Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [7]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-9381-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Podoplanin Monoclonal Antibody (NZ-1.3), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: Podoplanin is a 40-kDa type I transmembrane sialomucin-like glycoprotein involved in platelet aggregation and cancer metastasis. This protein is expressed on lymphatic endothelial cells and follicular dendritic cells, as well as on many human cancers, including squamous cell carcinoma, glioblastoma, seminoma, and mesothelioma. Podoplanin expression has been demonstrated to be regulated by the Fos protein of the Activating Protein-1 (AP-1) transcription factor complex. Much of its functionality arises via interaction with its endogenous receptor, C-type lectin-like receptor-2 (CLEC-2). This antibody has been reported to block the interaction between Podoplanin and CLEC-2. Applications Reported: This NZ-1.3 antibody has been reported for use in flow cytometric analysis. Applications Tested: This NZ-1.3 antibody has been pre-titrated and tested by flow cytometric analysis of U87 cell line. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- NZ-1.3
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Reprogramming bone progenitor identity and potency through control of collagen density and oxygen tension.
Dissecting human embryonic skeletal stem cell ontogeny by single-cell transcriptomic and functional analyses.
Directed induction of alveolar type I cells derived from pluripotent stem cells via Wnt signaling inhibition.
Stabilization of heterochromatin by CLOCK promotes stem cell rejuvenation and cartilage regeneration.
Kaposi's sarcoma-associated herpesvirus vFLIP promotes MEndT to generate hybrid M/E state for tumorigenesis.
Distinctive Properties of Endothelial Cells from Tumor and Normal Tissue in Human Breast Cancer.
Delayed Union of a Diaphyseal Forearm Fracture Associated With Impaired Osteogenic Differentiation of Prospectively Isolated Human Skeletal Stem Cells.
Geriatric fragility fractures are associated with a human skeletal stem cell defect.
SIRT7 antagonizes human stem cell aging as a heterochromatin stabilizer.
Podoplanin is a novel fos target gene in skin carcinogenesis.
Characterization of anti-podoplanin monoclonal antibodies: critical epitopes for neutralizing the interaction between podoplanin and CLEC-2.
Al Hosni R, Bozec L, Roberts SJ, Cheema U
iScience 2022 Apr 15;25(4):104059
iScience 2022 Apr 15;25(4):104059
Dissecting human embryonic skeletal stem cell ontogeny by single-cell transcriptomic and functional analyses.
He J, Yan J, Wang J, Zhao L, Xin Q, Zeng Y, Sun Y, Zhang H, Bai Z, Li Z, Ni Y, Gong Y, Li Y, He H, Bian Z, Lan Y, Ma C, Bian L, Zhu H, Liu B, Yue R
Cell research 2021 Jul;31(7):742-757
Cell research 2021 Jul;31(7):742-757
Directed induction of alveolar type I cells derived from pluripotent stem cells via Wnt signaling inhibition.
Kanagaki S, Ikeo S, Suezawa T, Yamamoto Y, Seki M, Hirai T, Hagiwara M, Suzuki Y, Gotoh S
Stem cells (Dayton, Ohio) 2021 Feb;39(2):156-169
Stem cells (Dayton, Ohio) 2021 Feb;39(2):156-169
Stabilization of heterochromatin by CLOCK promotes stem cell rejuvenation and cartilage regeneration.
Liang C, Liu Z, Song M, Li W, Wu Z, Wang Z, Wang Q, Wang S, Yan K, Sun L, Hishida T, Cai Y, Belmonte JCI, Guillen P, Chan P, Zhou Q, Zhang W, Qu J, Liu GH
Cell research 2021 Feb;31(2):187-205
Cell research 2021 Feb;31(2):187-205
Kaposi's sarcoma-associated herpesvirus vFLIP promotes MEndT to generate hybrid M/E state for tumorigenesis.
Chen W, Ding Y, Liu D, Lu Z, Wang Y, Yuan Y
PLoS pathogens 2021 Dec;17(12):e1009600
PLoS pathogens 2021 Dec;17(12):e1009600
Distinctive Properties of Endothelial Cells from Tumor and Normal Tissue in Human Breast Cancer.
Wilkus K, Brodaczewska K, Kajdasz A, Kieda C
International journal of molecular sciences 2021 Aug 17;22(16)
International journal of molecular sciences 2021 Aug 17;22(16)
Delayed Union of a Diaphyseal Forearm Fracture Associated With Impaired Osteogenic Differentiation of Prospectively Isolated Human Skeletal Stem Cells.
Goodnough LH, Ambrosi TH, Steininger H, DeBaun MR, Abrams GD, McAdams TR, Gardner MJ, Chan CK, Bishop JA
JBMR plus 2020 Oct;4(10):e10398
JBMR plus 2020 Oct;4(10):e10398
Geriatric fragility fractures are associated with a human skeletal stem cell defect.
Ambrosi TH, Goodnough LH, Steininger HM, Hoover MY, Kim E, Koepke LS, Marecic O, Zhao L, Seita J, Bishop JA, Gardner MJ, Chan CKF
Aging cell 2020 Jul;19(7):e13164
Aging cell 2020 Jul;19(7):e13164
SIRT7 antagonizes human stem cell aging as a heterochromatin stabilizer.
Bi S, Liu Z, Wu Z, Wang Z, Liu X, Wang S, Ren J, Yao Y, Zhang W, Song M, Liu GH, Qu J
Protein & cell 2020 Jul;11(7):483-504
Protein & cell 2020 Jul;11(7):483-504
Podoplanin is a novel fos target gene in skin carcinogenesis.
Durchdewald M, Guinea-Viniegra J, Haag D, Riehl A, Lichter P, Hahn M, Wagner EF, Angel P, Hess J
Cancer research 2008 Sep 1;68(17):6877-83
Cancer research 2008 Sep 1;68(17):6877-83
Characterization of anti-podoplanin monoclonal antibodies: critical epitopes for neutralizing the interaction between podoplanin and CLEC-2.
Ogasawara S, Kaneko MK, Price JE, Kato Y
Hybridoma (2005) 2008 Aug;27(4):259-67
Hybridoma (2005) 2008 Aug;27(4):259-67
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
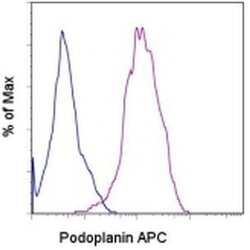
- Experimental details
- Staining of U87 cell line with Rat IgG2a K Isotype Control APC (Product # 17-4321-81) (blue histogram) or Anti-Human Podoplanin APC (purple histogram). Total viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
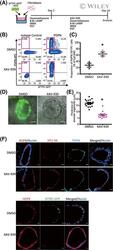
- Experimental details
- 6 FIGURE XAV-939 increases the population of iAT1 cells in FD-AOs. A, A schematic diagram of the analysis of the effect of XAV-939 on FD-AOs. B,C, Flow cytometric analyses of the PDPN and SFTPC expression of FD-EpCAM + cells (P2-5). FD-AOs (P2-5) were treated with 10 muM XAV-939 or DMSO from day 2 to 14, and then the proportion of SFPTC - PDPN + cells in FD-AOs was measured on day 14 (mean +- SEM, n = 3 from three independent experiments). The values indicate the percentage of each population. * P < .05 vs DMSO-treated hiPSC-derived SFTPC - PDPN + cells (unpaired two-tailed Student's t test). D,E, Representative bright-field and SFTPC-GFP fluorescence microscopic live cell images of FD-AOs (P2-5) treated with DMSO or 10 muM XAV-939 from day 2 to 14. Thickness of each FD-AO (P2-P5) treated with DMSO or 10 muM XAV-939 was measured (mean +- SEM, n = 18 spheres from three independent experiments). Scale bars = 50 mum. * P < .05 vs DMSO-treated FD-AO (unpaired two-tailed Student's t test). F, Immunostaining of AT1 markers (AGER, HOPX, HT1-56, and/or PDPN) and SFTPC-GFP in DMSO- or XAV-939-treated FD-AOs (P3). Scale bars = 10 mum. DMSO, dimethyl sulfoxide; FD-AOs, fibroblast-dependent alveolar organoids; hiPSC, human induced pluripotent stem cell
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
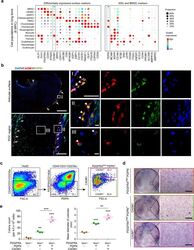
- Experimental details
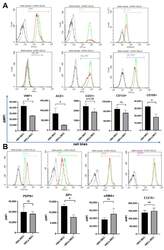
- Fig. 4 Identification of CADM1 as a phenotypic marker of eSSPCs. a Dot plots showing the expression of differentially expressed cell surface genes (left) and candidate SSC markers (right) in 8 WPC human long bone subsets. Asterisks indicated positive markers that were used to enrich eSSPCs. b Immunofluorescent images of PDPN + CADM1 + cells in 8 WPC human long bones. Overviews of PDPN + CADM1 + cells (arrows) in the articular (upper left) and POC (bottom left) regions were shown on the left. PDPN + CADM1 + cells were found in the inner layer of perichondrium in the articular regions (I) and surrounding POC (II). A few PDPN + CADM1 + cells were also found inside POC (III). Arrow heads indicated enlarged PDPN + CADM1 + cells. Merged and single-channel images of DAPI (blue), CADM1 (red) and PDPN (green) were shown ( n = 2 embryos). Scale bars in snapshot images, 50 mum; scale bars in magnified images, 5 mum. c Flow cytometry gating strategies for sorting different populations in 8 WPC long bones ( n = 3 embryos). d Representative crystal violet staining of CFU-F colonies generated by the sorted populations as indicated in c . Magnified images of the boxed areas were shown on the right. Scale bars, 25 mum. e Quantifications of the number (left) and mean diameter (right) of the CFU-F colonies ( n = 3 embryos). The statistical significance of differences was determined using one-way ANOVA with multiple comparison tests (LSD). * P < 0.05; ** P < 0.01; *** P < 0.001. Error bars indic
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
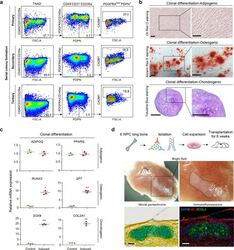
- Experimental details
- Fig. 5 Functional characterizations of eSSPCs in vitro and in vivo. a Flow cytometry plots showing the maintenance of phenotypic eSSPCs after serially passaging clonally expanded PDGFRA low/- PDPN + CADM1 + cells ( n = 3 clones). b Representative oil red O (top), alizarin red (middle) and toluidine blue (bottom) staining after adipogenic, osteogenic and chondrogenic differentiation of clonally expanded eSSPCs (PDGFRA low/- PDPN + CADM1 + ). Magnified images of the boxed areas were shown on the right. Scale bars, 200 mum. c qPCR analyses of adipogenic, osteogenic and chondrogenic marker genes in clonally expanded eSSPCs before and after trilineage differentiation in vitro ( n = 3 clones). The statistical significance of differences was determined using Wilcoxon signed rank test. * P < 0.05; ** P < 0.01. Error bars indicated SEM. d Renal subcapsular transplantation. The work flow for functional characterization of eSSPC in vivo (top). Subcapsular xenografts were dissected and sectioned 8 weeks after transplantation of culture expanded eSSPCs into immunodeficient mice. Bright field (middle), Movat pentachrome staining (bottom left; cartilage, blue; bone and fibrous tissue, yellow) and immunofluorescent staining images (bottom right; DAPI, blue; collagen I (COL1), red; collagen II (COL2), green) were shown ( n = 9 grafts from three embryos). Scale bars, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
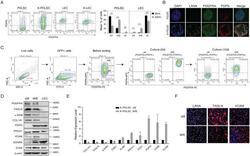
- Figure 2 Characterization of ECs by flow cytometry markers. The cells were stained for the following markers: VWF, ACE, CD31, CD34, CD133, CD105, PDPN, AP, alphaSMA, EGFR. Data were recorded for 10,000 events using CellQuest software (v.2.3.0.84) and presented as histogram overlays. ( A ) The most characteristic markers for ECs and EPCs. ( B ) Markers associated with cancerous phenotype. Histogram overlays display representative repetitions; gray--healthy ECs unstained, black--tumor ECs unstained, green--healthy ECs stained, red--tumor ECs stained. Y axis = the number of events; X axis = fluorescence intensity; the bar charts present delta MFI. Ns--not significant; * p < 0.05 in Student t test vs. HBH.MEC. Data are reported as the means +- SEM (n = 3).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- 10.1371/journal.ppat.1009600.g005 Fig 5 Characterization of KSHV-infected PDLSCs of distinct states in the mesenchymal-to-endothelial differentiation spectrum. (A) Mock- and KSHV-infected PDLSCs and LECs were examined for PDGFRA and PDPN expression profile by flow cytometry analysis. Three subpopulations (xM, hybrid M/E, and xE) were quantified based on the PDGFRA and PDPN profiles ( n = 3 independent experiments). Statistical analyses were performed using two-tailed Student's test and P-values were calculated by GraphPad Prism. *p < 0.05, **p < 0.01, ***p < 0.001. ( B) PDGFRA and PDPN expression in PDLSC and K-PDLSC spheroids at 4 days post-infection were analyzed by IFA. Scale bar, 50 mum. ( C) K-PDLSCs were stained for PDGFRA and PDPN and sorted by flow cytometry. The purified xM and M/E populations were cultured for 10 days and their PDGFRA/PDPN profiles were examined for their phenotypic plasticity. ( D) Western blot analysis of xM, M/E, and LECs for their mesenchymal and endothelial markers. ( E) The expression profiles of mesenchymal and endothelial markers in hybrid M/E and xM state cells were analyzed at the mRNA level by RT-qPCR. ( F) IFA analysis of LANA, TAGLN, and VCAM in xM and M/E state cells. Scale bar, 50 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Cell surface analysis of AMSCs within the transitional culture AMSCs were cultured in a 10% collagen type I gel for 7 days and embedded in a 0.2% collagen type I gel, making up the transitional culture for an additional 14 days. Flow cytometric analysis was conducted for PDPN, CD73, and CD146 (representative data presented).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
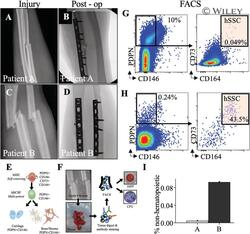
- 1 Fig ( A , C ) forearm radiographs demonstrating bending wedge diaphyseal ulna fracture at injury and immediately postoperatively. ( B , D ) fForearm radiographs demonstrating simple fracture patterns of diaphyseal radius and ulna at injury and immediately post-operatively. ( E ) Schematic of the human SSC lineage tree with a self-renewing stem cell at the top of the hierarchy giving rise to a human bone/cartilage/stromal cell progenitor (hBCSP) and to lineage committed cartilage and bone/stromal cells (PDPN: Podoplanin). ( F ) Experimental workflow for SSC isolation from patient tissue. ( G , H ) FACS plots demonstrating isolation of SSCs from fractures immediately after surgery. ( I ) Frequency of hSSCS at fracture sites as percentage of non-hematopoietic cells for normalization.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry