diphtheria toxin (DT) injected iDtr (Ctrl) and Cxcl4-Cre:iDtr ( Cre ) mice showing (f) representative IF staining of CD150 + megakaryocytes (red) at the indicated days post-5-FU, and (g) representative Ki67/DAPI staining of HSCs at d12 post-5-FU. Stars indicate pGMPs and dotted lines cGMPs. Results are expressed as mean +- S.D. (grey bars, reference range); *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. Egr2 and 3 function is cell intrinsic. (A-C) Irradiated WT mice were adoptively transferred with an equal number of BM cells from WT and CD2-Egr2/3 -/- (K2-3) mice. 8 wk after transfer, mice were infected with OVA-VV WR and analyzed 7 d after infection. (A) Splenic cells from chimeric mice were stained with CD45.1, CD45.2, CD4, and CD8, and the proportion of WT (CD45.1) and K2-3 (CD45.2) CD4 and CD8 cells was determined by flow cytometry. (B and C) Gated WT (CD45.1) and K2-3 (CD45.2) CD4 and CD8 cells were analyzed for expression of the activation marker CD44 and the proliferation marker Ki67 (left) and TNF and IFNgamma for CD4 cells and granzyme B for CD8 cells (right). The percentages of Ki67 + cells among the CD44 high population are indicated in parentheses in B. (D-G) WT and K2-3 OT1 retrogenic T cells were analyzed in recipient mice before and after infection. (D) GFP + CD8 + CD44 lo cells were isolated from WT and K2-3 OT1 retrogenic mice (left and middle) and confirmed as CD62L + Kb-SIINFEKL-tetramer + cells (right). 3 x 10 5 to 5 x 10 5 WT or Egr2/3 -/- retrogenic-OT1 cells were adoptively transferred to separate naive WT mice. 1 d after transfer, mice were infected with OVA-VV WR and analyzed 7 d after infection. (E and F) Retrogenic-OT1 GFP + CD8 + Kb-SIINFEKL-tetramer + cells among spleen and lymph node cells from recipient mice were identified (E), and the numbers of WT and K2-3 retrogenic-OT1 cells in spleen and lymph nodes (left) and lung (right) were
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
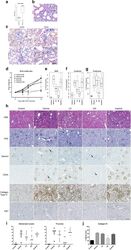
- Experimental details
- Fig. 3 Pharmacological inhibition of DDR1 activation protects animals against NTS-induced crescentic glomerulonephritis. a Quantitative RT-PCR for Ddr1 mRNA on whole kidney lysate of control mice (Control) and mice injected with nephrotoxic serum and treated with vehicle (vehicle). b Representative Ddr1 in situ hybridization (ISH) performed on tissue harvested from mice 14 days after NTS injection. * = crescent c representative DDR1 ISH double labelling with alpha smooth muscle actin (Acta2) or EGF-like module-containing mucin-like hormone receptor-like 1 (Emr-1) in control mice (Control) and mice injected with nephrotoxic serum and treated with vehicle (Vehicle). Arrows = cells labeled with Acta2 or Emr-1. d - g Body weight evolution (day 1, 4, 7 and 14) and renal function parameters ( e - g blood urea nitrogen (BUN), serum creatinine and proteinuria) measured at sacrifice (day 14). h Representative histopathology with Hematoxylin and Eosin (H&E) and Periodic Acid Schiff staining (PAS) and immunohistochemistry for desmin, CD44, Collagen type IV and Ki67. i Glomerular or tubulo-interstitial (TI) summary scores from semiquantitative histopathologic evaluation on H&E and PAS stained kidney sections respectively. j Morphometry analysis of collagen IV IHC. Statistically significant p value: p < 0.05 = *; p < 0.005 = **. Magnification x200, scale bar 100 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 6 AECs self-regenerate, and do not regenerate SECs. a Scheme of experiment design. Bmx -Cre ERT2 ;iTdtomato mice were injected with Tamoxifen to activate Cre expression. Four weeks later, 5-FU was given to these mice and BM ECs were analysed by FACS and immunofluorescence analysis. b Numbers of AECs and SECs 3 days after 5-FU treatment. Data are represented as mean +- SEM. c Representative FACS plot of cell cycle analysis of AECs and SECs from control mice and mice treated with 5-FU (9 days after 5-FU) using Hoechst 3334 and Ki-67. d FACS plot of the labelling of AECs and SECs by Bmx -Cre ERT2 at different time points after 5-FU treatment. e Representative image of whole-mount sternum from mice treated as in ( a ). The bone was harvested on day 17 after 5-FU injection. Mice were injected i.v. with anti-VE-cadherin and anti-CD31. All panels show the same area for different channels. Scale bar, 50 mum. f Scheme of experiment design. Bmx -Cre ERT2 ;iTdtomato mice were injected with Tamoxifen to activate Cre expression. Four weeks later, these mice were lethally irradiated and transplanted with BM cells from wild-type mice. BM ECs were analysed by FACS and immunofluorescence analysis at different time points after lethal irradiation. g Representative images from whole-mount sternum at 1 month (left panel) and 4 months (right panel) after lethal irradiation. Mice were injected i.v. with anti-VE-cadherin and anti-CD31. Scale bar, 200 mum. h Images from whole-mount sternum in w
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3 ERK activation in BRAF V600E microglia (a) CD68, YFP and pERK staining in spinal cord from 7-month old mice. Scale bars=500um, 10um for insets. n=4 per group. (b) pERK + microglia in brainstem. Circles represent individual mice. One-way ANOVA. (c) ERK phosphorylation in spinal cords and brains from 6-9 month-old mice. Top: representative western blot, bottom: pERK/ERK ratio, n=5 per group. One-way ANOVA. (d) pERK expression in YFP + microglia from BRAF VE mice. n=5 per group. Scale bars=5um. (e) Numbers of microglia from 5-9 month-old mice Circles represent individual mice. One-way ANOVA. (f) Heatmap representation of cell frequency among CD45 + cells in the brain. n=3 per group. (g) Ki67 + and cleaved Caspase 3 + (Casp3) expression in YFP + microglia from 5-9 month-old BRAF VE mice, n=6 per group. Unpaired two-tailed t -test. See also Extended data Fig. 7 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 ATF3 maintains intestinal homeostasis. (A) Comparison of colon length between naive mice as indicated. (B) Colon crypts from mice were isolated by shaking colon fragments in EDTA and counted under light microscopy. (C) Flow cytometry analysis of Ki67 and CD24 expression in ileum crypts, gated on the CD45 - EpCAM + populations, from the indicated naive mice. (D) Representative micrographs showing intestinal organoids derived from naive mice. (E) Quantitative real-time PCR analysis of cell cycle genes in naive ileum organoids at day 6 of culture (""n"" indicates organoids derived from 4 mice each group). (F) Representative confocal images of whole mount tissues with co-immunofluorescence staining of UEA-1 and WGA in naive ileum villi. Results were from at least two independent experiments and ""n"" refers to the number of mice unless indicated otherwise. All mice were at the age of 2~3 months old when analyzed. Statistical analysis was done using Multiple T -test on Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
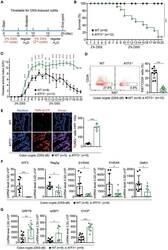
- Experimental details
- Figure 3 ATF3 protects mice from DSS-induced colitis. (A) Experimental protocol of DSS-induced colitis was shown. (B) Survival rate in mice after DSS treatment. (C) Disease activity index (DAI), a composite measurement of weight loss percentage, stool consistency, and blood in stools, was indicated in each group of mice during DSS colitis. (D-G) Analysis of colitis severity at day-8 post DSS treatment. (D) Flow cytometry of Ki67 + proliferating crypt cells in CD24 low/- cell population. (E) TUNEL assay showing apoptotic cells in colon tissues. Magenta positive apoptotic cells were quantified per 100x high-power field (HPF) from 10 different views of colon section from each mouse. (F-G) Quantitative real-time PCR analysis of crypt cells at day-8 post DSS. (F) Expression of ATF3 and anti-microbial peptide-related genes. (G) Expression of ER stress-related genes. Results were from two independent experiments. ""n"" refers to the number of mice analyzed. Survival curve was calculated using the Kaplan-Meier method and statistical significance was calculated using Log rank (Mantel-Cox) test. Statistical analysis was done using Multiple T -test on Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
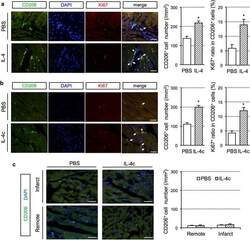
- Experimental details
- Figure 7 Source of increased cardiac M2-like macrophages by IL-4c treatment. ( a ) Viable heart slices were cultured with IL-4 (IL-4 group) or without IL-4 (PBS group) and subjected to immunolabeling for CD206 and Ki67 with DAPI nuclear staining. White arrows indicate CD206 + Ki67 + DAPI + cells. The numbers of CD206 + and Ki67 + ratios in CD206 + cells were shown in the graphs. N = 5 different hearts in each group. * P < 0.05 versus PBS group. ( b ) Two days after intraperitoneally injection of IL-4c or PBS (control) into normal mice, the hearts were subjected to labeling for CD206, Ki67 and DAPI. White arrows indicate CD206 + Ki67 + DAPI + cells. The numbers of CD206 + and Ki67 + ratios in CD206 + cells were counted and shown in the graphs. N = 5 hearts in each group. * P < 0.05 versus PBS group. ( c ) IL-4c or PBS was injected at 20 minutes after coronary artery ligation. At Day 1, the heart was subjected to immunofluorescence for CD206 with DAPI nuclear staining. The numbers of CD206 + cells were shown in the graphs. N = 5 hearts in each group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure S1 Administration of beta-Glucan Promotes Cell Proliferation of LT-HSCs, Related to Figure 1 (A and B) Cell cycle analysis was performed in LT-HSC at 24h after the administration of PBS or beta-glucan by staining for Ki67 and DAPI. (A) Representative flow cytometry plots and (B) frequency of LT-HSC at different phases of the cell cycle (n = 5 mice per group). Data presented as mean +- SEM. * p < 0.05, ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
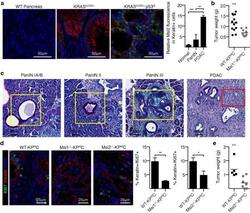
- Experimental details
- Extended Data Figure 4 Analysis of tumors from Msi KP f/f C mice ( a ) Msi2 (green) and Keratin (red) immunofluorescent staining was performed on tissue sections from WT pancreas (Normal, n=3 samples), KRAS G12D/+ ;Ptf1a Cre/+ (PanIN, n=2 samples), and KRAS G12D/+ ;p53 f/f ;Ptf1a Cre/+ (PDAC, n=3 samples) mice with quantification of Msi2 fluorescence in Keratin positive cells. ( b ) Average weights of WT-KP f/f C (n=13) and Msi1 -/- -KP f/f C tumors (n=9). See also Figure 2h-i . for tumor volume analysis ( c ) PAS and Alcian Blue stained sections of pancreata isolated from WT-KP f/f C represent areas used to identify the stages of PanINs (yellow boxes) and adenocarcinoma (red box). ( d ) Tumors from 11-13 week old WT-KP f/f C (n=6), Msi1 -/- -KP f/f C (n=3), and Msi2 -/- -KP f/f C (n=3) mice were stained and quantified for percent of Keratin+ tumor cells (red) expressing Ki67 (green); DAPI staining is shown in blue. ( e ) Average weights of WT-KP f/f C (n=5) and Msi2 -/- -KP f/f C tumors (n=7). See also Figure 2h-I for tumor volume analysis. Data are represented as mean +- SEM. * P < 0.05, ** P < 0.01 , *** P < 0.001 by Student's t-test or One-way ANOVA. Source Data for all panels are available online.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry