Antibody data
- Antibody Data
- Antigen structure
- References [6]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- ABIN258369 - Provider product page

- Provider
- antibodies-online
- Product name
- anti-Glycosylphosphatidylinositol Anchored High Density Lipoprotein Binding Protein 1 (GPIHBP1) (AA 100-170), (Internal Region) antibody
- Antibody type
- Polyclonal
- Description
- Immunogen affinity purified
- Reactivity
- Human, Mouse, Rat, Bovine
- Host
- Rabbit
- Epitope
- Internal Region, AA 100-170
- Vial size
- 0.025 mL
- Storage
- Aliquot and store at -20°C or -80°C. Avoid freeze-thaw cycles.
- Handling
- Avoid freeze-thaw cycles
Submitted references Transcription factor HBP1 is a direct anti-cancer target of transcription factor FOXO1 in invasive oral cancer.
Endothelial cells respond to hyperglycemia by increasing the LPL transporter GPIHBP1.
Highly conserved cysteines within the Ly6 domain of GPIHBP1 are crucial for the binding of lipoprotein lipase.
The acidic domain of GPIHBP1 is important for the binding of lipoprotein lipase and chylomicrons.
The expression of GPIHBP1, an endothelial cell binding site for lipoprotein lipase and chylomicrons, is induced by peroxisome proliferator-activated receptor-gamma.
Glycosylation of Asn-76 in mouse GPIHBP1 is critical for its appearance on the cell surface and the binding of chylomicrons and lipoprotein lipase.
Chan CY, Huang SY, Sheu JJ, Roth MM, Chou IT, Lien CH, Lee MF, Huang CY
Oncotarget 2017 Feb 28;8(9):14537-14548
Oncotarget 2017 Feb 28;8(9):14537-14548
Endothelial cells respond to hyperglycemia by increasing the LPL transporter GPIHBP1.
Pei-Ling Chiu A, Wang F, Lal N, Wang Y, Zhang D, Hussein B, Wan A, Vlodavsky I, Rodrigues B
American journal of physiology. Endocrinology and metabolism 2014 Jun 1;306(11):E1274-83
American journal of physiology. Endocrinology and metabolism 2014 Jun 1;306(11):E1274-83
Highly conserved cysteines within the Ly6 domain of GPIHBP1 are crucial for the binding of lipoprotein lipase.
Beigneux AP, Gin P, Davies BS, Weinstein MM, Bensadoun A, Fong LG, Young SG
The Journal of biological chemistry 2009 Oct 30;284(44):30240-7
The Journal of biological chemistry 2009 Oct 30;284(44):30240-7
The acidic domain of GPIHBP1 is important for the binding of lipoprotein lipase and chylomicrons.
Gin P, Yin L, Davies BS, Weinstein MM, Ryan RO, Bensadoun A, Fong LG, Young SG, Beigneux AP
The Journal of biological chemistry 2008 Oct 24;283(43):29554-62
The Journal of biological chemistry 2008 Oct 24;283(43):29554-62
The expression of GPIHBP1, an endothelial cell binding site for lipoprotein lipase and chylomicrons, is induced by peroxisome proliferator-activated receptor-gamma.
Davies BS, Waki H, Beigneux AP, Farber E, Weinstein MM, Wilpitz DC, Tai LJ, Evans RM, Fong LG, Tontonoz P, Young SG
Molecular endocrinology (Baltimore, Md.) 2008 Nov;22(11):2496-504
Molecular endocrinology (Baltimore, Md.) 2008 Nov;22(11):2496-504
Glycosylation of Asn-76 in mouse GPIHBP1 is critical for its appearance on the cell surface and the binding of chylomicrons and lipoprotein lipase.
Beigneux AP, Gin P, Davies BS, Weinstein MM, Bensadoun A, Ryan RO, Fong LG, Young SG
Journal of lipid research 2008 Jun;49(6):1312-21
Journal of lipid research 2008 Jun;49(6):1312-21
No comments: Submit comment
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
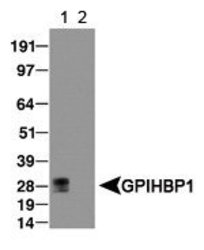
- Experimental details
- WB
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
- Experimental details
- IHC
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry Chromatin Immunoprecipitation
Chromatin Immunoprecipitation