Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 331588 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Occludin Monoclonal Antibody (OC-3F10), Alexa Fluor 488
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Mouse, Rat, Canine
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- OC-3F10
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C, store in dark
Submitted references A cellular and spatial map of the choroid plexus across brain ventricles and ages.
A human surfactant B deficiency air-liquid interface cell culture model suitable for gene therapy applications.
Establishment and Application of Peristaltic Human Gut-Vessel Microsystem for Studying Host-Microbial Interaction.
A Novel Three-Dimensional Glioma Blood-Brain Barrier Model for High-Throughput Testing of Tumoricidal Capability.
ZEB2, a master regulator of the epithelial-mesenchymal transition, mediates trophoblast differentiation.
Apelin-13 Is an Early Promoter of Cytoskeleton and Tight Junction in Diabetic Macular Edema via PI-3K/Akt and MAPK/Erk Signaling Pathways.
Involvement of CYP1B1 in interferon γ-induced alterations of epithelial barrier integrity.
The EPAC-Rap1 pathway prevents and reverses cytokine-induced retinal vascular permeability.
Electro-Magnetic Nano-Particle Bound Beclin1 siRNA Crosses the Blood-Brain Barrier to Attenuate the Inflammatory Effects of HIV-1 Infection in Vitro.
Size-selective opening of the blood-brain barrier by targeting endothelial sphingosine 1-phosphate receptor 1.
Alix-mediated assembly of the actomyosin-tight junction polarity complex preserves epithelial polarity and epithelial barrier.
Temporal Monitoring of Differentiated Human Airway Epithelial Cells Using Microfluidics.
Dani N, Herbst RH, McCabe C, Green GS, Kaiser K, Head JP, Cui J, Shipley FB, Jang A, Dionne D, Nguyen L, Rodman C, Riesenfeld SJ, Prochazka J, Prochazkova M, Sedlacek R, Zhang F, Bryja V, Rozenblatt-Rosen O, Habib N, Regev A, Lehtinen MK
Cell 2021 May 27;184(11):3056-3074.e21
Cell 2021 May 27;184(11):3056-3074.e21
A human surfactant B deficiency air-liquid interface cell culture model suitable for gene therapy applications.
Munis AM, Hyde SC, Gill DR
Molecular therapy. Methods & clinical development 2021 Mar 12;20:237-246
Molecular therapy. Methods & clinical development 2021 Mar 12;20:237-246
Establishment and Application of Peristaltic Human Gut-Vessel Microsystem for Studying Host-Microbial Interaction.
Jing B, Wang ZA, Zhang C, Deng Q, Wei J, Luo Y, Zhang X, Li J, Du Y
Frontiers in bioengineering and biotechnology 2020;8:272
Frontiers in bioengineering and biotechnology 2020;8:272
A Novel Three-Dimensional Glioma Blood-Brain Barrier Model for High-Throughput Testing of Tumoricidal Capability.
Sherman H, Rossi AE
Frontiers in oncology 2019;9:351
Frontiers in oncology 2019;9:351
ZEB2, a master regulator of the epithelial-mesenchymal transition, mediates trophoblast differentiation.
DaSilva-Arnold SC, Kuo CY, Davra V, Remache Y, Kim PCW, Fisher JP, Zamudio S, Al-Khan A, Birge RB, Illsley NP
Molecular human reproduction 2019 Feb 1;25(2):61-75
Molecular human reproduction 2019 Feb 1;25(2):61-75
Apelin-13 Is an Early Promoter of Cytoskeleton and Tight Junction in Diabetic Macular Edema via PI-3K/Akt and MAPK/Erk Signaling Pathways.
Li Y, Bai YJ, Jiang YR, Yu WZ, Shi X, Chen L, Feng J, Sun GB
BioMed research international 2018;2018:3242574
BioMed research international 2018;2018:3242574
Involvement of CYP1B1 in interferon γ-induced alterations of epithelial barrier integrity.
Alhouayek M, Gouveia-Figueira S, Hammarström ML, Fowler CJ
British journal of pharmacology 2018 Mar;175(6):877-890
British journal of pharmacology 2018 Mar;175(6):877-890
The EPAC-Rap1 pathway prevents and reverses cytokine-induced retinal vascular permeability.
Ramos CJ, Lin C, Liu X, Antonetti DA
The Journal of biological chemistry 2018 Jan 12;293(2):717-730
The Journal of biological chemistry 2018 Jan 12;293(2):717-730
Electro-Magnetic Nano-Particle Bound Beclin1 siRNA Crosses the Blood-Brain Barrier to Attenuate the Inflammatory Effects of HIV-1 Infection in Vitro.
Rodriguez M, Kaushik A, Lapierre J, Dever SM, El-Hage N, Nair M
Journal of neuroimmune pharmacology : the official journal of the Society on NeuroImmune Pharmacology 2017 Mar;12(1):120-132
Journal of neuroimmune pharmacology : the official journal of the Society on NeuroImmune Pharmacology 2017 Mar;12(1):120-132
Size-selective opening of the blood-brain barrier by targeting endothelial sphingosine 1-phosphate receptor 1.
Yanagida K, Liu CH, Faraco G, Galvani S, Smith HK, Burg N, Anrather J, Sanchez T, Iadecola C, Hla T
Proceedings of the National Academy of Sciences of the United States of America 2017 Apr 25;114(17):4531-4536
Proceedings of the National Academy of Sciences of the United States of America 2017 Apr 25;114(17):4531-4536
Alix-mediated assembly of the actomyosin-tight junction polarity complex preserves epithelial polarity and epithelial barrier.
Campos Y, Qiu X, Gomero E, Wakefield R, Horner L, Brutkowski W, Han YG, Solecki D, Frase S, Bongiovanni A, d'Azzo A
Nature communications 2016 Jun 23;7:11876
Nature communications 2016 Jun 23;7:11876
Temporal Monitoring of Differentiated Human Airway Epithelial Cells Using Microfluidics.
Blume C, Reale R, Held M, Millar TM, Collins JE, Davies DE, Morgan H, Swindle EJ
PloS one 2015;10(10):e0139872
PloS one 2015;10(10):e0139872
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent staining of Occludin in Human Caco-2 cells using mouse monoclonal antibody, Alexa Fluor® 488 (Product # 331588). DNA is counter stained with blue Hoechst 33258 (Product # H3569).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
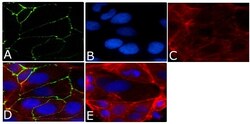
- Experimental details
- Immunofluorescence analysis of Occludin Antibody, Alexa Fluor® 488 conjugate was done on 90% confluent log phase CaCo2 cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Occludin Antibody, Alexa Fluor® 488 conjugate (Product # 33-1511) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cell junctional localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
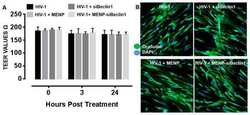
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
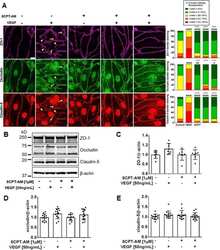
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
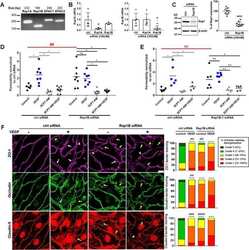
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
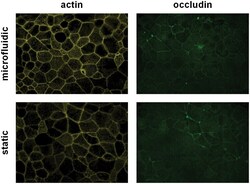
- Experimental details
- Fig 4 Cell viability and barrier integrity of differentiated PBECs in microfluidic compared to static culture conditions. After 24h in microfluidic or static culture conditions, actin filaments (yellow) and the tight junction protein occludin (green) were stained by immunofluorescence. Images are representative of 3 independent experiments using 3 different donors.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Inhibition of apelin receptor APJ using F13A decreased cell proliferation, migration, and the expression of cytoskeleton and tight junction proteins in HRMECs under high glucose condition . (a-c) According to the MTS and transwell assay results, the HRMECs proliferation and migration capability were decreased in a dose-dependent manner, significantly lower in the F13A group, compared with the control one, and the optimum concentration was 20 ng/ml. (d-e) IF staining showed that the phosphorylation of occludin and ZO-1 was downregulated after being treated with F13A in HRMECs under high glucose condition. Cells were stained with DAPI for visualization of nuclei (blue). Scale bar = 50 mu m for (d and e). (f-g) The western blot results showed that the phosphorylation of cytoskeleton and tight junction were also downregulated in a dose-dependent way after being stimulated with F13A. Protein intensity was quantified by Image J software and expressed as fold of change relative to control (mean +- SD, n = 3). *** P < 0.001, ** P < 0.01 versus control one, Student's t -test.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
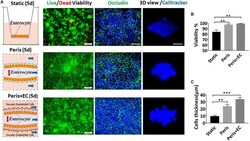
- Experimental details
- FIGURE 3 Impact of peristalsis and endothelial cells on epithelial organization growth. (A) Growth of the Caco-2 epithelial cells cultured in the static Transwell system (Static) versus in the peristaltic microfluidic chip without (Peris) or with endothelial cells (HUVECs; Peris + EC) for 5 days. Schematics (left) showing the system layout; the confocal fluorescence views (center) showing the viability of the Caco2 cells (live cells in green, dead in red) and the distribution of the tight junction protein - occludin in the epithelial monolayers (nuclei in blue, occludin in green); and the laser confocal scanning views (right) showing a vertical cross section of the epithelium highlighting cell shape (bar, 50 mum). Statistical analysis of the viability (B) and the height (C) of intestinal epithelial cell cultured in the static Transwell system or in the peristaltic microfluidic chip without or with endothelial cell for 5 days ( n = 3; ** p < 0.01, *** p < 0.05).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
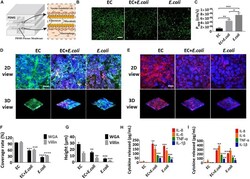
- Experimental details
- FIGURE 7 Intestinal damage and inflammatory responses caused by E. coli on the chip. (A) Conceptual diagram of the microsystem of human inflammatory bowel disease representing co-culture of vascular endothelial cells, enterocyte, macrophages with enterobacteria. (B) The confocal fluorescence views showing the distribution of the tight junction protein - occludin (green) in the epithelial monolayers cultured in the peristaltic human gut-vessel microsystem under control conditions (EC) versus presence of E. coli with (EC + E. coli ) or without ( E. coli ) endothelial cells for 24 h (bar, 50 mum). (C) Quantification of Papp measured by quantitating fluorescent dextran transport across intestinal epithelial layer under the conditions as described in (B) ( n = 3; * P < 0.05, ** p < 0.01, *** P < 0.001). (D) Confocal immunofluorescence views of the sugar chain portion (FITC-WGA, green) of the glycocalyx layer secreted by the Caco2 cells cultured under the conditions described in (B) (nuclei in blue, E. coli in red) (bar, 50 mum). (E) Confocal immunofluorescence views of the villin of the microvilli secreted by the Caco2 cells cultured under the conditions described in (B) (nuclei in blue, villin in red) (bar, 50 mum). Computerized quantification of the coverage (F) and height (G) of the sugar chain portion and villin measured under the conditions described in (B) ( n = 3; ** p < 0.01, *** P < 0.001, **** P < 0.0001). Polarized secretion of proinflammatory cytokines (IL-8, IL-6, TNF
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 8 Anti-inflammatory evaluation of L. casei and antibiotics on the chip. (A) The confocal fluorescence views showing the distribution of the tight junction protein - occludin (green) in the epithelial monolayers cultured in the peristaltic human gut-vessel microsystem with E. coli under control conditions (EC + E. coli ) versus presence of L. casei (EC + E. coli + L. casei ) or P/S (EC + E. coli + P/S) for 24 h (bar, 50 mum). (B) Quantification of Papp measured by quantitating fluorescent dextran transport across intestinal epithelial layer under the conditions described in (A) ( n = 3; * P < 0.05, ** p < 0.01). (C) Confocal immunofluorescence views of the sugar chain portion (FITC-WGA, green) of the glycocalyx layer secreted by the Caco2 cells cultured under the conditions described in (A) (nuclei in blue, E. coli in red) (bar, 50 mum). (D) Confocal immunofluorescence views of the villin of the microvilli layer secreted by the Caco2 cells cultured under the conditions described in (A) (nuclei in blue, villin in red) (bar, 50 mum). Computerized quantification of the coverage (E) and height (F) of the sugar chain portion and villin measured under the conditions described in (A) ( n = 3; ** p < 0.01, *** P < 0.001). Polarized secretion of proinflammatory cytokines (IL-8, IL-6, TNF-alpha, and IL-1beta) in the enteric cavity (H) and vessel lumen (G) under the conditions described in (A) ( n = 3; * P < 0.05, ** p < 0.01).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
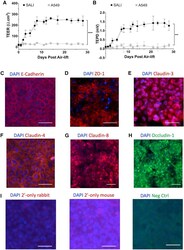
- Experimental details
- Figure 3 SALI forms cultures with strong barrier properties and tight junction markers (A and B) TEER (A) and TEPD (B) measurements taken from SALI cultures and A549 cells grown in ALI culture. SALI cultures established strong functional barrier properties approximately 7 days after air-lift with 249.3 +- 25.1 Omegacm 2 TEER and 1.35 +- 0.13 mV TEPD values. Data represent mean +- SD of six biological replicates. Kolmogorov-Smirnov test was performed to compare functional barrier properties of SALI and A549 ALI cultures (***p = 0.0001; ****p < 0.0001). Immunofluorescence microscopy analyses of tight-junction markers in SALI cultures. (C-H) Expression of (C) E-cadherin, (D) ZO-1, (E) claudin-3, (F) claudin-4, (G) claudin-8, and (H) occludin-1 was detected, confirming the presence of tight junctions in SALI cultures. Representative images from three biological replicates are shown. (I) Staining controls for immunofluorescence microscopy experiments, including secondary-only staining of SALI samples with Alexa Fluor 594-conjugated (left) anti-rabbit and (middle) anti-mouse secondary antibodies used in immunocytochemistry studies, and (right) HEK293T cells were stained with Alexa Fluor 488-conjugated anti-occludin-1 antibody as a negative control. Nuclei are stained blue. Representative images of three biological replicates. All scale bars indicate 50 mum.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Apelin induces the expression of cytoskeleton and tight junction proteins in C57/BL6 mice via PI-3K/Akt and MAPK/Erk signaling pathways . (a-e) The western blot results found that the expression of APJ and the phosphorylation of cytoskeleton (VE-Cadherin and FAK) and tight junction proteins (occludin and ZO-1) increased significantly in apelin-treated (10 and 100 ng/mL) C57BL/6J mice. (f-h) The phosphorylation of PI-3K/Akt and MEK/Erk signaling pathways, p38, Akt, and Erk also increased significantly. Protein intensity was quantified by Image J software and expressed as fold of change relative to control (mean +- SD, n = 3). *** P < 0.001, ** P < 0.01 versus control one, Student's t -test.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
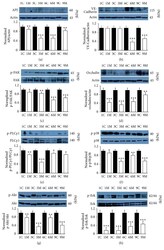
- Figure 8 Inhibition of apelin receptor APJ with F13A dramatically reduces pathological vascular permeability in db/db mice via PI-3K/Akt and MAPK/Erk signaling pathways . The western blot results revealed that the expression of apelin receptor APJ (a), phosphorylation of cytoskeleton (VE-Cadherin and FAK) (b and c), tight junction (occludin) (d), and PI-3K/Akt and MEK/Erk signaling pathways proteins (e-h) were higher in diabetic retinopathy mice, compared with the F13A treated groups. Protein intensity was quantified by Image J software and expressed as fold of change relative to control (mean +- SD, n = 3). *** P < 0.001, ** P < 0.01, * P < 0.05 versus control one, Student's t -test. 1C: one-month control; 1M: one month treated with F13A, 3C: 3-month control; 3M: 3 months treated with F13A; 6C: 6-month control; 6M: 6 months treated with F13A; 9C: 9-month control; 9M: 9 months treated with F13A.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
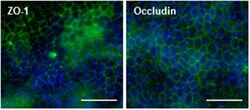
- Figure 2 Presence of tight junction proteins in MDCKII/MDR1 monolayer. Representative confocal images of ZO-1 (left) and occludin (right) immunofluorescence (green) showing characteristic tight junction localization between cells of the monolayer. Nuclei are counterstained with Hoechst (blue). Images were acquired with a 40X objective on the Thermo Scientific CX7 CellInsight and processed with ImageJ using the smoothing filter and rolling ball background subtraction (250 pixel radius). Scale bars = 50 mum.
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry